1 The First College of Clinical Medical Science, China Three Gorges University, Yichang Central People’s Hospital, 443003 Yichang, Hubei, China
2 Department of Pathology, The Fifth People’s Hospital of Yichang, 443007 Yichang, Hubei, China
3 Department of Ultrasound, The Fifth People’s Hospital of Yichang, 443007 Yichang, Hubei, China
Abstract
Adenomyosis involves epithelial-mesenchymal transition (EMT), yet the role of mitochondrial regulator Mitofusin 2 (Mfn2) remains unclear. This study investigated the role of Mfn2 in adenomyosis-related EMT and evaluated its potential as a therapeutic target.
Mfn2 expression was compared between human adenomyotic and normal endometrial tissues using immunohistochemistry. Transforming growth factor-beta 1 (TGF-β1)-induced EMT was established in Ishikawa cells, and Mfn2 was overexpressed to assess EMT markers using quantitative polymerase chain reaction (qPCR) and Western blot, as well as cell migration and invasion (via scratch and Transwell assays). A neonatal mouse model of tamoxifen-induced adenomyosis received intrauterine lentiviral Mfn2 overexpression. Uterine morphology, fibrosis, and EMT markers were evaluated after 20 days.
Mfn2 was significantly downregulated in adenomyosis (p < 0.05). Overexpression of Mfn2 reversed EMT, evidenced by increased E-cadherin and decreased N-cadherin and Vimentin (p < 0.05), suppressed cell migration and invasion in vitro (p < 0.05), improved uterine morphology (p < 0.05), reduced fibrosis (p < 0.05), and inhibited EMT in vivo.
Mfn2 suppresses EMT in adenomyosis, suggesting its protective role and potential as a therapeutic target.
Keywords
- adenomyosis
- epithelial-mesenchymal transition (EMT)
- Mitofusin 2 (Mfn2)
- transforming growth factor-beta 1 (TGF-β1)
Adenomyosis (ADS) is a common gynecological disease, and the main pathological feature is ectopic growth of endometrial glands and stroma into the myometrium [1]. The ectopic growth results in a cascade of clinical problems, including dysmenorrhea, dyspareunia, abnormal uterine bleeding, and infertility [2, 3], which seriously impair the quality of life of reproductive-age women [1]. The estimated prevalence of adenomyosis in women of reproductive age ranges from 20% to 34.6% on the basis of radiographic assessment to 10% to 88% on the basis of pathological diagnosis in resected uterine specimens, so this diagnostic heterogeneity underscores the underrecognition of this disease [4].
It has been demonstrated by recent studies that endometrial basal cell abnormal proliferation and invasion have intimate connections with epithelial-mesenchymal transition (EMT) [5]. EMT is a salient biological program: through reprogramming of some molecules, epithelial cells are able to transform into extremely migratory and invasive mesenchymal cells. Despite the inevitability of the process in embryonic development and tissue restoration, its pathological activation is widely implicated in inflammatory reactions and metastasis of tumors [6, 7]. In adenomyosis, EMT may lead to the phenotypic transformation of endometrial epithelial cells, which lose their original features step by step, acquire invasive stromal cell characteristics, and further breach the basal barrier and invade the myometrium to form the typical ADS lesions [8].
Because the dynamic equilibrium of mitochondria is in charge of cell homeostasis and even the body’s homeostasis, as the core center of cellular energy metabolism, the dynamic equilibrium of mitochondria is thus very crucial. In the regulating network, Mitofusin 2 (Mfn2) is also the core executor of mitochondrial fusion and an important safeguard of mitochondrial network structure [9]. By interacting with Mitofusin 1 (Mfn1) and other proteins to form functional complexes, Mfn2 initiates processes of membrane fusion between the mitochondria [10], thus ensuring mitochondrial function and morphology integrity. The regulation has a profound effect on basic processes such as energy metabolism, signaling by calcium, and cell life and death processes [11, 12, 13]. It should be noted that the abnormal expression or dysfunction of Mfn2 has been demonstrated to be closely related to a series of severe diseases, ranging from neurodegenerative diseases, metabolic disorders, to cardiovascular diseases, and even throughout the whole process of tumor occurrence, development, and metastasis [14, 15, 16, 17].
With an increasing depth of research on cell biology and molecular mechanisms, the regulatory network between Mfn2 and EMT has been progressively exposed. Present data suggest that Mfn2 has a tumor suppressor function by dynamically regulating the balance of mitochondrial fusion/fission in thyroid cancer progression and directly regulates the EMT process [18]. In ovarian cancer xenograft models, Mfn2 may be induced by genetic or drug approaches to cause mitochondrial fusion, which strongly inhibits the growth, migration, invasion and EMT phenotype of cancer cells by reducing the level of reactive oxygen species (ROS) [19]—the mechanism is mitochondrial fusion-mediated F-actin remodeling, decreasing the formation of lamella pseudopodia, a key connection in EMT. These findings collectively describe a multi-organ role of Mfn2 as an EMT mediator.
Surprisingly, despite accumulating evidence, the functional role of Mfn2 in ADS remains largely unknown. Based on confirmed Mfn2-EMT regulation in oncology models, and EMT’s established role in adenomyosis pathogenesis, we hypothesize that Mfn2 inhibits adenomyosis progression by suppressing EMT in endometrial cells. This study is the first to test this mechanism in benign gynecological disease. By combining the three-dimensional verification system of in vitro cell model, clinical sample analysis, and animal experiment, the principal role of Mfn2 in the occurrence and development of diseases will be studied deeply, which will provide a new understanding of ADS pathological mechanism and create the possibility of translational medicine for targeted intervention.
In the present study, samples were obtained from the Wujia District of Yichang Central People’s Hospital. The experimental group consisted of 15 cases of endometrial tissue of ADS patients, and the control group consisted of 8 cases of endometrial tissue of hysterectomy due to cervical intraepithelial neoplasia. No patients received any hormonal therapy, radiotherapy, or chemotherapy during the last 3 months before surgery. All the specimens were confirmed to be of the proliferative phase by histological examination, and other pathological conditions such as uterine fibroids and endometrial lesions were excluded.
Given the challenges in primary endometrial epithelial cell isolation and culture stability, we selected the Ishikawa human endometrial adenocarcinoma cell line as a well-established in vitro model. Although derived from malignancy, this cell line retains key endometrial epithelial characteristics and is widely used in adenomyosis/endometriosis EMT studies due to its hormone responsiveness and reliable transfection efficiency.
Mycoplasma contamination in the Ishikawa cell line was assessed using PCR and colorimetric assays, with results indicating no contamination. To verify genetic identity and purity, genomic DNA was extracted and amplified using a multiplex PCR system targeting 20 short tandem repeat (STR) loci and a gender identification marker. The amplified products were analyzed using a genetic analyzer and compared against the national database (CCRID, https://http://www.cellresource.cn/) for matching analysis.
The human endometrial cancer cell line Ishikawa (CL-0283, Pricella Biological,
Wuhan, Hubei, China) was cultured in DMEM supplemented with 10% FBS and 1%
penicillin-streptomycin at 37 °C in 5% CO2. Cells were divided
into four groups: Normal Control, transforming growth factor-beta 1
(TGF-
Total RNA was extracted using TRIzol. Purified RNA was reverse-transcribed to cDNA using the HiScript III reverse transcription kit (AKR-201, Accuri, Nanjing, Jiangsu, China). Quantitative real-time reverse transcription PCR (qRT-PCR) was performed using ChamQ Universal SYBR quantitative polymerase chain reaction (qPCR) Master Mix (AKR-301, Accuri, Nanjing, Jiangsu, China) on an Agilent Statagene Mx3000P instrument. Primers (Hunan Accuri BioEngineering Co., Ltd., Nanjing, Jiangsu, China) were used. The reaction conditions were: 50 °C for 30 min (reverse transcription); 95 °C for 3 min (pre-denaturation); followed by 40 cycles of 95 °C for 15 sec (denaturation) and 60 °C for 30 sec (annealing). Relative gene expression was calculated using the 2-ΔΔCt method. Primer sequences are provided in Supplementary Table 2.
Proteins were extracted using radioimmunoprecipitation assay (RIPA) lysis buffer
(Applygen Technologies Inc., Beijing, China). Protein concentration was
determined by bicinchoninic acid (BCA) assay (P0009; PINUOFEI, Beijing, China)
and samples were normalized. Samples were mixed with 5
Cell migration was assessed using a scratch wound healing assay. Ishikawa cells
(5
Ishikawa cells (2
ICR newborn female mice (
At PND 60, three mice per group were sacrificed. Uterine tissues were collected for hematoxylin and eosin H&E staining to confirm successful modeling, defined by histopathological observation of endometrial glands and stroma invading the myometrium to form characteristic adenomyosis lesions, accompanied by significant changes in EMT markers (down-regulated E-cadherin, up-regulated N-cadherin, Vimentin).
After successful modeling, mice were randomly divided into four groups (n =
9/group): Normal Control, ADS Model, Mfn2 Negative Control (LV-NC), and Mfn2
Overexpression (LV-Mfn2). Mice in the LV-NC and LV-Mfn2 groups received
intrauterine injections of empty vector lentivirus (LV-NC) or Mfn2-overexpressing
lentivirus (LV-Mfn2, 1
Uterine tissues were fixed in 4% paraformaldehyde (24 h), dehydrated, cleared, paraffin-embedded, and sectioned (5 µm). Sections were dried (60 °C, 2 h), deparaffinized in xylene I and II, and hydrated through graded ethanol (100%, 95%, 80%, 70%). After H&E staining, sections were dehydrated (70%, 80%, 95%, 100% ethanol), cleared in xylene, and mounted with neutral gum. Five random fields per section were imaged under a light microscope.
Following hematoxylin and eosin (HE) staining, tissue sections underwent
deparaffinization and hydration. Masson’s trichrome staining was performed
sequentially with: Weigert’s iron hematoxylin, ponceau acid fuchsin,
differentiation in 1% phosphomolybdic acid, and aniline blue. After dehydration
and mounting, collagen fibers (blue) and muscle fibers (red) were visualized
under a light microscope. Fiji ImageJ software was used to quantify the
percentage of collagen fiber area (5 random fields/section). Collagen regions
(stained blue) were identified by Red–Green–Blue (RGB) threshold segmentation
(threshold range: blue channel 60–255), and nuclei (dark blue, saturation
Paraffin-embedded tissue sections (5 µm) were fixed and subjected to H&E staining following standard protocol. For immunohistochemistry (IHC), sections underwent microwave antigen retrieval in 0.01 M sodium citrate buffer (pH 6.0), followed by endogenous peroxidase blocking with 3% H2O2. After blocking with 5% BSA, sections were incubated overnight at 4 °C with primary antibody: Mfn2 (1:1200, 12186-1-AP; Proteintech). After PBS washes, sections were incubated with HRP-conjugated secondary antibodies (1:500, G1213; Sevier) at room temperature, developed with 3,3’-diaminobenzidine (DAB), and counterstained with hematoxylin. Sections were then dehydrated, cleared, mounted, and imaged under a light microscope (5 random fields/section). Mfn2-positive area percentage and expression levels were quantified using ImageJ software.
Statistical analysis was performed with GraphPad Prism software (version 8.0,
San Diego, CA, USA). Continuous data are presented as mean
We found that Mfn2 protein levels were significantly lower in the endometrium of
adenomyosis patients compared to healthy women. This hints that Mfn2 might be
involved in the disease. Looking closer at a process called EMT—which is
important in diseases like this—we saw changes in key markers: the
“epithelial” marker E-cadherin (both mRNA and protein) was down (p
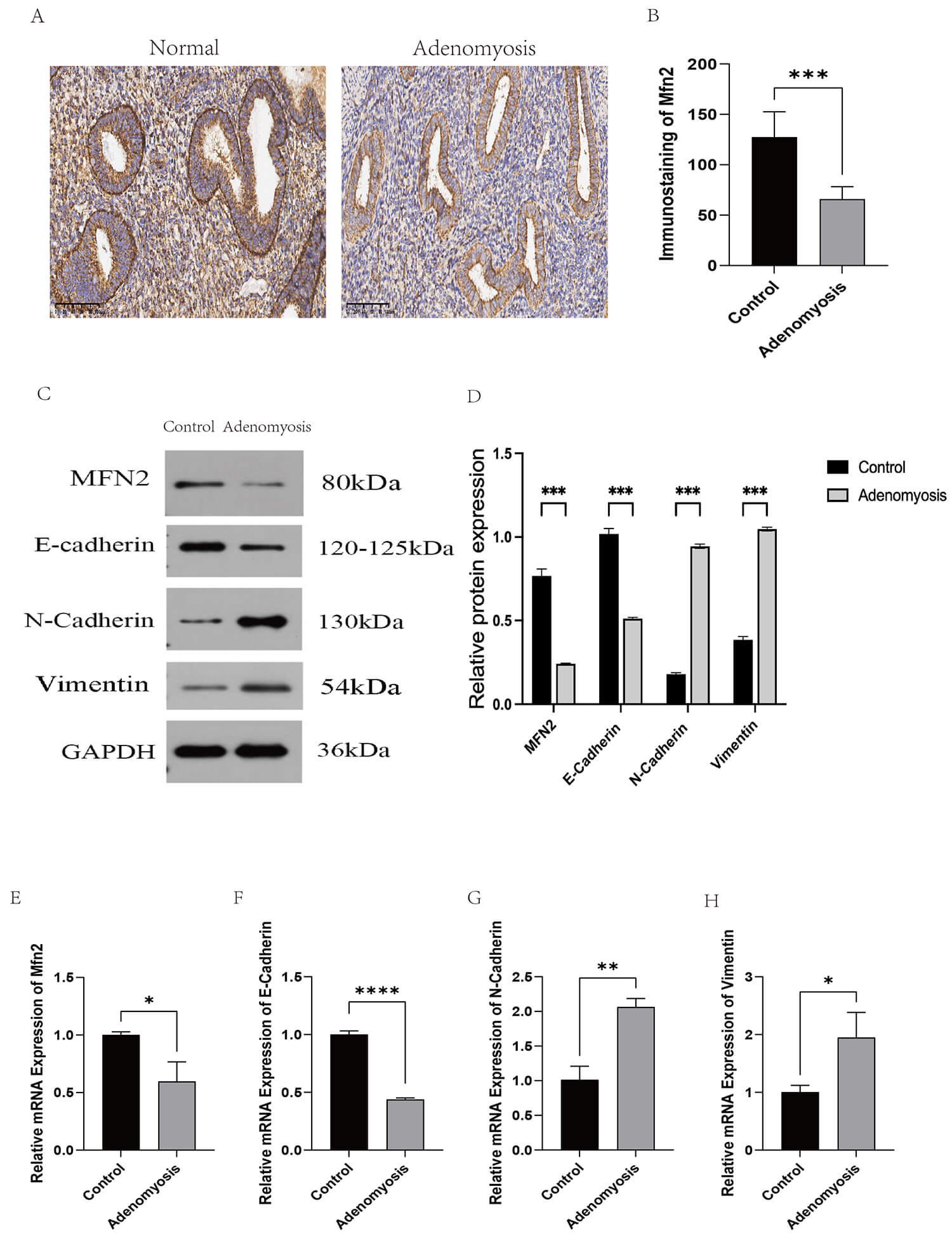 Fig. 1.
Fig. 1.
Immunohistochemical, Western Blot, and qPCR results of clinical
samples. (A,B) Immunohistochemistry was used to compare the expression of Mfn2
in normal endometrium and adenomyosis endometrium. Scale bar: 100 µm
(
To test this link, we used a lab model of EMT (treating Ishikawa cells with
TGF-
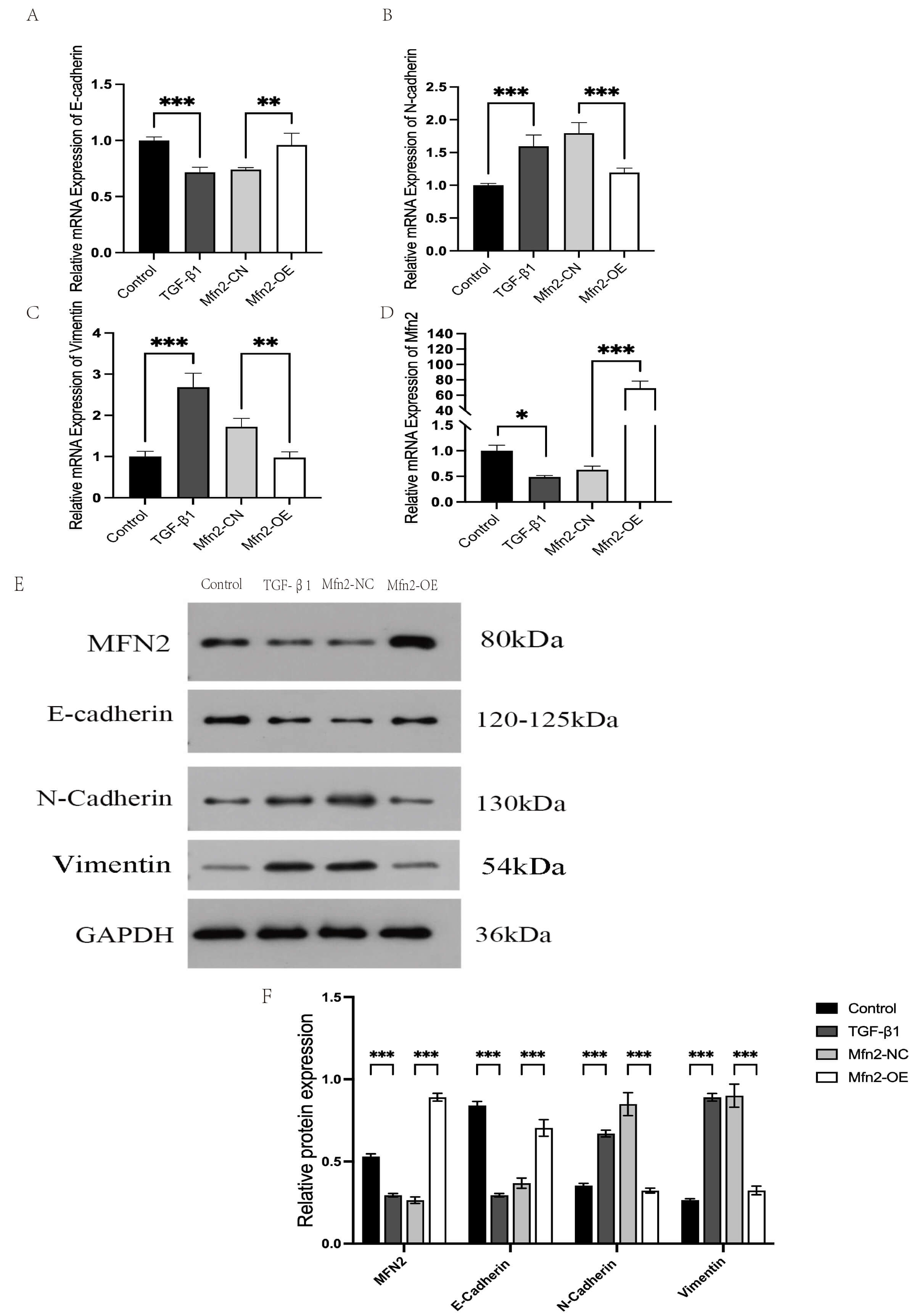 Fig. 2.
Fig. 2.
Ishikawa cells were treated with TGF-
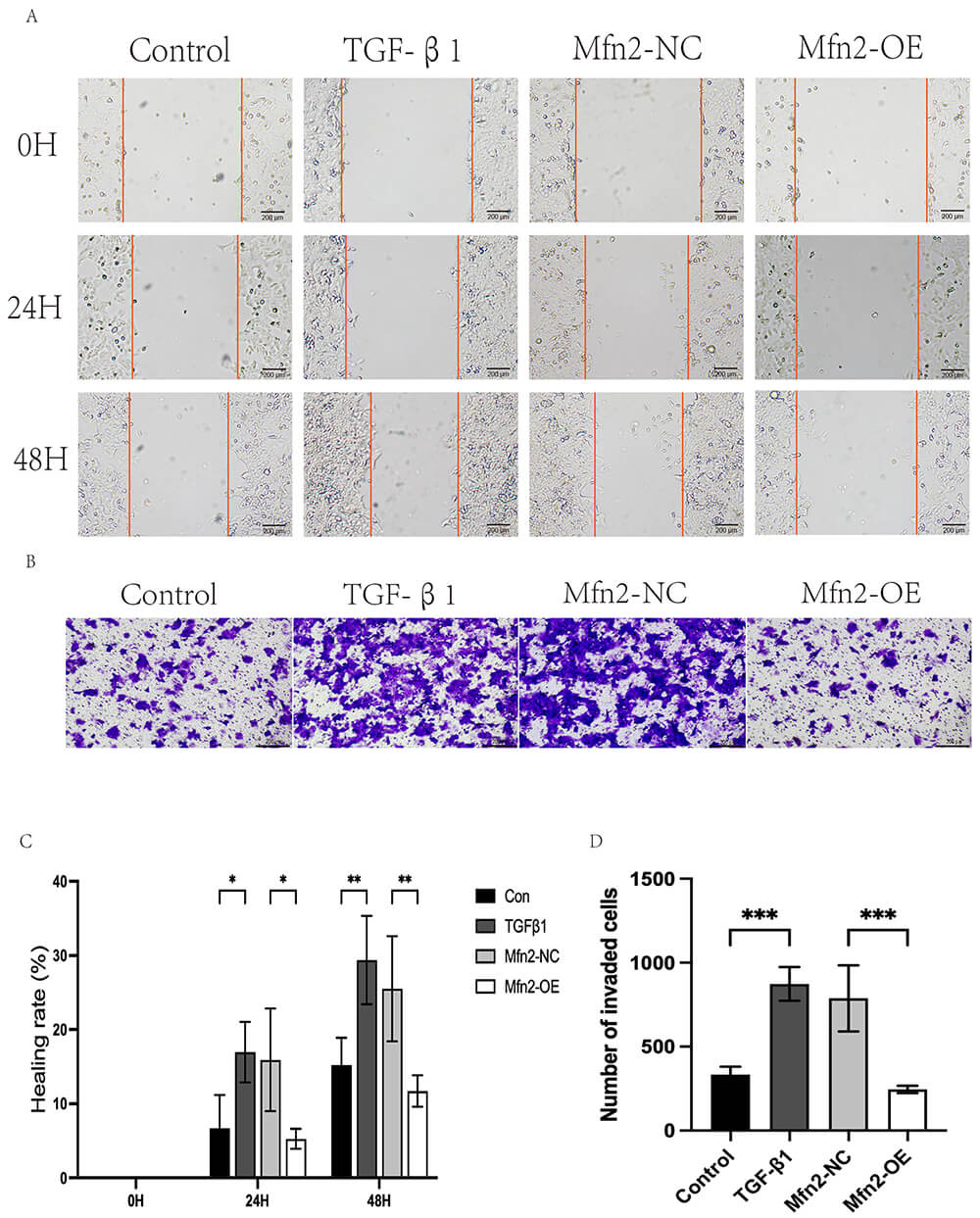 Fig. 3.
Fig. 3.
Cell scratch assay and Transwell invasion assay. Functional
verification: (A,C) cell scratch assay. Scale bar: 200 µm (
To see if Mfn2 protects against adenomyosis in a living system, we used a mouse
model of the disease. Mice with adenomyosis showed the expected problems:
disrupted boundary between the endometrium and muscle layer, glands invading the
muscle, and increased fibrosis (p
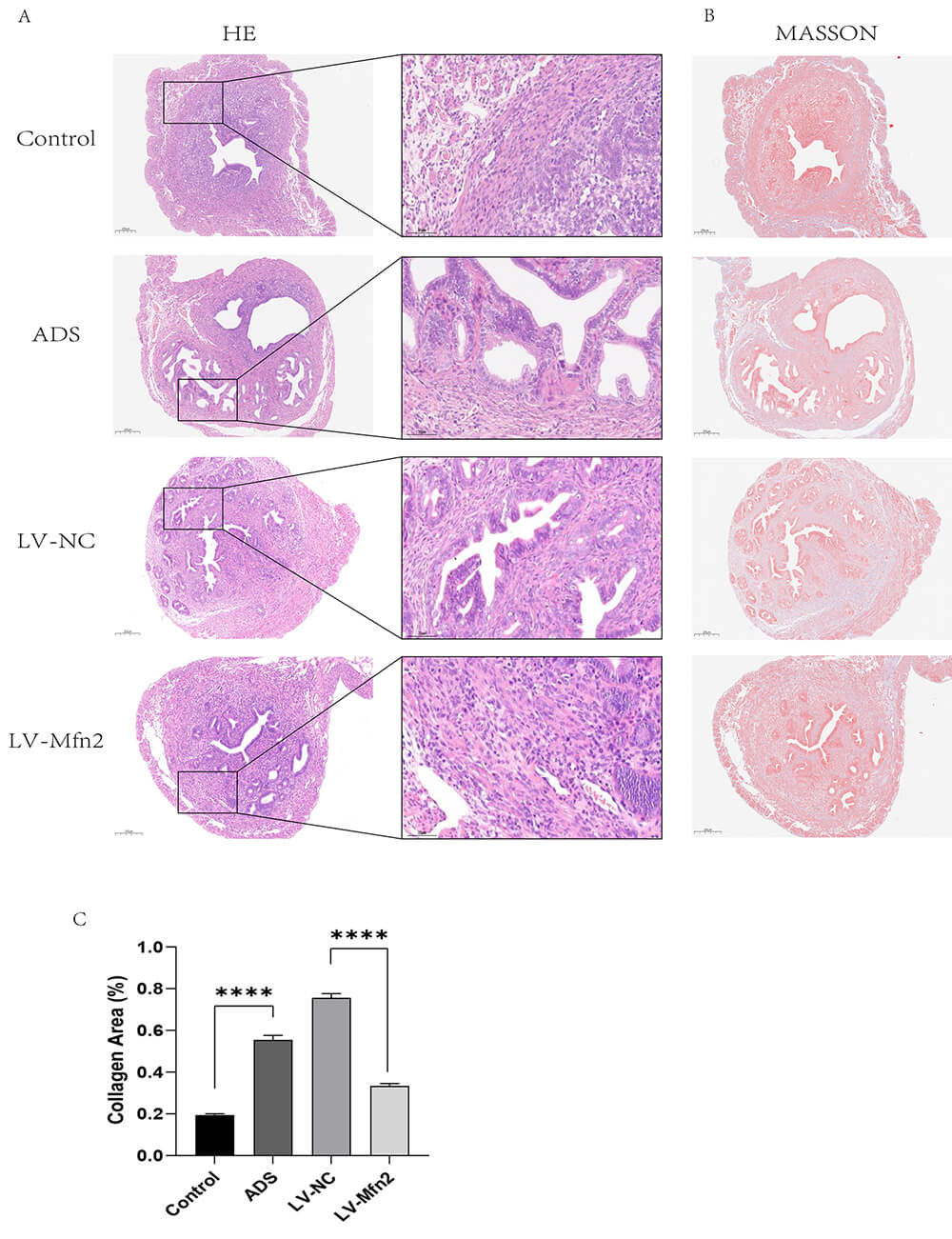 Fig. 4.
Fig. 4.
Related pathological changes in mouse uterus. (A) H&E staining
of the uterus of mice in each group: Control, adenomyosis model (ADS), Mfn2
negative control (LV-NC), lentiviral Mfn2 overexpression (LV-Mfn2). H&E staining
showed the endometrial-myometrial junction. Scale bar: 200 µm
(
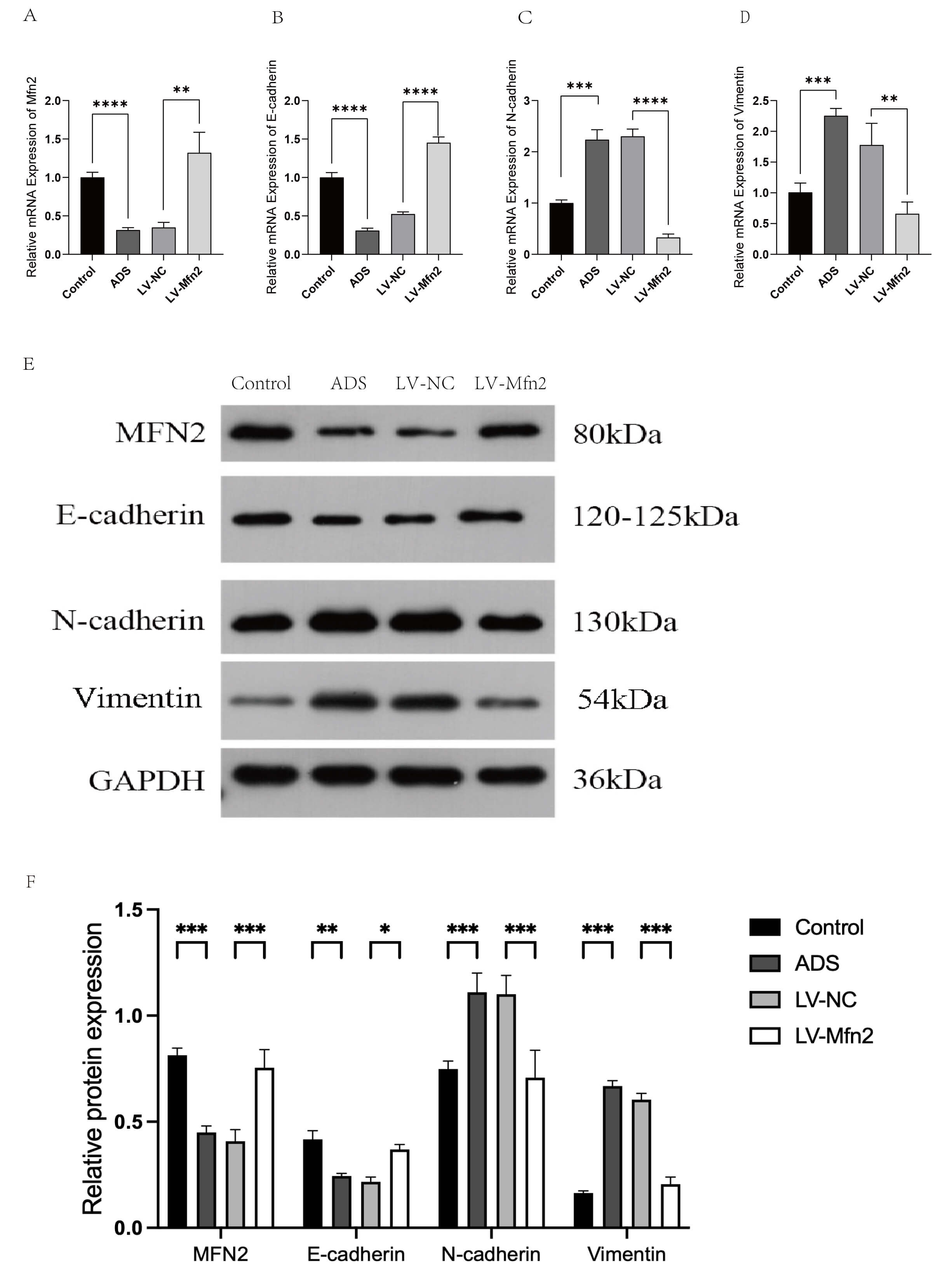 Fig. 5.
Fig. 5.
Method validation of relevant markers in mouse uterus by qPCR
and WB. (A–D) qPCR: the expression of E-cadherin, N-cadherin, Vimentin and Mfn2
mRNA in the uterus of the control vs model group (ADS) and Mfn2 negative control
(LV-NC) vs Mfn2 overexpression group (LV-Mfn2). Values represent mean
Together, our results show Mfn2 levels are consistently low in adenomyosis across different systems. Increasing Mfn2 counteracts the EMT process, reduces the invasive potential of endometrial cells, and significantly improves disease features in mice. This strongly indicates that Mfn2 plays a protective role in adenomyosis by inhibiting EMT, making it a promising potential target for treatment.
This study provides the first evidence that Mfn2 critically inhibits adenomyosis
progression by suppressing EMT. Through integrated clinical, cellular, and animal
analyses, we consistently demonstrated significant Mfn2 downregulation in
adenomyosis endometrium, which strongly correlated with aberrant EMT activation
(
Taken together with our findings, we propose a comprehensive hypothesis that Mfn2 may suppress ROS burst and reverse pathological metabolic reprogramming by inhibiting mitochondrial homeostasis (fusion/fission), ultimately inhibiting EMT. The basic mechanism may include the following interrelated levels:
1. Regulation of mitochondrial morphology and function: as a key regulatory
protein of mitochondrial outer membrane fusion, down-regulation of Mfn2
expression directly leads to excessive mitochondrial fission (fragmentation) [13, 23, 24]. The morphological defect impairs mitochondrial electron transport
chain (ETC) function, leading to the overproduction of ROS. Overproduced ROS act
as key signaling molecules, activating nuclear factor kappa B (NF-
2. Inhibition of metabolic reprogramming: Mitochondrial fragmentation not only leads to ROS accumulation (as described above), but also forces cells to forego efficient oxidative phosphorylation (OXPHOS) in favor of inefficient glycolysis (i.e., the Warburg effect) [25], a metabolic shift that is itself a hallmark feature of EMT. Overexpressed Mfn2 might restore the efficiency of OXPHOS by restoring the normal structure and function of mitochondrial cristae. At the same time, it is able to inhibit the activity of key glycolytic enzymes such as lactate dehydrogenase A (LDHA) [23, 26], effectively opposing the aberrant metabolic adaptation that EMT depends on.
3. Regulation of mitochondrial-endoplasmic reticulum coupling: In addition to regulating mitochondrial fusion, Mfn2 is also involved in the formation and maintenance of mitochondrial-endoplasmic reticulum contact sites (MERCs), which are important for calcium (Ca2+) signaling [27]. Calcium homeostasis disruption activates calcium-dependent proteases such as Calpain, which cleave E-cadherin and destabilize cell polarity, thereby inducing the EMT process. Therefore, Mfn2 could suppress the calpain-mediated EMT pathway through the stabilization of MERCs’ integrity and calcium signaling.
The major finding of the present study—that Mfn2 overexpression substantially
suppressed TGF-
In summary, this study demonstrates for the first time that Mfn2 plays a vital
protective role in adenomyosis by inhibiting the process of EMT, which introduces
new ideas for clinical diagnosis and treatment of adenomyosis. Studies have found
that the low expression mode of Mfn2 in the endometrium of patients shows that
Mfn2 can be used as an effective auxiliary diagnostic biomarker, and if combined
with serum EMT markers (such as Vimentin), it is expected to significantly
improve the accuracy of disease staging and invasiveness. In the therapeutic
field, therapies targeting Mfn2 have shown broad prospects. Small-molecule
compounds (e.g., natural product derivatives) with selective activation of Mfn2
can be developed through high-throughput screening and combined with in utero
sustained release systems to achieve lesion-precise intervention. Lentiviral or
adeno-associated virus (AAV) vector-mediated gene therapy has been shown to
improve pathological phenotypes in animal models. Maximizing delivery efficiency
or creating tissue-specific promoters may further improve efficacy and reduce
toxicity in the future. Of particular interest is whether the combination of Mfn2
activators with existing antifibrotic treatments, e.g., gonadotrophin-releasing
hormone (GnRH) agonists, can enhance efficacy through synergistic mechanisms.
However, it is still necessary to systematically tackle key problems to
facilitate clinical translation: to clarify the correlation of Mfn2 expression
with clinical stage and symptom severity, to stringently evaluate the in
vivo safety and off-target risk of Mfn2 agonists, and to fully explore the
mechanisms of interaction between Mfn2 regulating mitochondrial function and
Wnt/
Of course, we also know this study comes with several limitations that need to
be addressed in the future. For one, the current clinical sample size is rather
limited, and follow-up studies are urgently required to assess larger and more
representative patient cohorts to more reliably validate the role of Mfn2 in
adenomyosis and its clinical relevance. Although Ishikawa cells successfully
mimic the TGF-
For another, although the function of Mfn2 and its relationship with EMT have been preliminarily revealed, the exact molecular mechanism of Mfn2 remains to be fully revealed. Gene knockout and RNA interference can be employed in the future to manipulate Mfn2 expression in cells and animal models with high accuracy, and their downstream effector molecules and signaling pathways can be further analyzed. In addition, since the regulatory network of Mfn2 is predicted to involve multiple signaling pathways and complex molecular interactions, multi-omics analysis (e.g., transcriptomics and proteomics) will be a promising direction to systemically reveal the key mechanism and network association of Mfn2. Overcoming these limitations will be of great value to clarify a deeper insight into the pathophysiology of adenomyosis and also provide a good foundation for the further exploration of Mfn2-targeted therapeutic strategies.
This study confirms that Mfn2 is a key repressor of adenomyosis progression. We
consistently demonstrated that Mfn2 is significantly downregulated in human
adenomyosis endometrium, in a cell model of EMT, and in a mouse adenomyosis
model. Functionally, restoring Mfn2 expression effectively reversed
TGF-
The data supporting the results of this study are included in this paper and its online Supplementary Material.
XW: Responsible for the conceptualization and design of this study, and experimented. NNX: Responsible for writing the first draft of the manuscript and improving the study design. XQ: assisted in the H&E staining of histological samples, immunohistochemical staining, and the scoring of their results. QH and CD: participated in the execution of cell culture, animal model establishment, and tissue sampling. HY: supervised the whole research process, guided the implementation of the study, and analyzed and interpreted the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Ethics Committee of Yichang Central People’s Hospital approved this human subject/tissue sample study (Approval No. [2024-252-01]). All subjects gave written informed consent in compliance with the Declaration of Helsinki. Anonymity and confidentiality of the patients were strictly maintained during the study. All animal procedures were conducted according to the ARRIVE Guidelines for the Care and Use of Laboratory Animals and were endorsed by the Experimental Animal Ethics Committee of China Three Gorges University (Protocol No. [2024070I]). All animal experiments complied with ethical guidelines and the principles of Replacement, Reduction, and Refinement (3Rs). Efforts were made to minimize animal suffering, including the use of anesthesia during surgical procedures and the application of humane endpoints for euthanasia.
This study was strongly supported by the Central Laboratory of Wujia Branch of Yichang Central People’s Hospital in terms of research sites and equipment, which is greatly appreciated. At the same time, we sincerely thank the Laboratory Animal Center of China Three Gorges University for providing professional assistance in the feeding and management of laboratory animals and the technical operation of gavage.
This study was supported by the Natural Science Foundation of Hubei Province (Joint Fund Project: 2024AFD189).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG46464.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





