1 Department of Obstetrics and Gynecology, The Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou Municipal Hospital, 215002 Suzhou, Jiangsu, China
Abstract
Severe fetal growth restriction (sFGR) and preeclampsia (PE) are major pregnancy complications that often share placental dysfunction. This study aimed to compare maternal and neonatal outcomes among sFGR cases with or without gestational hypertensive disorders (GHD), with particular emphasis on PE.
A retrospective analysis was conducted on 371 singleton pregnancies with sFGR delivered at Suzhou Municipal Hospital between January 2020 and December 2024. Patients were categorized into three groups: sFGR with PE (sFGR-PE; n = 69), sFGR with gestational hypertension (sFGR-GH; n = 15), and normotensive sFGR (n = 287). Maternal characteristics, pregnancy complications, and neonatal outcomes were compared using Analysis of Variance (ANOVA), and chi-square tests, as appropriate. Multivariate logistic regression was used to obtain adjusted odds ratios (aORs).
The patients in PE group were older, and had higher body mass index (BMI), higher rates of gestational diabetes mellitus (GDM), and preterm delivery (p < 0.05). Neonates in the PE group had significantly lower gestational age and birth weight, and higher incidences of metabolic acidosis, and respiratory complications (p < 0.001). Multivariable regression analysis revealed that, compared with normotensive sFGR, the other two groups showed no significant increase in the risk of adverse neonatal outcomes but had a significantly higher risk of cesarean or instrumental delivery.
Although sFGR with hypertensive disorders showed increased crude risks of adverse outcomes, multivariable analysis indicated that most adverse effects were mediated by earlier delivery and lower birthweight. Only the risk of operative delivery remained independently elevated, highlighting the importance of optimal delivery timing in these pregnancies.
Keywords
- severe fetal growth restriction
- preeclampsia
- gestational hypertension disorders
- neonatal outcomes
Fetal growth restriction (FGR) refers to the failure of the fetus to achieve its genetically determined growth potential and may result from maternal, fetal, placental, or genetic factors. Severe fetal growth restriction (sFGR) is commonly defined as an estimated fetal weight (EFW) below the third percentile [1]. The severity of FGR strongly influences perinatal outcomes, with sFGR associated with the highest rates of adverse events [2].
Gestational hypertensive disorders (GHD), including preeclampsia (PE) and gestational hypertension (GH), are leading causes of maternal and neonatal morbidity and mortality, accounting for 9–26% of maternal deaths worldwide [3]. PE, the more severe form of GHD, and FGR are both classified as “placental syndromes” and share similar pathological mechanisms, such as impaired trophoblast invasion and placental oxidative stress [4].
Although the association between PE and FGR is well established, comparative data on maternal and perinatal outcomes in sFGR cases with or without GHD remain limited. This retrospective study aims to compare the clinical characteristics and outcomes of sFGR cases, stratified by the presence of PE or GH, providing evidence to support risk stratification and guide antenatal management.
This retrospective study included singleton pregnancies diagnosed with sFGR and delivered at Suzhou Municipal Hospital, Affiliated Suzhou Hospital of Nanjing Medical University, between January 2020 and December 2024. Of the 416 initially identified cases, those with incomplete medical records, stillbirths, chromosomal abnormalities, or major structural malformations were excluded. A final cohort of 371 cases was analyzed.
sFGR was defined as either an EFW or abdominal circumference (AC) below the 3rd percentile, or absent end-diastolic flow in the umbilical artery (UA-AEDF). EFW was calculated using the Hadlock formula [5]:
• FL = femur length
• BPD = biparietal diameter
• HC = head circumference
GH was defined as a systolic blood pressure
PE was diagnosed when hypertension was accompanied by proteinuria and/or signs
of organ dysfunction, such as thrombocytopenia, renal or liver dysfunction, or
pulmonary edema [3]. Gestational diabetes mellitus (GDM) was diagnosed based on
the 2018 American College of Obstetricians and Gynecologists (ACOG) guidelines
[6]. Neonatal acidosis was defined as an umbilical artery pH
Patients were categorized into three groups: Group A, sFGR with PE (sFGR-PE); Group B, sFGR with GH (sFGR-GH); Group C, normotensive sFGR.
Maternal demographics (age, body mass index [BMI], gravidity, parity, and mode of conception), pregnancy complications (GDM, preterm delivery, and delivery mode), and neonatal outcomes (gestational age at delivery, birthweight, acidosis, hyperbilirubinemia, and respiratory issues) were compared across the groups.
Statistical analyses were performed using SPSS 23.0 (IBM Corp., Chicago, IL,
USA). The Shapiro-Wilk test and Levene’s test were employed to assess the
normality of data and the homogeneity of variances, separately. Normally
distributed continuous variables were presented as mean
Among the 371 sFGR cases analyzed, 69 (18.6%) were complicated by PE, 15 (4.0%) by GH, and 287 (77.4%) were normotensive. A flowchart of patient selection is presented in Fig. 1.
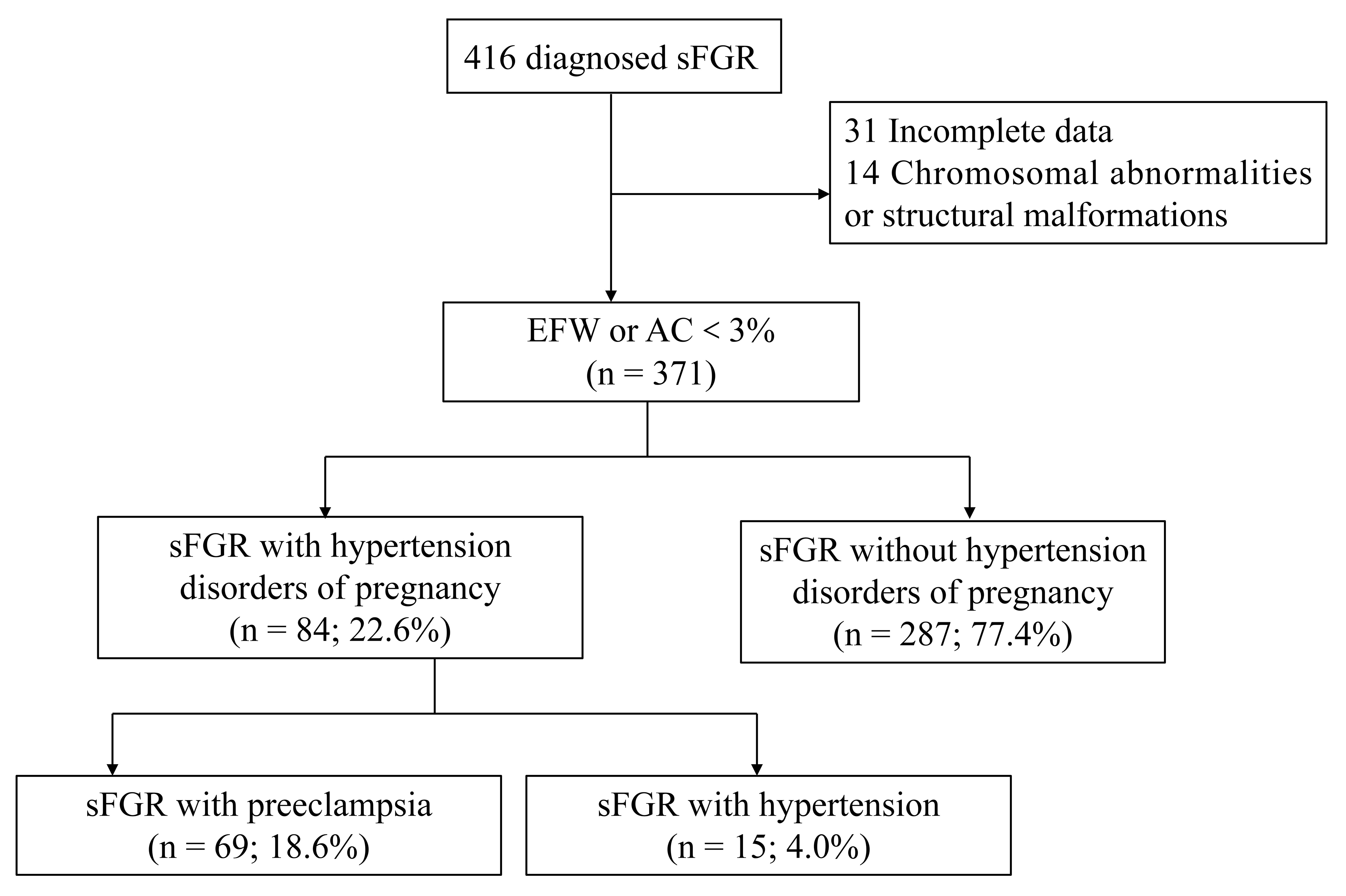 Fig. 1.
Fig. 1.
Flowchart of sFGR cases included in the study. sFGR, severe fetal growth restriction; EFW, estimated fetal weight; AC, abdominal circumference.
As shown in Table 1, maternal age and BMI were significantly higher in the PE
group compared with the other two groups (p = 0.03 and p
| Variables | Normotensive sFGR (n = 287) | sFGR-PE (n = 69) | sFGR-GH (n = 15) | p-value | |
| Maternal age (years) | 29.64 |
31.10 |
30.27 |
- | 0.027 |
| BMI (kg/m2) | 20.80 |
23.02 |
21.56 |
- | |
| ART | 11 (3.80%) | 6 (8.70%) | 1 (6.70%) | 2.96 | 0.228 |
Note: Data are expressed as mean
As shown in Table 2, the incidence of GDM and preterm birth was significantly
higher in the sFGR-PE group (p = 0.023 and p
| Variables | Total | Normotensive sFGR (n = 287) | sFGR-PE (n = 69) | sFGR-GH (n = 15) | p-value | |
| GDM | 72 | 47 (16.4%) | 21 (30.4%) * | 4 (26.7%) | 7.56 | 0.023 |
| Preterm | 90 | 46 (16.0%) | 40 (58.0%) *# | 4 (26.7%) | 53.31 | |
| Cesarean or instrumental delivery | 222 | 147 (51.2%) | 62 (89.9%) * | 13 (86.7%) * | 39.23 |
Note: Data are expressed as number (%); *p
Perinatal outcomes are summarized in Table 3. The mean gestational age at
delivery was significantly lower in the PE group compared with the other two
groups (36.27
| Variables | Total | Normotensive sFGR (n = 287) | sFGR-PE (n = 69) | sFGR-GH (n = 15) | p-value | ||
| Gestational age (weeks) | - | 38.47 |
36.27 |
37.99 |
- | ||
| Birthweight (g) | - | 2464.15 |
1978.55 |
2260.00 |
- | ||
| Gender | - | 1.02 | 0.600 | ||||
| Male | 140 | 108 (37.6%) | 28 (40.6%) | 4 (26.7%) | |||
| Female | 231 | 179 (62.4%) | 41 (59.4%) | 11 (73.3%) | |||
| Acidosis | 50 | 27 (9.4%) | 22 (31.9%) *# | 1 (6.7%) | 24.72 | ||
| Neonatal hyperbilirubinemia | 154 | 94 (32.8%) | 50 (72.5%) * | 10 (66.7%) * | 40.21 | ||
| Respiratory problems | 80 | 47 (16.4%) | 32 (46.4%) *# | 1 (6.7%) | 31.65 | ||
| APOs | 184 | 119 (41.5%) | 55 (79.7%) * | 10 (66.7%) | 34.37 | ||
| Stillbirth | 3 | 3 (1.0%) | 0 | 0 | 0.89 | 0.642 | |
Note: Data are expressed as mean
In the sFGR-PE group, there was a high incidence of neonatal metabolic acidosis (31.9%), hyperbilirubinemia (72.5%), and respiratory disorders (46.4%). Notably, no significant differences were observed in these complications between the sFGR-PE and sFGR-GH, except for respiratory disorders and acidosis. Three stillbirth cases occurred in the normotensive sFGR, and no statistically significant differences were found among the groups regarding the stillbirth incidence.
Table 4 illustrates the results of multivariable regression analysis after
adjusting for potential confounders. sFGR-PE and sFGR-GH were not associated with
a significantly increased risk of adverse neonatal outcomes but were associated
with a significantly higher risk of cesarean or instrumental delivery compared
with normotensive sFGR (adjusted odds ratios [aOR] = 6.70 and 3.71, p
| Group | Crude OR (95% CI) | aOR (95% CI) | |
| APOs | sFGR-PE | 5.55 (2.95–10.43) | 1.34 (0.58–3.12) |
| sFGR-GH | 2.82 (0.94–8.47) | 1.91 (0.43–8.40) | |
| Acidosis | sFGR-PE | 4.51 (2.37–8.57) | 1.42 (0.63–3.23) |
| sFGR-GH | 0.69 (0.09–5.44) | 0.38 (0.04–3.39) | |
| Neonatal hyperbilirubinemia | sFGR-PE | 5.40 (3.02–9.68) | 1.59 (0.77–3.30) |
| sFGR-GH | 4.11 (1.37–12.35) | 3.54 (0.94–13.27) | |
| Respiratory problems | sFGR-PE | 4.42 (2.51–7.79) | 0.77 (0.33–1.80) |
| sFGR-GH | 0.37 (0.05–2.84) | 0.11 (0.10–1.22) | |
| Cesarean or instrumental delivery | sFGR-PE | 8.44 (3.73–19.06) | 3.71 (1.47–9.37) |
| sFGR-GH | 6.19 (1.37–27.93) | 6.70 (1.31–34.13) |
Normotensive sFGR was taken as a reference category for the OR calculation. ORs were obtained from a logistic regression model (simple, multivariate analysis adjusted by maternal age, pre-pregnancy BMI, gestational age and birthweight). Abbreviations: OR, odds ratio; CI, confidence interval; aOR, adjusted odds ratio.
Fetal growth restriction remains a major contributor to perinatal morbidity and mortality and has long-term implications for cardiovascular and metabolic health. International guidelines vary in their diagnostic criteria for FGR [8], but there is consensus that fetuses with an EFW or AC below the 3rd percentile should be classified as sFGR due to their significantly elevated risk of adverse outcomes [9, 10].
PE and sFGR share common placental pathophysiology, particularly involving abnormal spiral artery remodeling, endothelial dysfunction, and placental insufficiency [11]. It has been hypothesized that different abnormalities of trophoblastic cells resulted in different disease phenotypes [12]. Huppertz [13] suggested that malfunction of the syncytiotrophoblast may increase oxidative stress markers and compromise the integrity of the placental barrier, thereby contributing to the pathogenesis of PE. In contrast, malfunction of the cytotrophoblast can impair placental proliferation and fusion, ultimately leading to FGR [13].
Previous clinical guidelines have primarily treated FGR as a consequence of PE [14]. However, our results suggest a more complex interplay: among sFGR cases, the incidence of PE (18.80%) far exceeded that of GH. This finding may indicate a distinct pathophysiological entity rather than a linear progression from GH to PE. The global prevalence of PE has been estimated at 4.6% of deliveries [15]. The incidence of GH in FGR strongly correlated with gestational age, declining from 39–43% in early preterm cases to 4–7% in term deliveries [16]. The International Society for the Study of Hypertension in Pregnancy (ISSHP) includes FGR as a diagnostic criterion for PE [17]. However, it remains controversial that cases diagnosed as PE with hypertension and FGR alone do not show an increased incidence of adverse maternal and neonatal outcomes compared with PE with hypertension accompanied by proteinuria or organ dysfunction [18]. Consequently, to delineate their distinct effects, GH and PE were analyzed separately in the sFGR population.
Our study demonstrated that sFGR-PE and sFGR-GH were associated with significantly worse maternal and neonatal outcomes compared with normotensive sFGR. However, after adjustment for potential confounders, such as gestational age and birthweight, these associations were no longer statistically significant, indicating that these variables largely account for the adverse outcomes observed in this group. The risk of cesarean or instrumental delivery remained significantly elevated, reflecting the vulnerability of growth-restricted fetuses to intrauterine hypoxia and the limited compensatory capacity of a compromised placenta during labor [19]. The timing of delivery is a critical consideration in the management of FGR, particularly when complicated by hypertensive disorders of pregnancy. Biochemical factors, such as soluble fms-like tyrosine kinase-1 (sFlt-1) and placental growth factor (PlGF), are increasingly incorporated into maternal evaluation [20]. In carefully selected near-term FGR fetuses without Doppler abnormalities, mechanical or pharmacological induction can be safely considered [21]. Clinicians should exercise particular caution when developing diagnostic strategies, surveillance protocols, and final recommendations for the timing of delivery of growth-restricted fetuses. While the univariable model indicated an increased likelihood of cesarean section in sFGR-PE compared with sFGR-GH, this relationship was reversed in the multivariable-adjusted model. This change, along with the large 95% CI in sFGR-GH, may be caused by the small sample size.
sFGR-PE pregnancies had significantly earlier deliveries in our study. This discrepancy may be attributed to the fact that the timing of delivery in sFGR-PE is influenced not only by fetal impairment but also by maternal conditions. Furthermore, abnormal umbilical artery Doppler waveforms appear earlier in sFGR-PE cases, which may contribute to the earlier gestational age at delivery [22]. Our study lacked data regarding the interval from disease diagnosis to delivery. Concomitant hypertensive disorders have been reported to shorten this diagnosis-to-delivery interval. However, only 24% of these deliveries were primarily due to maternal indications [23].
To our knowledge, this is one of the few retrospective studies that stratifies sFGR cases by both PE and GH. The relatively large sample size and use of a standardized diagnostic approach strengthen the reliability of our findings. However, several limitations should be noted. Due to the extended study duration and the retrospective design, it was difficult to precisely ascertain the timing of onset for both FGR PE, as well as to distinguish between early- and late-onset FGR and PE. A smaller sample size in the sFGR-GH group was a limitation that may have reduced statistical power. Furthermore, the timing of initiation and dosage of aspirin prophylaxis lacked standardization, and very few cases adhered to a daily regimen, resulting in data of limited reliability.
Compared with normotensive sFGR pregnancies, cases with coexisting GHD and PE did not show a significantly increased risk of adverse neonatal outcomes. However, poor neonatal outcomes may be influenced by prematurity. Future prospective, multicenter studies with larger sample sizes are needed to validate these findings and inform the clinical management of such pregnancies.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
MY and QN designed the research study. QN performed the research. MY analyzed the data. MY and QN wrote the manuscript. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The research was approved by the Ethics Committee of the Affiliated Suzhou Hospital of Nanjing Medical University (KL-2025-060-K01). Given the retrospective nature of the study, which involved only the review of existing medical records without any direct patient contact or intervention, the requirement for written informed consent was waived by the committee. All patient data were anonymized prior to analysis to ensure confidentiality. The study was carried out in accordance with the guidelines of the Declaration of Helsinki.
We acknowledged the assistance from the midwives and clinicians at Suzhou municipal hospital, the Affiliated Suzhou Hospital of Nanjing Medical University.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work the authors used Deepseek in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

