1 Department of Women’s Health, Wuhai Maternity and Child Healthcare Hospital, 016099 Wuhai, Inner Mongolia, China
†These authors contributed equally.
Abstract
Recurrent pregnancy loss (RPL) affects 1–5% of females of reproductive age, yet nearly half of these cases remain unexplained. This review synthesizes recent advances in understanding the immune-related causes of RPL and highlights how emerging technologies and precision medicine may help improve the diagnosis and treatment of RPL.
Increasing evidence implicates immune dysregulation at the maternal–fetal interface as a cause of RPL, including aberrant uterine natural killer cell activation, Th17/Treg imbalance, impaired macrophage polarization, and disrupted cytokine signaling. These alterations compromise immune tolerance and increase the risk of miscarriage. Autoimmune disorders such as antiphospholipid syndrome and systemic lupus erythematosus, as well as alloimmune intolerance to paternal antigens, further contribute to pathogenic immune responses.
Advances in single-cell RNA sequencing, spatial transcriptomics, and proteomic profiling have refined the understanding of decidual immune-cell states and identified candidate biomarkers—including human leukocyte antigen G (HLA-G), programmed death-ligand 1 (PD-L1), interleukin-10 (IL-10), and interleukin-17 (IL-17)—that distinguish tolerant from inflammatory microenvironments. Therapeutic approaches are shifting from the empirical use of glucocorticoids and anticoagulants toward mechanism-guided immunomodulation, incorporating calcineurin inhibitors, biologic agents, and emerging cellular therapies informed by immunophenotyping and molecular endotyping.
Integrating immunology, multi-omics technologies, and artificial intelligence offers a pathway toward precision immunotherapy in RPL. This convergence supports the development of safer, personalized, and more effective diagnostic and therapeutic strategies for females with immune-mediated reproductive failure.
Keywords
- recurrent pregnancy loss
- immune dysregulation
- precision immunotherapy
Recurrent pregnancy loss (RPL), which is defined as having two or more consecutive pregnancy losses, is still one of the most difficult reproductive disorders. It affects about 1–5% of females of reproductive age worldwide [1, 2, 3]. In addition to its physical effects, RPL increases mental and emotional stress for the couples involved. This stress is often accompanied by anxiety, depression, and fear of another failed pregnancy [4]. Even with progress in reproductive medicine, the cause of almost 40–50% of RPL cases is still unknown, and these cases are collectively referred to as unexplained recurrent pregnancy loss (URPL) [5, 6].
A growing body of evidence suggests that immune dysregulation at the maternal–fetal interface is a critical factor in these unexplained cases, leading to immune-related RPL [7, 8, 9]. During a typical pregnancy, a fragile equilibrium between immune activation and tolerance is essential for the acceptance of the semi-allogeneic fetus. Disruption of immune homeostasis via aberrant activation of uterine natural killer (NK) cells [10], an imbalance of T helper 17 cells (Th17)/Treg cells [11], or dysregulated macrophage polarisation [12, 13], can result in embryo rejection and miscarriage.
Autoimmune conditions such as antiphospholipid syndrome (APS) [14], systemic lupus erythematosus (SLE) [15], and undifferentiated connective tissue disease (UCTD) [16] have also been implicated as significant contributors to immune-related RPL. Furthermore, alloimmune mechanisms, characterised by insufficient maternal tolerance to paternal antigens, exacerbate the immunopathogenesis of RPL [11, 17].
Recent progress in immunology, molecular diagnostics, and systems biology has changed how we think about immune-related RPL. The development of high-throughput immunoassays, single-cell RNA sequencing, and omics-based profiling has provided unparalleled knowledge into the immune landscape of the endometrium and placenta [18, 19, 20, 21]. At the same time, new immunomodulatory and targeted treatments, such as low-dose aspirin and heparin, calcineurin inhibitors, biologics, and cellular therapies, have been studied to improve pregnancy outcomes and restore immune tolerance [22, 23, 24, 25]. However, clinical translation continues to be impeded by inconsistent diagnostic criteria, heterogeneous study populations, and an absence of tailored therapeutic strategies [3, 23, 26].
Although several reviews have discussed RPL, many of these publications largely reiterate well-established immunological concepts and do not fully incorporate recent technological advances or provide a cohesive framework that links basic science with clinical practice. This review seeks to address these gaps by offering several new perspectives. It introduces a precision-based approach to immunotherapy for immune-related RPL, connecting detailed immune-endotyping with individualised treatment strategies. It also brings together recent findings from single-cell transcriptomics, spatial immune profiling, and multi-omics studies to highlight the dynamic and complex immune environment at the maternal–fetal interface. In addition, this review explores the potential contribution of artificial intelligence (AI) and computational modelling to diagnostic stratification and treatment prediction—topics that have received little attention in previous reviews. By integrating these advanced concepts, this review aims to bridge the divide between immunological research and clinical application, and to outline a forward-looking path toward personalised diagnostic and therapeutic approaches.
This narrative review was designed to synthesise and contextualise current evidence on the immunopathogenesis, diagnostic evaluation, and therapeutic strategies for immune-related RPL. Although the structure partly follows elements of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 framework, the objective of a narrative review differs from that of a systematic review in that it emphasises conceptual integration rather than quantitative meta-analysis. The following subsections provide a detailed description of the search methodology, selection criteria, and evidence-handling approach.
A comprehensive search was carried out using PubMed, Web of Science, Scopus, and Embase, covering publications from January 2005 to September 2025. The strategy combined controlled vocabulary with free-text terms to ensure the search captured both foundational and emerging research. Key phrases included “recurrent pregnancy loss” and “recurrent miscarriage”, which were paired with immunology-related terms such as “immune dysregulation”, “natural killer cells”, “regulatory T cells”, “Th17”, “cytokines”, and “macrophages”.
To reflect the expanding landscape of reproductive immunology, the search also incorporated terms relating to autoimmune and alloimmune processes, antiphospholipid syndrome, maternal–fetal immune tolerance, biomarkers, molecular diagnostics, immunotherapies, precision medicine, and high-dimensional omics technologies. To ensure completeness, the reference lists of relevant articles and existing reviews were also manually screened for studies that did not appear in the initial database searches.
Studies were included if they provided clinical, mechanistic, diagnostic, or therapeutic insights into immune-related RPL. This encompassed original research, systematic and narrative reviews, and clinical guidelines. Preference was given to work involving females diagnosed with RPL or unexplained recurrent miscarriage and to studies that explored immune pathways, biomarkers, or immunomodulatory interventions.
Studies that did not contribute directly to the immunological understanding of RPL were not incorporated. This included case reports with very small sample sizes, studies limited to animal or in vitro systems without clear clinical relevance, and non-peer-reviewed materials such as conference abstracts or editorials. Articles focused exclusively on chromosomal, anatomical, or endocrine causes of pregnancy loss were also excluded, as these fall outside the scope of this review.
Because this work is a narrative review, its methodological priorities differ from those of a systematic review. The purpose was not to produce a quantitative synthesis or pooled effect estimate, but to offer an integrated perspective on how immune mechanisms, diagnostics, and treatments intersect in the context of RPL. As such, a formal risk-of-bias assessment, although valuable for systematic evaluations, was not applied. Instead, the review’s credibility rests on selecting studies that clearly describe their methods, report clinically meaningful outcomes, and contribute directly to understanding immune-related RPL.
To reduce selection bias, two reviewers independently screened titles and abstracts retrieved from the search. Articles that appeared relevant were examined in full, and discrepancies were resolved through discussion. When needed, a third reviewer helped reach a consensus. This process ensured that the included studies represented a balanced and coherent body of evidence.
The literature search initially found 250 articles. After screening and a full-text review, 112 publications met the criteria for inclusion. Of these, 70 were original research studies, and 42 were either review articles or clinical guidelines. The chosen literature collectively addressed four primary focal areas: the immunopathological mechanisms of RPL, advancements in the development of diagnoses and biomarkers, established and novel immunotherapeutic strategies, and the amalgamation of precision medicine with multidisciplinary management approaches.
Fig. 1 is a schematic summary of the literature selection process. It is based on the PRISMA flow diagram and shows the stages of identification, screening, and inclusion.
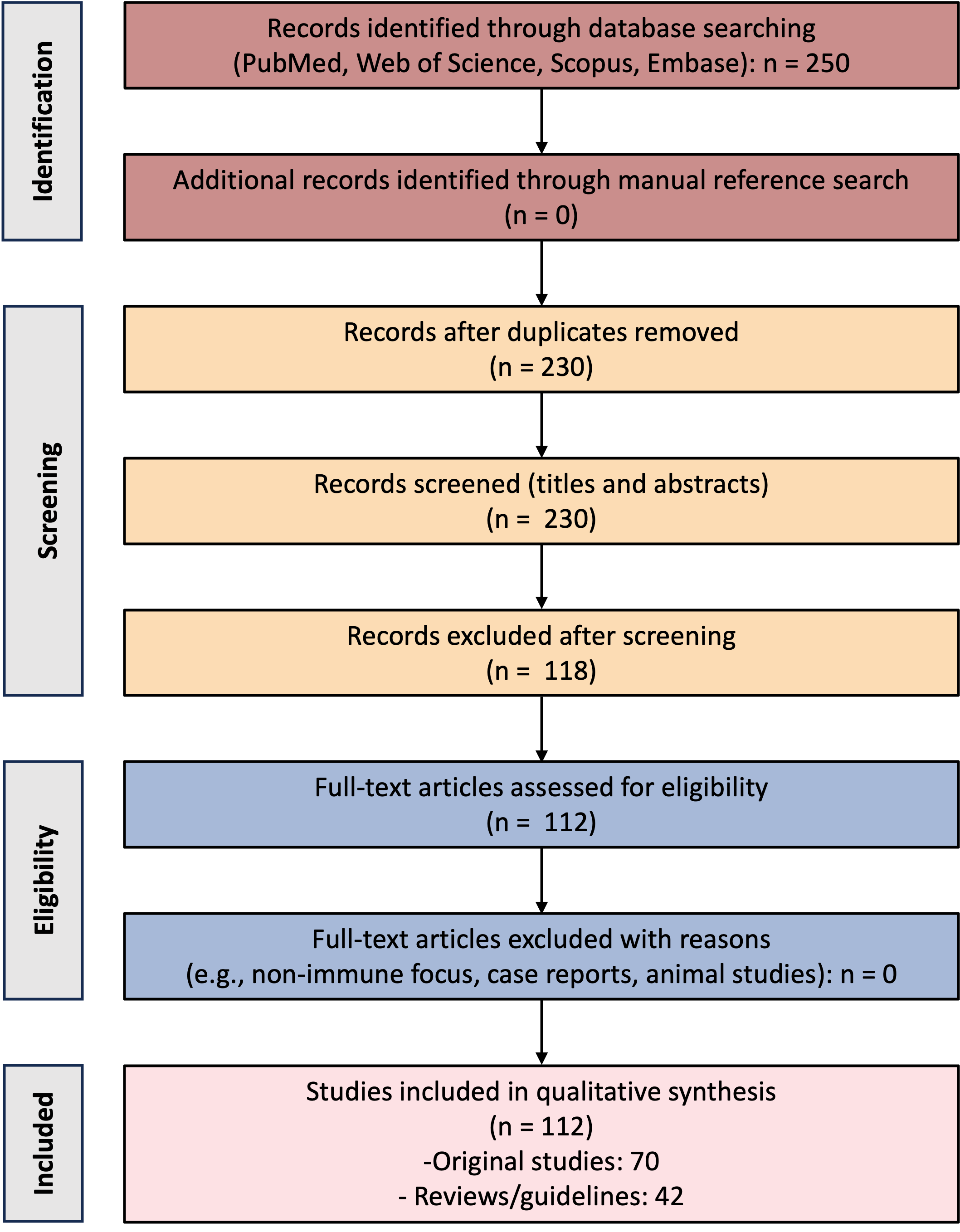 Fig. 1.
Fig. 1.
PRISMA 2020-based flow diagram illustrating the identification, screening, and inclusion of studies published between January 2005 and September 2025. A total of 112 eligible publications were included, comprising 70 original studies and 42 reviews or clinical guidelines. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Pregnancy represents a unique immunological state in which the maternal immune system must accept a semi-allogeneic fetus while still retaining the ability to defend against infection [27, 28]. When this delicate balance is disturbed, the consequences can include failure of implantation, placental dysfunction or RPL [29]. Immune-related RPL arises when autoimmune or alloimmune disturbances interfere with maternal–fetal immune tolerance, triggering abnormal immune-cell activity, cytokine imbalance and disrupted molecular signalling at the maternal–fetal interface [9, 30, 31].
Maternal–fetal immune tolerance depends on a closely coordinated network of decidual immune cells, such as uterine natural killer (uNK) cells, macrophages, dendritic cells and regulatory T cells working in concert with signals released by trophoblasts. Early pregnancy naturally involves a shift in immune tone: a short-lived inflammatory phase helps the embryo implant, after which the environment moves towards a calmer, tolerance-driven state that supports healthy fetal development. In females with RPL, this balance can be disrupted in several ways. One of the most commonly observed problems is increased activity of uNK cells, often accompanied by mismatches in NK receptor-ligand interactions, particularly killer cell immunoglobulin-like receptor (KIR)-human leukocyte antigen C (HLA-C) combinations. These disturbances can hinder trophoblast invasion and may contribute to early rejection of the embryo. Recent studies have also noted lower expression of receptors such as KIR2DL1/S1 and LILRB1 on uNK cells in females experiencing reproductive failure, suggesting alterations in how these cells help regulate local immunity [18].
Disturbances in T-cell balance are another recurrent issue. A higher Th17/Treg
ratio, indicating a tilt towards inflammation, has been described in RPL,
accompanied by an increased release of interleukin-17 (IL-17), tumor necrosis factor-
Together, these findings show that disturbances in maternal–fetal immune tolerance can arise from different immunological pathways. Some abnormalities, such as heightened Th17 activity, systemic cytokine shifts, and autoantibody formation, reflect autoimmune mechanisms, in which the maternal immune system becomes reactive to its own tissues. Others, such as impaired Treg induction in response to paternal antigens, reduced HLA-G signalling and altered KIR-HLA-C interactions, reflect alloimmune mechanisms, where the mother fails to establish tolerance toward the semi-allogeneic fetus. These distinct pathways form the foundation for the two major categories of immune-related RPL discussed below.
Autoimmune disorders are among the most significant causes of immune-related
RPL. In these cases, circulating autoantibodies and autoreactive immune cells
attack self-antigens in placental and decidual tissues, causing thrombosis,
complement activation, and inflammatory damage that impede fetal development
[14, 34]. APS is still the most common autoimmune cause of RPL. Antiphospholipid
antibodies, such as anticardiolipin, lupus anticoagulant, and
anti-
Autoimmune-mediated RPL signifies a condition of chronic systemic immune activation, characterised by the convergence of pathogenic antibodies, complement cascades, and localised decidual immune dysfunction, which collectively hinders placental development.
Alloimmune RPL occurs due to compromised maternal tolerance to paternal or fetal antigens, rather than from self-reactivity [46]. In normal physiological states, exposure to paternal antigens leads to maternal immunological adaptation, which is characterised by the generation of anti-paternal blocking antibodies and the formation of tolerogenic Treg cells that preserve feto-maternal immune balance [47, 48]. In some females, this adaptive process fails, leading the mother’s immune system to recognise the fetal tissues as alien and initiate immune-mediated rejection. The absence of anti-paternal blocking antibodies, an increase in cytotoxic T-lymphocyte responsiveness to paternal antigens, and a reduction in decidual Treg cell recruitment all indicate immunological dysfunction. In addition, there is diminished HLA-G-mediated signalling between trophoblasts and maternal immune cells, which influences local immunological tolerance at the implantation site [49]. Initial studies on lymphocyte immunotherapy utilising paternal or third-party leukocytes to re-establish tolerance yielded inconclusive outcomes in subsequent randomised controlled trials. Consequently, these interventions are presently not advised outside of research settings [50, 51]. Nonetheless, progress in reproductive immunology and antigen-specific tolerance strategies represents the potential for the formulation of future targeted therapies for alloimmune RPL.
Immune-related RPL is a multifactorial condition influenced by the interaction of autoimmune and alloimmune mechanisms, cellular immune imbalance, and cytokine dysregulation. Fig. 2 schematically summarises these overlapping immunopathological pathways at the maternal–fetal interface. Understanding these mechanisms provides the biological rationale for immunological testing and the development of targeted therapies described in the following sections.
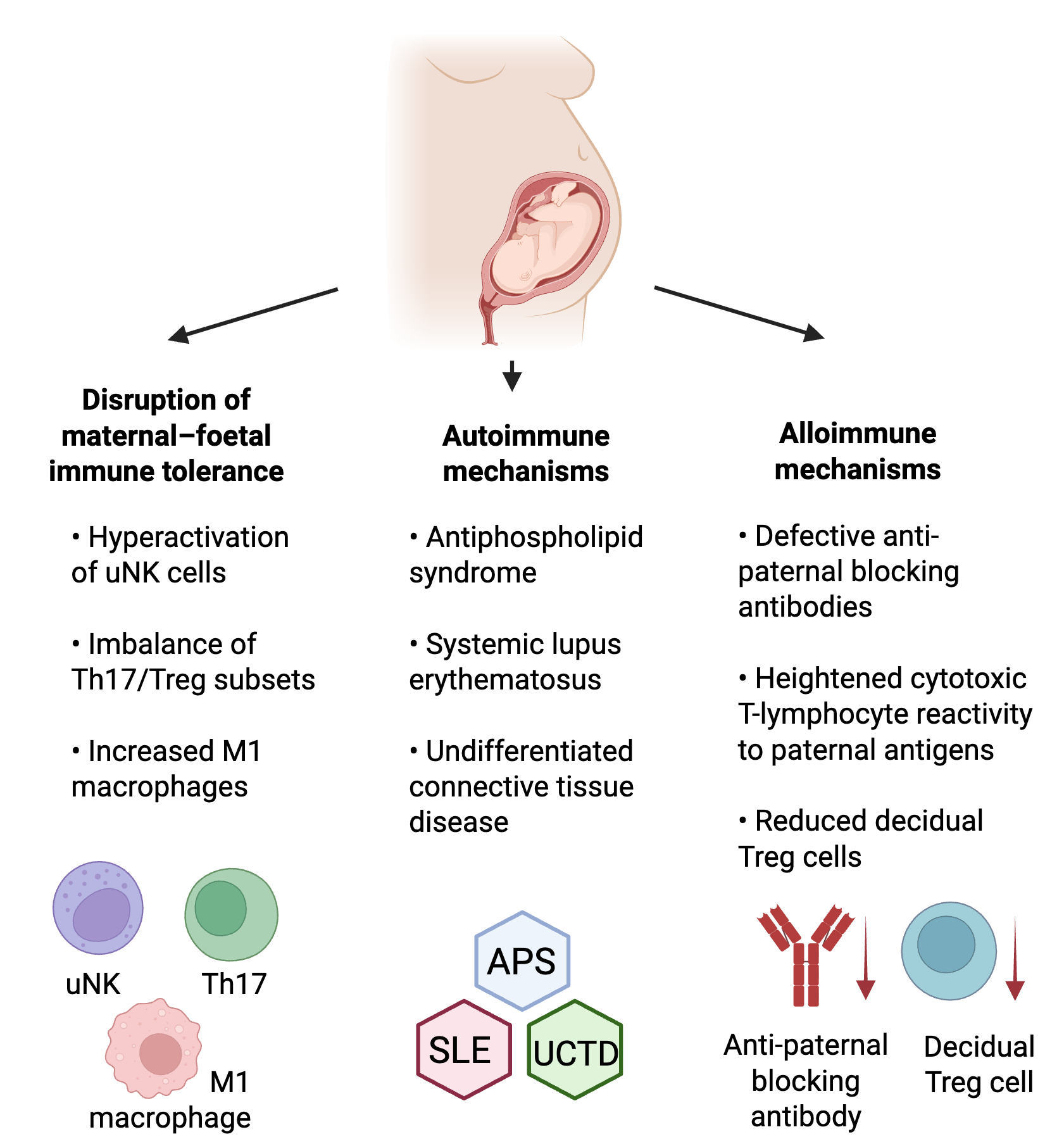 Fig. 2.
Fig. 2.
Overview of key immune pathways involved in recurrent pregnancy loss (RPL). This figure highlights how different types of immune imbalance can contribute to RPL. Disturbed maternal–fetal tolerance, such as overactive NK cells or an imbalance between Th17 and Treg cells, may interfere with healthy implantation. Autoimmune conditions like APS, SLE, and UCTD can cause harmful autoantibodies and inflammation that affect placental function. Alloimmune mechanisms arise when the mother has difficulty developing tolerance to paternal antigens, often linked to reduced HLA-G expression or altered KIR-HLA-C signalling. Together, these pathways show how both self-directed and tolerance-related immune disruptions can affect early pregnancy. Created in https://www.biorender.com/. NK, natural killer; APS, antiphospholipid syndrome; SLE, systemic lupus erythematosus; UCTD, undifferentiated connective tissue disease; HLA-G, human leukocyte antigen G; KIR-HLA-C, killer-cell immunoglobulin-like receptor–human leukocyte antigen C.
Diagnosing immune-related RPL is often challenging, largely because the underlying immune abnormalities can vary widely from one patient to another, and because no single international standard for diagnosis exists. Traditionally, evaluation has centered on detecting autoantibodies and performing routine coagulation tests. Although these remain important, recent advances in molecular immunology and various omics technologies have expanded what clinicians can look for and how they interpret immune dysfunction. Bringing together clinical findings with detailed immunological and molecular data now plays a crucial role in identifying each patient’s specific immune profile and helps to guide more personalised treatment decisions.
According to the European Society of Human Reproduction and Embryology (ESHRE) and the American Society for Reproductive Medicine (ASRM), the diagnostic work-up for RPL should begin with a comprehensive clinical assessment. This typically includes a detailed obstetric history, parental karyotyping, uterine imaging and evaluation for endocrine disorders and thrombophilia [3, 52]. Once structural, genetic and hormonal causes have been ruled out, clinicians can then turn their attention to potential immune-related factors.
Current clinical frameworks generally divide immune-related RPL into two main categories. The first is autoimmune-associated RPL, which is marked by disease-specific autoantibodies or established autoimmune conditions such as APS, SLE or UCTD. The second category is alloimmune-associated RPL, where the maternal immune system mounts an atypical response to paternal or fetal antigens despite the absence of autoimmunity [53]. This classification helps guide which tests are most appropriate and supports the identification of patients who may benefit from immunomodulatory treatment.
To help contextualise the range of diagnostic tools discussed in this review, we provide an overview of the main assays currently used to investigate immune-related RPL. As shown in Table 1 (Ref. [54, 55, 56, 57, 58, 59, 60, 61, 62]), these methods differ considerably in their clinical relevance, degree of validation, and reproducibility. Presenting them side by side highlights not only their potential contribution to understanding immune dysfunction but also the important limitations that restrict many of these tests to research settings rather than routine clinical practice.
| Diagnostic category | Key biomarkers/assays | Clinical implication | Limitations | Clinical status (ESHRE/ASRM) | References |
| Autoantibody testing | Antiphospholipid panel, ANA, ENA, anti-TPO | Identification of autoimmune RPL (APS, SLE, UCTD) | False positives; low specificity for outcome prediction | Limited prognostic value | [54, 55, 56] |
| Cellular assays | NK-cell cytotoxicity, Th1/Th2 and Th17/Treg balance | Assess cellular immune tolerance status | Lack of standardisation; inter-laboratory variability | Lack of standardisation; high variability | [57, 58] |
| Cytokine profiling | TNF- |
Reflects systemic or local inflammatory state | Influenced by menstrual phase and sampling timing | Influenced by cycle timing; poor reproducibility | [59, 60] |
| Omics-based diagnostics | scRNA-seq, proteomics, metabolomics, spatial transcriptomics | Discovery of novel biomarkers; immune-endotyping | High cost; limited clinical validation | High cost; limited clinical validation | [61, 62] |
ANA, antinuclear antibodies; ENA, extractable nuclear antigens; anti-TPO,
anti-thyroid peroxidase antibody; Th1, T helper 1 cell; TNF-
Testing for autoantibodies is still the main method to diagnose immune-related
RPL. The most clinically validated assays include the antiphospholipid antibody
panel, which consists of lupus anticoagulant, anticardiolipin IgG/IgM, and
anti-
A recent study indicates that multi-autoantibody profiling improves diagnostic sensitivity and facilitates the early identification of subclinical autoimmune conditions that might otherwise be missed in females experiencing unexplained RPL [54]. As a result, the tests for antiphospholipid and ANA profiles are suggested for patients with recurrent miscarriage of unknown cause.
Cellular immune assays provide additional insight into maternal immune imbalance, although their clinical utility remains limited. Tests such as peripheral or uterine NK-cell cytotoxicity, assessment of CD56bright/CD16dim subsets [59, 69], and flow-cytometric analysis of Th1/Th2 or Th17/Treg ratios, have been used in research to explore immune tolerance. Elevated NK-cell activity or a shift toward pro-inflammatory T-cell profiles has been associated with impaired implantation and pregnancy loss in some studies.
Cytokine profiling represents another potential tool. Multiplex assays allow
simultaneous measurement of cytokines such as TNF-
Importantly, major professional societies, including ESHRE and ASRM, do not recommend NK-cell testing, Th1/Th2 ratios, Th17/Treg profiling, or cytokine panels as part of routine evaluation for RPL. These assays should therefore be considered research tools rather than established diagnostics, and any results should be interpreted with caution.
Recent advancements in molecular diagnostics have markedly improved the understanding of the endometrial immune environment in RPL. Transcriptomic studies, particularly single-cell RNA sequencing (scRNA-seq), have revealed distinct transcriptional signatures in decidual immune cells, marked by increased interferon pathway activity and diminished regulatory T-cell signalling [61]. Proteomic and immuno-peptidomimetic studies have identified novel autoantigens and immune-related proteins linked to implantation failure and early pregnancy loss [62]. Metabolomic profiling has identified alterations in lipid and amino acid metabolism within the decidua that affect immunological tolerance and inflammatory homeostasis [71]. In addition, cutting-edge spatial multi-omics imaging methods now combine transcript and protein localisation to precisely characterise immune cell interactions at the maternal–fetal interface [72]. These technologies offer insight into potential biomarkers, although further validation is required.
Although techniques such as single-cell RNA sequencing and spatial transcriptomics have deepened our understanding of the maternal–fetal immune environment, these methods are still research tools rather than clinical tests. They provide valuable biological insight, but they remain expensive, technically complex, and are not validated for routine use. Their future promise is clear, but for now, their role is exploratory, not diagnostic.
Recent research indicates that the amalgamation of clinical parameters,
serological biomarkers, and high-throughput molecular signatures, when evaluated
through sophisticated bioinformatics and machine-learning algorithms, offers a
more holistic methodology for diagnosing immune-related RPL [73, 74]. Several
models have a high predictive accuracy (e.g., AUC
Advancements in diagnostics have redefined immune-related RPL from a serological disorder to a complex, multifaceted immune condition. Although traditional autoantibody testing is still the clinical standard, combining cellular assays with omics-based technologies could lead to earlier detection, a better understanding of the pathophysiology, and more accurate patient stratification. These new methods of diagnosing diseases are the basis for precision immunotherapy, which is discussed in the next section.
The management of immune-related RPL seeks to re-establish maternal–fetal immune tolerance, avert thrombosis, and foster a conducive endometrial environment for successful embryo implantation [22]. Because RPL has many causes, there is no one-size-fits-all treatment plan. Current clinical practice integrates empirical immunosuppressive, anticoagulant, and immunomodulatory approaches tailored to individual immune profiles. The rapid advancement of molecular immunology has led to the emergence of innovative biologics and cellular therapies that may enhance pregnancy outcomes.
For patients with obstetric APS or other hypercoagulable conditions, the standard treatment is still a combination of low-dose aspirin (75–100 mg daily) and low-molecular-weight heparin (LMWH). LDA inhibits platelet aggregation, and LMWH inhibits blood clots from forming and has mild anti-inflammatory effects on the maternal–fetal interface. Recent studies demonstrated that the LDA + LMWH regimen significantly enhances live-birth rates in comparison to LDA alone [76, 77, 78]. A recent review reaffirms that this combination continues to be the fundamental approach in the management of obstetric APS [79].
Low-dose prednisone or prednisolone (
HCQ has many different effects on the immune system, such as stopping Toll-like receptor signalling, activating the complement system, and causing oxidative stress. It has shown safety and effectiveness in the treatment of APS or SLE in females, and it has been accepted as a supplementary treatment to standard therapy. Recent studies indicate that HCQ enhances endothelial function and may decrease the recurrence of pregnancy loss in females with antiphospholipid antibodies [82, 83, 84].
IVIG has been reported to modulate immune responses by neutralising autoantibodies. It inhibits Fc-receptor–mediated phagocytosis and promotes Treg-cell activation. Although inconsistent outcomes were observed in early studies, meta-analyses have indicated a potential benefit for females with secondary RPL or proven immune dysfunction. Current guidelines recommend considering IVIG only for selected patients after exclusion of other etiologies and failure of first-line therapy [85, 86, 87].
In addition to its hormonal function, progesterone possesses immunoregulatory properties by enhancing Th2 cytokine dominance and suppressing NK-cell cytotoxicity. Starting progesterone in the luteal phase or right after ovulation has been linked to higher live-birth rates in idiopathic RPL [23, 88, 89].
With the advancements in knowledge about maternal–fetal immune-signalling pathways, more research is now exploring the biological and targeted therapies to modulate effectors implicated in RPL. One of these are the calcineurin inhibitors (CNIs) such as Tacrolimus and Cyclosporine, which suppress T-cell activation and down-regulate cytokine release via inhibition of the calcineurin NFAT (nuclear factor of activated T-cells) pathway. A recent randomised controlled trial of tacrolimus in patients with refractory RPL and elevated Th1/Th2 ratios showed that the tacrolimus group had a much higher live birth rate (80%) than the placebo group (63.5%; OR: 2.30, 95% CI: 1.10–4.81; p = 0.03) [25]. However, these findings remain preliminary, and these therapies are not recommended in the guidelines, and larger trials are needed before they can be considered for routine care.
Anti-TNF-
Granulocyte Colony-Stimulating Factor (G-CSF) is another immunomodulatory strategy, which may influence immune tolerance by increasing Tregs and improving endometrial receptivity via angiogenesis. However, a multicenter randomised trial of subcutaneous G-CSF in females with unexplained RPL found no significant improvement in live birth rates (59.2% vs. 64.9% in placebo; RR: 0.9, 95% CI: 0.7–1.2; p = 0.48) [91]. A recent meta-analysis, however, indicated that intra-uterine G-CSF, as opposed to subcutaneous G-CSF, may diminish the risk of miscarriage (RR: 0.48, 95% CI: 0.27–0.86; p = 0.01) [92].
Intravenous lipid emulsion (Intralipid) infusions have been administered to patients exhibiting elevated NK cell activity, purportedly by reducing NK cell cytotoxicity and altering cytokine secretion. A systematic review, however, revealed a lack of high-quality trials and concluded that the evidence is insufficient to support routine usage [93, 94].
These targeted therapies epitomise the shift towards precision immunomodulation in RPL. However, standardised protocols, particularly regarding patient selection criteria, dosing regimens, timing of initiation and large-scale randomised controlled trials, are still required to establish both safety and efficacy in routine clinical practice.
Adoptive transfer of Tregs is gaining attention as a means to restore maternal–fetal immune tolerance. Preclinical models demonstrate that maternal Treg insufficiency is associated with fetal loss, and that transfer of ex vivo-expanded Tregs can prevent miscarriage. For example, in a murine model of intra-uterine fetal death, adoptive transfer of pregnancy-induced Tregs reduced fetal loss and favourably altered uterine macrophage subsets [95]. While human studies are at an early phase and face ethical and technical hurdles, the feasibility of Treg-based therapy in the context of RPL is under active investigation.
Mesenchymal stem cell (MSC) therapy also holds promise, owing to its dual
immunosuppressive and tissue-repair capacities. MSCs secrete IL-10, transforming
growth factor-
Immune-checkpoint modulation represents another frontier: the expression of programmed cell death protein-1 (PD-1)/PD-L1 as well as cytotoxic T-lymphocyte associated antigen-4 (CTLA-4) pathways is recognised as essential for the establishment of maternal immune tolerance [98]. Investigational therapies aimed at enhancing these tolerance-inducing signals may represent future options. Conversely, the reproductive side effects of immune checkpoint inhibitors used in oncology emphasise the need for caution in translation to reproductive medicine.
Collectively, these therapeutic pathways reflect a shift towards precision immunomodulation in the management of RPL. Nonetheless, standardised selection criteria, optimal dosing and timing, and large-scale randomised controlled trials are required prior to routine clinical implementation.
Given the multifactorial nature of immune-related RPL, combination therapy is frequently required. For example, the dual regimen of LDA plus LMWH has been associated with markedly higher live-birth rates in cohorts of females with APS or persistent antiphospholipid antibodies (aPL) [99]. Moreover, in females who continued to suffer obstetric complications despite standard antithrombotic therapy, the addition of HCQ to LDA + LMWH resulted in an 83.8% live-birth rate in a selected cohort [99]. In another multicenter study, females with refractory APS treated with HCQ in addition to aspirin and heparin, achieved markedly improved outcomes compared with standard therapy alone [83].
These cases show that it is necessary to customise integrative treatment plans for patients and consider immunological test results, past pregnancies, and how well each patient can handle them. Furthermore, standard application of therapies devoid of validation in comprehensive randomised trials must be avoided, and each intervention should be supported by unique evidence-based justification.
Because immune-related RPL can arise from many different underlying etiologies, a single universal treatment approach is rarely effective. Instead, care is increasingly guided by a precision-medicine framework, in which therapy is tailored to the patient’s individual immune profile. This requires clinicians to consider the clinical history along with serological findings and molecular data. Immune phenotyping plays a central role in this process. By using antibody panels and immune-cell profiling, patients can be broadly grouped into autoimmune-dominant, alloimmune-dominant or mixed patterns of disease [31]. Biomarkers such as the Th1/Th2 ratio or NK-cell activity, can then help determine the intensity of the level of treatment needed [100]. There is also emerging evidence that carrying out repeated immune assessments before conception and during early pregnancy can offer valuable guidance, allowing treatment to be adjusted as the immune environment changes [101]. Given the complexity of immune interactions in reproduction, multidisciplinary considerations are discussed further in section 6.5. Looking ahead, artificial-intelligence models and integrated omics technologies are expected to improve personalisation even further by linking specific immune signatures with predicted treatment responses [100].
While such individualised regimens hold great promise, they must be founded on robust immunological evaluation and applied only when interventions are justified by evidence. Unvalidated therapies should be avoided in routine therapy as well.
In summary, immunotherapy for RPL has evolved from steroid administration to targeted and mechanism-based therapies. Standard treatments such as LDA, LMWH, and glucocorticoids remain the primary choice for autoimmune-associated RPL. Biologics and cellular therapies may also be options for cases that don’t respond to standard treatments or are alloimmune. Precision immunomodulation is the future of RPL management, and molecular diagnostics and cross-field collaboration are helping to make it happen.
To provide a clearer overview of how current treatments align with clinical evidence, we summarise the major therapeutic options for immune-related RPL in Table 2. This table distinguishes interventions supported by established guidelines from those that remain experimental or based on emerging data.
| Therapeutic category | Specific intervention | Evidence summary | Guideline status (ESHRE/ASRM) | Comment |
| Autoimmune-related, guideline-supported treatments | Low-dose aspirin + heparin (for APS) | Strong evidence for reducing pregnancy loss in APS; multiple RCTs and meta-analyses | Recommended | Standard of care for APS-associated RPL |
| Hydroxychloroquine (selected autoimmune profiles, SLE/UCTD) | Increasing observational evidence for reducing flares & improving pregnancy outcomes | Conditionally recommended in autoimmune disease | Not guideline-endorsed for unexplained RPL | |
| Thyroid management (e.g., levothyroxine in overt hypothyroidism) | Clear maternal–fetal benefit; well-established safety | Recommended | Applies to autoimmune thyroid disease with dysfunction | |
| Immunomodulatory therapies with emerging or limited evidence | Prednisone/glucocorticoids | Some benefit in autoimmune contexts; limited value in unexplained RPL; risks at higher doses | Not routinely recommended | Use with caution; evidence inconsistent |
| Tacrolimus/calcineurin inhibitors | Small observational studies suggest benefit in selected inflammatory phenotypes | Experimental | Requires large controlled trials | |
| Anti-TNF therapy (e.g., adalimumab, etanercept) | Mixed evidence; primarily case series and small trials | Not recommended | Considered research-only | |
| G-CSF | Conflicting RCT results; no consistent improvement in live-birth rate | Not recommended | Evidence graded as low to very low | |
| Intralipid | Heterogeneous findings; no confirmed clinical benefit | Not recommended | Research-only; not endorsed by ESHRE or ASRM | |
| Investigational/Research-stage approaches | MSC therapy | Early-phase data; limited safety and efficacy evidence | Experimental | Requires rigorous clinical trials |
| Intravenous immunoglobulin (IVIG) | Mixed evidence; high cost; inconsistent outcomes | Not recommended for routine use | Potential benefit in selected immune profiles still under investigation | |
| Omics-guided therapy/immune-endo-typing | Promising for future precision medicine | Research-only | Not validated for clinical decisions | |
| AI-based treatment stratification | Very early stage; no clinical application yet | Not applicable | Conceptual; requires validation |
Although the understanding of RPL immunopathogenesis has achieved significant advances, the clinical translation of immunological knowledge into effective diagnostic and therapeutic strategies remains incomplete. The complexity of maternal–fetal immune interactions and the patients’ variability are complicating standardised management. Future research should focus on mechanistic clarification, biomarker validation, precision medicine, and interdisciplinary collaboration to improve patient outcomes.
It is important to find reliable biomarkers for improving diagnostic precision and therapeutic performance in immune-related RPL. Emerging technologies such as single-cell RNA sequencing (scRNA-seq), spatial transcriptomics, and proteomic profiling are enabling high-resolution mapping of immune-cell states within the decidua and placenta. These techniques have discovered many potential diagnostic candidates, including altered expression of HLA-G, PD-L1, and cytokines such as IL-10 and IL-17 that reflect local immune tolerance or inflammation.
In addition, integrative immune signatures combining autoantibody panels, cytokine ratios, and immune-cell phenotypes have the potential to predict the risk of recurrence and treatment outcomes. However, clinical translation needs extensive multicenter validation to confirm sensitivity, specificity, and predictive value. Future diagnostic systems should strive to transform complex immune signatures into clinically applied tests, to promote the early identification of RPL patients who would benefit from immunotherapy.
The application of precision medicine in RPL seeks to tailor interventions based on individual immunological and genetic profiles. By integrating multi-omics data (genomic, transcriptomic, proteomic, and metabolomic) with clinical parameters, it becomes possible to delineate patient-specific immune endotypes. AI and machine-learning models have also been explored as tools to analyse complex datasets and identify immunological signatures associated with pregnancy outcomes. Early studies suggest that AI may one day help stratify patients or predict treatment responsiveness. However, these applications remain experimental, and their accuracy, reproducibility, and generalizability must be rigorously established. At present, AI should be considered a research instrument rather than a clinically deployable tool.
Taken together, precision medicine and AI point toward an exciting future for understanding immune-related RPL. However, these tools are still in the early stages of development. Before they can play a role in everyday clinical care, we will need stronger evidence, clearer standards, and agreement on how they should fit within existing diagnostic and treatment pathways. For now, their main value is in helping researchers make sense of complex data and generate new ideas, rather than directly guiding patient care.
Immunomodulatory treatments such as glucocorticoids, heparin and intravenous
immunoglobulin have shown clinical benefit in selected patients, but important
safety questions remain, particularly with newer biologic and cell-based
therapies. When immunotherapy is used during pregnancy, it must be both effective
and safe for the mother and the developing fetus, as excessive immune
suppression can increase susceptibility to infection or potentially interfere
with fetal development. For this reason, future research needs to focus on
optimising dosage, refining the timing of treatment, and establishing long-term
reproductive safety, especially for emerging options such as calcineurin
inhibitors, anti-TNF-
Management of immune-related RPL still differs markedly across countries and even between individual clinics. Major international guidelines, including those from ESHRE, ASRM and the Chinese Medical Association, often disagree on which diagnostic thresholds to use, which immune tests to include, and which treatments to recommend. As a result, females may receive very different evaluations and therapies depending on where they seek care. Moving forward, greater alignment of diagnostic criteria and treatment pathways will be necessary. This will require coordinated international efforts, supported by high-quality meta-analyses and consensus recommendations. Building shared registries and adopting standardised immunological assays would also strengthen data comparability and allow emerging biomarkers and therapies to be validated across diverse populations, ultimately improving care for patients with RPL.
Because RPL arises from several overlapping biological and emotional factors, it is rarely managed well by one clinician working alone. In practice, females benefit far more when different specialists, those in reproductive immunology, rheumatology, haematology, genetics and psychological support work together and share responsibility for care. This team-based approach makes it easier to address the many issues that can appear at the same time, whether immune disturbances, clotting problems, hormonal irregularities or the emotional strain that follows repeated losses. The psychological impact should not be underestimated; many females describe feelings of fear, uncertainty and exhaustion, and these can develop into anxiety, depression or tension within their relationships. Providing space for counselling, offering practical ways to manage stress, and giving clear information about treatment can help patients feel less alone and more in control, which in turn can improve adherence and overall wellbeing. Looking ahead, it would be valuable for research to explore more deeply how psychological stress interacts with the immune system, especially since several studies now suggest that stress can shift cytokine activity and may influence pregnancy outcomes in ways we do not yet fully understand.
Immune-related RPL remains a complex and emotionally difficult condition, but advances in reproductive immunology are beginning to clarify why some pregnancies fail repeatedly despite appearing healthy. Disturbances in maternal–fetal immune tolerance, whether driven by autoimmune disease, inadequate tolerance to paternal antigens, or imbalances in key immune-cell populations, represent an important subset of RPL that is increasingly recognised in clinical practice.
Diagnostic approaches are evolving from simple autoantibody testing toward more detailed immune profiling and exploratory omics technologies. These tools are improving our ability to identify meaningful immune patterns, although many remain research methods rather than routine clinical tests. Therapeutic strategies are also expanding, with established treatments such as low-dose aspirin, heparin, glucocorticoids and hydroxychloroquine supported by emerging options such as calcineurin inhibitors, biologics, and cell-based therapies. While these newer approaches show promise, their use should remain cautious and evidence-guided until validated by large, well-designed studies.
Looking forward, the integration of precision medicine, immune-endo-typing, and AI has the potential to reshape how RPL is diagnosed and treated. As research continues, collaboration across immunology, obstetrics, rheumatology and mental-health disciplines will be essential to ensure that advances in science translate into meaningful improvements in patient care.
LP, CW, and TW conceived and designed the research study. LP performed literature collection, organization, investigation, and visualization, and developed the methodology and software. LP also managed project administration, supervision, validation, and prepared the original draft of the manuscript. CW and TW acquired funding, supervised the overall project, and provided critical revisions of the manuscript for important intellectual content. PY, YG, QY, WL, and XL contributed to data collection and assisted in the acquisition of research materials. They also participated in reviewing and editing the manuscript for important intellectual content. All authors contributed to the interpretation of the results and the editorial revision of the manuscript. All authors read and approved the final version of the manuscript for publication. Each author has participated sufficiently in the work to take public responsibility for appropriate portions of the content and has agreed to be accountable for all aspects of the work, ensuring that questions related to the accuracy or integrity of any part are properly investigated and resolved.
Not applicable.
We would like to acknowledge the technical contributions of Professor Yumei Wu and Professor Yue He of Beijing Obstetrics and Gynecology Hospital, Capital Medical University. We further acknowledge Dr. Chengwei Jiang of University College London for his valuable academic input.
This research was funded by Wuhai City Science and Technology Project, grant number 2024WHSKJJN20.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


