1 Department of Obstetrics and Gynecology, The Affiliated Hospital, Southwest Medical University, 646000 Luzhou, Sichuan, China
2 Department of Reproductive Medicine, Sichuan Jin’xin Xi’nan Women’s and Children’s Hospital, 610011 Chengdu, Sichuan, China
3 Department of Obstetrics and Gynecology, Sichuan Provincial People’s Hospital, 610011 Chengdu, Sichuan, China
Abstract
Gonadotropin-releasing hormone (GnRH) antagonists (GnRH-ant) have been demonstrated to exert adverse effects on endometrial receptivity; however, the association between their cumulative dose and pregnancy outcomes in fresh embryo transfer cycles remains inconclusive. This study aimed to evaluate the effect of the cumulative dose of GnRH-ant on pregnancy outcomes in fresh embryo transfer cycles.
This retrospective cohort study evaluated 1795 in vitro fertilization/intracytoplasmic sperm injection (IVF/ICSI) cycles conducted between January 1, 2023 and March 31, 2024, at Sichuan Jin’xin Xi’nan Women’s and Children’s Hospital using flexible GnRH-ant protocols. The initial dose of antagonist (0.125–0.25 mg/day) was individualized according to age, body mass index (BMI), estradiol (E2), and basal luteinizing hormone (LH) levels. Antagonist doses were increased to 0.25 mg/day if LH levels exceeded 10 mIU/mL during stimulation. Participants were stratified by median total GnRH-ant dose into group A (≤0.75 mg, n = 909) and group B (>0.75 mg, n = 886). Propensity score matching (PSM) (1:1 nearest-neighbor method) was performed to balance baseline characteristics, yielding 719 matched pairs. Embryological parameters, including oocyte yield, fertilization rate, and embryo quality, and clinical outcomes, such as implantation, pregnancy, and live birth, were compared between the two groups.
Compared with group A, group B exhibited significantly higher levels of progesterone, E2, oocyte yield, 2-pronuclear (2PN) fertilization, and quality embryo counts (cleavage-stage and blastocyst) (p < 0.001). The embryo implantation rate was significantly lower in group B than in group A (adjusted odds ratio [OR] = 0.74, 95% CI: 0.55–0.99, p = 0.048). No differences in clinical pregnancy, miscarriage rates, or live birth were observed between the two groups (all p > 0.05). The total dose of GnRH-ant was positively correlated with oocyte and embryo parameters (r = 0.171–0.102, p < 0.001).
In women under 35 years of age with normal ovarian reserve, reduced GnRH-ant doses did not significantly improve the clinical pregnancy rate during fresh cycles.
Keywords
- GnRH antagonist
- IVF/ICSI
- embryo implantation rate
- clinical pregnancy rate
Infertility affects approximately one in six individuals of reproductive age globally, making it one of the most common reproductive health concerns worldwide [1]. Currently, assisted reproductive technology (ART) serves as the primary effective therapeutic modality for infertility. Notably, the gonadotropin-releasing hormone (GnRH) antagonist protocol confers substantial economic advantages owing to its shorter ovulation induction cycle and reduced gonadotropin (Gn) dosage, contributing to its widespread adoption in ovarian stimulation regimens [2]. A 2025 Cochrane systematic review, which incorporated 338 randomized controlled trials (RCTs) and conducted pairwise comparisons of 15 ovulation induction protocols across 59,086 women, indicated that the short-acting GnRH antagonist protocol can significantly mitigate the risk of ovarian hyperstimulation syndrome (OHSS) in women predicted to exhibit a normal ovarian response [3]. However, recent studies have shown that the clinical pregnancy rate associated with the GnRH antagonist protocol is relatively lower in fresh embryo transfer cycles compared to the GnRH agonist protocol [4, 5], and the underlying mechanisms remain controversial. Emerging evidence suggests that GnRH antagonists downregulate the expression of the c-kit receptor and inhibit its activation, thereby impairing the proliferative capacity of endometrial stromal cells (ESCs) and compromising endometrial receptivity [6]. Another study revealed that treatment with GnRH antagonist downregulates S100P expression and induces the apoptosis of endometrial epithelial cells. This process is recognized as a critical determinant of reduced endometrial receptivity [7]. Additionally, concerns have been raised regarding the potential adverse effects of GnRH antagonist on oocyte and embryo quality. An animal study has shown that GnRH antagonist reduces the expression of growth differentiation factor 9 (GDF9), which may disrupt the cytoplasmic maturation of oocytes and subsequently impair their developmental potential and implantation competence [8]. Wang et al. [9] also revealed that the use of the GnRH antagonist protocol is associated with a higher incidence of chromosomal aneuploidy in blastocysts and early miscarriage tissues.
Thus, clinicians have attempted to reduce the dosage of GnRH antagonists to
minimize their adverse effects. Liu et al. [10] reported that during
controlled ovarian stimulation (COS), luteinizing hormone (LH) levels can serve
as a biomarker for administering antagonists. Antagonist use may be unnecessary
for patients with persistently low levels of LH (LH levels
This retrospective cohort study enrolled patients undergoing in vitro
fertilization/intracytoplasmic sperm injection (IVF/ICSI) with a GnRH antagonist
(GnRH-ant) protocol at the Reproductive Medicine Center of our hospital between
January 1, 2023, and March 31, 2024. As this study did not intervene in patients’
diagnostic or treatment processes, all direct identifiers, including names,
medical record numbers, and contact details, were removed from the database
before analysis to protect participant privacy. Consistent with the Declaration
of Helsinki, the Ethics Committee of Sichuan Jin’xin Xi’nan Women’s and
Children’s Hospital approved a waiver of informed consent (Approval No.:
2025-14). The inclusion criteria were as follows: (1) age
The study cohort comprised 1795 patients, who were stratified by the median
total cetrorelix dose into group A (
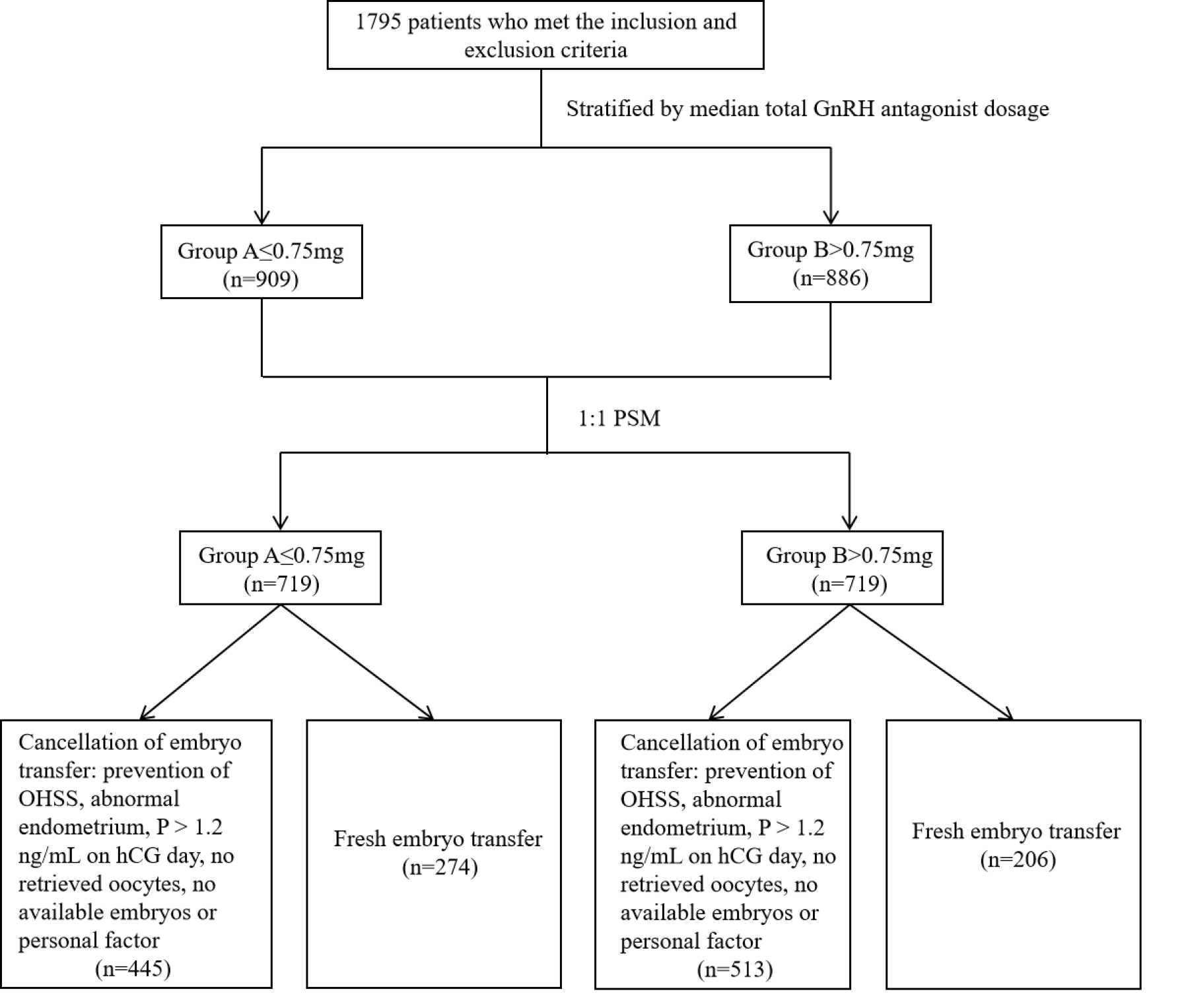 Fig. 1.
Fig. 1.
Flow chart of cycles included in the analysis. GnRH, gonadotropin-releasing hormone; PSM, propensity score matching; OHSS, ovarian hyperstimulation syndrome; hCG, human chorionic gonadotropin.
Based on the patient’s antral follicle count (AFC) and baseline levels of FSH,
LH, estradiol (E2), AMH, and BMI, ovarian stimulation was initiated with an
individualized dose of recombinant follicle-stimulating hormone (r-FSH) or highly
purified human menopausal gonadotropin (HP-HMG) on the second or third day of the
menstrual cycle. Cetrorelix (0.125 mg [13] or 0.25 mg daily) was introduced when
the lead follicles reached a diameter of
On day 3 or 5 of embryo culture, one to two fresh embryos were selected for
transfer based on their developmental status. Before transfer, patients were
instructed to achieve moderate bladder filling to facilitate ultrasound-guided
abdominal embryo transfer. Routine luteal phase support was initiated after
transfer, consisting of either progesterone soft capsules (200 mg three times
daily; Anqitian®, Zhejiang Medicine Co., Ltd., Shaoxing,
Zhejiang, China) combined with dydrogesterone (10 mg three times daily;
Duphaston®, Abbott Laboratories, Chicago, IL, USA), or vaginal
progesterone gel (90 mg daily; Crinone®, Merck & Co., Inc.,
Kenilworth, NJ, USA). The serum levels of
The primary endpoint was the embryo implantation rate. Secondary endpoints encompassed embryonic outcomes, specifically the number of oocytes retrieved, 2-pronuclear (2PN) zygotes, high-quality cleavage-stage embryos, and high-quality blastocysts, as well as clinical outcomes, including biochemical pregnancy, clinical pregnancy, early miscarriage, ongoing pregnancy, and live birth rates.
Biochemical pregnancy was defined as a serum level of
The outcomes were calculated using the following formulas: Embryo implantation
rate = (number of ultrasound-confirmed gestational sacs / the total number of
transferred embryos)
All analyses were conducted using R software (Version 4.4.2; R Foundation for Statistical Computing, Vienna, Austria). To minimize confounding bias caused by baseline differences, we applied PSM to identify comparable patients from the two groups. The propensity score was estimated using a multivariable logistic regression model, with the GnRH-antagonist group as the outcome and all baseline characteristics in Table 1 as covariates. The matched variables included age, BMI, infertility duration, infertility type, infertility cause, basal E2, LH, FSH, AFC, AMH, and fertilization type. A 1:1 nearest-neighbor matching algorithm with a caliper width of 0.03 was employed without replacement. Matching quality was assessed using standardized mean differences (SMDs). All post-matching SMDs were less than 0.1, suggesting adequate balance between groups. The post-matching comparability was validated through inter-group comparisons. Continuous variables are presented as median (interquartile range, IQR) due to non-normal distributions, which were assessed using the Kolmogorov-Smirnov test. The Mann-Whitney U test was applied for between-group comparisons. Categorical variables are expressed as frequencies and percentages and were compared using the chi-square test. The association between antagonist dosage and pregnancy outcomes was evaluated using a multivariable logistic regression model, adjusting for age, BMI, AFC, basal LH, AMH, the number of transferred embryos, and the number of high-quality cleavage-stage embryos and high-quality blastocysts. The embryo implantation rate was analyzed using a generalized estimating equation (GEE) model to account for the clustering of multiple embryos within patients. Correlations between antagonist dosage and clinical parameters were assessed using Spearman’s rank correlation analysis.
| Variables | Before PSM | After PSM | ||||||
| Group A (n = 909) | Group B (n = 886) | p-value | Group A (n = 719) | Group B (n = 719) | p-value | SMD | ||
| Female age, median (IQR), years | 31 (28, 33) | 30 (28, 33) | 0.780 | 31 (28, 33) | 30 (28, 33) | 0.837 | 0.004 | |
| BMI, median (IQR), kg/m2 | 21.48 (19.72, 23.37) | 21.51 (19.72, 23.56) | 0.606 | 21.48 (19.84, 23.44) | 21.48 (19.68, 23.51) | 0.551 | 0.034 | |
| Infertility duration, median (IQR), years | 3 (1, 4) | 3 (1, 4) | 0.461 | 3 (1, 4) | 3 (1.5, 4) | 0.747 | 0.029 | |
| Infertility type, n (%) | 0.392 | 0.832 | 0.014 | |||||
| Primary infertility | 524 (57.65%) | 492 (55.53%) | 399 (55.49%) | 404 (56.19%) | ||||
| Secondary infertility | 385 (42.35%) | 394 (44.47%) | 320 (44.51%) | 315 (43.81%) | ||||
| Infertility cause, n (%) | 0.953 | 0.016 | ||||||
| Tubal factor | 529 (58.20%) | 600 (67.72%) | 461 (64.12%) | 460 (63.98%) | ||||
| Male factor | 225 (24.75%) | 156 (17.61%) | 142 (19.75%) | 146 (20.31%) | ||||
| Others | 155 (17.05%) | 130 (14.67%) | 116 (16.13%) | 113 (15.72%) | ||||
| Basal E2, median (IQR), pg/mL | 31.00 (23.00, 40.00) | 30.00 (23.68, 38.89) | 0.714 | 30.92 (23.72, 39.88) | 30.00 (23.28, 39.00) | 0.824 | 0.011 | |
| Basal LH, median (IQR), mIU/mL | 3.72 (2.76, 4.84) | 4.08 (3.12, 5.40) | 3.93 (2.92, 5.06) | 3.89 (2.97, 5.08) | 0.601 | 0.028 | ||
| Basal FSH, median (IQR), mIU/mL | 6.72 (5.75, 7.86) | 6.91 (5.80, 8.11) | 0.017 | 6.84 (5.85, 8.02) | 6.83 (5.74, 8.06) | 0.705 | 0.019 | |
| AFC, median (IQR), n | 15 (11, 20) | 16 (12, 22) | 15 (11, 20) | 16 (11, 20) | 0.965 | 0.017 | ||
| AMH, median (IQR), ng/mL | 2.83 (1.92, 3.59) | 3.03 (2.11, 3.90) | 2.92 (2.02, 3.69) | 2.89 (2.04, 3.80) | 0.81 | 0.064 | ||
| Fertilization type, n (%) | 0.006 | 0.541 | 0.058 | |||||
| ICSI | 249 (27.39%) | 197 (22.23%) | 179 (24.90%) | 171 (23.78%) | ||||
| IVF | 654 (71.95%) | 688 (77.65%) | 540 (75.10%) | 547 (76.08%) | ||||
| Others | 6 (0.66%) | 1 (0.11%) | 0 | 1 (0.14%) | ||||
SMD, standardized mean difference; IQR, interquartile range; E2, estradiol; BMI, body mass index; LH, luteinizing hormone; FSH, follicle stimulating hormone; AFC, antral follicle count; AMH, anti-Müllerian hormone; ICSI, intracytoplasmic sperm injection; IVF, in vitro fertilization.
The following packages of R were employed for analysis: ‘MatchIt’ for PSM, the
built-in ‘glm’ function for logistic regression, and ‘geepack’ for GEE analysis.
A two-sided p-value
Before PSM, no significant differences existed between group A (n = 909) and
group B (n = 886) in terms of age, BMI, duration of infertility, infertility
type, or basal E2 levels (all p
After 1:1 PSM, 719 patients were included in each group, with all post-matching
SMDs for baseline characteristics being
Compared to group A, a longer duration of Gn administration (10 vs. 9 days,
p
Regarding embryological outcomes, group B had significantly higher numbers of
retrieved oocytes, 2PN zygotes, and high-quality cleavage-stage embryos compared
to group A (11 vs. 10, 8 vs. 7, and 3 vs. 2, all p
| Variables | Group A | Group B | p-value | |
| Gn usage daysa, median (IQR), days | 9.00 (8.00, 10.00) | 10.00 (9.00, 10.00) | ||
| Gn dosea, median (IQR), IU | 1725.00 (1425.00, 1950.00) | 1875.00 (1550.00, 2250.00) | ||
| Cetrorelix usagea, median (IQR), days | 3.00 (3.00, 4.00) | 5.00 (4.00, 5.00) | ||
| P level on hCG daya, median (IQR), ng/mL | 0.76 (0.51, 1.08) | 0.94 (0.68, 1.30) | ||
| LH level on hCG daya, median (IQR), mIU/mL | 2.11 (1.42, 3.19) | 2.17 (1.42, 3.20) | 0.882 | |
| E2 level on hCG daya, median (IQR), pg/mL | 1864.50 (1351.83, 2835.25) | 2621.50 (1898.07, 4011.00) | ||
| Incidence of premature follicular rupturea, n (%) | 8 (1.11) | 1 (0.14) | 0.045 | |
| Incidence of LH |
59 (8.21) | 93 (12.93) | 0.004 | |
| Number of retrieved oocytesb, median (IQR), n | 10.00 (7.00, 14.00) | 11.00 (8.00, 15.00) | ||
| Incidence of total embryo freezingc, n (%) | 388 (53.96) | 460 (63.98) | ||
| Number of 2PN fertilizationc, median (IQR), n | 7.00 (5.00, 10.00) | 8.00 (5.00, 11.00) | ||
| Number of high-quality embryos at cleavage stagec, median (IQR), n | 2.00 (1.00, 4.00) | 3.00 (1.00, 5.00) | ||
| Number of high-quality embryos in blastocyst stagec, median (IQR), n | 2.00 (0.00, 4.00) | 2.00 (1.00, 5.00) | 0.031 | |
| OHSSa, n (%) | 0 | 3 (0.42) | 0.249 | |
| Number of transferred embryosd, median (IQR), n | 2.00 (1.00, 2.00) | 2.00 (1.00, 2.00) | 0.607 | |
| Number of high-quality embryos transferredd, median (IQR), n | ||||
| Day 3 | 1.00 (1.00, 2.00) | 2.00 (1.00, 2.00) | 0.239 | |
| Day 5 | 1.00 (1.00, 1.75) | 1.00 (1.00, 1.00) | 0.108 | |
a: The sample size was 1438.
b: The sample size was 1420, excluding cases with canceled ovulation.
c: The sample size was 1419, excluding cases with canceled ovulation or failure to retrieve oocytes.
d: The sample size was 480 (Day 3: 323, Day 5: 157), excluding cases with cancelled or failed oocyte retrieval or cancelled embryo transfer.
2PN, 2-pronuclear.
Group A (cetrorelix
| Outcomes | Group A | Group B | p-value |
| Biochemical pregnancy ratea, n (%) | 68.98% (189/274) | 64.56% (133/206) | 0.308 |
| Clinical pregnancy ratea, n (%) | 62.77% (172/274) | 55.83% (115/206) | 0.124 |
| Embryo implantation rateb, n (%) | 47.25% (223/472) | 39.94% (143/358) | 0.036 |
| Early miscarriage ratec, n (%) | 8.14% (14/172) | 11.30% (13/115) | 0.368 |
| Ongoing pregnancy ratea, n (%) | 54.01% (148/274) | 46.60% (96/206) | 0.108 |
| Live birth ratea, n (%) | 54.01% (148/274) | 46.12% (95/206) | 0.087 |
a: The calculation of the variable is restricted to individuals who underwent embryo transfer (n = 480).
b: The denominator for this variable is the number of embryos transferred (n = 830).
c: The denominator for this variable is the number of individuals who achieved clinical pregnancy (n = 287).
We conducted a multivariable logistic regression analysis to control for potential confounding factors. The model was adjusted for key baseline characteristics (e.g., age, BMI, AFC, basal LH, and AMH) and treatment-related parameters, including the number of transferred embryos and the number of high-quality cleavage-stage embryos and blastocysts, all of which are well-established determinants of pregnancy outcomes [14, 15, 16].
The results showed that group A maintained a significantly higher embryo
implantation rate (adjusted odds ratio [aOR] = 0.74, 95% CI: 0.55–0.99,
p
| Outcomes | Group A | Group B | |
| aOR (95% CI) | aOR (95% CI) | p-value | |
| Biochemical pregnancy rate | 1 | 0.80 (0.54–1.19) | 0.269 |
| Clinical pregnancy rate | 1 | 0.72 (0.50–1.06) | 0.097 |
| Embryo implantation rate | 1 | 0.74 (0.55–0.99) | 0.048 |
| Early miscarriage rate | 1 | 1.66 (0.26–10.79) | 0.580 |
| Ongoing pregnancy rate | 1 | 0.72 (0.49–1.04) | 0.081 |
| Live birth rate | 1 | 0.70 (0.48–1.02) | 0.065 |
Model was adjusted for age, BMI, AMH, AFC, basal LH, the number of embryos transferred, number of high-quality embryos at cleavage stage, and number of high-quality embryos in blastocyst stage.
aOR, adjusted odds ratio.
Spearman correlation analysis revealed no significant associations between the
total cetrorelix dose and age, BMI, basal LH, AMH, or AFC (all p
| Variables | r | p-value |
| Female age | –0.007 | 0.799 |
| BMI | –0.024 | 0.373 |
| Basal LH | 0.046 | 0.081 |
| AMH | 0.024 | 0.367 |
| AFC | 0.036 | 0.171 |
| Number of retrieved oocytes | 0.171 | |
| Number of 2PN fertilization | 0.153 | |
| Number of high-quality embryos at cleavage stage | 0.134 | |
| Number of high-quality embryos in blastocyst stage | 0.102 |
In this retrospective cohort study of 1795 ovulatory women undergoing IVF/ICSI with a standardized GnRH-ant protocol, a higher cumulative antagonist dosage was associated with a dose-dependent increase in the risk of embryo implantation failure. However, between the dose-stratified groups, no significant differences were observed in terms of clinical pregnancy rates (aOR = 0.72, 95% CI: 0.50–1.06; p = 0.09) or live birth rates (aOR = 0.70, 95% CI: 0.48–1.02; p = 0.065). Our findings indicate that GnRH antagonist dosing during ovarian stimulation is not significantly correlated with patient age, BMI, or basal LH levels. Importantly, reducing the antagonist dose may increase the risk of premature follicular rupture.
Within COS protocols, GnRH antagonists competitively and reversibly bind to
pituitary GnRH receptors. This molecular interaction immediately suppresses
gonadotropin secretion, effectively preventing premature LH surges that can
trigger follicular luteinization or untimely ovulation [17]. The inhibitory
effect is dose-dependent, and the pituitary function can be restored within 2–4
days after drug withdrawal [18, 19]. Accumulating evidence suggests that GnRH
antagonists may exert dual endometrial impacts. First, by downregulating the
synthesis of vascular endothelial growth factor (VEGF) and insulin-like growth
factor 1 (IGF-1), these agents potentially compromise endometrial vascularization
and stromal decidualization, leading to diminished receptivity and lower fresh
embryo transfer success rates compared to agonist protocols [20]. Second,
treatment with dose-escalated GnRH antagonists significantly increases the levels
of endometrial interleukin-6 (IL-6) and monocyte chemoattractant protein-1
(MCP-1), establishing a pro-inflammatory milieu hostile to embryo implantation
[21]. Our clinical data reinforce this biological paradox. In 1795
normo-ovulatory women, although progesterone levels on the day of hCG
administration were higher in Group B than in Group A (0.94 ng/mL vs. 0.76 ng/mL,
p
Recent studies have increasingly focused on flexible, low-dose GnRH antagonist
protocols in ART. A prospective RCT by Kerimoğlu et al. [24]
indicated that a daily dose of 0.125 mg was as effective as the conventional 0.25
mg dose in preventing the premature surges of LH, with no significant difference
in clinical pregnancy or live birth rates. Another trial used an intermittent
strategy of 0.25 mg every other day, escalating to daily dosing based on LH level
monitoring. The trial achieved clinical outcomes comparable to those achieved by
the standard daily protocol [25]. A meta-analysis also showed that discontinuing
the antagonist on the trigger day can significantly increase the yield of mature
oocytes (OR = 1.26, 95% CI: 1.09–1.45) without elevating the risk of premature
ovulation, suggesting that a transient release from suppression may contribute to
final oocyte maturation [26]. Most recently, an RCT confirmed that stopping
antagonist administration on the hCG trigger day significantly improved live
birth rates after fresh-cycle single embryo transfer [27]. In contrast, Zhang
et al. [28] reported that reducing the antagonist dose did not improve
clinical pregnancy rates and increased the risk of premature follicular rupture.
Our findings align with this observation. Clinical pregnancy, ongoing pregnancy,
and live birth rates were numerically higher in the low-dose group; however, the
differences were not statistically significant (all p
Currently, whether the dose of GnRH antagonists correlates with BMI remains
controversial. In the study conducted by Al-Inany and Aboulghar et al. [29], in women
with a BMI of 18–30 kg/m2, the serum concentration of GnRH antagonists was
linearly and negatively correlated with body weight. A prospective RCT of GnRH
antagonists indicated that for low-weight Asian women, the minimum effective dose
of cetrorelix could be reduced to 0.2 mg [30]. Conversely, a study conducted in
Taiwan compared the clinical effects of different dosages of cetrorelix (0.15,
0.2, and 0.25 mg/day) on patients. The results indicated that 0.25 mg/day of
cetrorelix remained the minimum effective dose for low-weight (
Previous studies have proposed that GnRH antagonists may affect ovarian
steroidogenesis and granulosa cell function or directly affect the quality of
embryos [33, 34, 35]. In contrast, in our study, the number of retrieved oocytes, 2PN
fertilized embryos, cleavage-stage embryos, and high-quality blastocysts was
significantly higher in the high-dose group compared to the low-dose group
(p
Most previous studies have predominantly focused on the minimum effective daily dosage of antagonists. However, the association between the total dosage of antagonists and pregnancy outcomes remains incompletely understood. This research was the first to focus on the combined impact of GnRH antagonists in people with normal ovarian reserve. It unveiled a dose-dependent correlation between the total antagonist dosage and the likelihood of failed embryo implantation. We adopted cetrorelix as the sole antagonist type for dose analysis to address the common bias of medication heterogeneity frequently encountered in retrospective studies [39]. This approach has significantly enhanced the interpretability of the results. This study offers a novel perspective for optimizing antagonist regimens. In clinical practice, it is essential to dynamically monitor the characteristics of individual patients. It is of utmost importance to ensure a favorable embryo implantation rate and minimize the risk of premature follicular rupture. The results of this study underscore the need for individualized medications. They also provide a more refined evidentiary basis for formulating dose-optimization strategies grounded in evidence-based medicine.
However, this study still has several limitations. All of the included patients were managed with a flexible GnRH antagonist protocol, yet no stratified analysis was conducted based on the timing of antagonist initiation. Previous studies have indicated that either too early or too late addition of the antagonist can affect assisted reproductive outcomes [40, 41]. In our results, the proportion of patients experiencing an LH surge was higher in the high cumulative antagonist dose group. Evidence suggests that both hypo- and hyper-responders are prone to premature LH surges [42, 43], and an increased GnRH antagonist dose is often required to suppress such surges once they occur [44]. Moreover, previous research has found that premature LH surges can reduce clinical pregnancy rates in fresh embryo cycles [45]. Therefore, the potential confounding effects of ovarian response heterogeneity and LH surges on pregnancy outcomes cannot be excluded. Second, patients in group B had a longer duration of gonadotropin stimulation and antagonist use. The higher cumulative antagonist dose is inherently closely associated with the extended stimulation time. Importantly, increased gonadotropin exposure itself is a significant factor influencing endometrial receptivity [46]. Consequently, our findings cannot disentangle the potential confounding between antagonist dose and stimulation duration. Additionally, all patients in the current study received a dual trigger regimen, a strategy that has been shown to significantly increase the number of metaphase II (MII) oocytes, fertilized oocytes, and usable embryos, indicating favorable effects on oocyte maturation quality and developmental potential [47, 48]. Concurrently, accumulating evidence suggests that dual triggering positively modulates endometrial function by enhancing luteal function and endometrial receptivity [49]. Thus, the uniform administration of dual triggering may have partly masked or attenuated the true dose-dependent effects of GnRH antagonists on oocyte quality. Furthermore, as a single-center retrospective cohort study, this study was susceptible to selection bias and confounding variables. Therefore, the results should be considered preliminary exploratory insights, and further validation through large-scale, multi-center RCTs is warranted.
This study showed that in normo-ovulatory women, a higher cumulative dose of
GnRH antagonists (
The dataset generated during and analyzed during the current study is available from the corresponding author on reasonable request. All the clinical data involved in this study have not been shared with other studies.
XL and QL designed the study and were responsible for project development, XL wrote the main manuscript text, data collection and analysis. HX, LW and YJ contributed to the data collection. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was conducted in accordance with the principles of the Declaration of Helsinki. It has been approved by the Ethics Review Committee of Sichuan Jin’xin Xi’nan Women’s and Children’s Hospital (Approval No.: 2025-14). Each couple involved in the study signed the informed consent form and agreed to undergo assisted reproductive treatment.
We thank Edanz for professional language editing services.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

