1 Department of Gynecology, The Fourth Affiliated Hospital of School of Medicine, and International School of Medicine, International Institutes of Medicine, Zhejiang University, 322000 Yiwu, Zhejiang, China
2 Center for Reproductive Medicine, The Fourth Affiliated Hospital of School of Medicine, and International School of Medicine, International Institutes of Medicine, Zhejiang University, 322000 Yiwu, Zhejiang, China
Abstract
Endometriosis in adolescents is challenging to diagnose in its early stages, yet it is widespread among adolescent girls with chronic pelvic pain, posing a significant risk to their physical and mental health, as well as subsequent fertility. The treatment of endometriosis in adolescents primarily involves surgery and pharmacologic therapy, each with distinct advantages and disadvantages. Currently, there is no consensus on selecting the optimal treatment approach. Therefore, this study aimed to review the literature on the management of endometriosis in adolescents.
We conducted a narrative literature review to identify studies evaluating treatments for endometriosis in adolescents, using PubMed, MEDLINE, and Cochrane Library databases between January 2005 and December 2025. The final search was conducted on December 31, 2025.
Our review offers a critical analysis of current therapeutic strategies for endometriosis in adolescent populations and summarizes the characteristics of various management approaches, with an emphasis on comprehensive care and long-term management.
Although oral contraceptive therapy remains the first-line treatment for endometriosis in adolescents, surgery is indicated for those individuals unresponsive to pharmacologic therapy. Clinicians are advised to adopt a personalized approach combining pharmacological and surgical interventions, as well as to provide long-term follow-up to reduce disease recurrence and promote fertility.
Keywords
- adolescent endometriosis
- surgical treatment
- medical therapy
- fertility preservation
Endometriosis is a chronic gynecologic disorder characterized by the ectopic implantation and proliferation of functional endometrial-like tissue outside the uterine cavity. This pathological condition induces localized inflammatory responses, cyclic hemorrhage, and fibrotic lesions; it clinically manifests as secondary dysmenorrhea, chronic pelvic pain, and reproductive complications including infertility [1]. With rising incidence rates among adolescents, approximately 64% of those with chronic pelvic pain who undergo laparoscopy are found to have endometriosis [2]. Diagnostic delay in adolescent patients may extend from 5 to 12 years, and is attributable to nonspecific symptomatology (e.g., functional abdominal pain as the sole manifestation) and high rates of inconclusive imaging findings [3, 4, 5]. When treating young patients with the aforementioned symptoms, clinical gynecologists should screen for family history, an early menarche or short menstrual cycle, and obstructive genital malformations, which are suggestive of the presence of endometriosis [6]. Vaginal and/or rectal examination is crucial for the correct and timely diagnosis and treatment of adolescent patients with endometriosis, while age, sexual history, and cultural background should also be taken into consideration. Imaging examinations such as transvaginal/transrectal/transabdominal ultrasonography and magnetic resonance imaging are equally important in diagnosing endometriosis in adolescents; conversely, molecular biology assays, such as for serum biomarkers, lack relevant specificity [7, 8].
Endometriosis can lead to severe clinical sequelae that include refractory pain, significant quality-of-life impairment, and long-term reproductive health complications. Clinical management currently relies on non-opioid analgesics and hormonal medications that create a hypoestrogenic environment, with treatments focusing on managing pain and reducing recurrence [1, 9, 10]. Laparoscopic surgery can also effectively achieve significant pain relief by excising endometriotic lesions [2, 11, 12, 13, 14]. However, there are still significant controversies regarding the treatment strategies for endometriosis in adolescent girls, including when the best intervention time for surgical treatment is and what to define the surgical scope as, the safety of long-term hormone-suppressive therapy, determining the most appropriate course of treatment for adolescent patients, and what the long-term impact of various treatment modalities on the ovarian reserve are and what potential risks treatments pose to natural fertility. Thus, the management of endometriosis in adolescents necessitates a personalized therapeutic approach [15], and further research is warranted to optimize diagnostic and therapeutic strategies for this population. A literature review that outline the potential benefits of conducting genetic and epigenetic profiling studies on the genetic and epigenetic characteristics of early-onset diseases in this population, which may assist in providing personalized management for adolescents [16]. In this analysis, we evaluated various treatment modalities for endometriosis in adolescents, assessing their efficacy, safety profiles, and long-term outcomes. By synthesizing the currently available evidence, we herein sought to establish an evidence-based, decision-making framework for clinicians so as to ultimately improve prognostic outcomes in this patient cohort.
We conducted a narrative literature review of endometriosis treatments for adolescents using the following electronic databases: PubMed, MEDLINE, and Cochrane Library. We subsequently created a literature-screening flowchart by borrowing the structure of a Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart (Fig. 1).
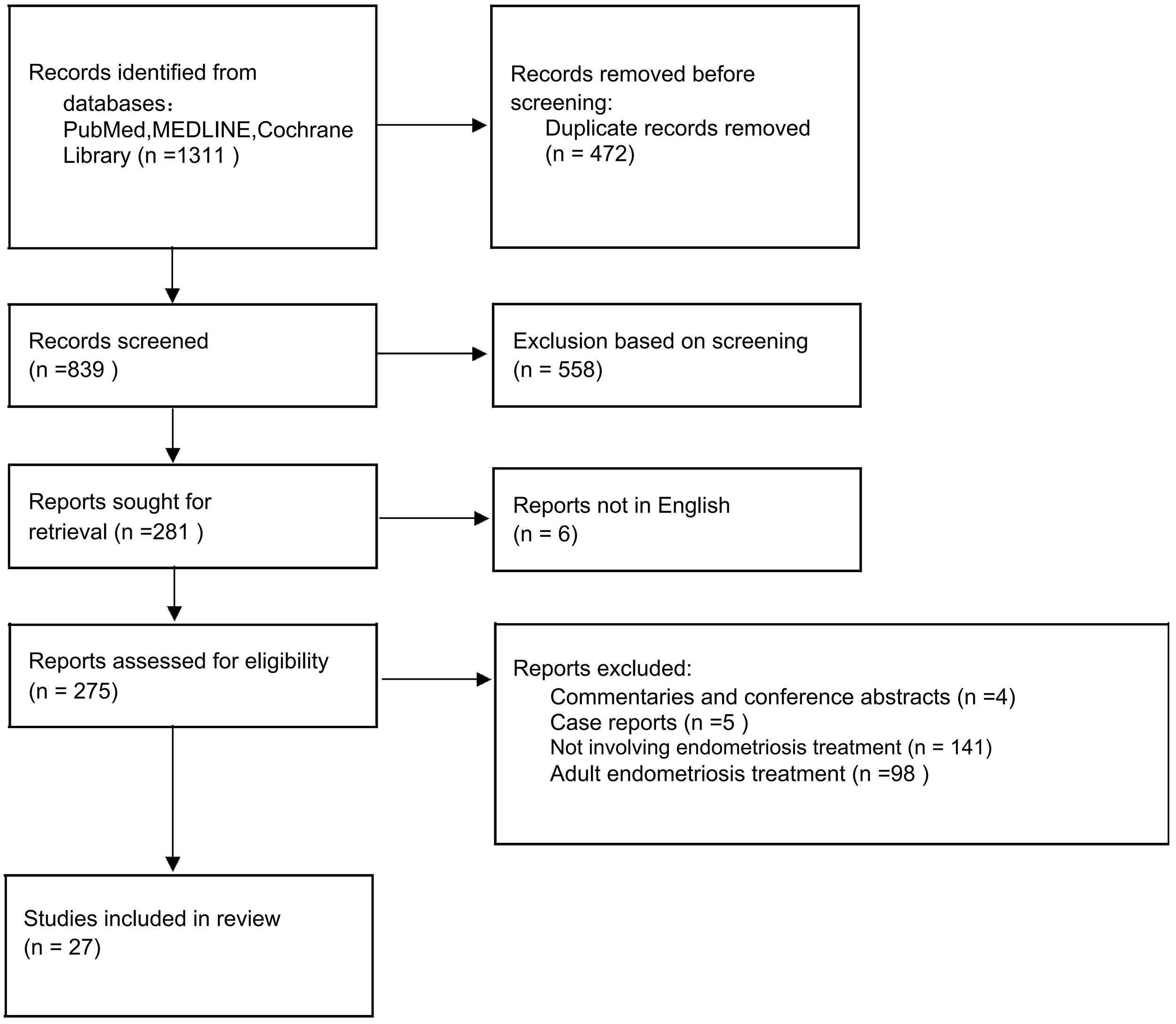 Fig. 1.
Fig. 1.
Flowchart of the literature-screening process.
Our search strategy incorporated controlled vocabulary (MeSH terms) and keywords, including “endometriosis”, “adolescent or adolescence or young or teenager”, and “therapy or therapeutic or management or treatment” to identify all relevant scientific publications. Boolean operators (“AND” and “OR”) were applied to optimize the sensitivity and specificity of the search. The time-frame we considered was from January of 2005 to December of 2025. The age range for adolescents was defined as 10 to 19 years based on the World Health Organization (WHO), based on the high-quality studies we reviewed that included the term “adolescents”, the age limit for patients with endometriosis was relaxed to 10 to 24 years old [2]. Therefore, this review will also discuss patients aged 10 to 24. Two reviewers independently screened all titles and abstracts retrieved through the initial search, and a third reviewer was available for any disputed literature. Additionally, all reference lists for included articles were manually reviewed to identify studies that may have been missed during database searching. The inclusion criteria for this review were as follows: eligible articles (1) pertained to uterine endometriosis treatment with the adolescent age group represented among the research subjects; (2) comprised original research articles, encompassing both prospective and retrospective observational studies, as well as review articles; (3) entailed specific treatment plans; and (4) were published in English. Studies were excluded if the research subjects did not include those aged 10–24; were animal or in vitro studies unrelated to the treatment of adolescent endometriosis; were case reports, editorials, commentaries, or conference abstracts without original data; or were studies with insufficient methodological quality, such as those with a high risk of bias.
The principal complaints of adolescents with endometriosis are endometriosis-associated pain and ovarian cysts, and the aims of long-term management are to improve symptom relief, protect the ovarian reserve and future fertility, delay progression, and prevent recurrence. First-line drug therapy for endometriosis in adolescent comprises hormonal contraceptives, progestogens with effective but small impacts on bone mineral density (BMD), and nonsteroidal anti-inflammatory drugs (NSAIDs) as the mainstays of pain relief [12]. Gonadotropin-releasing hormone (GnRH) agonists effectively reduce endometriosis-associated pain and are prescribed as second-line pharmaceuticals (particularly when hormonal contraceptives or progestogens are ineffective) due to their side-effect profile [12]. Clinical decision-making should therefore balance efficacy, side-effects, and individual patient characteristics in order to achieve medicinal precision (all medical treatments are presented in Table 1).
| Therapy | Mechanism(s) | Indication | Age limit | Impact on BMD | Fertility considerations | Therapeutic evaluation |
| NSAIDs | Inhibit prostaglandin synthesis by inhibiting the COX enzyme and prevent the formation and release of painful substances by directly acting on nociceptors. | All adolescents without severe liver or kidney diseases and no history of drug allergies, and unable to use any form of hormone therapy. | No age limit. | No impact on BMD, primarily gastrointestinal irritation or ulcers. | Lack of research on fertility. | It can initially relieve pain and is effective for mild endometriosis. However, if there is no evident effect, it is necessary to promptly use other hormonal drugs in combination. |
| OCPs | Inhibit ovulation, cause endometrial decidualization, and prevent the growth of endometrial ectopic lesions. | First-line treatment for endometriosis in adolescents, no history of or increased risk for thromboembolism, migraine with aura, hepatic disease. | No age limits. | No impact on BMD. | It may have a protective effect on fertility, but the long-term effects are uncertain. | First-line hormone therapy that effectively alleviates pain and can reduce the recurrence rate after surgery. |
| Progestins | Induce decidualization and atrophy of endometrial tissue; decrease estrogen-induced mitosis; suppress cellular proliferation; inhibit inflammatory pathways, angiogenesis, and neurogenesis. | First-line treatment for endometriosis in adolescents. | The target age tends to be younger adolescents. | Weight gain, bloating, mood lability, irregular bleeding; the impact on BMD is unclear. | Oral progestins principally protect the reproductive potential of adolescent patients indirectly by inhibiting the progression of the disease, but they cannot directly reverse the mechanism of infertility. | Similar to OCPs. |
| LNG-IUS | Reduces the expression of estrogen and progesterone receptors in eutopic (endometrium within the uterine lining) and ectopic endometrial tissues, causing glandular atrophy and decidualization of the stroma. | Long-term treatment after laparoscopic surgery. | Sexually active adolescents. | No impact on BMD has been found. | Limited data. | There is significant symptom relief. It should be combined with other treatments based on the severity of the patient’s symptoms, their mental health history, and their contraceptive needs. |
| DNG | Inhibits the growth, proliferation, and angiogenesis of endometriosis lesions in the uterus; shows anti-inflammatory effects. | Second-line treatment for endometriosis in adolescents. | There is no specific age limit. Tends to be more common among older adolescents. | The incidence of reduced bone density is lower than that with GnRH. | The impact of DNG on fertility is context-dependent. Long-term continuous use may temporarily inhibit ovulation or affect hormone balance. | Relieves pain and shrinks endometriotic cysts, irregular bleeding is more common. |
| GnRH agonists | Downregulates GnRH receptors in the pituitary gland, inhibiting gonadotropin release and ovarian hormone secretion, leading to a hypoestrogenic state. | Second-line treatment for endometriosis in adolescents. | Over the age of 18; avoid under the age of 16. | Perimenopausal symptoms, bone loss. | Postoperative use may increase the pregnancy rate. | Relieves moderate-to-severe pain, but it is necessary to combine with add-back. |
BMD, bone mineral density; NSAIDs, nonsteroidal anti-inflammatory drugs; OCPs, oral contraceptive pills; DNG, dienogest; GnRH, gonadotropin-releasing hormone; LNG-IUS, levonorgestrel-releasing intrauterine system; COX, cyclooxygenase.
NSAIDs are recommended when adolescents have contraindications to hormonal treatment and can be used either alone or in combination with hormonal drugs. In clinical practice, adolescents with dysmenorrhea frequently resort to over-the-counter NSAIDs for initial symptom relief. Due to the high prevalence of self-directed medication among adolescent dysmenorrhea patients and the real possibility of incorrect dosing and timing of sub-treatments, patient education is essential. As the endometriosis progresses, the underlying pain mechanism becomes more complex, and NSAIDs can exert little effect in relieving pelvic pain; patients ultimately seek formal medical care, where evaluation often confirms endometriosis. The standard clinical approach then shifts to initiating hormonal suppression. Opioids often possess ideal analgesic effects by directly acting on peripheral nociceptors and affecting opioid receptors in spinal dorsal horn neurons and higher-level neurons in the pain transmission pathway. However, opioids do not constitute first-line pharmacotherapy for endometriosis-associated pain in adolescents, owing to side-effects such as drug addiction, hypogonadism, and reproductive dysfunction [17, 18]. In a large cohort study conducted in the United States [19], the risk of developing chronic opioid use within 2 years was 4.4% among women aged 18–50 who were diagnosed with endometriosis, while for women without endometriosis the risk was only 1.1%. Moreover, the risk in this cohort was higher for women aged 18–25 relative to those aged 35–50 (odds ratio, 1.39; 95% confidence interval, 1.06–1.82). It can therefore be inferred that the risk of developing an opioid dependence is greater among adolescents than adults; consequently, opioids are not recommended for long-term treatment of endometriosis without professional guidance [20]. Analgesics can only alleviate symptoms, and as the disease progresses, their effectiveness in treating pain gradually diminishes. Moreover, analgesics exhibit almost no positive effect on subsequent fertility.
OCPs can effectively inhibit ovulation and create a low-estrogen environment by suppressing the hypothalamic–pituitary–ovarian axis, providing the dual benefits of dysmenorrheal relief and contraception. Postoperative use of OCPs can also effectively reduce the risk of endometriosis recurrence. In a multicenter study that followed adults for 24 months, postoperative OCP treatment reduced the recurrence of endometriotic cysts [21]. Current OCP regimens vary between continuous (non-cyclic) and cyclic administration (e.g., 21-day drospirenone/ethinyl estradiol followed by a 7-day withdrawal) and are associated with side-effects such as breakthrough bleeding, mastalgia, and weight gain. One study involved 136 adolescent users of continuous drospirenone (DRSP-C) oral contraceptives for menstrual suppression over a 12.3-month study period, of whom 44 exhibited endometriosis. Of these women, 11 withdrew from the study due to breakthrough bleeding. There were only three cases where the incidence of weight gain was a concern, and no one withdrew due to it [22]. In a small-scale, randomized double-blind experiment [23], 76 adolescents under the age of 19 with moderate or severe dysmenorrhea were administered either OCPs (20 micrograms of ethinyl estradiol/100 mg of levonorgestrel) or a placebo for 3 months. The authors ascertained that the side-effects of the OCPs were almost identical to those of the placebo. This might have been related to the adolescents’ susceptibility to psychological suggestion and the short duration of OCP use. In addition, Asians may be more conservative, and thus less likely to use contraceptives compared to Western countries [24]; for adolescents with no sexual history, they may be more likely to refuse contraceptives during their first treatment. This requires doctors to conduct adequate education for patients and their guardians during the consultation. Other, non-pill combined hormonal contraceptive options such as patches and vaginal rings can also be considered for adolescents who have no childbearing plans in the near future [25].
Progestogens induce ectopic endometrial decidualization. One example is
dydrogesterone, which is an orally absorbable synthetic progesterone that does
not possess estrogenic, androgenic, synthetic metabolic, or corticosteroid
properties. Progestogens and OCPs are used as first-line medications for
adolescents with endometriosis. The commonly used progestogens for adolescents
include dydrogesterone, progesterone, contraceptive implants, and a
levonorgestrel-releasing intrauterine system (LNG-IUS). A retrospective analysis
of 91 patients aged 14–25 with endometriosis showed that oral progestins were
more suitable for younger adolescents (15.7
Commonly used clinical agents include leuprolide acetate, nafarelin, buserelin, and goserelin. GnRH agonists are currently recognized as the most effective medication for alleviating endometriosis symptoms and reducing lesion severity by suppressing the release of gonadotropins, thereby reducing estrogen levels. However, there are a number of concerns among clinicians regarding the use of GnRH agonists by adolescents—most notably, the potential for reduction in bone density and perimenopausal symptoms. GnRH agonists are recommend for patients over 18 years of age, and physicians must pay close attention to bone density loss (D-grade evidence) and to supplement GnRH agonists with calcium and vitamin D [30]. Adolescent patients with endometriosis for whom conservative surgical and other drug treatments have been unsuccessful may also benefit from at least 6 months of GnRH agonist therapy [31]. For adolescents with laparoscopically-confirmed endometriosis and associated pain for whom hormonal contraceptives or progestogen therapy has failed, the use of GnRH agonists for up to 1 year may be carefully considered with informed consent, as they are effective and safe when combined with hormonal add-back therapy that mitigates their side-effects [32]. Add-back hormone therapy with estrogen–progestin combination regimens is necessary for adolescents treated with a GnRH agonists in order to improve bone health and quality of life [15]. A randomized controlled trial by DiVasta et al. [33] in adolescent patients (15–22 years of age) revealed that a GnRH agonist (leuprolide acetate at 11.25 mg/3 months) followed by add-back therapy (norethindrone acetate at 5 mg/day alone or combined with conjugated estrogens at 0.625 mg/day) after 21 days of treatment maintained bone health and effectively relieved pain.
Newer GnRH antagonists such as elagolix and linzagolix have shown significant efficacy in adults in some Phase II and III trials [34, 35, 36]. However, no trials aimed specifically at the adolescent population have yet been conducted. Thus, if GnRH agonist therapy is needed to treat endometriosis in adolescents, physicians must carefully consider and discuss potential side-effects and long-term health risks with the patient.
The authors of a recent literature review that encompassed 14 studies demonstrated the effectiveness and safety of DNG for adolescents [37]. DNG is an effective therapeutic option for endometriosis in adolescents, as it demonstrates comparable efficacy to GnRH analogues while avoiding hypoestrogenic side-effects, although long-term use may cause irregular bleeding [37]. In a 52-week multicenter study in Europe, 120 adolescent patients with endometriosis who were 12–18 years of age were enrolled and treated with 2 mg of DNG once daily. These authors’ results showed that pain symptoms related to endometriosis (such as pelvic pain and dysmenorrhea) were significantly reduced during treatment. Although DNG was associated with a decrease in lumbar bone density at the end of treatment, bone density levels recovered after 6 months of discontinuation, but remained below normal levels [38]. When two other patient samples were analyzed after postoperative use of dienogest for 6 or 12 months, both regimens relieved pain and improved quality of life, but the authors did not report on the effect on bone density [39, 40]. Attention should therefore be given to bone loss in adolescents with endometriosis who have been using DNG for an extended period of time.
Current preliminary evidence suggests that supplementation with vitamin D and omega-3 fatty acids may offer some benefit in alleviating pelvic pain in adolescents with endometriosis. There was also a clear placebo effect, which may be related to the fact that the subjects in these studies did not actually lack these nutrients, and that the supplements did not clearly target the mechanism(s) underlying endometriotic pain [41]. Emerging evidence indicates that melatonin may offer benefits which relieve pain and improve sleep quality for endometriosis-associated chronic pelvic pain (EACPP); e.g., acupuncture, herbal preparations, and yoga improved dysmenorrhea [42, 43]. Current evidence for these therapies, however, remains limited to small-scale trials (which are insufficient to confirm their efficacy), and acupuncture is costly. It is unknown whether such therapies could be widely applied to the future treatment of endometriosis in adolescents.
Long-term pharmacotherapy for endometriosis in adolescents requires careful balancing of efficacy against potential impacts on growth, bone health, and future fertility. A proactive management plan should therefore address the following key areas.
In most cases, the peak of bone density occurs between the ages of 18 and 20.
Bone density measurement is recommended to measure the hip and lumbar spine
(L1–L4) and calculate lumbar BMD Z-scores compared to age-matched controls. The
pharmaceutical used to treat endometriosis in adolescents that exerts the most
significant impact on bone density is the GnRH agonist. DiVasta et al.
[44] reported on a small-sample retrospective study comprising 36 patients with
endometriosis aged 13–21. When the reverse-addition regimen of leuprolide
acetate at 11.5 mg every 3 months and norethindrone acetate at 5 mg daily was
adopted for a treatment duration of 392
To reduce the low-estrogenic effect generated by GnRH agonists, progesterone or estrogen and progesterone should be added simultaneously based on the “estrogen-threshold hypothesis”. There is no current consensus as to the extent to which hormones should be added; there is a paucity of sufficient data for the adult population, and research on this aspect among adolescents is even scarcer. The accepted doses of add-back therapy 5 mg/day of norethindrone acetate, or 0.625 mg/day of conjugated estrogen with 5 mg/day of norethindrone acetate. Adolescents tend to favor the latter for its greater life-enhancing impact [47]. Some researchers have suggested starting the supplementary treatment 3 weeks after the first injection of GnRH agonist, based on their clinical experiences [48].
Most initial users of GnRH agonists and add-back patients do not need to measure bone density unless they manifest concurrent diseases that cause bone loss. According to the majority of studies conducted on adults, bone loss usually begins to occur at about the month 6–8 after treatment. Generally speaking, if the treatment lasts for more than 6–8 months, it is recommended to conduct a bone-density test; and bone density should then be monitored every 2 years by serial dual-energy x-ray absorptiometry (DEXA) scans [49]. For patients receiving preoperative or postoperative drug treatments, it is advisable to conduct follow-up visits every 3–6 months. Blood testing should also be implemented to check for severe liver and kidney function impairments, and transvaginal or transabdominal ultrasonography should be employed to assess changes in pelvic lesions.
A large-scale retrospective analysis revealed that drugs used in treatments for adolescents with endometriosis—especially hormones—are frequently changed and used in combination [50]. A retrospective analysis was conducted on the preoperative management of 305 patients under 22 years of age with pathologically confirmed endometriosis, and before the operation, various forms of hormonal therapy were selected, with a small number of opioid drugs used [51]. There are many types of treatment drugs and a wide range of choices. If side effects or unsatisfactory effects occur during the use, patients often change the drugs on their own. The multi-center management was complicated, and it was impossible to determine which was more suitable for the patients—surgery or drugs. We suggest that the management of endometriosis in adolescents involve long-term and fixed follow-up, with detailed records of drug use. It would be optimal if this treatment information was then disseminated. In addition, endometriosis has caused psychological problems of varying degrees to patients of all ages [52]. Among them, the difficulty in sexual intercourse caused by endometriosis brings obvious psychological damage to the group of teenagers, who are in a special period of forming opposite-sex interaction identities, special attention should be paid to the mental health of teenagers during the treatment process [53]. A survey was conducted among 21 patients aged 13 to 25 by filling out mental health questionnaires. It was found that over 79% of the patients had mild anxiety, and nearly half had moderate to severe anxiety. This inevitably had a negative impact on their life and study [54]. A review published in 2025 for teenagers emphasized that cognitive-behavioral interventions, mindful awareness practices, and youth-centered peer networks are crucial. Combined with timely detection and integrated, multi-specialty support, they help alleviate both physical distress and psychological impacts associated with the illness [55].
While laparoscopy is no longer the first-line diagnostic gold standard for endometriosis in adolescents due to the availability of non-invasive imaging modalities, it remains a valuable diagnostic tool in cases where imaging is inconclusive, where medical therapy fails, or when surgical intervention is indicated for symptom management or cyst excision [12]. If treatment with NSAIDs and hormonal drugs for more than 3 months fails, laparoscopic examination is recommended. It is noteworthy that nearly two-thirds of adolescents with chronic non-periodic pain who undergo laparoscopic examination suffer from endometriosis [32]. Surgical treatment can also effectively relieve pain. At a Chinese center specializing in female genital abnormalities, 85 patients under the age of 19 with endometriosis underwent surgery and were followed up with for 24–140 months. Pelvic pain was substantially alleviated postoperatively, with symptoms disappearing in 41.7% of patients and improving in another 38.3% [56]. If laparoscopic examination is conducted, it should be performed by an experienced surgeon so as to completely remove all endometriotic lesions if possible, and to confirm the diagnosis histologically [12]. The aim of surgery is to reconstitute the anatomy, eliminate painful symptoms, and protect fertility [15]; adequate preoperative evaluation is required for adolescent patients with endometriosis. Adolescents should not consider definitive and irreversible surgeries such as oophorectomy, or hysterectomy. Postoperative hormonal therapy should also be carefully considered to suppress symptom or endometrioma recurrence [56]. Additionally, the authors of one literature review proposed that sexually active women can undergo transvaginal endoscopy, which is an infrequently used technology [57].
Endometriosis in adolescents is believed to originate from the activation of dormant stem or progenitor cells seeded below the peritoneum through neonatal menstrual reflux. Thus, endometriosis in adolescents often presents as red, clear, or vascular implants and minimal fibrosis in peritoneal endometriosis; as dark, blood-stained-fluid in ovarian endometrioma; and, rarely, as deep endometriosis that differs from the characteristic advanced endometriosis in adults [58, 59]. During laparoscopic surgery, the surgeon can magnify the view to closely examine suspicious areas. By gently moving the lens or introducing saline into the abdominal cavity, subtle lesions can become more visible, thereby reducing the risk of misdiagnosis. Disease staging should ultimately be performed using the revised American Society for Reproductive Medicine (rASRM) classification system, which provides standardized criteria for lesion documentation and quantification of severity.
Conservative laparoscopic surgery represents the predominant intervention for adolescent patients, integrating meticulous lesion excision with targeted electrocoagulation. During an ovarian cyst-removal surgery, surgeons should provide close attention to the stripping technique used and deploy accurate cauterization to reduce ovarian damage and preserve healthy ovarian tissue. In a large, randomized double-blind trial of adults, superficial peritoneal lesions were shown to be more suitable for ablation [60]. Doyle et al. [61] conducted a retrospective case review that involved 90 adolescent patients and indicated that ablative surgery was performed only for superficial lesions, whereafter subsequent hormone therapy was implemented. There was no increase in the incidence of adhesions in the long term, and the disease did not show significant progression. Laufer et al. [62] reported a typical case: a 15-year-old girl with stage I endometriosis experienced no relief, but rather worsening pain after undergoing extensive radical peritoneal resection. Within a short period, extensive pelvic adhesions formed, and the disease recurred. Notably, the presentation of only one case does not suffice in demonstrating that the removal of superficial peritoneal lesions causes severe adhesions, since pre-existing adhesions constitute a risk factor for the occurrence of additional adhesions. A prospective observational case series of 20 cases [63] revealed that complete excision of endometriosis in adolescents with rASRM Stages I–III was safe and effective, and that successful treatment did not rely on postoperative hormonal therapy. In studies on adults, excision caused greater damage to ovarian function than ablation, and the damage to ovarian function was more significant when the endometriotic cysts were larger than 5 cm [64, 65]. Individuals with Stage I and II diseases were analyzed, and it was demonstrated that precise electrocoagulation of the lesion was more likely to be effective, and that excision was more suitable for deep infiltrative lesions [66].
The three-dimensional robotic system demonstrated superior detection rates for
small lesions (
While indocyanine green fluorescence imaging demonstrated limited diagnostic sensitivity in endometriosis (14.7%) [70], it provided intraoperative guidance for defining resection margins. Helium plasma coagulation also offered superior safety profiles for lesions that were adjacent to delicate anatomical structures compared with conventional thermal energy modalities [71].
It was reported that the recurrence rate after surgery for ovarian endometriotic cysts was approximately 10% annually, and that the younger the age, the greater the recurrence rate postoperatively [72]. International guidelines indicate that postoperative hormonal therapy should be considered in order to suppress endometriosis recurrence and its symptoms for adolescents [12]. All of the aforementioned hormonal regimens can be employed for the treatment of postoperative gynecological recurrence. For adolescent patients with pain symptoms and who are diagnosed with endometriosis through laparoscopy, if hormonal contraceptive or progesterone treatment fails, GnRH antagonist treatment can be applied for up to 1 year with reverse addition [12]. If adolescent patients with endometriosis undergo laparoscopic surgery due to dysmenorrhea or chronic pain, it is recommended that a levonorgestrel-releasing intrauterine device be emplaced after surgery to reduce the pain associated with both dysmenorrhea unresponsive to OCPs and dysmenorrhea and pain associated with endometriosis [15]. Postoperative use of OCPs can also effectively reduce the risk of recurrence after endometriosis, with a continuous regimen superior to a periodic regimen in preventing recurrence. A study involving 63 teenagers indicated that multi-site lesions were at dangerous risk for recurrence; of these 63, 5 cases were treated with three to six doses of GnRH agonists without any recurrence [73]. Due to the study’s small sample size, the authors did not specify whether their GnRH agonist exerted an improved effect in preventing recurrence. There is currently no consensus specifically indicating which drug manifests the lowest recurrence rate after surgery. Based upon clinical experience and for patients over 18 years of age with endometriosis Stages III–IV, a GnRH agonist is typically administered for three to six cycles first, followed by oral contraceptives or progesterone; a similar usage was reported for a small-sample retrospective study [74]. Sexually active adolescents can also be fitted with the LNG-IUS at the same time. It was reported that 58.10% of 284 patients under 22 who received the LNG-IUS experienced a safe application that did not increase complications and that reduced economic costs [75]. Another retrospective study revealed that of 224 individuals who employed the LNG-IUS after surgery, 208 (94.1%) added hormonal therapy or continued therapy post-surgery [76]; additionally, nearly half of the postoperative recurrence rates exhibited a median recurrence time of 3–5 years. It is now recommended to provide follow-up for at least 3–5 years postoperatively [73].
Several studies have indicated that some endometriosis in adolescents is diagnosed at Stages III–IV, which often implies lowered fertility [24, 77]. According to Audebert’s report [78], among 55 patients aged 12–19 diagnosed with endometriosis via surgery (60% at Stages I–II, 40% at Stages III–IV), 13 successfully gave birth; of these, 9 cases were at Stages I–II. Of the 55 cases, 5 were ascertained to reflect concomitant infertility problems during laparoscopic examination. Adolescent patients who already exhibit infertility or potential infertility issues can then avail themselves of oocyte freezing regimens for fertility preservation, as performed for adults [79].
There are no on-going, specialized, large-scale randomized controlled studies that specifically target adolescent girls, potentially due to the fact that patients do not always visit the same clinic or center, making data collection inconvenient. We suggest establishing a shared database that will allow for improved tracking, management, and research. When recording a patient’s condition, the demographic information, detailed surgical information, specific treatment measures, and subsequent reproductive status must be included as often as possible. It is critical to determine the indications for surgical treatment in adolescent patients with no symptoms or mild symptoms, but with a high suspicion of endometriotic cysts; however, the specific plan for reducing recurrence through postoperative combined medication remains unclear. In conclusion, no encompassing recommendations can be made for the treatment of endometriosis in adolescents. However, we posit that patients who undergo laparoscopic diagnosis or resection treatment, regardless of whether they receive combined drug therapy or not, should undergo long-term, postoperative follow-up.
Endometriosis represents a significant cause of chronic pelvic pain in adolescents, and clinicians face many challenges in its management in this age group. The relevant literature is often confusing, since providers adopt a wide variety of pharmaceutical agents and frequently modify treatment regimens. For patients with endometriosis or highly-suspected endometriosis, oral contraceptives, progesterone, or NSAIDs are recommended for initial treatment. Complete laparoscopic resection of deep lesions and electrocoagulation of superficial peritoneal lesions are preferred as a surgical treatment. The LNG-IUS can also be placed simultaneously during the operation, while long-term hormonal therapy is optimal postoperatively until the patient is interested in becoming pregnant. As endometriosis is a progressive disease, adolescent patients should be followed up with long term. More attention should also be given to the side-effects of drug treatment (including reduced bone density) and the protection of fertility. If necessary, oocyte freezing may be adopted as for adults with respect to preserving fertility. Adolescents with endometriosis thus benefit from the integration of ongoing education, support, and other multidisciplinary services. However, the selection of appropriate pharmacotherapy requires careful individualized assessments, particularly considering future fertility requirements.
rASRM, revised American Society for Reproductive Medicine; DNG, dienogest; NSAID, nonsteroidal anti-inflammatory drug; OCP, oral contraceptive pill; ENG, etonogestrel; GnRH, gonadotropin-releasing hormone; BMD, bone mineral density; VAS, visual analog scale; LNG-IUS, levonorgestrel-releasing intrauterine system; WHO, World Health Organization; ER, estrogen receptor; PR, progesterone receptor; EACPP, endometriosis-associated chronic pelvic pain; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; COX, cyclooxygenase; DEXA, dual-energy x-ray absorptiometry.
NC and XW, methodology and data collection; NC, writing—original draft preparation; NC and XW, writing—review and editing; and XW, project administration. Both authors have read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Not applicable.
Thanks go to my gynecology colleague, postdoctoral researcher Shaojie Ding, for the guidance on my thesis.
This research was supported by Zhejiang Provincial Natural Science Foundation of China under Grant No. LQ24H040003.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

