1 Department of Family-Centered Maternity Ward, Affiliated Hospital of Nantong University, 226001 Nantong, Jiangsu, China
2 School of Nursing and Rehabilitation, Nantong University, 226001 Nantong, Jiangsu, China
3 Department of Pediatrics, Affiliated Hospital of Nantong University, 226001 Nantong, Jiangsu, China
Abstract
Previous observational studies suggest that polyunsaturated fatty acids (PUFAs) may influence the risk of postpartum depression (PPD) by modulating neuroinflammation and synaptic function. However, causal evidence remains limited. This study employs a genetic approach to investigate the causal relationship between omega-3 and omega-6 PUFAs, including their subtypes, as well as the risk of PPD.
Genetic instrumental variables (IVs) associated with PUFA levels were selected based on genome-wide association study (GWAS) data from European populations. The inverse variance-weighted (IVW) method, along with additional Mendelian randomization (MR) approaches, were applied to analyze the associations between genetically predicted PUFA levels and PPD risk.
The results indicate that genetically predicted levels of omega-3, omega-6, and their subtypes were not causally associated with the risk of PPD in European women (odds ratio [OR] for omega-3 PUFAs = 1.026 [95% confidence interval [CI]: 0.949–1.109], p = 0.926; OR for docosahexaenoic acid (DHA) = 0.937 [95% CI: 0.808–1.087], p = 0.39; OR for eicosapentaenoic acid (EPA) = 0.798 [95% CI: 0.561–1.136], p = 0.211; OR for omega-6 PUFAs = 1.053 [95% CI: 0.939–1.181], p = 0.377; OR for linoleic acid (LA) = 1.046 [95% CI: 0.938–1.167], p = 0.418; and OR for arachidonic acid (AA) = 1.001 [95% CI: 0.941–1.063], p = 0.997).
The genetic evidence from this study does not support a causal role for omega-3 and omega-6 PUFAs, or their subtypes, in the prevention of PPD. Although the genetic evidence from this study does not support a causal role for omega-3 and omega-6 PUFAs, or their subtypes, in the prevention of PPD, these results do not preclude potential benefits in specific subgroups. Future biomarker-guided trials targeting individuals with baseline PUFA deficiency may still be warranted.
Graphical Abstract
Keywords
- postpartum depression
- omega-3 PUFAs
- omega-6 PUFAs
Postpartum depression (PPD) is a common psychological disorder experienced by women following childbirth, typically manifesting within the first four weeks postpartum [1]. The global prevalence of PPD is estimated to be between 10% and 15% [2], but there are significant variations across regions and cultural contexts. Notably, only half of PPD cases receive timely attention and intervention, whereas nearly half of affected individuals remain undiagnosed or untreated [3]. Untreated PPD not only affects maternal mental health and quality of life but can also impair mother‒infant bonding, leading to early neurodevelopmental delays in infants and long-term impacts on their emotional and behavioral development, including an increased risk of future emotional regulation disorders [4, 5].
While certain antidepressant medications have demonstrated efficacy in treating
adult depression, their specific effects on breastfeeding infants remain
inadequately understood. Given the limitations of pharmacological treatments for
PPD, interest in safer and more effective alternative therapeutic approaches is
increasing. Among various nutrients, polyunsaturated fatty acids (PUFAs)
[docosahexaenoic acid (DHA, C22:6n-3), eicosapentaenoic acid (EPA, C20:5n-3),
linoleic acid (LA, C18:2n-6), and arachidonic acid (AA, C20:4n-6)] have emerged
as biologically plausible candidates for the prevention of PPD [6]. As essential
fatty acids, LA and
DHA’s role in synaptic plasticity and EPA’s anti-inflammatory effects are
equally crucial in preventing PPD. DHA serves as a major structural component of
neuronal membranes and is crucial for synaptic plasticity, neurotransmission, and
neurogenesis—processes fundamental to mood regulation. As demonstrated by Cao
et al. [8], free DHA binds to its nuclear receptor, the retinoid X
receptor alpha (RXRA), directly upregulating the expression of immediate early
genes (IEGs). This signaling pathway promotes the development of dendritic spines
and the formation of functional synapses in vivo, representing a key
molecular mechanism through which DHA influences synaptic plasticity and
cognitive function. The core of EPA’s anti-inflammatory mechanism lies in its
role as a precursor metabolized by the lipoxygenase (LOX) and cytochrome P450
(CYP450) enzyme systems into a series of highly bioactive mediators, such as
5-Hydroxyeicosapentaenoic acid (5-HEPE), 18-Hydroxyeicosapentaenoic acid
(18-HEPE), and 17(18)-epoxyeicosatetraenoic acid (17(18)-EpETE) [9]. These potent
bioactive mediators exert their effects through a dual pathway: firstly, they
directly inhibit the activation of key pro-inflammatory signaling pathways such
as Nuclear factor kappa-light-chain-enhancer of activated B cells
(NF-
While preclinical evidence supports the neuroprotective effects of omega-3 fatty
acids, their clinical application in the perinatal period remains controversial
[10]. No international guidelines specify preventive or therapeutic doses for
PPD, likely owing to inconsistent clinical evidence. For example, Hoge et
al. [11] analyzed fatty acid levels in 72 Belgian pregnant women and reported
that DHA levels were inversely associated with PPD risk (p = 0.034).
Similarly, a Norwegian prospective cohort study (n = 43) reported inverse
correlations between late-pregnancy DHA levels/omega-3 highly unsaturated fatty
acids (HUFA) scores and Edinburgh Postnatal Depression Scale (EPDS) scores [12].
Conversely, a Swiss longitudinal case-control study [13] involving 54 pregnant
women with mental disorders and 40 healthy controls reported no significant
associations between PUFA levels and PPD (p
These inconsistencies may stem from methodological biases in traditional epidemiological studies, such as residual confounding from unadjusted factors such as FADS gene cluster polymorphisms, socioeconomic status (SES), cultural factors, and lipoprotein metabolism. Additionally, small sample sizes, recall bias from dietary questionnaires, and inconsistent PPD diagnostic criteria or EPDS in an intervention study may affect the validity of the results [15].
Mendelian randomization (MR) studies address these limitations by using genetic variants as instrumental variables (IVs), mimicking the randomization of RCTs to assess causal relationships between exposures (omega-3/omega-6 PUFA levels) and outcomes (PPDs). This method reduces confounding by leveraging genetic variants determined at conception and independent of behavioral/environmental factors while clarifying causal directionality and eliminating reverse causation [16].
In summary, while prior RCTs have explored the potential effects of omega-3 PUFAs on PPD, their results have been inconsistent, potentially due to methodological limitations such as residual confounding, reverse causality, or sample heterogeneity. More importantly, existing MR studies have not systematically investigated the modifying effect of polymorphisms in the FADS gene cluster (e.g., FADS1/FADS2) on the association between PUFAs and PPD. This represents a critical knowledge gap, given that the FADS genes encode desaturase enzymes that significantly influence PUFA metabolic efficiency and may contribute to the heterogeneity observed in previous studies.
To address this key gap, the present study was designed and implemented as a multivariable MR (MVMR) analysis. This approach systematically examines the causal relationship between genetically predicted PUFA levels and PPD risk, with a specific focus on evaluating the potential modifying role of FADS gene cluster polymorphisms on this relationship.
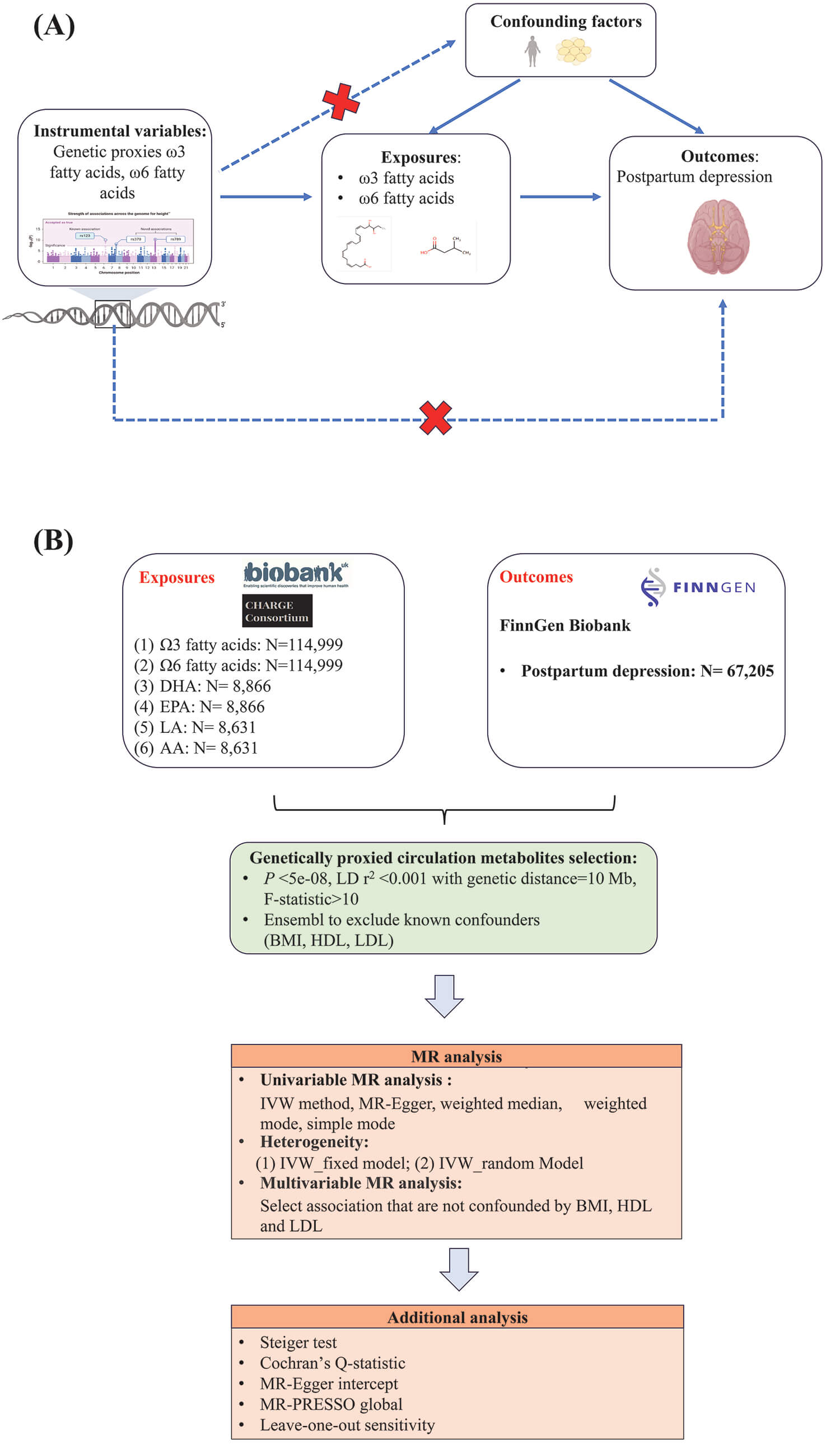
This study follows the MR_Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines, which are outlined in Supplementary Table 1, and is built upon the three foundational principles of MR. For genetic variants to function effectively as IVs in this analysis, it is imperative that they show a strong linkage to the omega-3 and omega-6 PUFAs examined in this research. Importantly, these IVs are not impacted by any other confounding factors that could distort the relationship between the omega-3/omega-6 PUFAs and the biological community of interest (e.g., PPD). Finally, it is imperative that the IVs do not have a direct effect on PPD, operating only through their influence on omega-3/omega-6 PUFAs.
The scope of this study is summarized below. Data Scope: The data sources utilized in this research are specified in Supplementary Table 2. To minimize sample overlap, primary analysis of PPD outcomes in Europeans was conducted via the FinnGen Biobank dataset [17]. The genome-wide association study (GWAS) data for omega-3/omega-6 PUFAs were sourced primarily from the UK Biobank, whereas the GWAS data for their subtypes (DHA, EPA, LA, and AA) were obtained from the CHARGE Consortium. Methodological Scope: The analysis employed multiple univariable MR methods, including the inverse variance-weighted (IVW), MR-Egger, weighted median, weighted mode, and simple mode approaches. Extensive sensitivity analyses were conducted to ensure robustness, which encompassed the exclusion of pleiotropic single-nucleotide polymorphisms (SNPs), adjustment for confounding factors [e.g., body mass index (BMI), blood lipids] via multivariable MR, control for horizontal pleiotropy using MR-PRESSO, and assessment of heterogeneity via Cochran’s Q test. Biological Focus Scope: The study placed a specific emphasis on the FADS gene locus, conducting stratified and interaction analyses to elucidate its potential role as an effect modifier in the relationship between PUFAs and PPD risk.
To ensure the independence of the IVs, SNPs were clumped for linkage
disequilibrium (LD) using criteria of r2
The primary analytical approach in this MR study was the IVW method. To address the issue of multiple comparisons, a Bonferroni correction was applied across all statistical tests. Additionally, genetic polymorphisms in the FADS gene cluster (e.g., FADS1/FADS2), which significantly influence the conversion efficiency of omega-3 PUFAs, were analyzed separately [18].
In MR studies, sensitivity analysis serves to re-examine the stability of causal inferences. The techniques applied encompass utilizing the Cochran’s Q test to evaluate potential heterogeneity and horizontal pleiotropy in the IVs [19], the MR-PRESSO method [20] to account for potential pleiotropy, and leave-one-out (LOO) sensitivity analysis to determine the impact of removing each IV individually on the relationship between omega-3/omega-6 PUFAs and the outcome of interest (PPD) [20].
BMI and lipid metabolic markers [high-density lipoprotein (HDL), low-density lipoprotein (LDL)] have been confirmed by multiple studies to be closely associated with the risk of developing PPD. Regarding BMI, research by Howard et al. [21] clearly indicated a significant positive correlation between PPD and BMI (r = 0.473, p = 0.020). The potential mechanism may involve hormone level disruptions and a chronic inflammatory state induced by obesity, thereby increasing the risk of PPD. Concerning blood lipid indicators, a cohort study by Teofilo et al. [22] found a positive correlation between low-density lipoprotein cholesterol (LDL-C) levels and PPD risk. Furthermore, Yang et al. [23], using MR methods, revealed an inverse causal relationship between genetically predicted high-density lipoprotein cholesterol (HDL-C) levels and PPD. These results collectively suggest that dyslipidemia may play a significant role in the pathogenesis of PPD. In summary, BMI, HDL, and LDL are closely related to the risk of PPD occurrence. Correcting for these confounding biases can enhance the accuracy and reliability of research results.
To mitigate potential confounding and horizontal pleiotropy, we conducted a
systematic screening of all IVs using the Ensembl genome browser
(http://www.ensembl.org, Release 110; European Molecular Biology
Laboratory-European Bioinformatics Institute, Hinxton, UK). SNPs exhibiting
genome-wide significant associations (p
Furthermore, we carried out an additional analysis known as MVMR, as outlined by Yun et al. [24]. The analysis of reverse causality aims to determine whether the outcomes of PPD could influence the exposure of omega-3/omega-6 PUFAs rather than the exposure causing the outcome. This investigation typically involves the Steiger filtering method [25] (Supplementary Methods).
The identified SNPs as IVs associated with exposures (omega-3 PUFAs, DHA, EPA, omega-6 PUFAs, LA, and AA) and confounding information (BMI, LDL, and HDL) are summarized in Supplementary Table 3 and Supplementary Table 4, respectively. F-statistics for each exposure or confounding were over 10, indicating that there is no weak instrument bias (Supplementary Table 3 and Supplementary Table 4).
Based on the outcome dataset sourced from the FinnGen Biobank, the initial univariable MR analysis demonstrated an absence of statistically significant causal links between genetically predicted levels of total omega-3 PUFAs, DHA, or EPA and a decreased risk of PPD in European populations (OR for omega-3 PUFAs = 1.026 [95% CI: 0.949–1.109], pIVW_omega-3 PUFAs = 0.926; OR for DHA = 0.937 [95% CI: 0.808–1.087], pIVW_DHA = 0.39; OR for EPA = 0.798 [95% CI: 0.561–1.136], pIVW_EPA = 0.211) (Fig. 1A). The results of four additional MR analyses are presented in Supplementary Table 4.
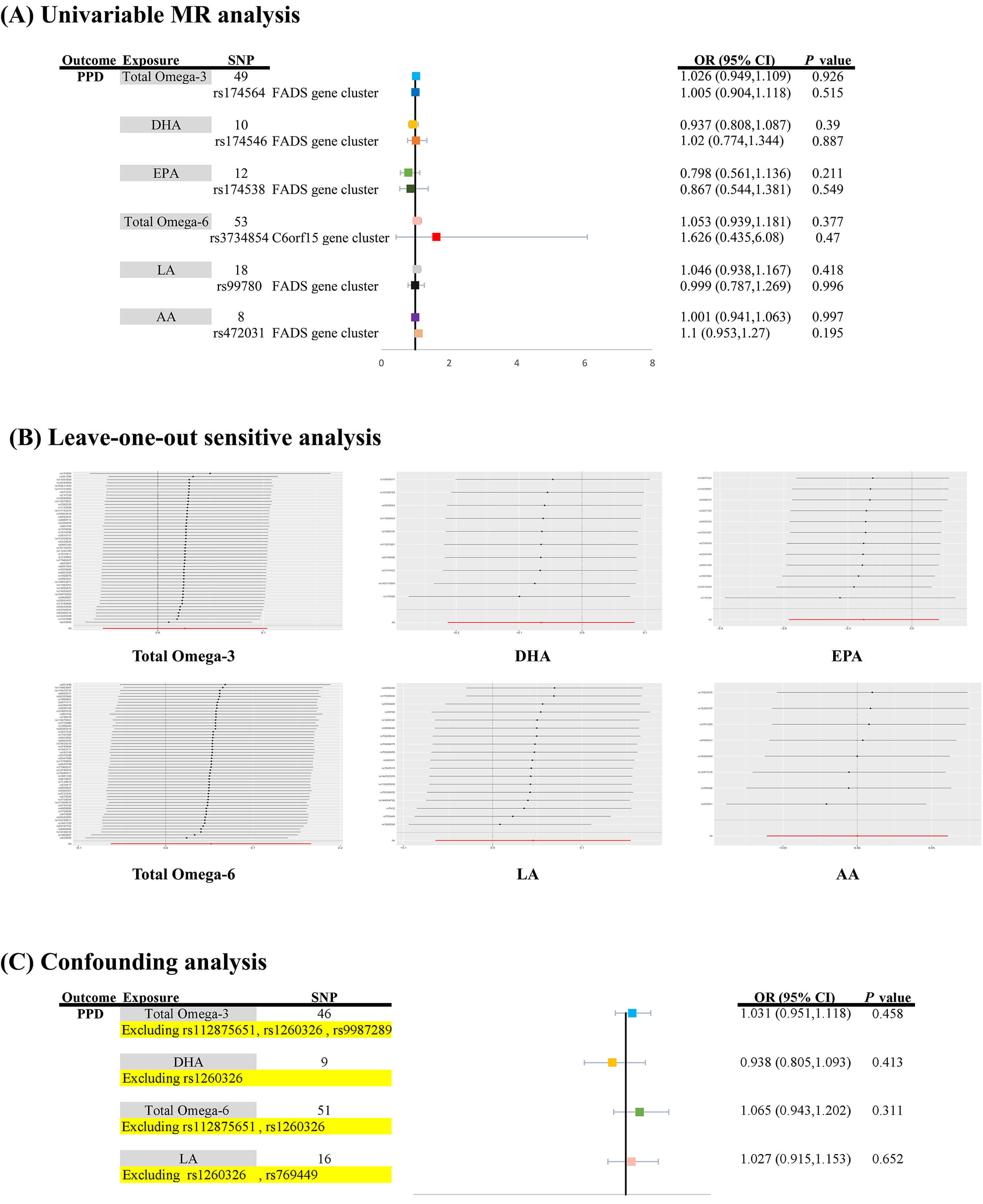 Fig. 1.
Fig. 1.
Causal associations between genetically predicted polyunsaturated fatty acids and postpartum depression risk. (A) Causal estimates from univariable MR analyses examining the associations of genetically predicted levels of total omega-3 PUFAs, DHA, EPA, total omega-6 PUFAs, LA, and AA with PPD risk, using genetic instruments from the FADS or chromosome 6 open reading frame 15 (C6orf15) gene clusters. (B) Sensitivity analysis using the leave-one-out approach was performed to evaluate the robustness of the causal inferences for each PUFA subtype. (C) Assessment of potential confounding influence by pleiotropic SNPs associated with BMI orHDL; the analysis excluded specific variants for each PUFA exposure (e.g., rs112875651 and rs1260326 for total omega-3 PUFAs; rs1260326 for DHA; see Supplementary Table 5 for the complete list of excluded SNPs and their corresponding confounders). MR, Mendelian randomization; AA, arachidonic acid; DHA, docosahexaenoic acid; EPA, eicosapentaenoic acid; FADS, fatty acid desaturase; LA, linoleic acid; PPD, postpartum depression.
Regarding additional PUFAs, specifically total omega-6 PUFAs, LA, and AA, no statistically significant links with PPD risk were identified in the European cohort derived from the FinnGen Biobank dataset (OR for omega-6 PUFAs = 1.053 [95% CI: 0.939–1.181], pIVW_omega-6 PUFAs = 0.377; OR for LA = 1.046 [95% CI: 0.938–1.167], pIVW_LA = 0.418; OR for AA = 1.001 [95% CI: 0.941–1.063], pIVW_AA = 0.997) (Fig. 1A and Supplementary Table 5). Moreover, Supplementary Table 6 includes the outcomes of four supplementary MR analyses.
Follow-up analysis of the FADS region demonstrated no association between PPD risk and genetic proxies for omega-3 PUFAs, DHA, or EPA in Europeans [pIVW_omega-3 PUFAs = 0.515, pIVW_DHA = 0.887, and pIVW_EPA = 0.549] (Fig. 1A). Moreover, single-locus MR analysis revealed no causal effect of the C6orf15 gene cluster of total omega-6 PUFAs or the FADS gene cluster of LA or AA on the risk of PPD [pIVW_omega-6 PUFAs = 0.470, pIVW_LA = 0.996, and pIVW_AA = 0.195] (Fig. 1A).
LOO analysis reinforced the stability of the findings related to omega-3 and omega-6 PUFAs (Fig. 1B).
In the confounding analysis, eight pleiotropic SNPs (total omega-3: rs112875651 and rs1260326 pertaining to BMI and rs9987289 pertaining to HDL; total omega-6: rs112875651 and rs1260326 pertaining to BMI; DHA: rs1260326 pertaining to BMI; LA: rs1260326 and rs769449 pertaining to BMI) were identified and are summarized in Supplementary Table 5. The removal of these specific SNPs did not alter the initial results, as the associations remained non-significant [pIVW_omega-3 PUFAs = 0.458, pIVW_DHA = 0.413, pIVW_omega-6 PUFAs = 0.311 and pIVW_LA = 0.652] (Fig. 1C and Supplementary Table 5).
Furthermore, the MVMR analysis in this study utilized a joint model; consistent
with the results of univariable MR analysis, MVMR analyses controlling for BMI,
HDL, and LDL also revealed no causal associations between six types of PUFAs and
the risk of PPD (pMVMR
| Exposure | Outcome | Adjusted covariates | MVMR_IVW method | Raw p value | Bonferroni corrected p value | Statistically significant results |
| OR (95% CI) | ||||||
| Omega-3 fatty acids | Postpartum depression | BMI | 1.016 (0.926, 1.113) | 0.742 | 0.0028 | None |
| HDL | 1.030 (0.947, 1.120) | 0.494 | 0.0028 | None | ||
| LDL | 1.046 (0.950, 1.151) | 0.361 | 0.0028 | None | ||
| DHA | Postpartum depression | BMI | 1.180 (0.988, 1.408) | 0.070 | 0.0028 | None |
| HDL | 0.782 (0.281, 2.175) | 0.638 | 0.0028 | None | ||
| LDL | 1.881 (0.440, 8.048) | 0.394 | 0.0028 | None | ||
| EPA | Postpartum depression | BMI | 1.170 (0.980, 1.397) | 0.082 | 0.0028 | None |
| HDL | 1.043 (0.381, 2.859) | 0.934 | 0.0028 | None | ||
| LDL | 1.243 (0.391, 3.951) | 0.713 | 0.0028 | None | ||
| Omega-6 fatty acids | Postpartum depression | BMI | 1.084 (0.949, 1.239) | 0.235 | 0.0028 | None |
| HDL | 1.065 (0.935, 1.213) | 0.344 | 0.0028 | None | ||
| LDL | 1.100 (0.920, 1.313) | 0.298 | 0.0028 | None | ||
| LA | Postpartum depression | BMI | 1.042 (0.909, 1.194) | 0.553 | 0.0028 | None |
| HDL | 1.023 (0.899, 1.163) | 0.733 | 0.0028 | None | ||
| LDL | 1.063 (0.895, 1.262) | 0.489 | 0.0028 | None | ||
| AA | Postpartum depression | BMI | 1.231 (0.642, 2.361) | 0.531 | 0.0028 | None |
| HDL | 1.026 (0.891, 1.181) | 0.722 | 0.0028 | None | ||
| LDL | 1.418 (0.705, 2.850) | 0.327 | 0.0028 | None |
BMI, body mass index; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
The Bonferroni correction was applied for 18 independent tests [6 exposures:
(omega-3, DHA, EPA, omega-6, LA, and AA)
Consequently, the absence of a significant genetic association between genetically proxied PUFAs and PPD risk was consistently demonstrated through additional confounding factor analysis and MVMR validation. Collectively, the evidence from this MR investigation does not support a causal role for genetically determined PUFA levels on PPD susceptibility within the European population.
Supplementary sensitivity analyses further confirmed the robustness of the
primary findings, indicating no substantial heterogeneity
(pheterogeneity
| Exposure | Outcome | Heterogeneity | Pleiotropy | MRPRESSO | Steiger filtering (Steiger_p value) | ||||||
| Method | Q value | Q_df | I2 (%) | Q_p value | Intercept | p value | Global test p value | Corrected results for outlier SNPs | |||
| Omega-3 fatty acids | Postpartum depression | MR Egger | 43.409 | 47 | 0.00 | 0.622 | 0.005 | 0.877 | 0.670 | No outliers were identified, the results for the outlier-corrected MR are set to NA | TRUE |
| (p = 3.89942 × 10−161) | |||||||||||
| Inverse variance weighted | 43.433 | 48 | 0.00 | 0.660 | |||||||
| DHA | Postpartum depression | MR Egger | 0.776 | 8 | 0.00 | 0.999 | –0.017 | 0.438 | 0.997 | No outliers were identified, the results for the outlier-corrected MR are set to NA | TRUE |
| (p = 1.836 × 10−8) | |||||||||||
| Inverse variance weighted | 1.444 | 9 | 0.00 | 0.998 | |||||||
| EPA | Postpartum depression | MR Egger | 4.690 | 10 | 0.00 | 0.911 | –0.012 | 0.716 | 0.998 | No outliers were identified, the results for the outlier-corrected MR are set to NA | TRUE |
| (p = 3.423 × 10−17) | |||||||||||
| Inverse variance weighted | 4.830 | 11 | 0.00 | 0.939 | |||||||
| Omega-6 fatty acids | Postpartum depression | MR Egger | 53.138 | 51 | 4.02 | 0.392 | 0.007 | 0.719 | 0.439 | No outliers were identified, the results for the outlier-corrected MR are set to NA | TRUE |
| (p = 1.099 × 10−125) | |||||||||||
| Inverse variance weighted | 53.274 | 52 | 2.39 | 0.425 | |||||||
| LA | Postpartum depression | MR Egger | 19.547 | 16 | 18.15 | 0.241 | –0.024 | 0.150 | 0.161 | No outliers were identified, the results for the outlier-corrected MR are set to NA | TRUE |
| (p = 3.226 × 10−46) | |||||||||||
| Inverse variance weighted | 22.345 | 17 | 23.92 | 0.172 | |||||||
| AA | Postpartum depression | MR Egger | 3.480 | 6 | 0.00 | 0.747 | –0.015 | 0.588 | 0.792 | No outliers were identified, the results for the outlier-corrected MR are set to NA | TRUE |
| (p = 3.193 × 10−41) | |||||||||||
| Inverse variance weighted | 3.807 | 7 | 0.00 | 0.802 | |||||||
NA, not applicable.
In recent years, the effects of omega-3 PUFA supplements on perinatal depression
have been studied in several RCTs, with conflicting results. Su et al.
[26] reported that in pregnant women with severe depression (n = 36), high-dose
omega-3 supplementation (3.4 g/day) for 8 weeks significantly reduced HAMD scores
and increased the clinical remission rate by 2.3-fold, indicating good safety.
This success may be due to the active placental transfer of DHA and increased
blood‒brain barrier permeability during pregnancy, allowing high-dose omega-3
fatty acids to exert anti-inflammatory [inhibiting IL-6/tumor necrosis
factor-
In contrast, Rees et al.’s RCT [28] on late-pregnancy women with
moderate to severe depression (n = 26) reported no significant difference in EPDS
score reduction or remission rates after 6 weeks of EPA + DHA supplementation (6
g/day) (p
Further exploration of the conflicting results from previous RCTs revealed that PPD diagnosis in all studies was based on self-reported EPDS scores. Although the EPDS has been widely validated and performs well, factors such as the subjects’ emotional state, cognitive ability, and understanding of the scale may influence the results. Additionally, the use of nutritional supplements in RCTs typically relies on self-reported food frequency questionnaires, which may introduce recall bias [15]. In a clinical study examining the correlation between omega-3/omega-6 PUFAs and PPD, RCTs often report only overall omega-3 PUFA intake and fail to focus on specific dietary adjustments for particular types (e.g., EPA or DHA), potentially overlooking differences in bioavailability and efficacy among different types of omega-3 PUFAs [31]. Moreover, some RCTs have small sample sizes (e.g., Rees et al.’s study [28] included only 26 participants), which may have resulted in insufficient statistical power. Furthermore, intervention durations in different RCTs vary from 4–65 weeks, and in some studies, omega-3 supplements were combined with psychotherapy, which may mask the specific effects of omega-3 supplements [29]. Finally, heterogeneity in factors such as the severity of depressive symptoms, baseline omega-3 levels, and socioeconomic background across study samples may also affect intervention outcomes.
MR, which uses genetic variants as IVs, is determined at birth and is not influenced by later environmental and lifestyle factors. This method can reduce the interference of confounding factors on causal relationships and avoid reverse causation issues. MR studies are typically based on large-scale GWASs and can provide assessments of long-term or even lifelong effects.
The discrepancy between the negative results in this MR study and the positive outcomes reported in some RCTs may arise from the following aspects: First, both observational studies and some RCTs may be influenced by residual confounding factors that are challenging to fully control, such as variations in the severity of depressive symptoms, baseline omega-3 levels, and socioeconomic backgrounds across study samples [32]. SES is a critical confounding factor; individuals with higher SES typically benefit from healthier dietary patterns (including higher fish and omega-3 intake), superior healthcare access, lower stress levels, and stronger social support networks, all of which are independent protective factors against PPD [32]. MR mitigates such biases by using lifelong genetic predispositions as instruments, effectively circumventing the interference of postnatal environmental and socioeconomic variables.
Thus, our findings suggest
that a substantial portion of the associations observed in prior observational
studies may reflect the influence of confounders like SES rather than a direct
causal effect of PUFAs. Second, certain RCTs demonstrating positive outcomes
exhibit methodological limitations. For instance, a study has a small sample
sizes (e.g., n
A previous study by Sallis et al. [33], which was based on the
ALSPAC cohort study in southern England (n = 2663), investigated the associations
between omega-3 PUFAs and perinatal depression via two-stage analysis. In the
baseline model without adjusting for covariates, no significant associations were
observed between DHA or EPA and antenatal depression (p
The univariable MR (UVMR) analysis in this study further confirmed that
genetically predicted total omega-3 PUFA levels were not significantly associated
with PPD risk (p = 0.926). Further subgroup analysis revealed that
circulating DHA and EPA levels were also unrelated to PPD risk (p
To address the limitations of Sallis et al.’s study [33], which
did not include omega-6 PUFAs, this study incorporated total omega-6 PUFAs and
their key metabolites, LA and AA. The results revealed no significant causal
associations between these PUFAs and PPD risk (p
This study not only addressed the aforementioned gaps but also explored the impact of genetic polymorphisms in desaturase-coding genes (e.g., FADS) and C6orf15 on the conversion efficiency of omega-3 PUFAs. Additionally, a multivariable confounding analysis was innovatively employed, incorporating covariates such as HDL, LDL, and BMI, to more precisely evaluate the causal relationships between omega-3 and omega-6 PUFAs and PPD, further validating the robustness of the univariable MR results.
Genetic polymorphisms in desaturase-encoding genes (e.g., FADS) and C6orf15 significantly affect the conversion efficiency of omega-3 PUFAs. Specific variants in FADS and C6orf15 may reduce desaturase activity, thereby impairing the synthesis of sufficient levels of omega-3 PUFAs and their subtypes, such as EPA and DHA [34]. Individuals carrying specific SNPs in FADS or C6orf15 may have reduced efficiency in synthesizing omega-3 PUFAs, necessitating exogenous supplementation of these fatty acids. Previous MR studies by Sallis et al. [33] did not account for the effects of FADS or C6orf15 genetic polymorphisms. Genetic variants (e.g., SNPs in the FADS gene cluster) may simultaneously regulate multiple lipoproteins (HDL/LDL) and fat distribution, violating the assumption of IV independence. Even after adjusting for FADS/C6orf15 genotypes in this MR study, no significant associations were found between omega-3 PUFAs and their subtypes DHA and EPA, as well as between omega-6 PUFAs and their subtypes LA and AA, and PPD (Fig. 1A).
In this MR study, although genetic polymorphisms in the FADS gene
cluster have been confirmed to significantly alter the activity of
Furthermore, the univariable MR model used in previous studies did not fully
account for collinearity within the lipid metabolism network. Covariates such as
HDL, LDL, and BMI may influence PPD through pathways such as inflammation and
lipid metabolism, creating collinearity with the target exposure (e.g., omega-3
PUFAs) [16]. To address this confounding bias, a multivariable MR model was
constructed [37], incorporating genetic IVs for HDL, LDL, and BMI. The results
revealed no substantial changes in the direction of effect or confidence
intervals (pIVW
Finally, through confounding analysis, SNPs with pleiotropic associations were excluded, further validating the assumption of instrument variable independence (Fig. 1C). These findings collectively suggest that, despite genetic polymorphisms significantly altering PUFA metabolic pathways, their causal effects on perinatal depression may be buffered by endogenous compensatory mechanisms. This provides a molecular basis for reconciling discrepancies between observational studies and MR results.
The maternal diet and maternal BMI are significant confounding factors that cannot be overlooked in studies of PPD. We candidly recognize this as an inherent limitation of our study: the use of publicly available summary-level GWAS data, rather than individual-level data, which were collected without detailed individual dietary information or BMI measurements. Consequently, it was not possible to directly adjust for “maternal diet” or “maternal BMI” as covariates in the model, as would be done in traditional regression analyses. This limitation may affect the interpretation and generalizability of our findings. Nevertheless, we employed several genetic methodologies to indirectly assess the potential influence of these confounding factors. First, although we could not control for measured BMI values, we were able to adjust for the genetic predisposition to BMI. We conducted an MVMR analysis, simultaneously incorporating genetic instruments for BMI, HDL, and LDL alongside the genetic instruments for PUFAs into a single model. The results, presented in Table 1, demonstrate that the association between PUFAs and PPD remained non-significant even after adjusting for genetically predicted BMI.
Furthermore, in our confounding analysis, we used the Ensembl database to
identify SNPs exhibiting genome-wide significant associations (p
The inherent design of the MR approach itself helps mitigate the impact of confounding factors. Our genetic IVs are randomly assigned at conception and are therefore less susceptible to influence by postnatal dietary or behavioral factors [38].
However, it is important to note that MR controls for the genetic predisposition to confounding (e.g., the baseline BMI tendency determined by genetics) rather than measured confounding (e.g., actual BMI changes measured directly during pregnancy). The latter may still introduce residual confounding through non-genetic pathways. Therefore, in future research, our team plans to initiate prospective cohort studies with individual-level data, enabling direct adjustment for baseline maternal diet and maternal BMI to validate the causal genetic effects of PUFAs on PPD.
Several limitations should be acknowledged in this study. First, the study data
were derived primarily from European populations, which may limit the
generalizability of the results, particularly in contexts where racial or
regional differences exist in PUFA metabolism and PPD risk. For instance,
research by Mathias et al. [39] demonstrated that among African
Americans, approximately 80% of individuals carry two copies of the alleles
associated with elevated AA levels. This genetic background correlates with
higher levels of LC-PUFAs and greater enzymatic efficiency, as indicated by
higher AA/dihomo-
Second, although MR methods can reduce confounding bias, unmeasured environmental or genetic factors may still influence the results. Additionally, the study relied on existing GWAS data, which may be subject to biases and limitations inherent in the original data collection processes.
Third, exploring the modifying role of baseline inflammatory status on the
PUFA-PPD relationship could provide deeper insight into our MR findings. However,
because our MR analysis relied on publicly available GWAS summary statistics
rather than individual-level phenotypic data, we were unable to retrieve or match
baseline inflammatory measures for participants, rendering a direct subgroup
analysis by inflammatory status infeasible. This is an intrinsic limitation of
the current study design. Future investigations should therefore prioritize the
construction of large, multicenter, prospective birth cohorts that repeatedly
quantify inflammatory biomarkers (e.g., CRP, IL-6, TNF-
Fourth, it is important to note that MR aims to estimate the average effect of ‘lifelong exposure’ across the general population by utilizing genetic IVs as proxies for variations in exposure levels to infer causal relationships. While this method offers significant advantages in identifying potent and universal causal effects, its capacity to detect effect modifications that exist only under specific environmental or physiological contexts may be limited [42]. Consequently, although the present study did not find a significant average causal effect of PUFAs on PPD risk across the entire population, this finding does not entirely preclude the potential benefits of PUFAs for certain subgroups of women. For instance, individuals with low baseline omega-3 levels, elevated inflammatory markers, or those carrying functional-decreasing variants of the FADS gene might still derive benefits from PUFA supplementation. In the presence of subgroup effects, if an intervention is effective only for a specific subpopulation, its overall effect will be diluted in a full population-level analysis, potentially leading to insufficient statistical power for detection [43]. This diminution of statistical power due to effect heterogeneity is a recognized limitation of MR studies utilizing summary-level data. Furthermore, the present study relied on publicly available GWAS summary data, which precluded access to individual-level covariate information. This limitation impeded further subgroup analyses based on phenotypic indicators such as inflammatory status or baseline nutritional status, representing a constraint for the in-depth exploration of heterogeneity using such datasets. Based on these considerations, future research should focus on identifying and validating potentially benefitting subgroups. By designing prospective, randomized, placebo-controlled intervention trials targeting specific female populations, such as those with low omega-3 levels, high inflammatory markers, or particular genetic backgrounds (e.g., FADS gene functional variants), we can facilitate early, precise identification and targeted intervention. Given the implication from this study that a universal supplementation strategy may lack significant public health benefits for the general population, directing limited scientific and clinical resources toward high-risk subgroups with genuine potential for benefit will be crucial for advancing the prevention and management of PPD toward a new model of precision nutrition intervention.
Fifth, this study has limitations stemming from insufficient statistical power, as evidenced by the wide confidence intervals observed for certain estimates (e.g., DHA-adjusted LDL: OR = 1.881, 95% CI: 0.440–8.048; EPA-adjusted LDL: OR = 1.243, 95% CI: 0.391–3.951; AA-adjusted LDL: OR = 1.418, 95% CI: 0.705–2.850). The broad confidence intervals likely result from a relatively small sample size, which reduces statistical power. In MR studies, a small sample size increases the standard error of the effect estimates, thereby widening the confidence intervals and compromising the precision of the causal inferences [44].
A key challenge in current genetic studies on specific PUFA subtypes, such as DHA, EPA, and AA, is the relatively limited sample size of their respective GWAS, despite the data being sourced from the authoritative CHARGE Consortium [45, 46]. Specifically, the sample sizes for DHA and EPA are both 8866, and for AA, the sample size is 8631. This constraint in sample size somewhat reduces the statistical power, which may consequently affect the precision of the effect estimates. Therefore, to enhance the reliability of future research, it is crucial to conduct larger-scale GWAS. This effort should particularly focus on expanding the sample coverage for specific PUFA subtypes like DHA, EPA, and AA through primary data-level collaboration and updates. Such initiatives will contribute to obtaining more precise genetic effect estimates and strengthen the persuasiveness of causal inferences.
Furthermore, although the MVMR framework employed in this study effectively controls for confounding bias, it inevitably introduces the issue of multicollinearity. Since the genetic IVs used to predict PUFAs and metabolic covariates (e.g., BMI, HDL, LDL) are associated through biological pathways, this leads to variance inflation in model estimates, manifesting as widened confidence intervals. Consequently, the statistical power for causal inference in this study is reduced. Nonetheless, as emphasized by Sanderson [47], the joint adjustment model remains the most appropriate method for estimating independent direct effects in the presence of potential covariates.
Thus, the attenuation of statistical precision, manifested as wider confidence intervals in MVMR, is an inherent limitation of the model when disentangling direct causal effects in the presence of correlated exposures. This is principally driven by the variance inflation factor introduced by conditioning on genetically correlated covariates. Thus, the imprecision is not a flaw but rather an expected statistical consequence of rigorously controlling for pleiotropic pathways, thereby providing less biased but more uncertain estimates of the direct effect.
The genetic evidence from this study does not support a causal role for omega-3 and omega-6 PUFAs or their subtypes in the prevention of PPD. While the genetic evidence from this study does not support a causal role for omega-3 and omega-6 PUFAs or their subtypes in the prevention of PPD, these results do not preclude potential benefits in specific subgroups. Future biomarker-guided trials in deficient subgroups (e.g., individuals with baseline PUFA deficiency) may still be warranted.
The data used in this study were obtained from publicly available databases. Specifically, summary-level data from the Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) Consortium are available at http://www.chargeconsortium.com/main/results , and data from FinnGen are available at https://www.finngen.fi/en.
XZ designed the research study and wrote the manuscript. LD, XC and JG performed the research, collected data, and analyzed the data. LF contributed substantially to the study conception and design, critically interpreted the results, and supervised the entire study. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
As the data are publicly accessible and contain no identifiable personal information, ethical approval and informed consent were not required.
Thanks to all the peer reviewers for their opinions and suggestions.
This work was funded by the Special Research Project of Nantong Municipal Health Commission (QNZ2025002).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG44542.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

