1 Department of Obstetrics and Gynecology, Hangzhou Lin’an Fourth People’s Hospital, 311321 Hangzhou, Zhejiang, China
2 Department of Obstetrics and Gynecology, Wenzhou Medical University, 325015 Wenzhou, Zhejiang, China
Abstract
Intrauterine adhesions (IUAs) pose a persistent challenge following hysteroscopic submucosal myomectomy, frequently compromising reproductive potential and menstrual health. While postoperative inflammation is implicated in endometrial fibrosis, the predictive capacity of inflammatory biomarkers for IUA formation is not well established.
In this prospective cohort study, 200 women of reproductive age undergoing hysteroscopy resection of submucosal fibroids were enrolled. Serum concentrations of C-reactive protein (CRP), interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) were quantified preoperatively, 48 hours post-surgery, and at a 3-month follow-up. Adhesion development was assessed via follow-up hysteroscopy at 12 weeks and graded using the European Society for Gynecological Endoscopy (ESGE) criteria. Multivariate logistic regression and receiver operating characteristic (ROC) analyses evaluated biomarker-adhesion correlations.
Among the 180 participants who completed follow-up, 45 (25.00%) with mild-to-severity, which were included in predictive modeling. Median CRP and IL-6 levels were consistently elevated in the adhesion cohort at all intervals (p < 0.01), with IL-6 exhibiting the strongest correlation to adhesion severity (Spearman’s ρ = 0.51, p < 0.001). Adjusted regression identified IL-6 (odds ratio [OR] = 1.18, 95% confidence interval [CI]: 1.08–1.29) and CRP (OR = 1.33, 95% CI: 1.07–1.65) as independent predictors. TNF-α levels were significantly higher at 48 hours and 3 months postoperatively and showed moderate predictive ability in univariate analysis (area under the curve [AUC] = 0.71). However, TNF-α did not remain an independent predictor after multivariable adjustment (OR = 1.12, 95% CI: 0.97–1.29, p = 0.112). ROC curves demonstrated robust discriminative accuracy for IL-6 (AUC = 0.82) and moderate predictive performance for CRP (AUC = 0.74).
Sustained perioperative increases in IL-6 and CRP levels correlate strongly with IUA risk after hysteroscopic submucosal myomectomy, highlighting their potential as early, non-invasive indicators of adhesion development. TNF-α may serve as an auxiliary marker in univariate analyses but is not an independent predictor after adjustment. Integrating these biomarkers into postoperative surveillance may facilitate timely interventions to mitigate fibrosis, optimize endometrial healing, and safeguard fertility.
Keywords
- intrauterine adhesions
- hysteroscopy
- inflammatory markers
- IL-6
- C-reactive protein
- TNF-α
- postoperative fibrosis
Intrauterine adhesions (IUAs), also known as Asherman’s syndrome, represent an important cause of acquired infertility and menstrual disturbance in reproductive-age women. These adhesions consist of fibrous bands that partially or completely obliterate the uterine cavity, often resulting from trauma to the endometrial basalis layer following procedures such as curettage, myomectomy, or endometrial ablation [1, 2]. The true prevalence of IUAs remains underestimated, though rates as high as 20–35% have been reported following hysteroscopic surgery, particularly in women undergoing submucosal myomectomy or repeated intrauterine procedures [3, 4]. IUAs can lead to a spectrum of adverse outcomes, hypomenorrhea (reduced menstrual flow) or secondary amenorrhea, as well as recurrent pregnancy loss [5].
The pathogenesis of IUAs is increasingly recognized as a process fundamentally
driven by aberrant inflammation and impaired wound healing. Injury to the
endometrial basalis initiates a cascade of local and systemic inflammatory
responses, characterized by neutrophil and macrophage infiltration, release of
pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis
factor-
Despite advancements in hysteroscopic techniques and preventive interventions,
IUAs remain a significant clinical challenge, with recurrence rates after
adhesiolysis reported to reach up to 30% in some series [12]. Early
identification of women at risk for adhesion formation is critical to guide
preventive and therapeutic strategies. Inflammatory biomarkers offer a promising
avenue for risk stratification, as they are readily accessible, reflect real-time
endometrial milieu, and may predict both the incidence and severity of IUAs [13, 14]. Recent studies have shown that perioperative elevations in CRP, IL-6, and
TNF-
Despite growing evidence for the role of inflammation in IUA pathogenesis, there is still a lack of consensus regarding which biomarkers provide the best predictive value and how they can be integrated into clinical protocols. Most existing studies are retrospective or cross-sectional, with limited prospective evidence linking dynamic changes in inflammatory markers to IUA development and severity [15, 16]. In addition, the influence of surgical factors such as myoma type, operative duration, and intraoperative trauma on the inflammatory response and subsequent adhesion risk remains incompletely understood [17, 18].
If validated and integrated into clinical practice, perioperative biomarker
surveillance could enable earlier identification of high-risk patients,
facilitate timely interventions to prevent adhesion-related infertility, and
potentially reduce the need for repeat surgeries and costly fertility treatments.
To our knowledge, this is among the first large, prospective cohort studies to
longitudinally assess IL-6, CRP, and TNF-
The primary aim of this prospective cohort study is to evaluate the association
between perioperative inflammatory markers (CRP, IL-6, and TNF-
Women of reproductive age scheduled for hysteroscopic submucosal myomectomy were assessed for eligibility. Participants were recruited through the gynecologic outpatient clinic and underwent preoperative evaluation including ultrasonography, hormonal profile testing, and infection screening. A total of 200 participants were enrolled with consideration of a 10% –15% attrition rate to maintain adequate statistical power for final analysis.
The required sample size was determined a priori based on published
post-hysteroscopic recurrence rates of IUAs. In a randomized controlled trial of
98 women undergoing hysteroscopic transcervical resection of adhesions, Qu and
Zhou reported a 25%–40% re-adhesion rate at three months despite the use of
barrier therapy [18]. Based on this rate, we calculated that a minimum of 170
participants would provide 80% power at
The primary outcome was the incidence of IUAs, diagnosed by follow-up hysteroscopy at 3 months postoperatively. Secondary outcomes included IUA severity, changes in inflammatory biomarker levels, and their associations with clinical and surgical variables.
All hysteroscopic submucosal myomectomies were performed using a standardized bipolar resectoscope system (Karl Storz, Tuttlingen, Germany) under general anesthesia induced with propofol (AstraZeneca, Shanghai, China). Intrauterine distension was achieved with isotonic saline, and careful resection of the myoma was performed to minimize injury to the surrounding endometrium. Operative videos were archived for documentation and teaching purposes.
To allow unbiased assessment of adhesion formation, no postoperative anti-adhesion barriers or estrogen therapy were administered. All patients received routine postoperative monitoring for pain, bleeding, and infection and were given standard discharge instructions. Follow-up hysteroscopy was scheduled 12 weeks postoperatively to evaluate adhesion formation, and any adverse events were recorded prospectively.
To standardize postoperative care and avoid confounding effects, no prophylactic anti-adhesion barrier or postoperative estrogen therapy was administered to any participant. In our institution, such measures are not routinely used following hysteroscopic myomectomy but are reserved for high-risk or recurrent cases.
Peripheral blood samples were collected at three time points: 24 hours
preoperatively, 24–48 hours postoperatively, and at 3 months post-surgery. At
the 3-month follow-up, participants completed a short clinical questionnaire and
were screened for recent febrile illnesses, infections, or new inflammatory
conditions. If an acute illness was present at the time of the scheduled blood
draw, sampling was postponed until recovery, whenever feasible. Primary
inflammatory markers assessed included CRP, IL-6, and TNF-
Follow-up hysteroscopy was scheduled for all participants at 12 weeks following the index procedure. IUAs were evaluated and graded based on the European Society for Gynecological Endoscopy (ESGE) classification system, which stratifies adhesions into grades I–V based on their density, extent, and anatomic involvement. Evaluations were conducted by two independent reviewers blinded to inflammatory marker data. Discrepancies in scoring were resolved through consensus review.
Patient-level data on potential confounders were recorded, including age, body mass index (BMI), parity, smoking history, hormonal phase at the time of surgery (follicular vs. luteal), and presence of anemia. Menstrual phase was determined using the date of LMP and ultrasound endometrial thickness, and was corroborated by mid-cycle serum estradiol and progesterone measurments when needed.
Missing data were minimal: CRP (1.7%), IL-6 (2.2%), TNF-
All statistical analyses were performed using SPSS Statistics for Windows,
Version 26.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were used to
summarize baseline characteristics. Continuous variables were assessed for
normality using the Shapiro-Wilk test and compared with Student’s t-test
or the Mann-Whitney U test, as appropriate. Categorical variables were analyzed
using Pearson’s chi-square test when all expected cell counts were
Both unadjusted and adjusted ORs with 95% confidence intervals (CIs) were
calculated. Unadjusted ORs were obtained from univariable logistic regression
models; adjusted ORs from multivariable logistic regression models controlling
for prespecified confounders (BMI, smoking status, menstrual phase, surgical
complexity, and FIGO type 0–II myoma type). Candidate variables for
multivariable modeling were selected based on univariable p
p-values for biomarker comparisons across time points were adjusted
using the Benjamini–Hochberg false discovery rate (FDR) procedure. ROC curves
were used to quantify the discrimination of biomarkers for moderate-to-severe
IUAs. Area under the curve (AUCs) with 95% CIs (DeLong method) were reported.
Optimal cut-off values for CRP, IL-6, and TNF-
Missing data were minimal, and sensitivity analyses confirmed that excluding
cases with missing values did not materially alter the primary results. A
two-sided p
Of 220 women screened, 20 were excluded (n = 12 did not meet inclusion criteria; n = 5 refused participation; n = 3 other reasons). 200 participants were enrolled. Postoperative attrition included 15 lost to follow-up and 5 withdrawals, leaving 180 for final analysis (Fig. 1).
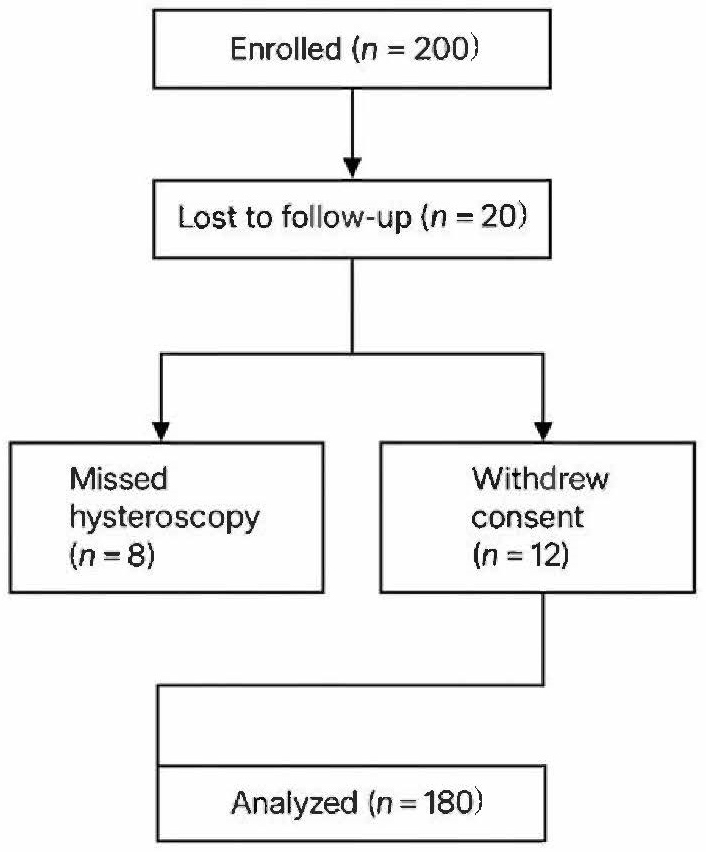 Fig. 1.
Fig. 1.
STROBE flow diagram. STROBE, Strengthening the Reporting of Observational studies in Epidemiology.
Flowchart of participant enrollment, attrition, and final analysis cohort. Of 220 women screened, 200 met eligibility criteria and underwent hysteroscopic submucosal myomectomy. 20 participants were excluded due to failure to meet inclusion criteria (n = 12), refusal to participate (n = 5), or other reasons (n = 3). Postoperative attrition included 15 participants lost to follow-up and 5 withdrawals, resulting in 180 participants for final analysis.
Baseline demographic and clinical characteristics are summarized in Table 1a.
The mean age of participants was 34.5
| Characteristic | Value | |
| Age (years) | 34.5 | |
| BMI (kg/m2) | 24.3 | |
| Parity | ||
| Nulliparous | 112 (62%) | |
| Parous | 68 (38%) | |
| Myoma type (FIGO) | ||
| Type 0 | 40 (22%) | |
| Type I | 122 (68%) | |
| Type II | 18 (10%) | |
| Smoking status | ||
| Current smoker | 27 (15%) | |
| Non-smoker | 153 (85%) | |
| Menstrual phase | ||
| Follicular phase | 108 (60%) | |
| Luteal phase | 72 (40%) | |
| Presence of anemia | 36 (20%) | |
SD, standard deviation; BMI, body mass index; FIGO, International Federation of Gynecology and Obstetrics; n, of patients.
| Characteristic | Mild-to-Severity IUAs (n = 45) | Absent or mild IUAs (n = 135) | p-value | |
| Age (years), mean |
35.1 |
34.3 |
0.31 | |
| BMI (kg/m2), mean |
24.7 |
24.2 |
0.48 | |
| Nulliparous, n (%) | 29 (64.4%) | 83 (61.5%) | 0.73 | |
| Myoma type | ||||
| Type 0, n (%) | 7 (15.6%) | 33 (24.4%) | 0.30 | |
| Type I, n (%) | 30 (66.7%) | 92 (68.1%) | 0.85 | |
| Type II, n (%) | 8 (17.7%) | 10 (7.4%) | 0.08* | |
| Current smoker, n (%) | 8 (17.8%) | 19 (14.1%) | 0.54 | |
| Menstrual phase at surgery | ||||
| Follicular, n (%) | 26 (57.8%) | 82 (60.7%) | 0.72 | |
| Luteal, n (%) | 19 (42.2%) | 53 (39.3%) | 0.74 | |
| Presence of anemia, n (%) | 10 (22.2%) | 26 (19.3%) | 0.68 | |
IUAs, intrauterine adhesions. *Statistically significant at p
Out of 200 enrolled patients, 180 completed the full follow-up, including follow-up hysteroscopy at 3 months postoperatively. Based on ESGE grading, 83 patients (46.10%) exhibited no IUAs, while 46 (25.62%) had mild IUAs, 34 (18.92%) had moderate IUAs, and 17 (9.40%) had severe IUAs. The distribution of IUA severity is summarized in Table 2.
| S. No | IUA severity | (n = 180) | Percentage of patients % |
| 1. | No IUA | 135 | 75.00 |
| 2. | Mild | 22 | 12.22 |
| 3. | Moderate | 16 | 8.89 |
| 4. | Severe | 7 | 3.89 |
S. No, serial number.
Inflammatory marker levels varied significantly across the different adhesion
severity groups. The average levels and SDs for CRP, IL-6, and TNF-
| IUA severity | CRP (mg/L) | IL-6 (pg/mL) | TNF- |
| No IUA | 5.01 |
14.98 |
9.68 |
| Mild IUA | 6.25 |
14.66 |
9.39 |
| Moderate IUA | 6.79 |
18.01 |
10.00 |
| Severe IUA | 7.03 |
20.26 |
11.85 |
CRP, C-reactive protein; IL-6, interleukin-6; TNF-
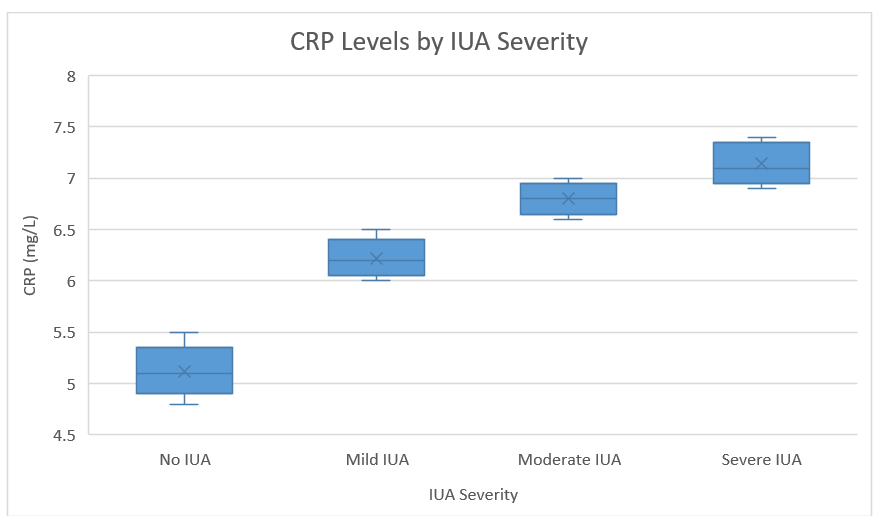 Fig. 2.
Fig. 2.
Boxplot showing CRP levels increase progressively with IUA severity.
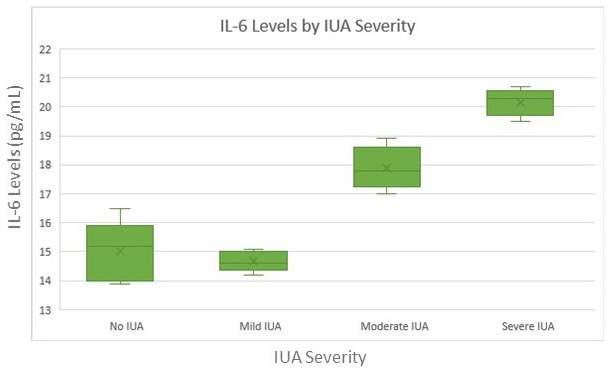 Fig. 3.
Fig. 3.
IL-6 levels were significantly elevated in moderate and severe IUA groups.
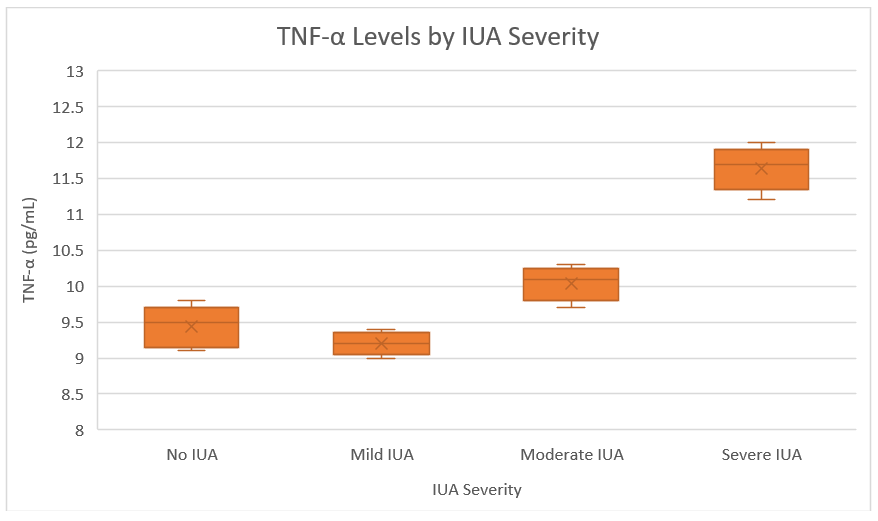 Fig. 4.
Fig. 4.
TNF-
| Marker | Timepoint | Mild-to-Severity IUAs (n = 45) | Absent or mild IUAs (n = 135) | p-value |
| CRP (mg/L) | Pre-op | 5.15 |
4.92 |
0.280 |
| 48 h Post-op | 8.61 |
6.02 |
||
| 3 months Post-op | 6.37 |
5.04 |
||
| IL-6 (pg/mL) | Pre-op | 15.93 |
14.76 |
0.230 |
| 48 h Post-op | 22.75 |
15.77 |
||
| 3 months Post-op | 18.08 |
14.23 |
||
| TNF- |
Pre-op | 10.22 |
9.61 |
0.230 |
| 48 h Post-op | 13.21 |
9.97 |
||
| 3 months Post-op | 11.48 |
9.33 |
Spearman’s correlation analysis demonstrated a moderate positive correlation
between inflammatory marker levels and IUA severity scores: IL-6 exhibited the
strongest correlation (
These results are detailed in Table 4 and visually summarized via the heatmap in Fig. 5.
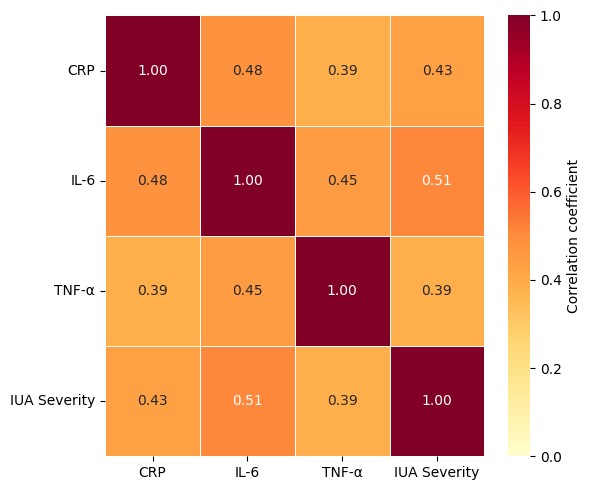 Fig. 5.
Fig. 5.
Heatmap displaying Spearman’s correlation between inflammatory biomarkers and IUA severity. Strongest correlation observed between IL-6 and adhesion grade.
| Marker | Correlation with IUA severity |
| CRP | 0.43 |
| IL-6 | 0.51 |
| TNF- |
0.39 |
To assess predictive power, a logistic regression model was developed to identify patients at risk of developing moderate-to-severe IUAs based on perioperative biomarker levels. IL-6 emerged as the strongest independent predictor:
These findings are presented in Table 5a, with the ROC curves for IL-6, CRP, and
TNF-
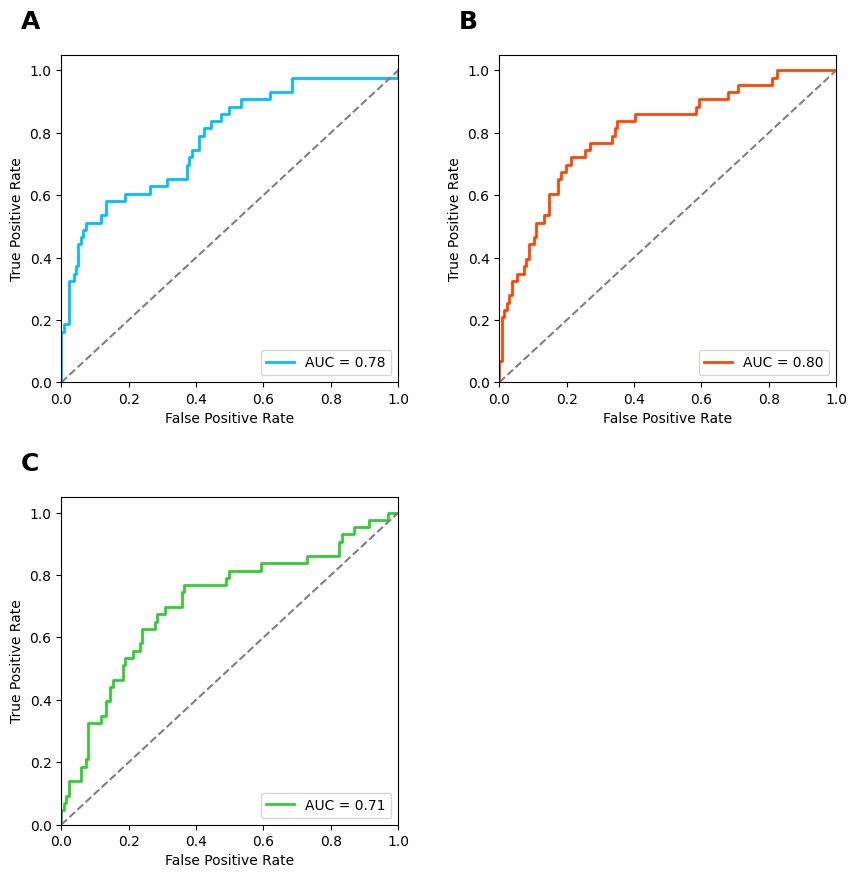 Fig. 6.
Fig. 6.
Receiver operating characteristic (ROC) curves for
prediction of IUA using inflammatory markers. (A) CRP. (B) IL-6. (C)
TNF-
| Marker | Coefficient | p-value |
| CRP | 0.63 | 0.004 |
| IL-6 | 0.84 | 0.001 |
| TNF- |
0.42 | 0.019 |
In multivariate analysis, both IL-6 and CRP remained independent predictors of IUA development after adjustment for surgical complexity, FIGO myoma type, BMI, parity, smoking history, menstrual phase, and anemia. The risk associated with elevated IL-6 levels was especially pronounced in women with FIGO type II myomas.
Table 5b presents both unadjusted and adjusted ORs for all predictors. In
univariable models, IL-6 and CRP showed strong crude associations with IUA
development, which remained significant after multivariable adjustment. In
contrast, TNF-
| Variable | Unadjusted OR (95% CI) | Adjusted OR (95% CI) | p-value |
| IL-6 (per 1 pg/mL increase) | 1.21 (1.11–1.32) | 1.18 (1.08–1.29) | |
| CRP (per 1 mg/L increase) | 1.38 (1.11–1.70) | 1.33 (1.07–1.65) | 0.010 |
| TNF- |
1.16 (1.01–1.34) | 1.12 (0.97–1.29) | 0.112 |
| Myoma type II vs. I/0 | 2.85 (1.08–7.55) | 2.70 (1.01–7.25) | 0.048 |
| Operative time (per 10 min) | 1.20 (1.02–1.43) | 1.18 (0.98–1.43) | 0.084 |
| BMI (per 1 kg/m2 increase) | 1.05 (0.96–1.15) | 1.03 (0.94–1.13) | 0.520 |
Unadjusted ORs derived from univariable logistic regression models; adjusted ORs derived from multivariable models controlling for BMI, smoking status, menstrual phase, surgical complexity, and myoma type.
Histograms were created to visualize the overall distribution of inflammatory
biomarker levels in the study population. CRP levels exhibited a mildly
right-skewed distribution, with most values clustering between 5.5–6.0 mg/L and
a longer tail toward higher concentrations (Fig. 7). The mean exceeding the median
is consistent with the right-skewed distribution and aligns with the summary
statistics. IL-6 levels showed a normal distribution with a higher tail in severe
cases (Fig. 8). Histogram demonstrates a slightly right-skewed distribution of
IL-6, with most values between 14 and 20 pg/mL, and a tail extending beyond 21
pg/mL. TNF-
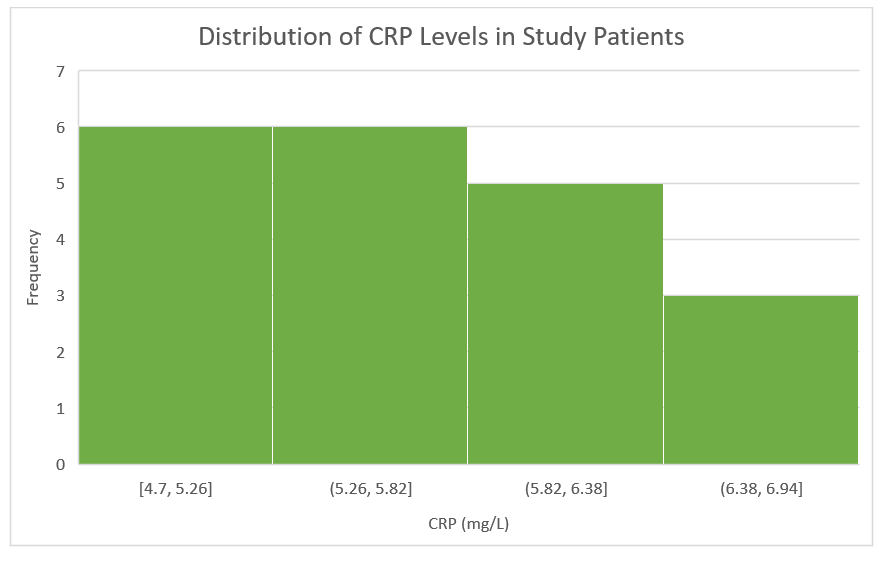 Fig. 7.
Fig. 7.
Histogram of CRP levels (n = 180). The distribution is
mildly right-skewed (mean
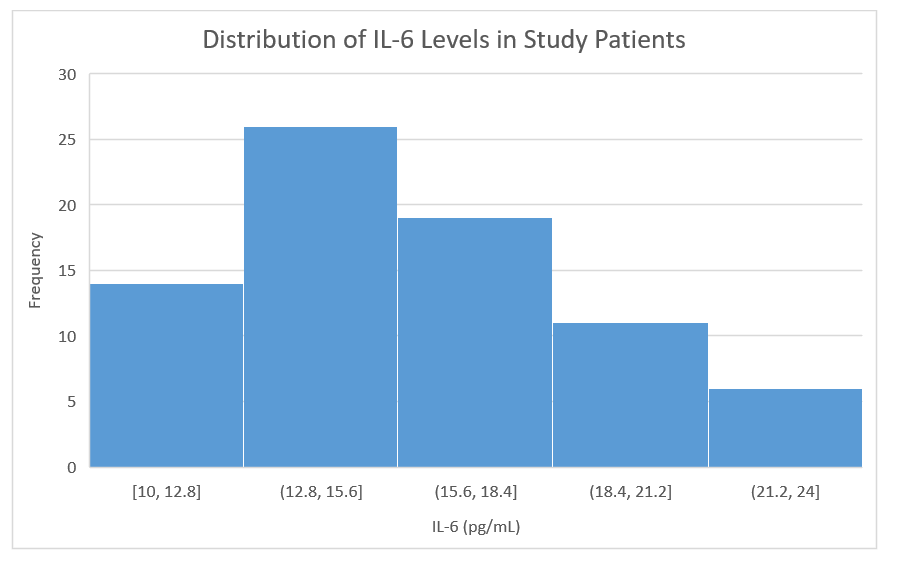 Fig. 8.
Fig. 8.
Histogram of IL-6 levels (n = 180). The distribution
is slightly right-skewed with a tail extending beyond 21 pg/mL. Shapiro-Wilk test
p
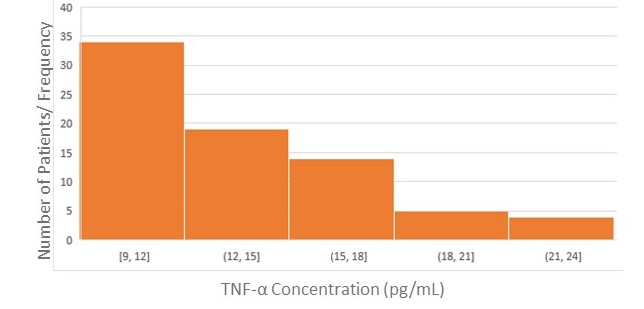 Fig. 9.
Fig. 9.
Histogram of TNF-
Table 6a summarizes the AUC values and significance levels of inflammatory
markers for predicting moderate-to-severe IUAs. IL-6 showed the highest
predictive performance (AUC = 0.82), showing strong discrimination for
moderate-to-severe IUAs. Both CRP and TNF-
| Marker | AUC | 95% CI | Coefficient | p-value |
| IL-6 | 0.82 | 0.74–0.90 | 0.84 | |
| CRP | 0.74 | 0.66–0.83 | 0.63 | 0.004 |
| TNF- |
0.71 | 0.62–0.80 | 0.42 | 0.019 |
| Marker | Cut-off value | Sensitivity (%) | Specificity (%) | Youden index |
| IL-6 (pg/mL) | 18.0 | 82 | 73 | 0.55 |
| CRP (mg/L) | 6.3 | 76 | 68 | 0.44 |
| TNF- |
10.5 | 70 | 65 | 0.35 |
| Marker | Cut-off | Sens (%) | Spec (%) | LR+ | LR− | PPV (%) | NPV (%) | 95% CI (AUC) |
| IL-6 (pg/mL) | 18.0 | 82 | 73 | 3.04 | 0.25 | 68 | 85 | 0.74–0.90 |
| CRP (mg/L) | 6.3 | 76 | 68 | 2.38 | 0.35 | 62 | 80 | 0.66–0.83 |
| TNF- |
10.5 | 70 | 65 | 2.00 | 0.46 | 57 | 77 | 0.62–0.80 |
Sens, sensitivity; Spec, specificity; LR+, positive likelihood ratio; LR–, negative likelihood ratio; PPV, positive predictive value; NPV, negative predictive value; AUC, area under the ROC curve.
Table 7 summarizes surgical outcomes, including operative time and intraoperative and postoperative complications, comparing the IUA group and non-IUA group. Operative time was significantly longer in patients who developed IUAs (p = 0.02), possibly due to more complex resection or endometrial trauma. Fluid deficit was also greater in the IUA group (p = 0.04), suggesting higher intrauterine irrigation volume needs. Complication rates (bleeding, uterine perforation, and postoperative pain) were higher in the IUA group, though not all reached statistical significance.
| Surgical outcome | IUA group (n = 45) | Non-IUA group (n = 135) | p-value |
| Operative time (minutes), mean |
48.60 |
43.10 |
0.02* |
| Fluid deficit (mL), mean |
520.00 |
470.00 |
0.04* |
| Cervical dilation needed, n (%) | 29 (64.40%) | 65 (48.10%) | 0.06 |
| Intraoperative bleeding |
5 (11.10%) | 6 (4.40%) | 0.14 |
| Uterine perforation, n (%) | 1 (2.20%) | 0 (0%) | 0.25 |
| Immediate postoperative pain (VAS |
16 (35.60%) | 31 (23.00%) | 0.11 |
| Readmission within 30 days, n (%) | 3 (6.70%) | 4 (3.00%) | 0.36 |
VAS, visual analog scale. * shows statistically significant values.
Fig. 10 shows a line graph comparing trends of CRP, IL-6, and TNF-
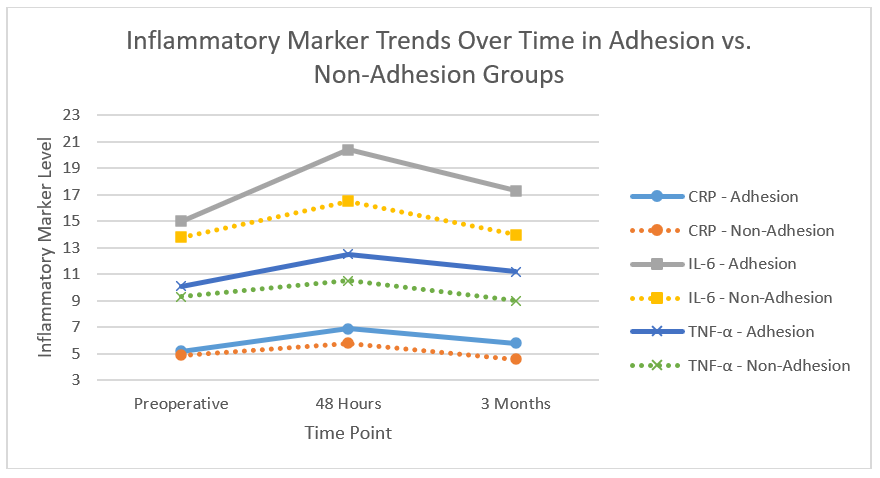 Fig. 10.
Fig. 10.
Trends of CRP, IL-6, and TNF-
Next, we developed a Venn diagram that shows the overlap of the three
inflammatory markers (IL-6, CRP, and TNF-
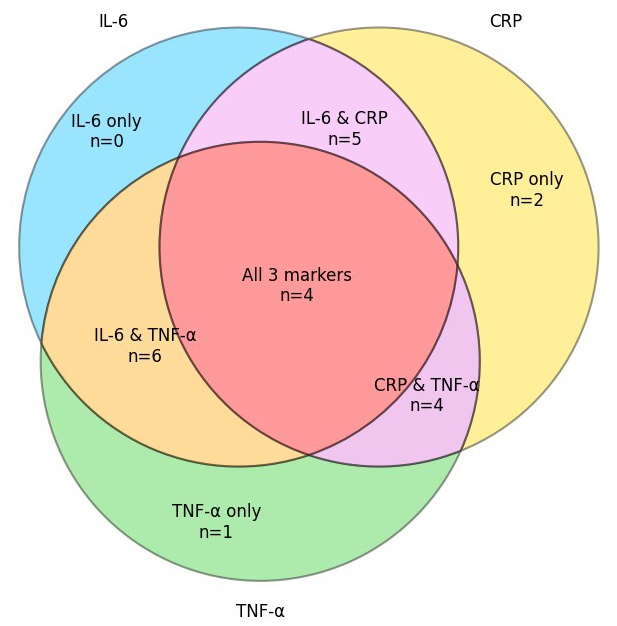 Fig. 11.
Fig. 11.
Overlap of elevated inflammatory biomarkers in IUA patients.
Venn diagram with the number of patients with elevated IL-6, CRP, and
TNF-
This prospective cohort study demonstrates that perioperative elevations in
IL-6, CRP, and TNF-
Our results align with the established understanding that abnormal inflammatory
responses play a pivotal role in endometrial fibrosis and adhesion development
[6, 7, 8]. Elevated perioperative IL-6 and CRP levels were found to correlate not
only with IUA incidence, but also with adhesion severity, supporting evidence
from previous clinical and translational studies [10, 11, 13, 14].
Mechanistically, IL-6 and TNF-
Experimental models and clinical tissue analyses have confirmed that unresolved
inflammation disrupts cyclical endometrial regeneration, resulting in fibrous
bands and reproductive dysfunction [8, 9]. For instance, Wang et al. [6]
demonstrated that experimental endometrial injury in rodents induced persistent
upregulation of IL-6, TNF-
Early identification of women at risk for IUA is essential for optimal
management, yet remains a clinical challenge given the often asymptomatic or
delayed presentation of adhesions [4, 5]. The present study reinforces recent
evidence showing that CRP, IL-6, and TNF-
Recent research by Hong et al. [13] showed that postoperative CRP and
IL-6 are predictive of adhesion recurrence following hysteroscopy adhesiolysis,
while Miyazaki et al. [14] reported CRP as a key predictor for severe
IUAs. Incorporating such biomarkers into routine practice may enhance clinical
decision-making and support personalized care. Using the Youden index, we derived
optimal cut-off values for IL-6, CRP, and TNF-
Consistent with prior literature, our findings show that patients with FIGO type II myomas, greater operative times, and higher intraoperative fluid deficits are at higher risk of developing IUAs [17, 18]. These features likely reflect greater surgical complexity and deeper myometrial involvement, both of which contribute to tissue trauma and amplify the inflammatory cascade [3, 18]. Careful surgical technique—including minimization of endometrial injury, careful use of energy devices, and optimized resection strategies—remains essential to reducing postoperative adhesion risk [18].
Subgroup analyses further suggested that operative time and surgical complexity, when combined with elevated perioperative biomarkers, can identify a subset of patients at especially high risk for significant adhesions. This layered approach to risk stratification may be particularly useful in tailoring postoperative surveillance and preventive measures.
Despite advances in surgical technique and postoperative care, IUA recurrence remains a significant challenge, with rates exceeding 30% in some cohorts [12]. The identification of reliable biomarkers for early detection could enable more aggressive or preemptive interventions. In addition to anti-inflammatory therapies and hormonal support, physical anti-adhesion barriers have shown clinical efficacy [19, 20]. Randomized trials and meta-analyses confirm that hyaluronic acid gel, in particular, significantly reduces both incidence and recurrence of IUAs when applied after hysteroscopic procedures [19, 20]. Given its safety and practicality, hyaluronic acid gel could be selectively utilized in patients identified as high-risk based on elevated IL-6 or CRP levels.
Cost-effectiveness and feasibility of biomarker-based monitoring remain to be fully established, particularly in low-resource settings. Nonetheless, integrating these markers into clinical pathways may optimize the allocation of resources by targeting preventive strategies to those most likely to benefit. Emerging technologies such as artificial intelligence and machine learning are increasingly influencing risk prediction and management in gynecology. Recent models that integrate perioperative biomarker data with clinical and surgical variables have achieved high predictive accuracy for postoperative IUAs. These tools could enable real-time, individualized risk assessment and support shared decision-making with patients [16].
Future research should prioritize multicenter, prospective validation of biomarker cut-offs and machine learning models [16]. Randomized controlled trials are needed to determine whether interventions targeted to high-risk biomarker profiles—such as anti-cytokine therapies, regenerative biomaterials, or individualized follow-up—can significantly reduce adhesion rates and improve reproductive outcomes [9, 20, 21]. Mechanistic studies using endometrial fluid sampling, tissue-level cytokine analysis, and multi-omics approaches will further elucidate the inflammatory-fibrotic continuum in IUA pathogenesis [8, 21].
In addition to the robust associations between IL-6, CRP, and IUA risk, specific clinical and procedural variables warrant further attention. The number, size, and location of submucosal fibroids—particularly larger or multiple lesions on opposing uterine walls—may exacerbate endometrial trauma and potentiate postoperative fibrosis. Although our cohort was stratified by FIGO classification, future analyses should incorporate more granular metrics such as cumulative myoma burden and uterine cavity geometry to refine risk prediction.
Similarly, the depth and extent of tissue injury during resection are influenced by the type and settings of electrosurgical energy. In our study, the use of a bipolar resectoscope was standardized; however, wattage and specific power settings were not recorded. Higher thermal energy can exacerbate tissue injury and may amplify inflammatory signaling. Reporting these parameters in future research will improve reproducibility and allow evaluation of dose–response relationships between energy delivery and adhesion risk.
Although perioperative IL-6 and CRP showed consistent associations with IUA presence and severity in our cohort, our thresholds are derivation cutoffs and have not been externally validated. Prior studies support the association and potential predictive value of these biomarkers (e.g., CRP and IL-6 after hysteroscopic procedures [10, 13, 14]; narrative and clinical reviews on IUA pathogenesis and management [15, 16]; and emerging prediction models, but none have established practice-changing cut points across diverse settings. Prospective, multicenter external validation, model calibration, and cost-effectiveness analyses are required before considering routine clinical use.
Beyond their potential role in predicting IUA risk after hysteroscopic myomectomy, systemic inflammatory indices are increasingly being applied across gynecological oncology and reproductive medicine. For example, recent work has demonstrated that combining systemic inflammatory indices with tumor markers such as CA-125 (SIR-125 and SIRI-125) significantly improves the preoperative differentiation of borderline ovarian tumors from early-stage ovarian carcinoma, achieving AUCs above 0.80 [22]. Similarly, the SIR-En index, which integrates systemic inflammation measures with endometrial thickness, distinguished endometrial carcinoma from atypical hyperplasia in postmenopausal women presenting with abnormal uterine bleeding, demonstrating high specificity [23]. Collectively, these findings highlight the broader applicability of inflammation-based biomarkers in gynecologic settings, including ovarian and endometrial disease, and support our conclusion that perioperative monitoring of inflammatory markers in intrauterine surgery may represent a clinically relevant and cost-effective extension of this diagnostic paradigm.
This study has several limitations. First, although our study was prospective and included standardized biomarker assessment, detailed fibroid metrics beyond FIGO classification or operative energy settings were not captured, which may represent important modifiers of adhesion risk. Second, chronic endometritis and other baseline inflammatory conditions were not assessed histologically, limiting our ability to distinguish pre-existing from procedure-induced inflammation. Third, our follow-up was limited to 3 months after surgery, which may underestimate late or progressive adhesion formation; longer surveillance (e.g., at 6 or 12 months) could capture delayed cases and refine predictive cut-offs. Fourth, while we recalculated all p-values using Fisher’s exact test or Yates’ continuity correction for low-cell-count categorical variables, some residual risk of type-I error remains due to multiple comparisons. Finally, as an observational study, our findings establish an association but not causality; randomized controlled trials are needed to determine whether modifying perioperative inflammation can reduce adhesion rates. Our study focused on adhesion incidence and severity, without directly assessing fertility outcomes such as conception rates, pregnancy maintenance, or live birth. Because IUAs primarily affect reproductive health, longer-term follow-up capturing fertility and obstetric outcomes is essential to establish the clinical utility of perioperative biomarker monitoring. Another limitation is that we did not conduct histological examinations to definitively rule out endometriosis or chronic endometritis. Although women with known or clinically suspected conditions were excluded based on history, imaging, and laboratory testing, subclinical cases may have been present and could act as unmeasured confounders. Future studies incorporating histopathologic screening may help clarify the association between perioperative biomarkers and adhesion risk.
This study underscores the potential of inflammation-guided, personalized risk
stratification for women undergoing intrauterine surgery. Serial perioperative
measurement of IL-6 and CRP, combined with established clinical risk factors, can
identify patients at increased risk of IUAs who may benefit from targeted
preventive strategies such as early follow-up hysteroscopy, application of
anti-adhesion barriers, or adjunctive hormonal therapy. Although the
cost-effectiveness and operational feasibility of such protocols require further
evaluation, our findings indicate that perioperative monitoring of IL-6, CRP, and
TNF-
Our findings demonstrate that perioperative elevations in IL-6, CRP, and
TNF-
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
JT conceptualized the study, conducted the investigation, analyzed the data, and wrote the original draft. JT reviewed and approved the final manuscript and agrees to be accountable for all aspects of the work.
The study was approved by the Institutional Ethics Committee of Wenzhou Medical College, vide letter No. WMCH/Ethics/2799, and all participants provided informed consent before enrollment. Clinical procedures and data handling adhered strictly to the principles of the Declaration of Helsinki.
The author sincerely thanks the staff of the Department of Obstetrics and Gynecology, Wenzhou Medical College, for their assistance with participant recruitment, data collection, and laboratory analyses.
This research received no external funding.
The author declares no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.











