1 Department of Human Reproductive Medicine, Beijing Obstetrics and Gynecology Hospital, Capital Medical University. Beijing Maternal and Child Health Care Hospital, 100026 Beijing, China
Abstract
The selection of an optimal thawing and transfer strategy is a critical determinant of success in frozen embryo transfer (FET) cycles. To investigate the optimal FET strategy, this study analyzed the effects of extended culture duration after warming on clinical outcomes following embryo transfer.
We retrospectively analyzed 9981 FET cycles following either an unsuccessful fresh embryo transfer or cycle cancellation. In these cycles, embryos were warmed and transferred on varying days, spanning from Day 2 to Day 5. We compared baseline characteristics, thawing recovery, and embryo development across groups. Additionally, we performed regression analyses to examine the relationship between clinical outcomes and in vitro culture conditions.
The post-warming embryo survival rate was 98.60%, with an available embryo rate of 86.88%. For warmed Day 2 embryos, extending culture to Day 3 was significantly associated with higher clinical pregnancy and live birth rates. Similarly, Day 3 warmed embryos cultured to Day 4 or Day 5 were associated with better outcomes compared with Day 3 transfer. However, there was no significant difference between transferring embryos on Day 4 or Day 5 after warming on Day 4. Likewise, no significant differences were observed when all embryos were cultured to Day 5, regardless of the initial warming day.
Tailoring FET strategies with an optimized post-warming culture duration is associated with improved clinical outcomes in assisted reproductive technology. Direct transfer of frozen-warmed blastocysts yielded similar success rates to those undergoing extended culture to the blastocyst stage.
Keywords
- blastocyst
- clinical outcomes
- extend culture
- frozen embryo transfer
- in vitro embryo culture
Embryo cryopreservation has become a cornerstone of assisted reproductive technologies (ART). Initially developed to preserve surplus embryos after fresh in vitro fertilization and embryo transfer (IVF-ET) cycles, frozen embryo transfer (FET) has been shown to be associated with superior clinical outcomes in certain patient populations. Controlled ovarian stimulation disrupts normal endometrial physiology and receptivity, leading to impaired receptivity and embryo-endometrial asynchrony [1, 2]. In contrast, FET cycles better mimic natural endocrine conditions, thereby restoring endometrial receptivity and improving synchrony [3, 4]. The ‘freeze-all’ strategy further optimizes outcomes in high responders and reduces the treatment burden by avoiding repeated ovarian stimulation. Particularly for older patients with diminished ovarian reserve, FET can effectively mitigate the detrimental impact of supraphysiologic stimulation on endometrial receptivity, thereby increasing the likelihood of a live birth [5]. Nevertheless, optimizing pregnancy rates after FET remains a significant challenge in reproductive medicine.
Vitrification can be used to preserve embryos at different developmental stages, and the survival rate of warmed embryos is closely associated with the developmental stage. Although current methods for assessing embryonic developmental potential remain limited, advances in culture systems have improved the evaluation of viability through extended in vitro culture, which helps to avoid unnecessary transfers and reduces intrauterine interventions. Consequently, selecting the optimal developmental stage for cryopreservation is critical for maximizing ART success. Currently, the most commonly cryopreserved stages are cleavage-stage (Day 3) and blastocyst-stage (Day 5 or Day 6) embryos, with most studies focusing on the extended culture from cleavage-stage embryos to blastocysts or overnight culture before transfer [6, 7, 8, 9]. Blastocyst-stage transfers have been shown to yield better clinical outcomes than do cleavage-stage embryo transfers [6, 10, 11]. However, practical considerations such as patient-scheduling constraints or embryology-laboratory schedules often necessitate cryopreservation at alternative times (Day 2 or Day 4) [12, 13], a practice reflected in Department of Human Reproductive Medicine, Beijing Obstetrics and Gynecology Hospital, Capital Medical University where embryos are cryopreserved from Day 2 to Day 6.
Although some studies have been done on this topic [14, 15], no previous study has systematically analyzed a substantial number of cases across these developmental periods. In the present large-scale retrospective study, we analyzed 9981 frozen-warmed embryos cultured for varying durations (Day 2 to Day 5). The choice of freezing timing is influenced by multiple factors (e.g., laboratory and clinical scheduling, weekend adjustments), particularly on Day 2 and Day 4. We performed a detailed stratification based on embryo-warming day and transfer timing to systematically evaluate their impact on clinical outcomes and to identify optimal transfer strategies.
We conducted a retrospective analysis of FET cycles performed at the Department of Human Reproductive Medicine, Beijing Obstetrics and Gynecology Hospital, Capital Medical University, between January 2016 and December 2023. The study included infertile couples undergoing IVF-ET or intracytoplasmic sperm-injection embryo transfer (ICSI-ET) for tubal or male-factor infertility. From an initial pool of 10,130 FET cycles, we excluded 149 cycles: 23 due to poor post-warming embryo quality and 126 due to embryo unavailability after extended culture. The final analysis comprised 9981 cycles (Fig. 1). All procedures complied with relevant regulations and institutional guidelines, and the study protocol was approved by the Ethics Committee of Beijing Obstetrics and Gynecology Hospital, Capital Medical University (Approval date: January 30, 2024; Approval No.: 2024-KY-024-01). This study is a retrospective analysis based on existing medical records and data. It involved no direct intervention in human subjects and posed no direct risks. All data used were de-identified to protect patient privacy. The study protocol was rigorously reviewed and approved by the Ethics Committee of Beijing Obstetrics and Gynecology Hospital, Capital Medical University. The committee confirmed that the study fully complied with ethical standards and waived the requirement for informed consent.
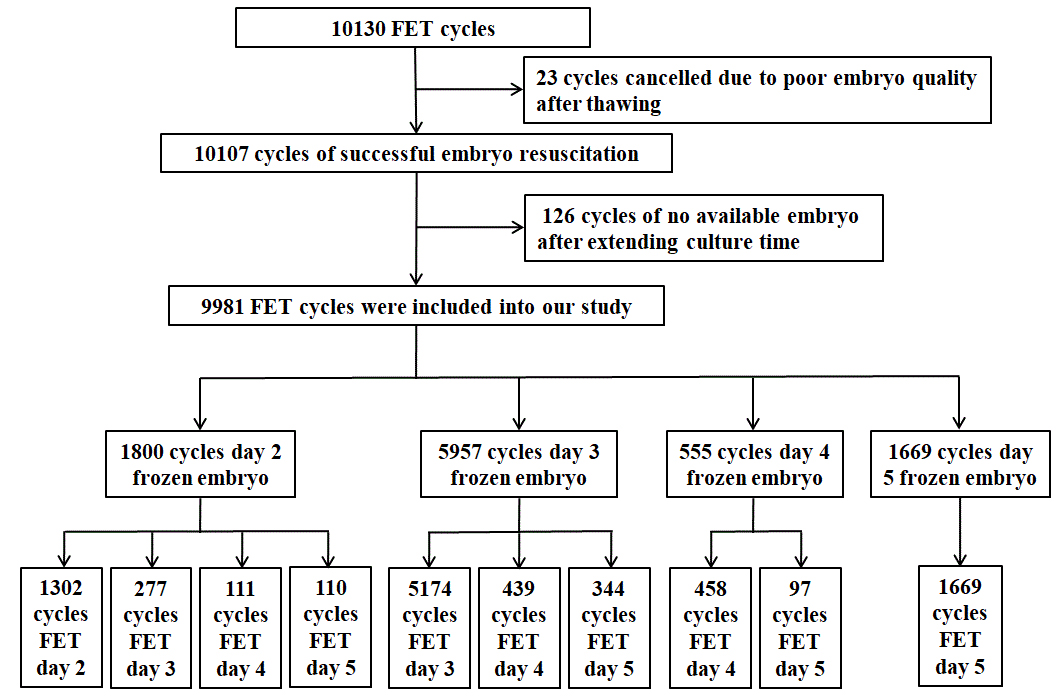 Fig. 1.
Fig. 1.
Selection of patients for the study. FET, frozen embryo transfer.
We included only patients
We excluded patients diagnosed with polycystic ovary syndrome, symptomatic
uterine leiomyoma or submucous myoma, endometriosis, significant metabolic
disorders, uterine malformations, or a history of major ovarian or pelvic surgery
or pelvic irradiation. Patients experiencing repeated implantation failure or
with decreased ovarian reserve (basal serum follicle stimulating hormone (FSH)
The patients were stratified into groups based on the day of embryo warming and the day of transfer: Groups A1–A4 = embryos warmed on Day 2 and transferred on the same day (A1), Day 3 (A2), Day 4 (A3), or Day 5 (A4); Groups B1–B3 = embryos warmed on Day 3 and transferred on the same day (B1), Day 4 (B2), or Day 5 (B3); Groups C1–C2 = embryos warmed on Day 4 and transferred on the same day (C1) or Day 5 (C2); and Group D = embryos warmed and transferred on Day 5.
Embryo warming was performed using Kitazato Thawing Mwarming media (Lot numbers varied across the study period, with the primary lots used being D0414, E0808, and G0704; Kitazato Corporation, Fuji, Shizuoka, Japan) in accordance with the manufacturer’s protocol. The freezing carriers were retrieved from liquid nitrogen, and the front portion was immersed in the warming solution. After 1 min, the embryos were transferred to diluent solution for 3 min, followed by sequential washes in washing solution 1 and washing solution 2 for 5 min each.
Once warmed, the embryos were placed in culture medium for further cultivation until the optimal transfer time. Embryos designated for same-day transfer were cultured for 2–4 h prior to transfer. Assisted laser hatching was performed on all embryos prior to transfer.
The culture duration was individualized based on the patient’s condition and clinic schedule. Embryos warmed on Day 2 and cultured to Day 5 had their medium replaced on Day 3 to mitigate metabolic stressors. Embryo morphology was evaluated according to the Istanbul Consensus criteria [16]. Blastocysts were graded according to the Gardner standards [17].
The embryo-grading criteria on Days 2 and 3 were as follows: Grade 1
The grade criteria for Day 4 morulae were as follows: Grade 1 =
Complete embryonic compaction after the fourth mitosis; Grade 2 = Partial
compaction (
Blastocysts were graded according to the stage of expansion: Stage 1 = Cavity
formation occupying
The endometrium-preparation protocol (NC or HRT) was personalized based on
patient characteristics. Embryo-transfer timing was precisely matched to the
developmental stage of the embryo to ensure synchronization between embryonic
development and endometrial receptivity. During extended culture, when more than
two embryos reached transferable quality, the most advanced one or two embryos
were selected for transfer, and the remaining viable embryos were re-vitrified.
The embryo developmental potential was assessed by the available-embryo rate,
defined as: (Number of transferred and re-vitrified embryos)/(Number of warmed
embryos)
Pregnancy was confirmed by a positive serum-human Chorionic Gonadotropin (hCG)
test (
The primary outcome measure was the clinical-pregnancy rate. The secondary outcome measure was the live-birth rate. We recorded demographic information [age, FSH, AMH, body mass index (BMI), infertility duration, infertility type, and causes of infertility], and endometrial characteristics (thickness, preparation protocols). We also analyzed embryo-quality indicators at the time of warming (including the number of warmed embryos and survived embryos) and after culture (including the number of available, transferred, and discarded embryos).
Statistical analyses were performed using SPSS Statistics software (version
23.0; IBM Corporation, Armonk, NY, USA). The Shapiro-Wilk test was used to assess
the normality of continuous data. Homogeneity of variances was evaluated using
Levene’s test. Continuous data were presented as mean
For comparisons across multiple groups, 1-way ANOVA was used for
normally distributed variables. If a significant difference was identified,
post-hoc pairwise comparisons were conducted using Tukey’s test. For
comparisons between two groups, the independent samples Student’s t-test
was used for normally distributed data. The chi-squared (
A total of 9981 FET cycles were analyzed, with patients stratified according to
embryo-warming and transfer timing. The demographic and clinical characteristics
are presented in Table 1. No significant differences were observed in these
baseline characteristics across groups (p
| Warming day | Day 2 | Day 3 | Day 4 | Day 5 | p | |
| Warm cycles | 1800 | 5957 | 555 | 1669 | ||
| Age (years) | 32.97 |
32.83 |
32.66 |
32.70 |
0.087 | |
| FSH (IU/L) | 6.71 |
6.74 |
6.62 |
6.71 |
0.588 | |
| AMH (ng/mL) | 3.24 |
3.23 |
3.19 |
3.20 |
0.616 | |
| BMI (kg/m2) | 21.51 |
21.49 |
21.47 |
21.49 |
0.933 | |
| Infertility duration (years) | 3.48 |
3.52 |
3.50 |
3.44 |
0.495 | |
| Infertility type, n (%) | 0.812 | |||||
| Primary infertility | 875 (48.6) | 2877 (48.3) | 257 (46.3) | 802 (48.1) | ||
| Secondary infertility | 925 (51.4) | 3080 (51.7) | 298 (53.7) | 867 (51.9) | ||
| Causes of infertility, n (%) | 0.730 | |||||
| Tubal | 1105 (61.4) | 3647 (61.2) | 329 (59.3) | 1006 (60.3) | ||
| Male | 695 (38.6) | 2310 (38.8) | 226 (40.7) | 663 (39.7) | ||
| Endometrial thickness (mm) | 9.93 |
9.87 |
9.85 |
9.89 |
0.333 | |
| Endometrial preparation protocols [n (%)] | 0.340 | |||||
| NC | 795 (44.2) | 2516 (42.2) | 231 (41.6) | 689 (41.3) | ||
| HRT | 1005 (55.8) | 3441 (57.8) | 324 (58.4) | 980 (58.7) | ||
FSH, follicle stimulating hormone; AMH, anti-Müllerian hormone; BMI, body mass index; NC, natural cycles; HRT, hormone replacement therapy.
The study included 9981 warming cycles encompassing 20,907 embryos. After
warming, 20,615 embryos survived, yielding a survival rate of 98.60%. After
extended culture, 18,163 embryos remained viable, resulting in a post-warm
culture available rate of 86.88%. The survival rate did not differ significantly
between subgroups (p
| Group | No. of warmed embryos | Survived embryos1, n (%) | Available embryos2, n (%) | Transferred embryos, n (%) | Discarded embryos, n (%) | ||
| Day 2 warming | 0.985 | 0.000 | 0.000 | 0.000 | |||
| Day 2 ET | A1 | 2377 | 2342 (98.53)a | 2342 (98.53)a | 2342 (98.53)a | 35 (1.47)a | |
| Day 3 ET | A2 | 698 | 687 (98.42)a | 514 (73.64)b | 492 (70.49)b | 184 (26.36)b | |
| Day 4 ET | A3 | 410 | 403 (98.29)a | 222 (54.15)c | 197 (48.05)c | 188 (45.85)c | |
| Day 5 ET | A4 | 387 | 381 (98.29)a | 190 (49.10)c | 182 (47.03)c | 197 (50.90)c | |
| Day 3 warming | 0.991 | 0.000 | 0.000 | 0.000 | |||
| Day 3 ET | B1 | 9675 | 9536 (98.56)a | 9536 (98.56)a | 9351 (96.65)a | 139 (1.44)a | |
| Day 4 ET | B2 | 1756 | 1731 (98.58)a | 783 (44.59)b | 759 (43.22)b | 973 (55.41)b | |
| Day 5 ET | B3 | 1360 | 1340 (98.53)a | 603 (44.34)b | 559 (41.10)b | 757 (55.66)b | |
| Day 4 warming | 0.894 | 0.000 | 0.000 | 0.000 | |||
| Day 4 ET | C1 | 838 | 827 (98.69)a | 827 (98.69)a | 827 (98.69)a | 11 (1.31)a | |
| Day 5 ET | C2 | 409 | 404 (98.78)a | 182 (44.50)b | 161 (39.36)b | 227 (55.50)b | |
| Day 5 warming | |||||||
| Day 5 ET | D | 2997 | 2964 (98.90) | 2964 (98.90) | 2964 (98.90) | 33 (1.10) | |
1: Cleavage stage embryos survival was assessed by observing
2: The number of available embryos includes the number of transferred and re-vitrified embryos after extended culture.
a.b.c: Different letters indicate statistically significant
differences (p
ET, embryo transfer.
We compared clinical pregnancy rate between subgroups (Table 3). To adjust for
potential confounding factors, variables including maternal age, BMI, infertility
type, endometrial thickness and preparation protocol, the number of good-quality
embryos after thawing, and the quality/quantity of transferred embryos were
included in the multivariate regression analysis. For embryos warmed on Day 2,
extending the culture to Day 3 was significantly associated with a higher
clinical pregnancy rate (p
| Baseline parameter | Group | Clinical pregnancy rate | Univariate | Multivariate1 | ||||
| p | p | OR | 95% CI | |||||
| Lower | Upper | |||||||
| Day 2 thawing | 0.000 | |||||||
| Day 2 ET | A1 | 34.5 (449/1302)a | 0.000 | 1.000 | ||||
| Day 3 ET | A2 | 53.4 (148/277)b | 0.000 | 3.104 | 1.777 | 5.423 | ||
| Day 4 ET | A3 | 45.9 (51/111)a,b | 0.382 | 1.273 | 0.741 | 2.189 | ||
| Day 5 ET | A4 | 47.3 (52/110)b | 0.105 | 1.634 | 0.903 | 2.956 | ||
| Day 3 thawing | 0.006 | |||||||
| Day 3 ET | B1 | 33.7 (1742/5174)a | 0.000 | 1.000 | ||||
| Day 4 ET | B2 | 34.4 (151/439)a,b | 0.002 | 1.640 | 1.206 | 2.231 | ||
| Day 5 ET | B3 | 42.2 (145/344)b | 0.000 | 1.850 | 1.355 | 2.526 | ||
| Day 4 thawing | 0.516 | |||||||
| Day 4 ET | C1 | 39.7 (182/458)a | 0.000 | 1.000 | ||||
| Day 5 ET | C2 | 43.3 (42/97)a | 0.105 | 1.915 | 0.873 | 4.203 | ||
OR, odds ratio.
a.b: Different letters indicate statistically significant differences
(p
1: Covariates including: maternal age, BMI, infertility type, endometrial thickness and preparation protocol, the number of good-quality embryos after thawing, and the quality/quantity of transferred embryos.
The live-birth rates of the subgroups are compared in Table 4. For the
multifactorial analysis, the same set of covariates was used for both live birth
and clinical pregnancy rates. Extending the culture to Day 3 for embryos warmed
on Day 2 was associated with a higher live birth rate (p
| Baseline parameter | Group | Live birth rate | Univariate | Multivariate1 | ||||
| p | p | OR | 95% CI | |||||
| Lower | Upper | |||||||
| Day 2 thawing | 0.000 | |||||||
| Day 2 ET | A1 | 28.3 (369/1302)a | 0.000 | 1.000 | ||||
| Day 3 ET | A2 | 43.0 (119/277)b | 0.005 | 2.175 | 1.263 | 3.745 | ||
| Day 4 ET | A3 | 37.8 (42/111)a,b | 0.672 | 1.120 | 0.663 | 1.890 | ||
| Day 5 ET | A4 | 40.9 (45/110)b | 0.200 | 1.455 | 0.820 | 2.580 | ||
| Day 3 thawing | 0.022 | |||||||
| Day 3 ET | B1 | 26.9 (1391/5174)a | 0.001 | 1.000 | ||||
| Day 4 ET | B2 | 27.3 (120/439)a,b | 0.001 | 1.723 | 1.254 | 2.367 | ||
| Day 5 ET | B3 | 33.7 (116/344)b | 0.002 | 1.662 | 1.204 | 2.295 | ||
| Day 4 thawing | 0.847 | |||||||
| Day 4 ET | C1 | 33.0 (151/458)a | 0.000 | 1.000 | ||||
| Day 5 ET | C2 | 32.0 (31/97)a | 0.372 | 1.452 | 0.641 | 3.291 | ||
a.b: Different letters indicate statistically significant differences
(p
1: Covariates including: maternal age, BMI, infertility type, endometrial thickness and preparation protocol, the number of good-quality embryos after thawing, and the quality/quantity of transferred embryos.
Embryos frozen on Days 2–4 during the fresh cycle were cultured further to
blastocysts before transfer in the frozen cycle. No significant differences in
clinical pregnancy or live birth rates were observed among these groups
(p
| Day 5 FET | Group | Clinical pregnancy rate | p | Live birth rate | p |
| 0.745 | 0.504 | ||||
| Day 2 warming | A4 | 47.3 (52/110)a | 40.9 (45/110)a | ||
| Day 3 warming | B3 | 42.2 (145/344)a | 33.7 (116/344)a | ||
| Day 4 warming | C2 | 43.3 (42/97)a | 32.0 (31/97)a | ||
| Day 5 warming | D | 44.8 (748/1669)a | 35.2 (588/1669)a |
a: Different letters indicate statistically significant differences
(p
To our knowledge, this study was the first to compare pregnancy outcomes after cryopreservation at multiple developmental stages [cleavage-stage (including Days 2 and 3), morula, and blastocyst] in a large patient cohort. Our research demonstrated that embryos warmed on Day 2 and subsequently cultured to Day 3 exhibited significantly higher clinical-pregnancy and live-birth rates. Furthermore, embryos warmed on Day 3 and cultured to Days 4 or 5 also showed better clinical outcomes. No significant differences were observed in clinical outcomes among groups when all embryos were cultured to Day 5, irrespective of the original warming day.
In clinical practice, cleavage-stage embryo transfer is typically performed on
Day 3, yet no consensus exists on whether transfer on Day 2 or Day 3 yields the
better outcome. A previous study suggested that Day-3 transfers are associated
with lower survival rates and higher miscarriage rates than were Day-2 transfers;
they attributed this to damage during freeze-thaw procedures [18]. That led to
the hypothesis that Day-2 embryos may tolerate freeze-thaw procedures better.
However, some studies reported no statistical difference in clinical outcomes
when Day-2 warmed embryos underwent overnight culture [14, 19]. With advancements
in vitrification techniques, freeze-warm-related damage has been substantially
minimized. A study has indicated that transferring embryos with
Morula-stage embryo transfer represents a clinically feasible strategy, though it remains underutilized in ART. After fertilization, embryos undergo compaction typically at the 8-cell stage, forming a morphologically distinct morula—a transitional phase between cleavage and blastocyst stages. Characterized by a tightly packed cellular mass, the morula is often perceived as fragile due to its lack of clear morphological markers [26]. This morphological ambiguity, coupled with stage-specific biological complexity, has limited clinical research on morula transfer, resulting in its frequent oversight as a valuable transfer option [13]. At this developmental juncture, embryos exhibit heightened DNA-synthesis activity, priming for subsequent cell-lineage differentiation critical for implantation [27]. The compaction process involves multiple self-correcting mechanisms that may enhance developmental competence [28]. One study showed that there was no statistical difference in clinical pregnancy outcomes for embryos frozen on Day 3, whether they were transferred after warming or after overnight culture [8]. Our study indicated that extended culture of thawed cleavage-stage (Day 2/3) embryos to morula stage (Day 4) was associated with better clinical outcomes. This association was significantly stronger for embryos thawed on Day 3 compared to those thawed on Day 2. No significant differences in clinical pregnancy rates have been shown to result from embryos transferred on Day 4 and Day 5 [13, 29]. Our findings align with those observations. Our study showed that there were no significant differences in clinical outcomes between embryos transferred on Day 4 immediately after warming and those transferred after overnight culture. However, A study suggested that Day-5 transfers have higher success rates than Day-4 transfers [30]. It is possible that Day 4 transfer is often not based on clinical indications, but on logistical limitations such as public holidays, or scheduling issues. This nonrandom selection introduces potential bias. Therefore, the research on Day 4 transfer is relatively limited, and further research is needed.
The transfer of blastocysts has been reported to have better clinical outcomes than does the transfer of cleavage-stage embryos [31, 32]. Our experiments showed that although extended culture to Day 5 was associated with better outcomes, this association was significant only for embryos thawed on Day 3, not for those thawed on Day 2. Early embryo development primarily reflects oocyte quality, which does not fully predict subsequent embryo development. During development into a blastocyst, the genome regulates the transitions from maternal to zygote control [33]. Blastocysts, with their higher cell numbers and structured organization (ICM and TE), offer enhanced viability and more reliable implantation potential than do cleavage-stage embryos [15]. With the success of blastocyst culture, chromosomal abnormalities become less frequent, facilitating improved screening of suitable embryos for transfer. Physiologically, embryos with 4–8 cells initially reside in the fallopian tubes, entering the uterus upon reaching the blastocyst stage. Therefore, transferring blastocysts rather than cleavage-stage embryos may align more closely with the natural timing of the embryo to enter the uterus. Better synchronization with the endometrial-receptivity window may potentially improve pregnancy rates [31, 34].
The present study showed that no significant differences exist between transferring frozen-warmed blastocysts and transferring blastocysts cultured from warmed cleavage-stage embryos. This was consistent with another study showing that extending culture of cleavage-stage embryos to blastocysts is comparable to using frozen-warmed blastocysts, with similar live-birth rates [35]. For patients with recurrent implantation failure who have blastocysts available, prioritizing frozen-warmed blastocyst FET is recommended. Otherwise, in cases without blastocyst preservation, extended culture of frozen-warmed Day 3 embryos to Day 5 for blastocyst transfer is advisable, as both strategies yield similar pregnancy outcomes [10]. However, extending embryo culture without selection is not beneficial, as it increases the transfer-cycle cancellation rate and reduces the cumulative pregnancy rate [36]. Some embryos may fail to develop into blastocysts in vitro, but they may develop and implant in vivo. For patients with diminished ovarian reserve or a strong desire for expedited treatment, early transfer/freezing (Day 2/3) may optimize embryo availability.
Although an extended post-warming culture entails additional costs and the risk of embryo attrition, it offers significant advantages by eliminating developmentally compromised or genetically abnormal embryos, thereby selecting those with higher implantation potential. This selective approach reduces the financial and emotional burdens associated with transferring non-viable embryos. Furthermore, extended culture may decrease ectopic-pregnancy risks by minimizing the embryo transit time within the uterine cavity. Clinicians should therefore adopt a patient-centered approach when recommending extended culture, carefully weighing individual circumstances and preferences.
A limitation of this study is the imbalanced sample sizes among subgroups, which may reduce the statistical power of the comparisons. As a single-center retrospective study, our findings may have been influenced by local protocols and patient demographics. To strengthen the generalizability of our conclusions, multicenter randomized trials with larger cohorts are warranted. Future collaborations with other reproductive centers could help validate whether these results apply across diverse patient populations and laboratory settings.
Selective cryopreservation and FET are safe and effective strategies for optimizing cumulative live birth rates in ART. After adjustment for confounding factors, a significant correlation was found between extended embryo culture and superior clinical outcomes. The statistical significance was particularly strong in two groups: embryos thawed on Day 2 and cultured to Day 3, and those thawed on Day 3 and cultured to Day 4 or 5. Direct transfer of frozen-warmed blastocysts and transfer of blastocysts cultured from warmed cleavage-stage embryos both achieve comparable clinical success. Furthermore, when logistically necessary (e.g., due to clinic or patient scheduling constraints), Day 4 transfer can also achieve comparable clinical outcomes. Therefore, individualized extended culture strategies were associated with FET outcomes. However, if no blastocysts are available, cleavage-stage embryo transfer should be considered in subsequent cycles to maximize reproductive potential.
The data used and analyzed during the current study are available from the corresponding authors on reasonable request.
Study design was proposed by ZX, XY and QW. Material preparation and data collection were performed by LY, RS and LW. Data check was done by YF and QW. Data analysis and the first draft of the manuscript were written by QW. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of Beijing Obstetrics and Gynecology Hospital, Capital Medical University (date: January 30, 2024. No. 2024-KY-024-01). The ethics committee granted a waiver of informed consent for this retrospective study, given the use of anonymized data and the absence of risk to individuals.
We would like to express our gratitude to all those who helped us during the writing of this manuscript.
This study was supported by the Beijing Hospitals Authority’ Ascent Plan (DFL20191401) and Specialized Youth Foundation Project of Beijing Obstetrics and Gynecology Hospital, Capital Medical University (FCYYQN-202104).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

