1 Department of Obstetrics and Gynecology, Peking University First Hospital, 100034 Beijing, China
2 Beiing Key Laboratory of Innovations and transformations in intelligent and precise diagnosis and treatment technologies for reproductive health, 100026 Beijing, China
3 Department of Medical Statistics, Peking University First Hospital, 100034 Beijing, China
4 School of Health Policy and Management, Chinese Academy of Medical Sciences & Peking Union Medical College, 100006 Beijing, China
Abstract
Timely and accurate prenatal diagnosis of placenta accreta spectrum (PAS) is pivotal for improving maternal and fetal outcomes. However, the timing of PAS detection and its association with outcomes have rarely been explored. This study examined the association between the gestational week at which PAS was first detected by ultrasound and the clinical characteristics and outcomes of the disorder.
This retrospective cohort study included PAS patients at a tertiary referral center over a 5-year period. Patients who underwent cesarean section with prenatal PAS diagnosis were classified according to the gestational age of the earliest ultrasound PAS detection (<28 weeks or ≥28 weeks). Patient characteristics and outcomes were obtained from the inpatient medical records. Univariate and multivariate robust-error-variance Poisson regression models were used to explore the associations between the timing of ultrasound diagnosis and the risk of intraoperative hemorrhage (≥1500 mL) as well as blood-product transfusion volume (≥800 mL).
The study included 166 PAS patients, of whom 39.2% were diagnosed before 28 weeks, and 77.1% delivered at or beyond 34 weeks (median: 35 weeks). Patients with PAS detected before 28 weeks (early detection group) experienced significantly higher intraoperative blood loss volume (median: 1500 mL vs. 800 mL) and red blood cell transfusion volume (median: 586 mL vs. 70 mL). In regression analysis, patients in the early-detection group were significantly more likely to experience blood loss ≥1500 mL and red blood cell transfusion ≥800 mL, and these associations remained significant after adjustment for invasive PAS and hysterectomy. When ultrasound-detection weeks were divided into 6 categories, early detection (<20, 20–24, and 24–28 weeks) was associated with a significantly higher risk of excessive blood loss and increased transfusion volume.
PAS detected during the second trimester was associated with more adverse outcomes than cases identified later, highlighting the need for tailored management strategies. The association between gestational age of PAS detection and clinical outcomes warrants further investigation at both clinical and etiological levels.
Keywords
- hemorrhagic morbidity
- placenta accreta spectrum
- prenatal diagnosis
- ultrasound
Placenta accreta spectrum (PAS) disorders are severe obstetric complications that could cause severe and life-threatening hemorrhage, even maternal death [1]. Parallel to the increased use of cesarean section, the incidence of PAS has gone up in recent years, to about 1 per 272–730 deliveries [2, 3, 4]. Uterine scarring is considered to be one of the main risk factors for PAS that results from previous cesarean section, abortions, or other uterine operations like myomectomy or hysteroscopy [5]. According to the International Federation of Gynecology and Obstetrics (FIGO) classification, PAS disorders are grouped into placenta accreta, increta, and percreta based on clinical features at delivery and the invasion depth of the extravillous trophoblasts (EVT) as determined by histopathology [6]. Given the operative difficulties derived from invasive placenta and disturbed local anatomy, the current guidelines emphasize the importance of etiological preterm delivery to improve maternal-fetal outcomes, in the context of timely and accurate antenatal diagnosis of PAS, multi-disciplinary team engagement, and sufficient storage of blood components.
Considering the limited resources, a contingency plan should be formulated based on the prenatal ultrasonic assessment, featuring thin muscles, subplacental hypervascularity, placenta lacunae, and vesicouterine interruption [7]. A history of previous cesarean section and the location of the placenta also need to be considered. Current guidelines recommend an ultrasound examination right before the planned delivery of PAS patients to determine placental location and blood flow [8], when considering the appropriate surgical strategy [9, 10]. Since ultrasound manifestations of PAS were reported to be consistent during pregnancy [11], some characteristics can be observed as early as the first trimester [12, 13, 14]. The gestational age at which the ultrasound characteristics for PAS become detectable could be of clinical importance. It signifies different disease progression stages or courses, and affects the subsequent clinical decision of delivery timing and treatment modality, thus affecting peripartum outcomes. Ultrasound-based PAS detection during all trimesters is a logical conclusion [11, 13, 15]; first-trimester detection of cesarean-scar pregnancy (CSP)/PAS leads to about 90% hysterectomy and only rarely to a live birth [12, 16]. Although the role of detection time in PAS patients is important, there is currently little evidence regarding the association between the gestational age of PAS antenatal detection and patient outcomes.
This study examined the association between the gestational age of the first detection and the clinical characteristics and outcomes of PAS. The findings of the study could provide suggestions for pre-surgical preparation and ultrasound-surveillance protocols for suspected PAS patients.
This was a single-center retrospective cohort study using the electronic medical records of Peking University First Hospital, Beijing, China, and was approved by the ethics committee of Peking University First Hospital (ID: 2020-411). The hospital is a tertiary referral center with approximately 6000 deliveries per year. PAS patients included those transferred from Northern China, primarily Beijing, Tianjin, and Hebei provinces. The inclusion criteria included singleton delivery, live birth, and previous cesarean section with antenatal PAS diagnosis from November 2016 to October 2021.
All patients received routine ultrasound examination during pregnancy, at 11–13 weeks, 21–24 weeks, 28–32 weeks, 34–36 weeks, and 40 weeks (if applicable). Both transvaginal and transabdominal ultrasound were performed by experienced ultrasound doctors (senior attending) after systematic training, following the FIGO recommendations on ultrasonic features [17]: placenta lacunae with feeder vessels, loss of hypoechoic space, abnormalities of the uterus-bladder interface, and color Doppler abnormalities, including hypervascularity and bridging vessels.
If PAS was suspected, routine extra ultrasound examinations were performed every 1–4 weeks. For patients who were first suspected of PAS at other hospitals and were referred, further diagnosis was performed within 1 week, for a timely and comprehensive assessment. To identify the gestational age of the first detection of PAS by ultrasound examination, we screened all the ultrasound images and reports within (n = 98) or outside (n = 68) our hospital for each patient throughout the pregnancy.
Details were extracted from the surgical records regarding the position of the placenta and the degree and extent of invasion. All patients were diagnosed according to the intraoperative FIGO classification. Prenatal care and subsequent operations were performed by the same group of senior doctors specialized in PAS disorders. When the recommended gestational age of 34–36 weeks could not be reached [18], gestational age at delivery was determined according to the severity and progression of ultrasonic features. A multi-disciplinary surgery team was assembled at the planned delivery, including experts from the Obstetrics and Gynecology Department, Anesthesia Department, and Neonatal Department. Urologists and general surgeons would participate if necessary. Patients showing hypervascularity during color Doppler imaging would be given a pre-operative intra-aortic balloon placement (IABO) to mitigate major hemorrhage [19]. Hysterectomy was decided either preoperatively or during laparotomy.
The primary independent variable was the detection week of PAS, as determined by
ultrasound. As a continuous variable, the timing was also grouped in relation to
time points of prenatal examinations, first into 2 categories (
The primary outcomes were intra-operative blood loss and red-blood-cell
(RBC)-transfusion volume. Intraoperative hemorrhage was estimated by suction and
weighing of swabs. It was recoded as a binary variable (blood loss
The following variables were also extracted: socio-demographic characteristics, gestational complications (gestational diabetes mellitus and hypertension), pregnancy history and characteristics (cesarean section and abortion history, use of assisted reproductive technology, placenta previa, etc.), clinical manifestations (antepartum hemorrhage, abdominal pain, placenta position [anterior or not]), PAS severity (accreta, increta, percreta), FIGO clinical grading of PAS, use of operational procedure (abdominal aorta balloon, tourniquet, uterine artery ligation, uterine cavity tamponade, and hysterectomy), maternal perinatal outcomes (gestational weeks at delivery, length of stay, maternal complications [which included post-partum bleeding, amount of blood transfusion, acute transfusion reaction, disseminated intravascular coagulation (DIC), hemorrhagic shock, infection, anemia, other organ damage]). In our study, we defined placenta previa (partial and complete placenta previa) according to the latest guideline [21]. Fetal outcomes, including neonatal complications (dyspnea, pneumonia, infection), were based on the pediatric records. No maternal or fetal deaths occurred in the study.
For descriptive analysis, categorical data were presented as frequency (percentage). Median (interquartile range, IQR) was reported for continuous variables. Kruskal-Wallis tests and Chi-square tests were used for group comparisons.
To illustrate the association between ultrasound-detection timing and the
outcomes, we plotted box charts of blood loss and RBC-transfusion volume over the
6 categories of ultrasound-detection week (
For statistical inference, we applied univariate and multivariate Poisson
regression models with robust error variance to examine the associations between
the gestational week at ultrasound detection (categorized into 2 and 6 groups)
and the outcomes of major intraoperative hemorrhage (
To understand the influence of gestational age at delivery on maternal outcomes,
we stratified the sample according to PAS-detection-week categories (
A two-tailed p
For the 166 included PAS patients, the median age was 34 (IQR: 31–37), and
48.8% were of advanced maternal age (
| Characteristics (N (%)) | All (n = 166) | PAS detection week | p | ||
| Age, median (IQR) | 34 (31, 37) | 33 (31, 37) | 35 (31, 37) | 0.920 | |
| Age |
81 (48.8%) | 30 (46.2%) | 51 (50.5%) | 0.630 | |
| Ultrasound detection week, median (IQR) | 30 (24, 34) | 23 (20, 25) | 33 (31, 36) | ||
| Gestational week at delivery, median (IQR) | 35 (34, 36) | 34 (32, 35) | 35 (34, 36) | ||
| Gestational week at delivery |
128 (77.1%) | 40 (61.5%) | 88 (87.1%) | ||
| Pregnancy history | |||||
| Gravity, median (IQR) | 2 (1, 4) | 3 (2, 4) | 2 (1, 3) | 0.002 | |
| Parity, median (IQR) | 1 (1, 1) | 1 (1, 1) | 1 (1, 1) | 0.130 | |
| Abortion | 125 (75.3%) | 56 (86.2%) | 69 (68.3%) | 0.010 | |
| Abortion times, median (IQR) | 1 (1, 2) | 2 (1, 3) | 1 (0, 2) | 0.004 | |
| Previous cesarean section | 132 (79.5%) | 59 (90.8%) | 73 (72.3%) | 0.005 | |
| Cesarean section times, median (IQR) | 1 (1, 1) | 1 (1, 1) | 1 (0, 1) | 0.011 | |
| Cesarean section gap (year), median (IQR) | 6 (4, 8) | 6 (4, 8) | 6.5 (4, 9) | 0.550 | |
| Endometrium damage history | 25 (15.1%) | 8 (12.3%) | 17 (16.8%) | 0.510 | |
| Pregnancy characteristics | |||||
| Transferred from other hospital | 68 (41.0%) | 1 (1.5%) | 67 (66.3%) | ||
| IVF | 9 (5.4%) | 2 (3.1%) | 7 (6.9%) | 0.480 | |
| Antepartum bleeding | 42 (25.3%) | 18 (27.7%) | 24 (23.8%) | 0.590 | |
| Antepartum abdominal pain | 10 (6.0%) | 3 (4.6%) | 7 (6.9%) | 0.740 | |
| HBP | 10 (6.0%) | 2 (3.1%) | 8 (7.9%) | 0.320 | |
| GDM | 36 (21.7%) | 12 (18.5%) | 24 (23.8%) | 0.450 | |
| Placenta previa | 0.040 | ||||
| No | 29 (17.5%) | 12 (18.5%) | 17 (16.8%) | ||
| Partial | 13 (7.8%) | 1 (1.5%) | 12 (11.9%) | ||
| Complete | 124 (74.7%) | 52 (80.0%) | 72 (71.3%) | ||
| Placenta previa (First ultrasound report) | |||||
| Without sign of placenta previa | 14 (8.4%) | 6 (9.2%) | 8 (7.9%) | ||
| With sign of placenta previa | 106 (63.9%) | 59 (90.8%) | 47 (46.5%) | ||
| Only had 1 ultrasound record | 46 (27.7%) | 0 (0.0%) | 46 (45.5%) | ||
| Anterior placentation | 138 (83.1%) | 61 (93.8%) | 77 (76.2%) | 0.003 | |
| Anterior placentation and cesarean section history | |||||
| Not anterior + No cesarean section history | 10 (6.0%) | 0 (0.0%) | 10 (9.9%) | ||
| Anterior + No cesarean section history | 24 (14.5%) | 6 (9.2%) | 18 (17.8%) | ||
| Not anterior + cesarean section history | 18 (10.8%) | 4 (6.2%) | 14 (13.9%) | ||
| Anterior + cesarean section history | 114 (68.7%) | 55 (84.6%) | 59 (58.4%) | ||
| FIGO clinical classification | 0.004 | ||||
| FIGO 1 | 29 (17.5%) | 5 (7.7%) | 24 (23.8%) | ||
| FIGO 2 | 54 (32.5%) | 16 (24.6%) | 38 (37.6%) | ||
| FIGO 3a | 49 (29.5%) | 27 (41.5%) | 22 (21.8%) | ||
| FIGO 3b | 25 (15.1%) | 13 (20.0%) | 12 (11.9%) | ||
| FIGO 3c | 9 (5.4%) | 4 (6.2%) | 5 (5.0%) | ||
| PAS types | |||||
| Accreta | 29 (17.5%) | 5 (7.7%) | 24 (23.8%) | ||
| Increta | 54 (32.5%) | 16 (24.6%) | 38 (37.6%) | ||
| Percreta | 83 (50.0%) | 44 (67.7%) | 39 (38.6%) | ||
IQR, interquartile range; IVF, in vitro fertilization and embryo transfer; HBP, high blood pressure; GDM, gestational diabetes mellitus; FIGO, The International Federation of Gynecology and Obstetrics; PAS, placenta accrete spectrum disorders.
The median intra-operative blood-loss volume was 1000 mL (IQR: 600–1800 mL), and 62.7% of the patients had blood-product transfusion. The median volume of RBC transfusion was 238 mL (IQR: 0–1000 mL) and 14.5% had more than 8 units (1600 mL). Patients with PAS ultrasound detection before 28 gestational weeks had higher intra-operative blood-loss volume (median: 1500 mL vs. 800 mL) and RBC-transfusion volume (median: 586 mL vs. 70 mL) (Table 2 and Fig. 1A–C). Severe maternal morbidity occurred very rarely in the sample; the incidence of reoperation, organ injury, DIC, and infection were not different in the various PAS ultrasound-detection week groups (Table 2 and Supplementary Table 2).
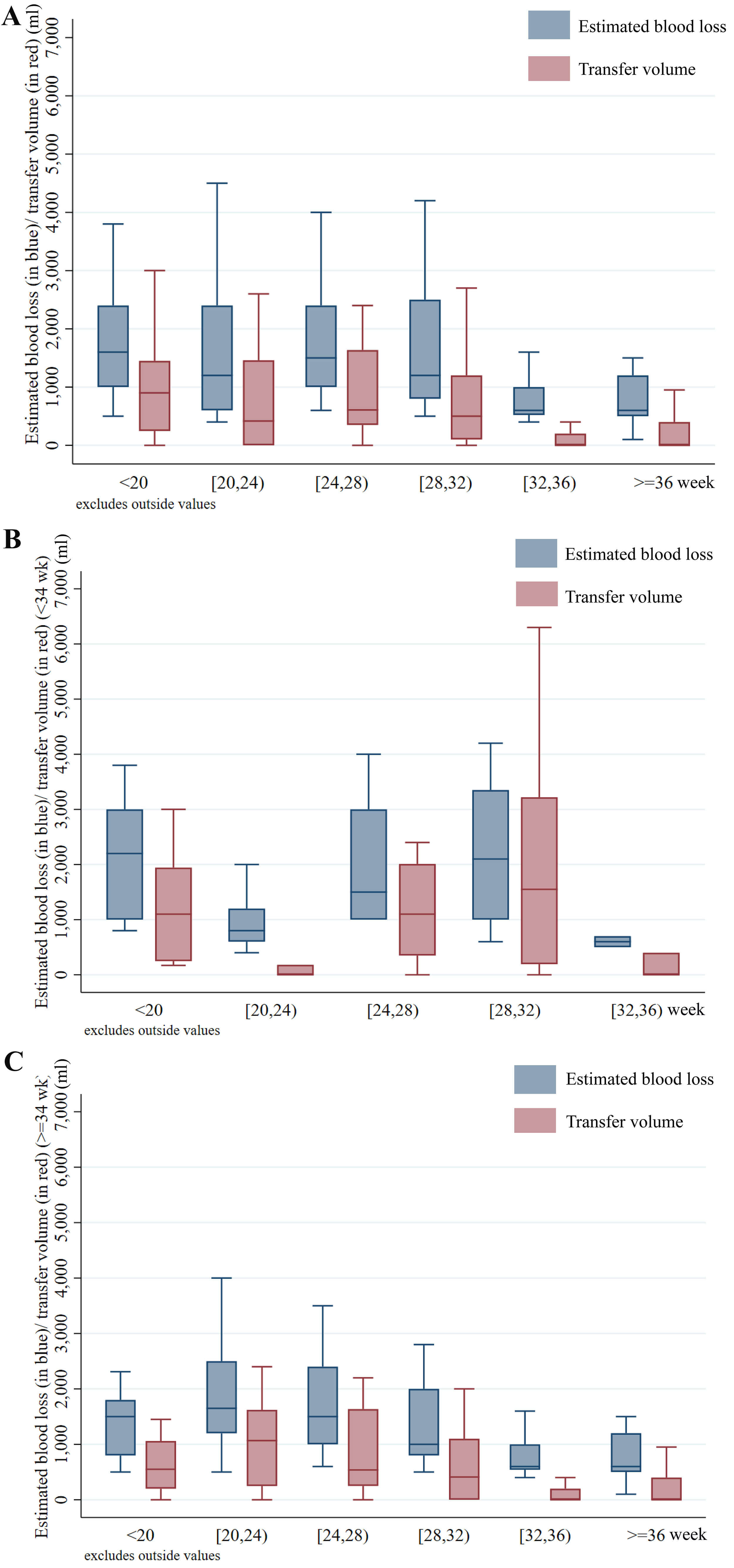 Fig. 1.
Fig. 1.
The trend of intraoperative blood loss and RBC-transfer volume
over the 6 ultrasound-detection-week categories. (A) The overall trend showed much
lower intraoperative blood loss and RBC transfer volume for patients with
ultrasound-detection week over 32. (B,C) After stratification by
gestational-age categories, we found that in the delivery
| Characteristics (N (%)) | All (n = 166) | PAS detection week | p | ||
| Interoperative hemorrhage | |||||
| Intraoperative blood loss, median (IQR) | 1000 (600, 1800) | 1500 (1000, 2400) | 800 (500, 1400) | ||
| Intraoperative blood loss |
58 (34.9%) | 33 (50.8%) | 25 (24.8%) | ||
| Blood product transfusion | 104 (62.7%) | 52 (80.0%) | 52 (51.5%) | ||
| Red blood cell, median (IQR) | 238 (0, 1000) | 586 (200, 1450) | 70 (0, 600) | ||
| Red blood cell transfusion |
24 (14.5%) | 14 (21.5%) | 10 (9.9%) | 0.044 | |
| Red blood cell transfusion |
51 (30.7%) | 30 (46.2%) | 21 (20.8%) | ||
| Plasma transfusion, median (IQR) | 0 (0, 400) | 0 (0, 400) | 0 (0, 0) | 0.014 | |
| Maternal complications* | 142 (85.5%) | 61 (93.8%) | 81 (80.2%) | 0.022 | |
| Maternal complications excluding intraoperative bleeding | 119 (71.7%) | 48 (73.8%) | 71 (70.3%) | 0.720 | |
| Acute kidney injury | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | - | |
| Embolism | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | - | |
| Pulmonary edema | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | - | |
| Acute transfusion reaction | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | - | |
| DIC | 3 (1.8%) | 1 (1.5%) | 2 (2.0%) | 1.000 | |
| Hemorrhagic shock | 4 (2.4%) | 2 (3.1%) | 2 (2.0%) | 0.640 | |
| Infection | 2 (1.2%) | 1 (1.5%) | 1 (1.0%) | 1.000 | |
| Anemia | 118 (71.1%) | 48 (73.8%) | 70 (69.3%) | 0.600 | |
| Reoperation | 3 (1.8%) | 1 (1.5%) | 2 (2.0%) | 1.000 | |
| Location of uterine incision | 0.160 | ||||
| Lower segment | 103 (62.0%) | 36 (55.4%) | 67 (66.3%) | - | |
| Uterine corpus | 62 (37.3%) | 28 (43.1%) | 34 (33.7%) | - | |
| Other | 1 (0.6%) | 1 (1.5%) | 0 (0.0%) | - | |
| Other organ injury | 8 (4.8%) | 4 (6.2%) | 4 (4.0%) | 0.710 | |
| Bowel injury | 2 (1.2%) | 2 (3.1%) | 0 (0.0%) | 0.150 | |
| Bladder injury | 6 (3.6%) | 2 (3.1%) | 4 (4.0%) | 1.000 | |
| Peri-operative procedure use | |||||
| IABO | 52 (31.3%) | 30 (46.2%) | 22 (21.8%) | 0.001 | |
| Tourniquet | 134 (80.7%) | 58 (89.2%) | 76 (75.2%) | 0.028 | |
| Uterine artery ligation | 120 (72.3%) | 50 (76.9%) | 70 (69.3%) | 0.370 | |
| Intrauterine balloon packing | 11 (6.6%) | 4 (6.2%) | 7 (6.9%) | 1.000 | |
| Neonatal complications# | 98 (59.0%) | 47 (72.3%) | 51 (50.5%) | 0.006 | |
| Neonatal asphyxia | 69 (41.6%) | 32 (49.2%) | 37 (36.6%) | 0.150 | |
DIC, disseminated intravascular coagulation; IABO, intra-aortic balloon occlusion.
* Maternal complications included: postpartum bleeding, amount of blood transfusion, acute transfusion reaction, DIC, hemorrhagic shock, infection, anemia, or other organ damage.
# Neonatal complications included: dyspnea, pneumonia, or infection.
Patients with ultrasound detection week
| Intra-operative blood loss volume |
RBC transfusion volume | |||||||
| Unadjusted RR | Model A | Model B | Model C | Unadjusted RR | Model A | Model B | Model C | |
| (95% CI) | Adjusted RR | Adjusted RR | Adjusted RR | (95% CI) | Adjusted RR | Adjusted RR | Adjusted RR | |
| (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | |||
| Ultrasound detection week (2 categories) (Ref: | ||||||||
| 2.05 (1.35–3.11) | 1.63 (1.09–2.44) | 1.52 (0.98–2.37) | 1.47 (0.94–2.29) | 2.22 (1.40–3.53) | 1.74 (1.12–2.73) | 1.60 (0.98–2.63) | 1.61 (0.98–2.64) | |
| Ultrasound detection week (6 categories) (Ref: [32–36] week) | ||||||||
| 4.32 (1.73–10.79) | 3.04 (1.22–7.55) | 2.97 (1.15–7.66) | 2.82 (1.04–7.66) | 6.40 (1.96–20.95) | 4.32 (1.33–14.06) | 4.09 (1.19–14.12) | 4.91 (1.31–18.43) | |
| [20–24] | 2.70 (1.03–7.10) | 2.28 (0.89–5.83) | 2.28 (0.88–5.93) | 2.19 (0.83–5.78) | 5.50 (1.71–17.74) | 4.58 (1.44–14.59) | 4.44 (1.36–14.53) | 3.73 (1.08–12.94) |
| [24–28] | 4.15 (1.72–10.02) | 3.29 (1.35–8.05) | 3.46 (1.41–8.48) | 3.38 (1.32–8.61) | 5.08 (1.57–16.46) | 3.99 (1.21–13.10) | 4.02 (1.21–13.40) | 4.15 (1.13–15.23) |
| [28–32] | 3.49 (1.44–8.49) | 2.88 (1.19–6.94) | 3.07 (1.22–7.72) | 3.04 (1.19–7.73) | 5.09 (1.60–16.20) | 4.17 (1.32–13.22) | 4.09 (1.21–13.77) | 3.80 (1.05–13.70) |
| 0.90 (0.26–3.08) | 1.08 (0.35–3.33) | 1.13 (0.36–3.51) | 1.14 (0.38–3.44) | 1.50 (0.36–6.23) | 1.75 (0.46–6.62) | 1.90 (0.53–6.86) | 1.96 (0.54–7.09) | |
Model A: adjusted for PAS severity (invasive PAS or not) and hysterectomy; Model
B: additionally adjusted for IABO and gestation age (
RR, risk ratio.
For patients in the early detection group (ultrasound-detection week
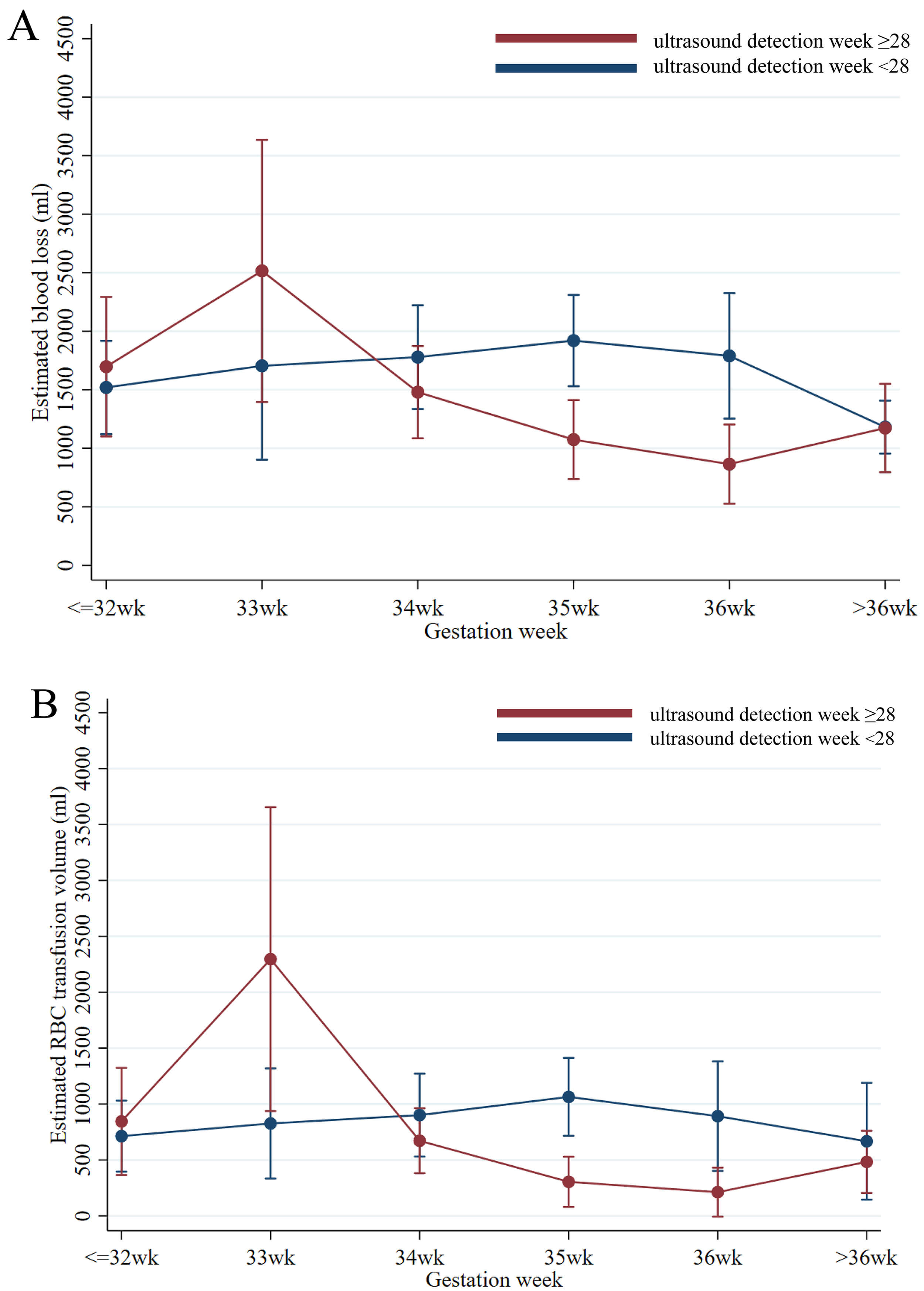 Fig. 2.
Fig. 2.
The predicted blood loss (A) or RBC-transfusion volume
(B) (95% confidence interval [CI]) for each gestational week at delivery
category (
When we restricted our sample to those with blood-loss volume less than 5000 mL (n = 164) to minimize the influence of outliers, and the sample (n = 98) in which transferred patients were excluded, the trends were preserved (Supplementary Table 3, Supplementary Figs. 1,2).
The present study found that the gestational age of PAS detection was associated with the clinical characteristics and intraoperative hemorrhagic outcome. Patients with ultrasound detection before 28 weeks were significantly more likely to have more intra-operative blood loss and blood-product-transfusion volume after adjusting for PAS severity, peri-operational procedures, and gestational week at delivery, which indicated that the timing of PAS appearance could be an independent risk factor for intra-operative outcomes of PAS.
In this study, the timing of first detection of PAS was associated with maternal-hemorrhage outcome at delivery. Our study mainly focused on the patients that had PAS diagnoses in the second and third trimesters.
The earliest ultrasound features that could predict PAS disorders include the number and size of the lacunae, abnormal uteroplacental interface, and lower uterine-segment hypervascularity [23, 24]. Shih et al. [25] described an ultrasound manifestation and termed it “rail sign”, defined as parallel “neovascularizations”, which are abnormally dilatated vessels (on color Doppler detection) with linking bridging vessels, which were related to greater blood loss and longer hospital stay. However, the associations between ultrasound manifestations and clinical outcomes rarely combined the ultrasound characteristics with the gestational age, making it hard to observe the progression pattern. In the present study, the PAS patients with an early ultrasound-detection week were also found to have more risk factors that could induce decidual damage, including a previous history of cesarean section along with anterior placentation, which is consistent with a previous study on the parallel trends of the incidence of PAS and the rate of cesarean section [26]. For a cesarean section number of 1, 2, or 3, the odds ratio (OR) of developing PAS was 6.699, 12.914, or 9.383, respectively [26]. This made cesarean-section history a strong indicator for searching for PAS ultrasound characteristics in the early stages of pregnancy.
The association between PAS-detection week and outcomes may challenge the
current practice of delivery time and surgical planning for PAS patients. Since
this study found that patients with PAS that was detected before 28 weeks were
more likely to have invasive PAS, together with more intraoperative bleeding and
adverse maternal or fetal outcomes, these patients could be in need of more
attention on disease-progress monitoring, emergency response planning, earlier
planning of delivery time, and extensive blood-product preparation. For patients
who had ultrasound PAS diagnosis after 28 weeks of gestation, the risk of major
hemorrhage and adverse outcomes could be much lower, especially for those with
gestational age at delivery
The disease course and etiology of PAS may differ by the timing of PAS detection. Given the substantial differences in risk factors, the pathogenesis of PAS detected before versus after 28 weeks of gestation may differ, potentially reflecting differences in the development and structural changes of the lower uterine segment during late pregnancy. During the development of the lower segment initiated from 28 gestational weeks, uterus myometrium layer becomes stretched and thinner. In patients with a cesarean section scar, the chance of developing the “uterine window” increases; this is known as uterine dehiscence. This phenomenon might also be recognized as placenta percreta because of the thinner myometrium and extending bulge, which raises the importance of standardization of PAS diagnosis with histopathology.
In addition, the pathogenesis of the early- and late-detection patients might be different, it could be dominated by either the aberrant invasion of EVT, or the defect of decidualization during placentation [27]. Some studies confirmed the role of cell-cycle control of cytotrophoblast and EVT, as the different expression of senescence markers was found in PAS delivered before or after 34 weeks [28, 29].
The association of gestational age of ultrasound detection and clinical outcomes of PAS was investigated in a tertiary hospital, which used standardized management of PAS and detailed data, including clinical features and ultrasound reports. The heterogeneity and confounding factors were carefully controlled.
The present study had several limitations. As a retrospective study based on routine pregnancy ultrasound screening, the frequency of ultrasound and the gestational age at delivery varied within a 4-week range, which might have confounded the results. For those referred PAS patients from other hospitals at the late third trimester, the ultrasound features before referral could be neglected, even though every patient was re-assessed in our center. We also did not include images and diagnoses by other diagnostic modalities, such as magnetic resonance imaging (MRI). Since we only included patients with prenatal diagnosis of PAS, other missing cases could affect the generalizability of the study. We only reported on patients with a singleton live birth, and mainly focused on their postpartum hemorrhage as the outcome criterion. Multicenter design to boost sample size and adopt a prospective study design should be considered with a further standardized ultrasound information collection process.
This study examined an important question: is the detection time of PAS associated with maternal-fetal morbidity, especially intraoperative hemorrhage morbidity. The findings highlighted the need for further evaluation of the predictive value of PAS-detection time on outcomes. Accurate prenatal diagnosis of PAS is crucial for the individualized plan of delivery; the timing of such a diagnosis could make a difference in both patient outcome at the clinical level and disease progression at the etiological level. For those detected before 28 weeks, further investigation of the ultrasonic features, along with the underlying mechanism for poor prognosis, is warranted.
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
XY: Conceptualization, methodology, formal analysis. XH: Data curation, software, validation, writing- reviewing and editing, visualization. JM: Interpretation of data for the work, supervision, review and editing the draft. WZ: Data curation. ZG: Data curation. HY: Conception and design of the work, supervision, review and editing the draft. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Peking University First Hospital (approval number: 2020-411). The project, using clinical databases with anonymous data only, does not involve any personal privacy or commercial interests, with a waiver of informed consent.
Not applicable.
We want to acknowledge the supports of the Strategic Collaborative Research Program of the Ferring Institute of Reproductive Medicine (Grant No. FIRMA181104), the National Key Research and Development Program of China (No. 2021YFC2700700), the National Natural Science Foundation of China (Grant No. 72104002) and the Scientific Research Seed Fund of Peking University First Hospital (Grant No. 2021SF62).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG47844.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


