1 Shandong University of Traditional Chinese Medicine, First School of Clinical Medicine, 250355 Jinan, Shandong, China
2 Hongjitang Pharmaceutical Group Co., Ltd., 250100 Jinan, Shandong, China
3 Department of Traditional Chinese Medicine, Shandong Maternal and Child Health Hospital, 250013 Jinan, Shandong, China
4 Department of Traditional Chinese Medicine, Shandong Provincial Hospital Affiliated to Shandong First Medical University, 250021 Jinan, Shandong, China
Abstract
This review summarizes global advances in antibody-drug conjugate (ADC) research for the treatment of gynecologic cancers and highlights China’s rapidly evolving role in developing these agents, aiming to contextualize their growing impact on future therapeutic standards.
ADCs represent a transformative class of targeted therapies, coupling monoclonal antibodies with potent cytotoxic payloads through linkers, thereby enabling selective drug delivery to tumor cells while minimizing systemic toxicity.
Globally, ADCs such as mirvetuximab soravtansine and tisotumab vedotin have demonstrated significant clinical benefits in platinum-resistant ovarian and recurrent cervical cancers. Parallel progress in other ADCs has expanded therapeutic options across ovarian, endometrial, and cervical cancers. In China, ADC development is accelerating, with several domestically developed agents targeting folate receptor alpha, human epidermal growth factor receptor 2, trophoblast cell surface antigen 2, cadherin-6, nectin-4, B7 homolog 4, and epidermal growth factor receptor/human epidermal growth factor receptor 3 advancing into phase II and III trials. Early results show encouraging efficacy and safety, underscoring China’s emergence as a key contributor to global anticancer innovation. Nevertheless, variability in biomarker assay, regulatory differences, and future challenges related to pricing and equitable access remain important considerations.
ADCs are redefining the treatment landscape for gynecologic cancers. China’s expanding pipeline, supported by domestic innovation and international collaboration, is poised to significantly influence future standards of care. Continued progress in clinical validation, regulatory alignment, and the development of diagnostic and reimbursement infrastructures will be essential to ensure that these novel therapies achieve meaningful and equitable global impact.
Keywords
- antibody-drug conjugates
- gynecologic cancers
- ovarian cancer
- cervical cancer
- endometrial cancer
- clinical trials
- China
An antibody-drug conjugate (ADC) is a targeted anticancer agent that comprises a monoclonal antibody linked via a specialized chemical linker to a potent cytotoxic payload, enabling selective delivery of the latter to tumor cells (Fig. 1) [1]. In gynecological oncology, ADCs have emerged as a promising strategy for improving objective response rates (ORRs), progression-free survival (PFS), and overall survival (OS) in patients with challenging cases (e.g., relapsed or metastatic cancers) whose tumors are resistant to conventional therapies such as platinum-based chemotherapy.
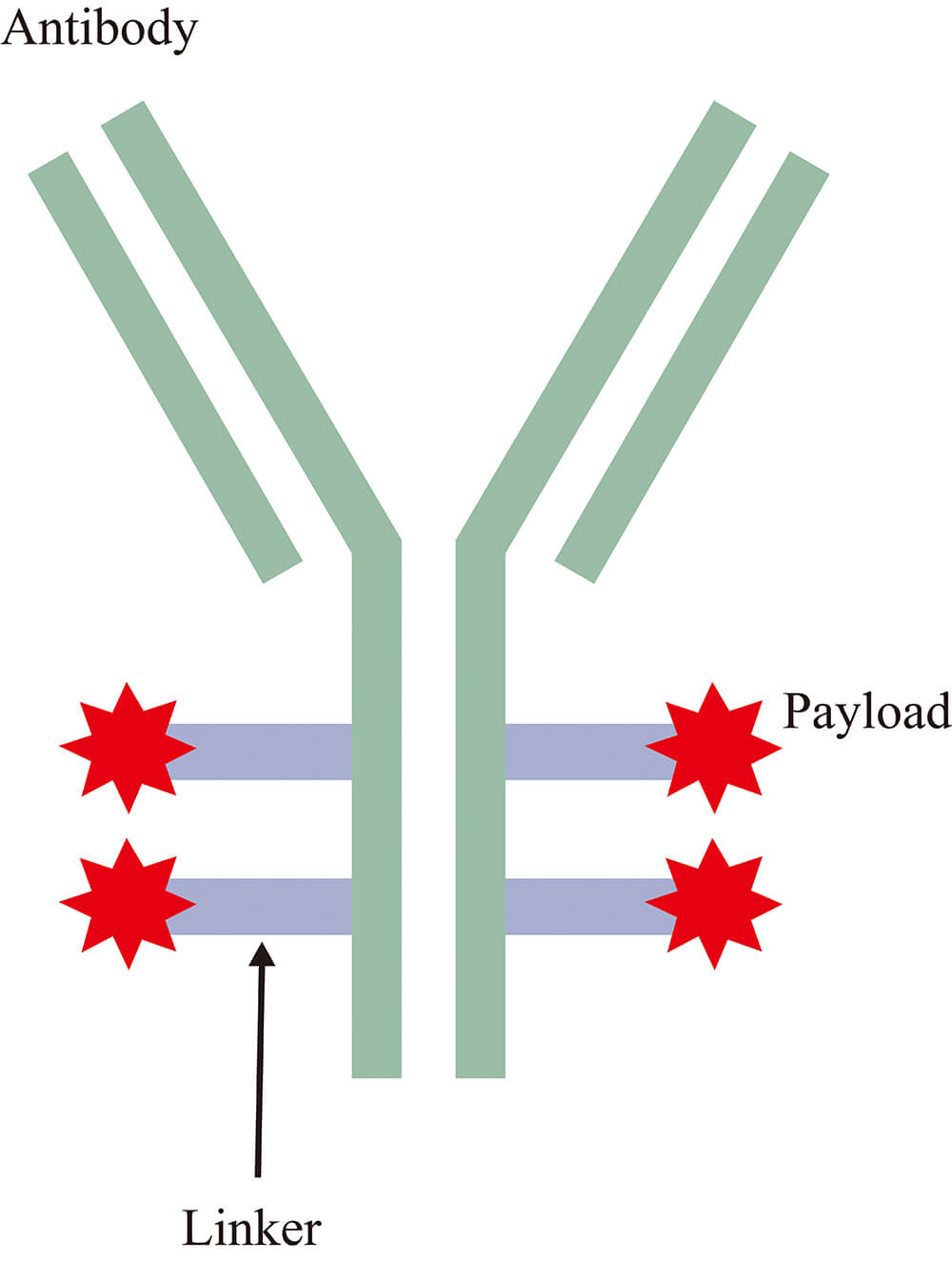 Fig. 1.
Fig. 1.
Schematic illustration of the structural components of an antibody-drug conjugate.
Globally, several ADCs have gained regulatory approval for treating
gynecological malignancies. For example, mirvetuximab soravtansine (MIRV), a
folate receptor alpha (FR
In contrast, China’s regulatory landscape for ADC use in patients with gynecological malignancies remains nascent, with only MIRV approved by the National Medical Products Administration (NMPA) for PROC in November 2024. Despite this, China is swiftly positioning itself as a pivotal contributor to anticancer therapies through a rapid expansion in ADC development; this effort is being driven by substantial investments, abundant patient resources, expedited regulatory pathways (e.g., the NMPA’s 30-day fast-track mechanism), and cost-effective clinical environments [6]. This surge has positioned China as a key player in global oncology research, with several domestically produced ADCs against novel targets like nectin-4 and epidermal growth factor receptor (EGFR)/human epidermal growth factor receptor 3 (HER3) entering late-stage trials.
This comprehensive review describes China’s contribution to the global progress in the research and development of ADCs that target gynecological tumors. Emphasis is placed on current phase III clinical trials for patients with ovarian, cervical, and endometrial cancers being conducted in China and on assessing the future accessibility of these novel therapies for women with gynecologic malignancies in the country.
Although a formal systematic review framework was not applied for this descriptive review, we adopted a structured approach to ensure transparency in literature identification and performed a targeted search of ClinicalTrials.gov and the Chinese Clinical Trial Registry (http://www.chinadrugtrials.org.cn) to identify phase III clinical trials conducted in China involving ADCs used to treat gynecologic malignancies. Disease-related search terms included “ovarian cancer”, “cervical cancer”, “endometrial cancer”, “vulvar cancer”, “vaginal cancer”, and “gestational trophoblastic neoplasia”. Trials were screened for relevance based on cancer type, phase, study design, and availability of ADC-related interventions.
For each ADC that had progressed to phase III trials in China, we conducted additional searches in PubMed and Embase to collect supporting evidence from earlier-phase (I/II) studies. Search terms included combinations of the ADC name, molecular target, and the gynecologic cancer terms listed above. Both peer-reviewed publications and major conference abstracts were considered if they reported efficacy, safety, or mechanistic data relevant to the ADCs being evaluated in China.
Literature and trial information published or updated as of August 31, 2025, were included. Reference lists of key articles were also reviewed to ensure completeness. As this is a narrative review, no Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) flow diagram was generated; however, care was taken to avoid selective reporting by cross-checking trial identifiers and verifying that all ADCs entering phase III investigations in China were acquired.
Twenty-one phase III clinical trials of ADCs in patients with gynecological
malignancies are currently underway in China, reflecting an unprecedented
expansion in late-stage development. These studies span three major cancer types:
endometrial cancer (six trials), cervical cancer (three trials), and ovarian
cancer (12 trials). Moreover, the trials encompass eight distinct molecular
targets, highlighting the diversity of China’s ADC pipeline: HER2 (seven trials);
trophoblast cell surface antigen 2 (TROP-2) (five trials); FR
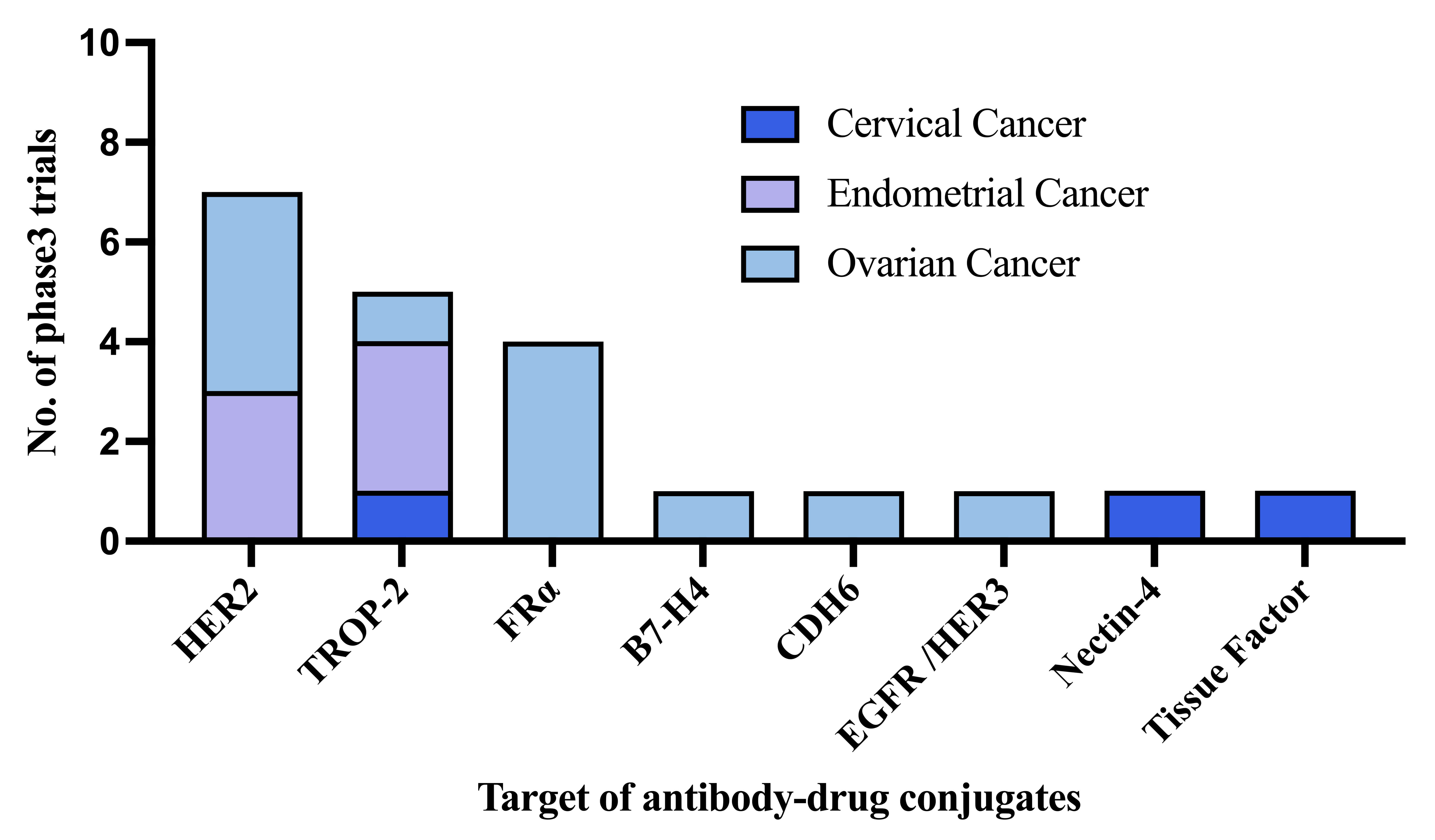 Fig. 2.
Fig. 2.
Landscape of phase III antibody–drug conjugate trials in
patients with gynecologic malignancies in China. HER2, human epidermal growth
factor receptor 2; TROP-2, trophoblast cell surface antigen 2; FR
FR
| Target | ADC | Payload | Linker | Study design and phase | Cancer type/sample size | Key outcomes |
| FR |
Mirvetuximab soravtansine (MIRV) | Tubulin inhibitor DM4 | Cleavable disulfide-containing linker | Randomized controlled Phase III (MIRASOL) | PROC 453 | PFS 5.6 mo; OS 16.5 mo |
| FR |
BAT8006 | Top1 inhibitor Exatecan | Cleavable linker | Single-arm Phase II | PROC 108 | ORR 32.4%; PFS 6.9 mo |
| Tissue factor | Tisotumab vedotin (TV) | Tubulin inhibitor MMAE | Valine–citrulline cleavable linker | Randomized controlled Phase III (InnovaTV-301) | Second-line CC 502 | PFS 4.2 mo; OS 11.5 mo |
| HER2 | Trastuzumab deruxtecan (T-DXd) | Top1 inhibitor deruxtecan | Cleavable GGFG tetrapeptide linker | Single-arm Phase II | OC 40; EC 40; CC 40 | ORR EC 57.5%, CC 50.0%, OC 45.0% |
| HER2 | SHR-A1811 | Top1 inhibitor | Cleavable tetrapeptide linker | Single-arm Phase II | OC 30; EC 23; CC 24 | ORR EC 43.5%, CC 66.7%, OC 66.7% |
| HER2 | JSKN003 | Top1 inhibitor | Cleavable tetrapeptide linker | Single-arm Phase II | OC 46 | ORR 64.4%; PFS 7.1 mo |
| HER2 | IBI354 | Top1 inhibitor: amptothecin derivative | Cleavable linker | Single-arm Phase I | OC 40 | ORR 55.0%; PFS 7.1 mo |
| HER2 | BNT323/DB-1303 | Top1 inhibitor P1003 | Cleavable linker | Single-arm Phase I/II | EC 17 | ORR 58.8%; DCR 94.1% |
| TROP-2 | Sacituzumab tirumotecan (MK-2870) | Top1 inhibitor Belotecan derivative | Cleavable tetrapeptide linker | Single-arm Phase II | EC 44; OC 40 | ORR EC 34.7%, OC 40%; PFS EC 5.7 mo; OC 6.0 mo |
| TROP-2 | Sacituzumab govitecan (SG) | Top1 inhibitor SN-38 | Hydrolysable linker | Single-arm Phase II | CC 40; EC 41 | ORR CC 43–48%, EC 22%; PFS CC 7.4 mo; EC 4.8 mo |
| TROP-2 | SHR-A1921 | Top1 inhibitor | Cleavable tetrapeptide linker | Single-arm Phase I | OC 43 | ORR 48.8%; DCR 97.7%; PFS 7.2 mo |
| CDH6 | Raludotatug deruxtecan (R-DXd) | Top1 inhibitor DXd | Cleavable linker | Single-arm Phase I | OC |
ORR 38% |
| Nectin-4 | 9MW2821 | Tubulin inhibitor MMAE | Valine–citrulline cleavable linker | Single-arm Phase I/II | CC 45 | ORR 35.6%; DCR 75.6% |
| B7-H4 | HS-20089 | Top1 inhibitor | Cleavable tetrapeptide linker | Single-arm Phase I | OC 44 | ORR 24.2%; DCR 63.6% |
Abbreviations: mo, months; CC, cervical cancer; DCR, disease control rate; DXd, deruxtecan; DM4, maytansinoid DM4; EC, endometrial cancer; MMAE, monomethyl auristatin E; OC, ovarian cancer; ORR, objective response rate; PROC, platinum-resistant ovarian cancer; PFS, progression-free survival; Top1, topoisomerase I.
BAT8006, discovered by Bio-Thera Solutions, Ltd. in China, is an ADC consisting
of a humanized anti-FR
| Target | ADC | Clinical trial identifier | Patients/estimated enrollment | Setting | Interventions | Primary outcome measures | Estimated study completion date |
| TROP-2 | *Sacituzumab Tirumotecan/MK-2870 | CTR20252559/NCT06952504 | EC/1123 | First-line Maintenance Treatment | MK-2870 + Pembrolizumab vs. Pembrolizumab | PFS and OS | May, 2032 |
| TROP-2 | Sacituzumab Tirumotecan/MK-2870 | CTR20241248/NCT06132958 | EC/710 | Post Platinum and Immunotherapy | MK-2870 vs. Chemotherapy | PFS and OS | January, 2028 |
| TROP-2 | Sacituzumab Tirumotecan/MK-2870 | CTR20243424/NCT06459180 | CC/686 | as second-line treatment | MK-2870 vs. Chemotherapy (PC) | OS | October, 2028 |
| TROP-2 | Sacituzumab Govitecan (SG) | CTR20244107/NCT06486441 | EC/640 | Post Platinum-based Chemotherapy and Immunotherapy | SG vs. Chemotherapy (PC) | PFS and OS | June, 2029 |
| Tissue factor | Tisotumab Vedotin | CTR20223290/NCT04697628 | CC/502 | second-line recurrent or metastatic | Tisotumab Vedotin vs. Chemotherapy (PC) | OS | Completed |
| HER2 | Trastuzumab Deruxtecan (T-DXd) | CTR20244095/NCT06989112 | EC/600 | First-Line Treatment | T-DXd Plus Rilvegostomig or Pembrolizumab vs. Chemotherapy Plus Pembrolizumab | PFS | January, 2029 |
| HER2 | Trastuzumab Deruxtecan (T-DXd) | CTR20251716/NCT06819007 | OC/582 | First-line Maintenance Therapy | T-DXd With Bevacizumab vs. Bevacizumab | PFS | November, 2028 |
| HER2 | Trastuzumab Deruxtecan (T-DXd) | CTR20253443/NCT07022483 | EC/600 | Post-surgery adjuvant therapy | T-DXd vs. SoC Chemotherapy | DFS | March, 2032 |
| FR |
Mirvetuximab soravtansine (MIRV) | CTR20211628/NCT04209855 | OC/453 | Post Platinum-based Chemotherapy | MIRV vs. Chemotherapy (PC) | PFS | Completed |
| FR |
Mirvetuximab soravtansine (MIRV) | CTR20241552/NCT05445778 | Platinum-sensitive OC/520 | Maintenance after platinum-based Chemotherapy | MIRV + Bevacizumab vs. Bevacizumab | PFS | March, 2027 |
| CDH6 | Raludotatug Deruxtecan (R-DXd) | CTR20240778/NCT06161025 | PROC/710 | Post Platinum-based Chemotherapy | R-DXd vs. Chemotherapy (PC) | PFS | December, 2027 |
Abbreviations: DFS, disease-free survival; PC, physician’s choice; SoC, standard of care.
*Sacituzumab tirumotecan was developed in China and licensed to Merck, Sharp & Dohme.
Tissue factor is expressed on normal mesenchymal cell types such as endothelial
cells; however, it is also expressed on the surfaces of tumor cells in up to 95%
of patients with cervical cancers [10]. Tisotumab vedotin is an ADC that targets
tissue factor using a cleavable linker attached to the microtubule-disrupting
agent monomethyl auristatin E [11]. It was granted full FDA approval in April
2024 by Hong Kong’s Department of Health in September 2024 for recurrent or
metastatic cervical cancer progressing during or after chemotherapy after results
from the InnovaTV-301 trial were published. The InnovaTV-301 phase III trial
(NCT04697628) aimed to compare tisotumab vedotin to the investigator’s choice
chemotherapy and showed a significant improvement in PFS (4.2 vs. 2.9 months; HR:
0.67, 95% CI: 0.54–0.82; p
HER2 expression data vary widely across studies; its overexpression has been observed in 12–31% of endometrial cancers, 20–38% in ovarian cancers, and 2–6% in cervical cancers [13]. Trastuzumab deruxtecan (T-DXd) is a HER2-directed ADC with a potent Top1 inhibitor payload; it has been approved by the FDA for HER2-positive (IHC 3+) solid tumors based on the DESTINY-PanTumor02, DESTINY-Lung01, and DESTINY-CRC02 trials. The DESTINY-PanTumor02 trial included patients with cervical, endometrial and ovarian cancers with HER2 scores of 2+/3+ on IHC, with 40 patients in each cohort. The ORRs in patients with endometrial, cervical, and ovarian cancer with IHC scores of 2+/3+ were 57.5%, 50.0%, and 45.0%, respectively; those in the subset of patients with IHC scores of 3+ were 84.6%, 75.0%, and 63.6%, respectively (Table 1) [14]. The most common treatment-related AEs (TRAEs) owing to T-DXd were nausea, anemia, diarrhea, and vomiting, which were generally manageable. Interstitial lung disease (ILD)/pneumonitis occurred in 10.5% of patients, with most cases being low-grade (1 and 2).
The ongoing phase III DESTINY-Endometrial01 (NCT06989112/CTR20244095) trial is evaluating the efficacy of T-DXd in combination with rilvegostomig or pembrolizumab as a first-line therapy for patients with HER2-expressing (IHC 2+/3+), mismatch repair-proficient, primary advanced or recurrent endometrial cancer. The DESTINY-Endometrial02 study (NCT07022483/CTR20253443) will enroll post-surgical patients with HER2-expressing (IHC scores of 2+ or 3+) endometrial cancer who have not received prior systemic or neoadjuvant therapy and will assess the efficacy and safety of T-DXd compared with those of standard-of-care chemotherapy with or without radiotherapy as adjuvant treatment. Additionally, the DESTINY-Ovarian01 trial (CTR20251716/NCT06819007) is a phase III, multicenter, randomized study designed to evaluate the efficacy and safety of T-DXd in combination with bevacizumab vs. bevacizumab monotherapy as a first-line maintenance therapy in patients with HER2-expressing (IHC score of 1+, 2+, or 3+) high-grade epithelial ovarian cancer. All three trials are international, multicenter studies that include Chinese participants from mainland China (Table 2).
In addition to T-DXd, several HER2-targeted ADCs developed in China have
demonstrated encouraging clinical activity, and phase III trials of these agents
in patients with gynecologic malignancies are currently underway (Table 2). In a
phase II study of SHR-A1811, a novel HER2-directed ADC conjugated to a Top1
inhibitor payload, the ORR was 66.7% among 30 patients with ovarian cancer,
43.5% among 23 patients with endometrial cancer, and 66.7% among 24 patients
with cervical cancer exhibiting HER2 expression (IHC score 2+ or 3+) (Table 1)
[15]. Grade
| Target | ADC | Clinical trial identifier | Patients/estimated enrollment | Setting | Interventions | Primary outcome measures | Estimated study completion date |
| TROP-2 | SHR-A1921 | CTR20241535/NCT06394492 | PROC/440 | Post Platinum-based Chemotherapy | SHR-A1921 vs. Chemotherapy (PC) | PFS | May, 2026 |
| Nectin-4 | 9MW2821 | CTR20243246/NCT06692166 | CC/420 | Post Platinum-based Chemotherapy | 9MW2821 vs. Chemotherapy (PC) | OS | December, 2027 |
| HER2 | BNT323/DB-1303 | CTR20252691/NCT06340568 | EC/540 | Post Platinum and Immunotherapy | BNT323/DB-1303 vs. Chemotherapy | PFS | October, 2027 |
| HER2 | SHR-A1811 | CTR20250550/NCT06990503 | PROC/300 | Post Platinum-based Chemotherapy | SHR-A1811 vs. Chemotherapy (PC) | PFS | June, 2030 |
| HER2 | JSKN003 | CTR20244822/NCT06751485 | PROC/430 | Post Platinum-based Chemotherapy | JSKN003 vs. Chemotherapy (PC) | PFS | December, 2026 |
| HER2 | IBI354 | CTR20250364/NCT06834672 | PROC/450 | Post Platinum-based Chemotherapy | IBI354 vs. Chemotherapy (PC) | PFS and OS | December, 2027 |
| FR |
BAT8006 | CTR20251345 | PROC/476 | Post Platinum-based Chemotherapy | BAT8006 vs. Chemotherapy | PFS | NA |
| FR |
BAT8006 | CTR20252907 | Platinum-sensitive OC/100 | Maintenance after platinum-based Chemotherapy | BAT8006 + Bevacizumab vs. Bevacizumab | PFS | NA |
| EGFR/HER3 | BL-B01D1 | CTR20251986/NCT06994195 | PROC/84 | Post Platinum-based Chemotherapy | BL-B01D1 vs. Chemotherapy (PC) | PFS and OS | June, 2027 |
| B7-H4 | HS-20089 | CTR20250629/NCT06855069 | PROC/468 | Post Platinum-based Chemotherapy | HS-20089 vs. Chemotherapy (PC) | PFS | March, 2027 |
Abbreviations: DB-1303, BNT323/DB-1303; NA, not available.
JSKN003 is a biparatopic HER2-targeting ADC conjugated to a Top1 inhibitor payload. Results from two clinical studies conducted in Australia and China demonstrated its promising efficacy in patients with PROC. Among 46 patients who received JSKN003 every 3 weeks (Q3W) at varying doses, the ORR was 64.4% with a median PFS of 7.1 months among all patients. In a subset of patients with HER2-expressing tumors (IHC scores of 1+/2+/3+), the ORR reached 72.2% with a median PFS of 9.4 months (Table 1). JSKN003 showed good tolerability, with predominantly low-grade toxicities and nausea as the most common TRAEs. Low-grade ILD was observed in 8.7% of the patients, with no higher-grade cases identified [16]. A confirmatory phase III trial (NCT06751485) is ongoing in an all-comer population with any level of HER2 expression, evaluating the efficacy and safety of JSKN003 vs. the investigators’ choice of chemotherapy (Table 3).
IBI354 is another HER2-targeted ADC bearing a Top1 inhibitor payload. In a phase I study of 40 patients with ovarian cancer treated with IBI354 (12 mg/kg Q3W), the ORR was 55.0%, and the median PFS was 7.1 months. Notably, the ORR remained 55.6% in patients with low HER2 expression (IHC 1+) (Table 1) [17]. IBI354 showed a manageable safety profile, with most TRAEs being hematologic with generally low-to-moderate severity. ILD/pneumonitis occurred in one patient (1.1%); this grade 2 event was deemed unrelated to treatment, supporting the overall tolerability of IBI354. A confirmatory phase III trial (CTR20250364/NCT06834672) is currently underway to compare IBI354 monotherapy with the investigators’ choice of chemotherapy in patients with HER2-expressing PROC (Table 3).
BNT323/DB-1303, developed by DualityBio, is a HER2-directed ADC with a cleavable linker and a TOP1 inhibitor payload (P1003). It has been granted a ‘Breakthrough Therapy’ designation by the FDA for the treatment of advanced or metastatic HER2-expressing endometrial cancer that has progressed following immune checkpoint inhibitor therapy. This decision was supported by topline results from a phase I/II study (NCT05150691) in which BNT323/DB-1303 achieved an ORR of 58.8% and a disease control rate (DCR) of 94.1% among 17 evaluable patients treated at doses of 7.0 mg/kg or 8.0 mg/kg; the safety profile was manageable (Table 1) [18]. An ongoing global phase III trial (CTR20252691/NCT06340568) is evaluating BNT323/DB-1303 vs. the investigators’ choice of chemotherapy in patients with HER2-expressing endometrial cancer previously treated with immune checkpoint inhibitors (Table 3).
TROP-2 is a transmembrane glycoprotein that is widely expressed in different
cancer types including endometrial, cervical, and ovarian tumors [19, 20].
Sacituzumab tirumotecan (sac-TMT; also known as MK-2870/SKB264) is a
TROP-2-targeted ADC developed with a hydrolytically cleavable linker conjugated
with a Belotecan-derivative Top1 inhibitor; this agent was developed by China’s
Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. and licensed to Merck, Sharp &
Dohme in May 2022. Sac-TMT has shown promising anti-tumor activity with a
manageable safety profile in patients with pre-treated advanced endometrial,
cervical, and ovarian cancers. The ORR among 44 patients with endometrial cancer
treated with Sac-TMT was 34.1%, although it was higher (41.7%) among those with
a TROP-2 IHC H-score
Sacituzumab govitecan (SG) is another TROP-2-targeting ADC composed of an
anti-TROP-2 monoclonal antibody conjugated to the cytotoxic payload SN-38, a Top1
inhibitor. In the China-based EVER-132-003 study, SG demonstrated encouraging
antitumor activity in patients with recurrent or metastatic cervical cancer,
achieving an ORR of 43–48%, along with a manageable safety profile consistent
with that of prior studies [23]. In the endometrial cancer cohort of the
TROPiCS-03 study, SG achieved an ORR of 22% and a median PFS of 4.8 months, with
a safety profile also consistent with those previously observed (Table 1) [24].
SG demonstrated a manageable safety profile; the most common TEAEs were
gastrointestinal and included diarrhea (56%), nausea (54%), and fatigue (51%).
Grade
SHR-A1921 is a novel TROP-2-targeted ADC comprising a humanized anti-TROP-2
Immunoglobulin G1 (IgG1) antibody conjugated via a cleavable tetrapeptide linker
to a DNA Top1 inhibitor payload. This ADC demonstrated promising efficacy in
patients with PROC in a phase I study, achieving an ORR of 48.8% and a DCR of
97.7% (Table 1) [25]. Grade
CDH6 is a single-pass transmembrane protein expressed in several malignancies,
including renal cell carcinoma and ovarian cancer. Up to 85% of advanced ovarian
tumors overexpress CDH6, and this has been associated with a poor prognosis [26].
Raludotatug deruxtecan (R-DXd; DS-6000) is a CDH6-targeted ADC that employs a DNA
Top1 inhibitor payload (DXd). In a phase I study, R-DXd demonstrated encouraging
antitumor activity and a manageable safety profile in patients with ovarian
cancer who had previously undergone extensive therapies but were not preselected
for CDH6 expression, achieving an ORR of 38%, a cancer antigen 125 (CA-125) level
decrease in 52%, and durable responses across multiple dose levels [27]. Grade
Nectin-4 is a cell adhesion molecule that is highly expressed in various solid tumors, particularly in cervical and breast cancers [28]. A novel nectin-4-targeted ADC, 9MW2821, was developed by Mabwell Bioscience Co., Ltd. in Shanghai to selectively deliver the cytotoxic payload monomethyl auristatin E to tumor cells. In a phase I/IIa study, 9MW2821 demonstrated encouraging antitumor activity and a manageable safety profile in 45 patients with advanced cervical cancer, achieving an ORR of 35.6% and a DCR of 75.6% (Table 1) [29, 30]. The most common TRAEs were hematologic toxicities, elevations in liver transaminases, rash, and gastrointestinal symptoms such as decreased appetite and nausea. Based on these promising results, an open-label phase III trial (CTR20251843/NCT06946527) is currently evaluating 9MW2821 vs. the physicians’ choice of chemotherapy in patients with recurrent or metastatic cervical cancer who previously received platinum-based therapy (Table 3).
B7-H4 is a transmembrane immune checkpoint glycoprotein that is highly expressed in several solid tumors; its expression is limited in normal tissues [31, 32, 33]. B7-H4 is expressed in approximately 70–90% of ovarian cancers, 60–80% of endometrial cancers, and 50–70% of cervical cancers [34, 35, 36]. Currently emerging B7-H4-targeting ADCs include SGN-B7H4V, XMT-1660, and AZD8205 [37, 38, 39].
HS-20089 is a B7-H4-targeted ADC with a Top1 inhibitor payload developed by Hansoh Pharmaceutical. In a phase I study, 44 patients with advanced solid tumors (including two with ovarian cancer) achieved an ORR of 24.2% and a DCR of 63.6% following HS-20089 treatment, demonstrating encouraging efficacy in B7-H4-expressing tumors (Table 1) [40]. The most common TRAEs were hematologic toxicities, gastrointestinal symptoms (nausea and vomiting), fatigue, and elevations in liver transaminases. HS-20089 was granted a ‘Breakthrough Therapy’ designation by the NMPA of China for the treatment of PROC, based on results from the phase I and II HS-20089-201 trials. HS-20089 is also being evaluated internationally by GlaxoSmithKline under an exclusive licensing agreement. A phase III trial (NCT06855069) is ongoing in patients with PROC to evaluate the efficacy and safety of HS-20089 vs. the investigators’ choice of chemotherapy.
EGFR (also known as Erythroblastic Leukemia Viral Oncogene Homolog erb-b2 receptor tyrosine kinase 1 [ERBB1] or
human epidermal growth factor receptor 1 [HER1]) and HER3 (also known as erb-b2 receptor tyrosine kinase 3 [ERBB3]) are receptor tyrosine kinases that belong to
the ERBB family [41, 42]. Both are highly expressed in multiple solid tumor types
including lung and gynecologic malignancies [43, 44]. Therefore, therapeutic
strategies that simultaneously target EGFR and HER3 are sensible treatment
approaches for patients with these cancers. BL-B01D1 is a first-in-class
EGFR-HER3 bispecific ADC developed by Sichuan Baili Tianheng Pharmaceutical; it
comprises a bispecific antibody, a tetrapeptide-based cleavable linker, and the
toxin Ed-04 (a camptothecin derivative Top1 inhibitor) [45]. In a first-in-human
phase I clinical trial (NCT05194982) involving 195 patients with heavily
pretreated advanced solid tumors, BL-B01D1 treatment resulted in an ORR of 34%.
It also had an acceptable safety profile, with 71% of 195 patients experiencing
grade
Based on the results of the Phase Ib/II study of BL-B01D1 in patients with recurrent metastatic ovarian cancer, a phase III trial (CTR20251986/NCT06994195) is currently underway to compare BL-B01D1 with the investigators’ choice of chemotherapy in patients with PROC (Table 3).
The safety profiles of ADCs are heavily influenced by the types of payloads and linkers used. Tubulin inhibitors (e.g., monomethyl auristatin E in tisotumab vedotin or DM4 in MIRV) are commonly associated with ocular toxicities (e.g., keratopathy and blurred vision), peripheral neuropathy, and gastrointestinal toxicities (nausea and diarrhea). Topoisomerase I (Top1) inhibitors (e.g., deruxtecan in T-DXd) cause hematologic and gastrointestinal toxicities, although ocular toxicities are less common. A major concern with these agents is ILD and pneumonitis, which require close monitoring.
Currently, most ADCs targeting gynecologic cancers use cleavable linkers, which are chemically or enzymatically labile, enabling payload release at the tumor site. Less-stable linkers (e.g., Gly-Gly-Phe-Gly in T-DXd) can cause earlier payload release into circulation, increasing the risk of chemotherapy-related toxicities such as cytopenia, alopecia, and gastrointestinal issues owing to higher peak cytotoxin concentrations. In contrast, more-stable linkers (e.g., valine-citrulline in tisotumab vedotin) lead to longer circulation times for the intact ADC but release the payload more slowly; this can lead to ocular toxicities and other off-target effects owing to the drug being released in tissues other than the tumor [46].
Strategies for managing ADC-related adverse events include (i) dose modifications for ocular maladies (often combined with steroidal eye drops) and for hematologic toxicities (which may be combined with growth factor treatment), (ii) supportive care that includes symptom control for peripheral neuropathy and antiemetics for gastrointestinal issues, and (iii) frequent monitoring of ocular health, pulmonary function, and blood counts, which is essential for early detection.
The expression levels of biomarkers such as FR
As for patients with 2+/3+ IHC expression scores for HER2, the DESTINY-PanTumor02 trial revealed ORRs of 57.5%, 50.0%, and 45.0% in patients with endometrial, cervical, and ovarian cancers, respectively; among those with HER2 IHC 3+ expression, the ORRs were higher at 84.6%, 75.0%, and 63.6%, respectively. These results demonstrate that HER2 IHC expression is linked to treatment efficacy [5]. However, there is currently no standardized companion diagnostic for HER2 IHC, nor are there established evaluation criteria for IHC testing in patients with gynecologic cancers, leading to differences in testing methods that can make it difficult to compare results across clinical trials. This underscores the need for standardized HER2 IHC assessment to optimize treatment and improve trial comparability.
China’s rapidly expanding ADC pipeline holds significant potential to reshape the treatment landscape for gynecologic cancers. However, its clinical impact remains constrained by heterogeneous biomarker testing, regional regulatory differences, high costs, and unequal access to diagnostics and therapies. Intense competition among developers of ADCs that target similar antigens, as well as the challenges of aligning with global trial designs, further delay progress.
Future efforts should prioritize standardizing key biomarker assays (e.g.,
FR
ADCs have become a transformative class of therapeutics in gynecologic oncology that demonstrate high clinical efficacy across multiple settings including PROC and recurrent cervical cancer. The rapid advancement of China’s ADC pipeline highlights its growing contribution to global anticancer treatment innovation. Continued clinical validation and multi-stakeholder collaboration will be critical for ensuring the effective and equitable integration of these promising therapies into routine care.
All data and materials referenced in this review are derived from publicly available publications and clinical trial databases cited in the manuscript.
PC and ZM conceived and designed the review. DT and YS were responsible for the literature search, data extraction, and analysis. XK made significant contributions to the conceptual design, literature interpretation, and critical revision of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We gratefully acknowledge the guidance and support of Professor Jingzhen Tian from Shandong Hongjitang Pharmaceutical Group Co., Ltd.
This research was funded by the 2023 Qilu Biancang Traditional Chinese Medicine Talent Training Project (Lu Health Letter No. [2024] 78) and the Fifth Batch of National Traditional Chinese Medicine Clinical Outstanding Talents Training Project (National Administration of Traditional Chinese Medicine Talent Education Letter No. [2022] 1).
Dongmei Tan and Zhaoqing Meng are employees of Hongjitang Pharmaceutical Group Co., Ltd. However, the company had no role in the handling or conduct of the study. The authors had full access to all data in the study and take full responsibility for the integrity of the data and the accuracy of the data analysis.
During the preparation of this manuscript, the authors used ChatGPT-5 to assist with spelling and grammar checking. After using this tool, the authors carefully reviewed and edited the content as necessary and take full responsibility for the final version of the manuscript.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


