1 Department of Gynaecology and Obstetrics, Yantai Mountain Hospital in Yantai City, 264003 Yantai, Shandong, China
2 Department of Radiotherapy, Yantai Mountain Hospital in Yantai City, 264003 Yantai, Shandong, China
Abstract
Approximately one-third of patients with locally advanced cervical cancer (LACC) experience treatment failure with concurrent chemoradiotherapy (CCRT), underscoring the need for reliable predictive biomarkers. This study aimed to elucidate the clinical value of serum inflammatory factors, including tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and C-reactive protein (CRP) in predicting the short-term efficacy of CCRT in patients with LACC.
Patients with LACC treated with CCRT were categorized into remission and non-remission groups based on their short-term treatment response. Pre-treatment serum levels of TNF-α, IL-6, and CRP were measured, and their predictive value for treatment efficacy was assessed. Multivariate analyses were performed to identify independent factors associated with short-term treatment outcomes.
The non-remission group (nRG) showed significantly raised serum levels of TNF-α, IL-6, and CRP compared to the remission group (RG). Receiver operating characteristic (ROC) curve analysis confirmed that these serum levels could predict the short-term efficacy of CCRT. Furthermore, lymph node metastasis (LNM), maximum tumor diameter, and elevated serum levels of TNF-α, IL-6, and CRP were identified as independent risk factors for poor short-term treatment efficacy. Consequently, patients with LACC with elevated serum levels of TNF-α, IL-6, and CRP following CCRT showed a significantly lower 12-month progression-free survival rate.
Serum levels of TNF-α, IL-6, and CRP serve as valuable predictors of the short-term efficacy of CCRT in LACC patients. Maximum tumor diameter, LNM, and elevated serum levels of TNF-α, IL-6, and CRP are independent risk factors influencing treatment outcome.
Keywords
- local advanced cervical cancer
- concurrent chemoradiotherapy
- TNF-α
- IL-6
- CRP
- short-term efficacy
- area under the curve
- multivariate logistic regression analysis
Cervical cancer (CC) remains a major global health burden and ranks among the most common gynecological malignancies worldwide [1], representing the 4th leading cause of cancer incidence and mortality globally and the 2nd in developing countries [2, 3]. A substantial proportion of patients present with locally advanced CC (LACC, International Federation of Gynecology and Obstetrics (FIGO) cervical Cancer Clinical Staging System, Stages IB through IVA) at diagnosis [4], whom concurrent chemoradiotherapy (CCRT) is the standard treatment modality [5, 6]. However, approximately one-third of patients experience treatment failure, resulting in unsatisfactory locoregional control and compromised prognosis [7], identifying reliable biomarkers that predict the therapeutic response to CCRT is therefore crucial for optimizing individualized treatment strategies.
Inflammation has been recognized as an important contributor to tumor
progression, including CC [8, 9]. Several inflammatory mediators such as tumor
necrosis factor-alpha (TNF-
Despite these observations, the predictive value of pre-treatment inflammatory
markers for short-term CCRT efficacy in LACC remains insufficiently investigated.
Therefore, this study aimed to evaluate whether baseline serum levels of
TNF-
This study was conducted in accordance with the principles of the Declaration of Helsinki and complied with all relevant ethical guidelines and regulations. Ethical approval was obtained from the Ethics Review Committee of Yantai Mountain Hospital(YTS2005001), and written informed consent was signed by the patients themselves or their guardians. This study was undertaken before the requirement for prospective registration of cohort studies. To minimize potential confounding factors that could influence systemic inflammatory status, treatment tolerance, or clinical outcomes, strict exclusion criteria were applied. Patients with autoimmune diseases, chronic inflammatory conditions, or active infections were excluded to avoid non-tumor-related inflammatory activation that might bias biomarker assessment. Patients with a history of prior malignancy, defined as any previous diagnosis of malignant tumors other than CC regardless of disease-free interval or treatment outcome, were also excluded to reduce heterogeneity related to cancer-associated inflammatory responses. In addition, individuals with severe hepatic, renal, or hematologic dysfunction were excluded because such conditions may alter cytokine metabolism, compromise treatment safety, or interfere with the delivery of CCRT. Furthermore, patients who had received prior pelvic radiotherapy or chemotherapy were excluded to ensure a treatment-naïve cohort, as previous anticancer therapies may modify baseline inflammatory profiles and treatment responsiveness. Collectively, these eligibility criteria were implemented to enhance cohort homogeneity and improve the internal validity of the biomarker-based prognostic analyses.
A total of 227 patients with LACC were prospectively enrolled at Yantai Mountain Hospital between June 2020 and June 2022. All patients were admitted for and treated with CCRT.
Patients were eligible for inclusion if they met all of the following criteria:
(1) Histopathologically confirmed CC, including squamous cell carcinoma and non-squamous cell carcinoma (primarily adenocarcinoma and adenosquamous carcinoma);
(2) Diagnosed with LACC at FIGO stage IIB–IVA according to the 2018 FIGO classification;
(3) Availability of baseline imaging data, including magnetic resonance imaging (MRI), pelvic examination findings, positron emission tomography/computed tomography (PET/CT), and/or CT, for assessment of primary tumor extent and lymph node status;
(4) Planned to receive and completed platinum-based CCRT as initial definitive treatment;
(5) Availability of complete clinical, laboratory, treatment, and follow-up data.
Patients were excluded if they met any of the following conditions:
(1) History of prior malignancy, defined as any previous diagnosis of malignant tumors other than CC, regardless of disease-free interval or treatment outcome;
(2) Presence of autoimmune diseases, chronic inflammatory disorders, or active infections at baseline;
(3) Severe hepatic, renal, or hematologic dysfunction, or other serious systemic diseases that could affect treatment tolerance or inflammatory marker levels;
(4) Prior pelvic radiotherapy, chemotherapy, or surgical treatment for CC;
(5) Receipt of neoadjuvant chemotherapy, targeted therapy, or planned surgical intervention after admission;
(6) Inability to complete the full course of radiotherapy or concurrent chemotherapy;
(7) Acute infectious conditions within 2 weeks before or after treatment, or use of antimicrobial or anti-tuberculosis drugs during the peri-treatment period;
(8) Known allergy or intolerance to chemotherapeutic agents used in the study;
(9) Pregnancy or lactation;
(10) Incomplete follow-up data or loss to follow-up.
The sample size estimation was performed using a statistical efficiency-based
approach by the G*Power 3.0.10 software (University of Düsseldorf,
Nordrhein-Westfalen, Germany). Firstly, the statistical parameters were set to
two-tailed test,
The sample size estimation was conducted using G*Power 3.0.10 based on Cohen’s established framework for detecting group differences in biomedical research. A medium effect size (d = 0.5) was selected for two main reasons. First, previous clinical studies investigating inflammatory biomarkers in CC have consistently reported moderate differences in baseline cytokine levels between responder and non-responder groups, supporting the appropriateness of a medium effect size assumption. Second, because no pilot data were available prior to study initiation, adopting Cohen’s conventional benchmark for a medium effect size provided a conservative and methodologically sound basis for sample size determination, avoiding overly optimistic or excessively restrictive assumptions. This approach is widely recommended in prospective clinical research when empirical estimates are limited. Furthermore, the final enrolled sample size exceeded the minimum requirement derived from the power analysis, ensuring that the study maintained adequate statistical power.
After carefully ruling out contraindications to radiotherapy and chemotherapy, all patients received standardized platinum-based CCRT according to institutional protocols. Radiotherapy consisted of external beam radiotherapy (EBRT) followed by intracavitary or combined intracavitary/interstitial brachytherapy. EBRT was delivered primarily using image-guided intensity-modulated radiotherapy (IMRT); three-dimensional conformal radiotherapy (3D-CRT) was applied when clinically indicated. Patients were positioned supine and immobilized with a thermoplastic mask. Simulation was performed using contrast-enhanced computed tomography with a slice thickness of 5 mm, extending from approximately 10 cm below the ischial tuberosities to the superior border of the tenth thoracic vertebra. The clinical target volume (CTV) included the primary cervical tumor and relevant adjacent structures (uterine cervix, uterine corpus, parametria, and partial or complete vagina), as well as regional lymphatic drainage areas (obturator, internal iliac, external iliac, common iliac, and presacral lymph nodes). Para-aortic or inguinal lymph node regions were included when clinically indicated. The planning target volume (PTV) was generated by adding a 3–5 mm margin to the CTV. EBRT was delivered using a 6 MV photon beam generated by a Varian Clinac IX linear accelerator (Varian Medical Systems, Inc., Palo Alto, CA, USA), with a total pelvic dose of 45.0–50.4 Gy administered in 25–27 fractions (1.8–2.0 Gy per fraction), five days per week. For patients with parametrial involvement, a simultaneous integrated boost was delivered to a total dose of 58–62 Gy over the same fractionation schedule. After completion of EBRT or when the primary tumor diameter was reduced to less than 3 cm, high-dose-rate (HDR) brachytherapy was performed using a Nucletron MicroSelectron-HDR Ir-192 remote afterloading system (Nucletron B.V. Eindhoven, Noord-Brabant, Netherlands). Brachytherapy was delivered under CT or MRI guidance, using intracavitary applicators or combined intracavitary/interstitial techniques as appropriate based on tumor extent and anatomical considerations. A total brachytherapy dose of 24–26 Gy was administered in 4–5 fractions (2 fractions per week), corresponding to an equivalent dose in 2 Gy fractions (EQD2) of approximately 82–88 Gy when combined with EBRT. Concurrent chemotherapy was administered weekly throughout the course of radiotherapy. The preferred regimen was cisplatin at a dose of 30–40 mg/m2 administered intravenously once per week for 5–6 cycles. In patients with intolerance to cisplatin, carboplatin was used as an alternative. In selected cases, a combination regimen of weekly cisplatin (Guangdong Lingnan Pharmaceutical Co., Ltd., Shaoguan, Guangdong, China) (25–30 mg/m2) plus paclitaxel (Harbin Pharmaceutical Group Bioengineering Co., Ltd., Harbin, Heilongjiang, China) (60–80 mg/m2) was administered on days 1, 8, 15, 22, 29, and 36 of radiotherapy. Chemotherapy doses were adjusted according to treatment-related toxicities, with the overall principle of maintaining uninterrupted radiotherapy whenever possible.
Short-term efficacy: all patients were subjected to post-CCRT routine
monitoring. Three months after the end of treatment, subjects were evaluated for
short-term efficacy in accordance with the Response Evaluation Criteria in Solid
Tumors Version 1.1 (RECIST 1.1) [21] based on the findings of MRI, CT, or PET-CT
before and following treatment. Recent outcomes were classified as complete
remission (CR), with complete disappearance of target lesions; partial remission
(PR), with at least a 30% reduction in the sum of target lesion diameters from
baseline levels; stable disease (SD), between PR and CR; progression disease
(PD), with more than a 20% increase in the sum of lesion diameters from baseline
levels and one or more additional lesions occuring. Overall remission rate = CR +
PR/total number of patients
Long-term efficacy: patients underwent routine telephone check-up and medical examinations every 3–6 months post treatment. Follow-up visits consisted of whether patients had local recurrence or distant metastasis of the tumor, the diagnosis time of recurrence or metastasis, patient survival and adverse reactions [22]. All subjects were followed up for at least 12 months. Following CCRT, patients were recorded for 12-month progression-free survival (PFS) (the period from the start of treatment to observed disease progression or the occurrence of death because of any reason in subjects with tumor diseases).
General and clinical information were collected from all patients including age,
body mass index (BMI), maximum tumor diameter, smoking history, pathological
staging, FIGO stage, and lymph node metastasis (LNM). Before three to seven days
of treatment, 10 mL of fasting elbow venous blood was gathered from patients, and
centrifuged at 4 °C and 1000
Statistical analyses and data visualization were performed using SPSS version
22.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 8.0 (GraphPad
Software Inc., San Diego, CA, USA). The Shapiro-Wilk test was used to assess the
normality of continuous variables. Normally distributed continuous data were
expressed as mean
Nominal categorical variables were compared between groups using the Chi-square test. Tumor grade, as an ordinal variable, was analyzed using statistical methods appropriate for ordered data. The Mann-Whitney U test was applied to compare overall distributional differences in tumor grade between the remission group and the nRG, while the Cochran-Armitage trend test was considered as an alternative approach for assessing ordinal trends when appropriate.
The predictive performance of serum TNF-
Variables with a p value
PFS was analyzed using the Kaplan-Meier method, and
differences between groups were assessed using the log-rank test. All statistical
tests were two-sided, and a p value
For survival analysis, patients were stratified into high- and low-level groups
based on the median values of TNF-
We analyzed 227 LACC patients who received CCRT. The mean age of the patients
was 52.1
| Parameters | Patients | RG | nRG | p value | |
| Case [n (%)] | 227 | 162 (71.4) | 65 (28.6) | - | |
| Age (years) | 52.1 |
51.7 |
52.9 |
0.409 | |
| BMI (kg/m2) | 23.2 |
23.0 |
23.7 |
0.145 | |
| Maximum tumor diameter (cm) | 4.6 |
4.3 |
5.2 |
||
| Smoking history [n (%)] | 0.215 | ||||
| Yes | 27 (11.9%) | 22 (13.6%) | 5 (7.7%) | ||
| No | 200 (88.1%) | 140 (86.4%) | 60 (92.3%) | ||
| Pathological type [n (%)] | 0.251 | ||||
| Squamous cell carcinoma | 182 (80.2%) | 133 (82.1%) | 49 (75.4%) | ||
| Non-squamous cell carcinoma | 45 (19.8%) | 29 (17.9%) | 16 (24.6%) | ||
| FIGO stage [n (%)] | 0.006 | ||||
| IIB-IIIB | 130 (57.3%) | 102 (62.9%) | 28 (43.1%) | ||
| IIIC-IVA | 97 (42.7%) | 60 (37.1%) | 37 (56.9%) | ||
| Lymph node metastasis [n (%)] | 0.006 | ||||
| Yes | 65 (28.6%) | 38 (23.5%) | 27 (41.5%) | ||
| No | 162 (71.4%) | 124 (76.5%) | 38 (58.5%) | ||
| Tumor grade [n (%)] | 0.173 | ||||
| G1 | 37 (16.30) | 30 (18.52) | 7 (10.77) | ||
| G2 | 81 (35.68) | 60 (37.04) | 21 (32.31) | ||
| G3 | 109 (48.02) | 72 (44.44) | 37 (56.92) | ||
Note: LACC, locally advanced cervical cancer; RG, remission group; nRG, non-remission group; BMI, body mass index; FIGO,
the International Federation of Gynecology and Obstetrics. Normally-distributed
measurement data were expressed as mean
To explore the clinical significance of serum inflammatory indicators
TNF-
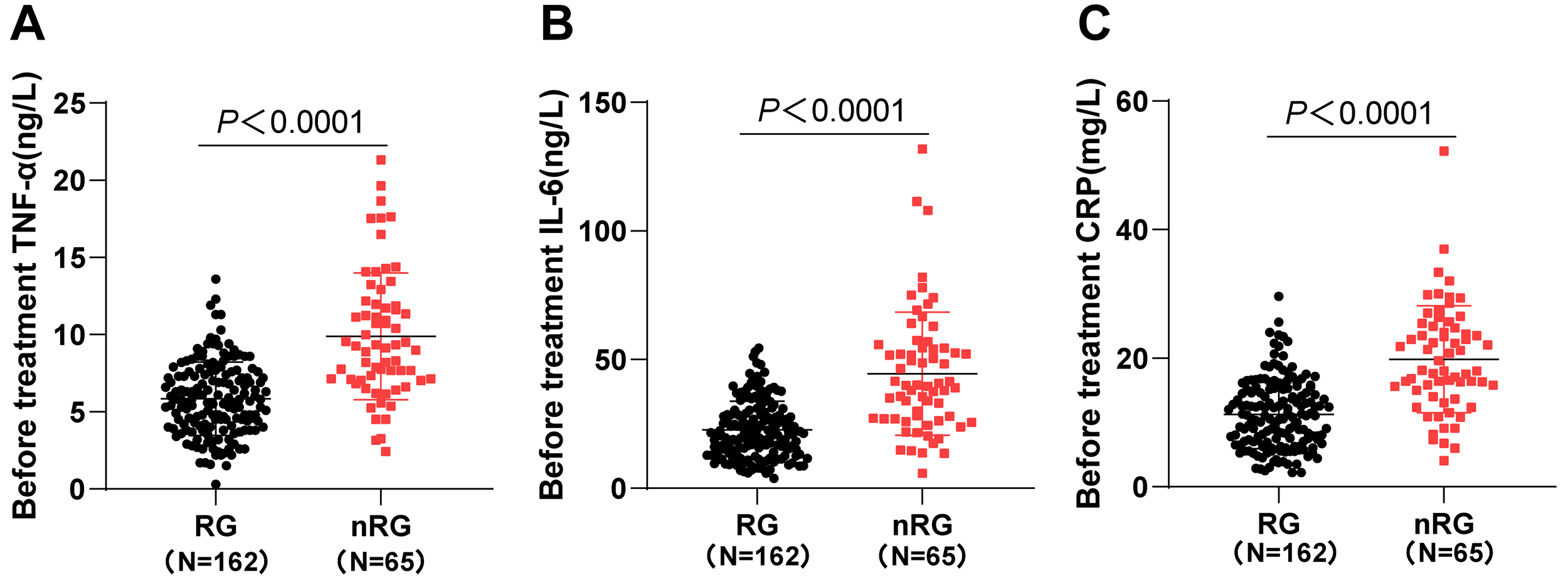 Fig. 1.
Fig. 1.
Comparisons of serum TNF-
Moreover, we used ROC curves to assess the predictive value of serum levels of
TNF-
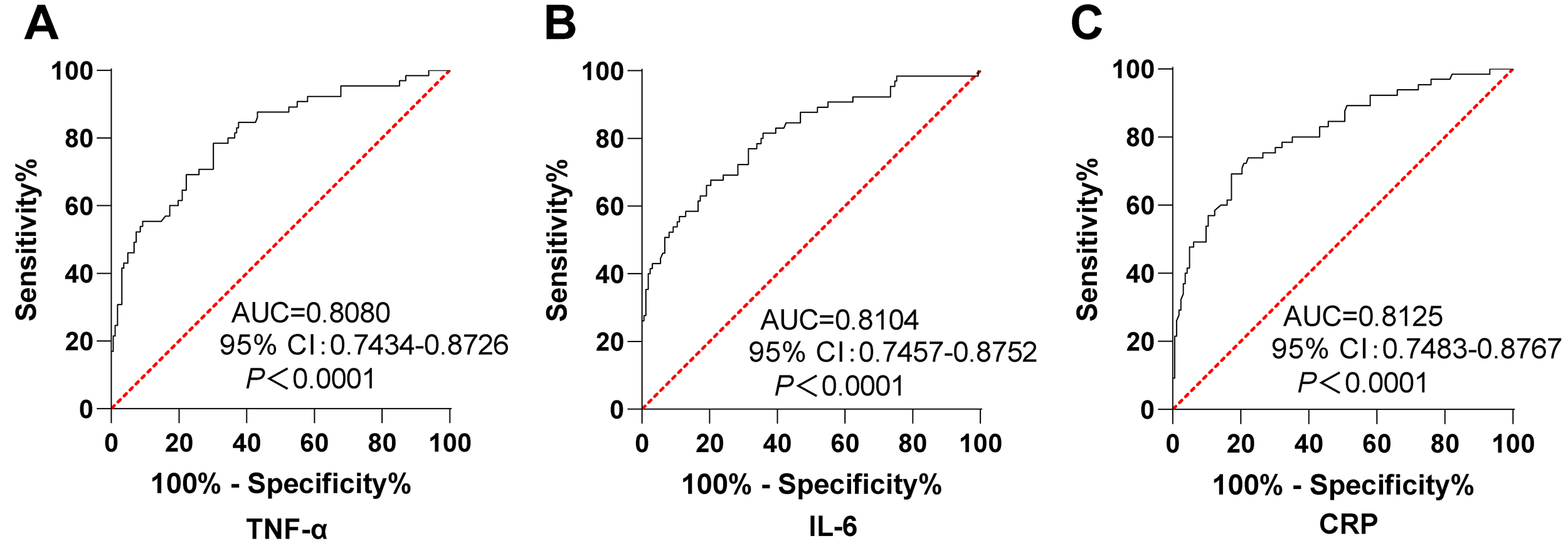 Fig. 2.
Fig. 2.
Serum TNF-
We divided 227 LACC patients into High-TNF-
| Group | Remission rate | p value |
| Low-TNF- |
113/127 (89.0%) | |
| High-TNF- |
49/100 (49.0%) | |
| Low-IL-6 group | 129/150 (86.0%) | |
| High-IL-6 group | 33/77 (42.9%) | |
| Low-CRP group | 134/154 (87.0%) | |
| High-CRP group | 28/73 (38.4%) |
Note: Enumeration data were expressed as number of cases (n) and percentages (%), and the Chi-square test was implemented for comparison between the two groups.
To evaluate the independent risk factors influencing the short-term efficacy of
CCRT in LACC patients, variables with p
| Variable | OR | 95% CI | p value |
| Maximum tumor diameter | 1.979 | 1.063–3.687 | 0.031 |
| FIGO stage | 2.333 | 0.748–7.278 | 0.144 |
| Lymph node metastasis | 3.512 | 1.041–11.843 | 0.043 |
| TNF- |
1.245 | 1.022–1.518 | 0.030 |
| IL-6 | 1.041 | 1.003–1.080 | 0.036 |
| CRP | 1.101 | 1.011–1.198 | 0.026 |
Note: CI, confidence interval.
We further investigated the association between baseline serum inflammatory
markers and 12-month PFS in patients with LACC
following CCRT. Patients were stratified into high and low groups for
TNF-
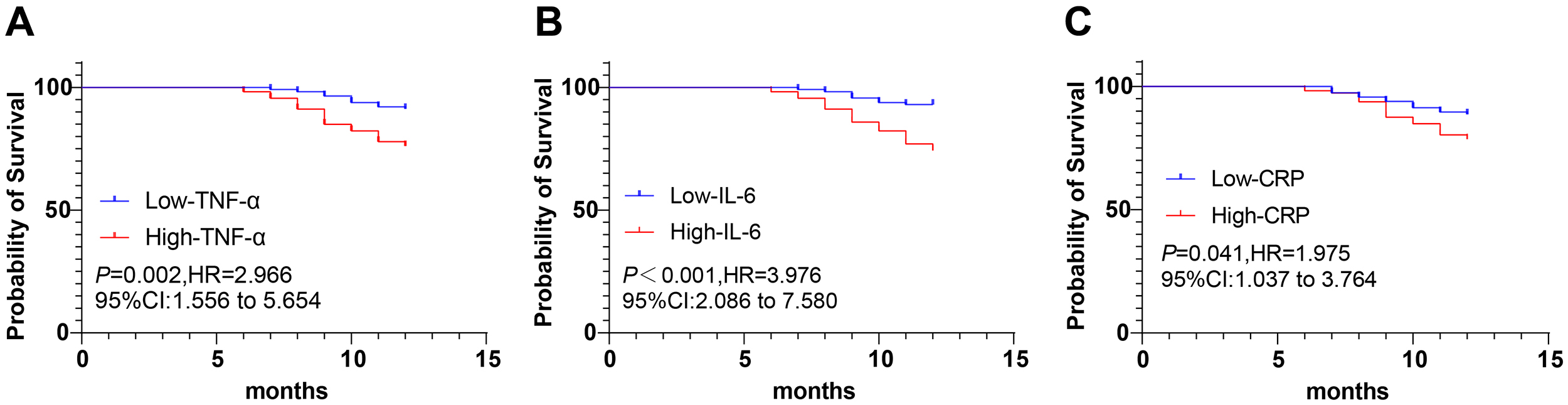 Fig. 3.
Fig. 3.
Kaplan–Meier analysis of progression-free survival (PFS)
according to pre‑treatment serum levels of TNF‑
Early identification of patients who are unlikely to respond adequately to CCRT
remains a major challenge in the management of LACC. Although CCRT is the
standard of care, treatment resistance still contributes to poor survival
outcomes [23, 24]. In this context, our study provides new evidence that
pre-treatment systemic inflammatory status, reflected by serum TNF-
Another emerging area that warrants consideration is the relationship between
systemic inflammatory biomarkers and functional imaging parameters, particularly
the metabolic response assessed by 18F-FDG PET/CT. PET-based markers such as
SUVmax, metabolic tumor volume, and total lesion glycolysis have shown prognostic
relevance in LACC after definitive chemoradiotherapy, reflecting tumor viability,
hypoxia, and microenvironmental alterations. Given that cytokines including
TNF-
Beyond demonstrating that TNF-
In a previous animal study, TNF-
Another important aspect worth considering is the growing role of imaging guidance during brachytherapy procedures. Recent frameworks, such as the COMIRI (COMplexity Index of interventional Radiotherapy Implants), have highlighted how procedural complexity in image-guided brachytherapy may influence treatment delivery and tissue response. The COMIRI classification provides a standardized approach to quantify implant complexity based on technical, anatomical, and organizational factors, thereby facilitating objective comparison across different interventional radiotherapy procedures and institutions. The COMIRI index provides a structured and standardized method to quantify brachytherapy complexity based on implant type, imaging modality, equipment requirements, and multidisciplinary team involvement, thereby enabling objective comparison across different institutions and procedures. By accounting for these procedural factors, COMIRI offers an important perspective on how technical and organizational aspects of brachytherapy may interact with biological responses, including treatment-related inflammation. In this context, incorporating COMIRI-based assessments may help clarify whether variations in procedural complexity contribute to heterogeneity in inflammatory activation and treatment outcomes in patients undergoing CCRT.
Recent advances in CC management have also highlighted the potential value of
integrating immunotherapy with chemoradiotherapy. Immune checkpoint inhibitors,
particularly those targeting PD-1/PD-L1 pathways, have shown promising activity
in recurrent and metastatic CC, leading to growing interest in their
incorporation into definitive treatment for LACC. Early-phase studies evaluating
agents such as pembrolizumab, nivolumab, and camrelizumab combined with
concurrent CRT have demonstrated encouraging signals of enhanced tumor regression
and sustained immune activation, suggesting that modulation of the immune
microenvironment may sensitize tumors to radiation and chemotherapy. Given that
systemic inflammatory cytokines such as TNF-
There were strengths in our article, we found that that serum levels of
inflammatory parameters TNF-
This study has several limitations. First, it was conducted at a single center
with a relatively limited sample size, which may restrict the generalizability of
the findings. Multicenter prospective studies with larger cohorts are needed to
validate the predictive value of serum TNF-
In addition to the limitations previously noted, several methodological issues
warrant further consideration. First, this was a single-center study, which may
limit the generalizability of the findings due to institutional differences in
patient characteristics, treatment protocols, and imaging practices. External
validation in multicenter cohorts is essential to confirm the reproducibility and
stability of the predictive value of TNF-
In conclusion, this study demonstrates that pre-treatment systemic inflammatory
status, reflected by serum TNF-
Importantly, integrating inflammatory biomarkers with routine clinical parameters may facilitate early risk stratification and identification of patients at higher risk of poor response to CCRT. Such information could support more individualized clinical management, including closer surveillance and timely consideration of treatment optimization strategies. Overall, these findings highlight the potential application value of simple and accessible serum inflammatory markers as adjunctive tools for early efficacy assessment in LACC.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
YL contributed to the study concepts, study design; YL contributed to the literature research; YL and XW contributed to the experimental studies and data acquisition; YL and XW contributed to the data analysis and statistical analysis; YL revised the manuscript, and XW contributed to editing and review. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The research was conducted in accordance with the Declaration of Helsinki, and all methods were performed following relevant guidelines and regulations. The research was approved by the Ethics Review Committee of Yantai Mountain Hospital (YTS2005001), and written informed consent was signed by the patients themselves or their guardians.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



