1 Department of Ultrasound Medicine, Wuxi People’s Hospital Affiliated to Nanjing Medical University, 214023 Wuxi, Jiangsu, China
Abstract
Gestational diabetes mellitus (GDM) elevates the risk of neonatal respiratory distress syndrome (NRDS), highlighting the need for robust predictive tools. Current assessments of fetal lung maturity assessments are invasive, creating a clinical demand for non-invasive alternatives. This study presents a dual-parameter framework that combines artificial intelligence (AI)-derived fetal lung texture signatures with uterine artery pulsatility index (PI) to predict NRDS risk in GDM pregnancies.
A prospective cohort of 50 patients with GDM patients was enrolled. Standardized four-chamber view ultrasound images were processed using a TensorFlow-based framework to extract 342 gray-level co-occurrence matrix (GLCM) texture features from the fetal lungs. A support vector machine (SVM) classifier was then employed for NRDS risk stratification. Concurrently, uterine artery PI was measured transvaginally following International Society of Ultrasound in Obstetrics and Gynecology (ISUOG) guidelines. The predictive performance of the AI model, uterine artery PI, and their combination was evaluated for predicting NRDS.
The uterine artery PI was significantly elevated in the NRDS group (n = 22) compared with controls (n = 28) (median 1.52 [interquartile range, IQR: 1.35–1.70] vs. 1.16 [IQR: 0.95–1.30]; p < 0.001). The standalone AI-based pulmonary texture analysis achieved 86.4% sensitivity and 78.6% specificity for NRDS prediction, with substantial agreement with clinical diagnosis (κ = 0.67). The synergistic integration of an AI-based high-risk classification with a uterine artery PI ≥1.28 yielded superior predictive performance, attaining 92.0% overall accuracy (46/50). Decision curve analysis confirmed that the combined model provided a superior net benefit across clinically relevant threshold probabilities (10%–50%).
The integration of AI-quantified fetal lung texture analysis with uterine artery Doppler hemodynamics provides a refined, non-invasive tool for NRDS risk stratification in pregnancies complicated by GDM. This dual-pathway framework effectively captures the interplay between placental vascular insufficiency and pulmonary immaturity, offering high diagnostic accuracy and clinical utility to guide perinatal management decisions.
Keywords
- gestational diabetes mellitus
- neonatal respiratory distress syndrome
- artificial intelligence
- ultrasonographic texture analysis
- uterine artery pulsatility index
Gestational diabetes mellitus (GDM) is a critical gestational metabolic disorder with a growing global prevalence. It imposes a dual burden due to its association with maternal complications such as preeclampsia and cesarean, as well as adverse neonatal outcomes [1] such as macrosomia, hypoglycemia, and neonatal respiratory distress syndrome (NRDS). The latter warrants particular attention given its pathogenesis rooted in pulmonary surfactant deficiency, which precipitates alveolar collapse and impaired ventilation. Crucially, emerging evidence indicates that maternal hyperglycemia simultaneously drives placental vascular dysfunction (elevated uterine artery pulsatility index (PI)) and impairs fetal surfactant synthesis [2, 3]. This dual-pathway etiology justifies our integrated hemodynamic-pulmonary framework and explains the sustained elevation of NRDS risk even at term gestations. This mechanistic duality underscores the imperative for advanced predictive strategies capable of identifying high-risk pregnancies early in the clinical course.
Although amniotic fluid phospholipid analysis has historically been considered the gold standard for diagnosing fetal lung maturity (FLM) [4], its invasive nature and associated complications, such as infection [5], preterm rupture of membranes, and fetal loss, limit routine clinical implementation [6]. Consequently, ultrasonography has emerged as a pivotal noninvasive alternative [7], with recent advances in artificial intelligence (AI) demonstrating remarkable potential for quantifying subtle parenchymal textures indicative of surfactant distribution [8]. These AI-driven approaches, exemplified by gray-level co-occurrence matrix (GLCM) feature extraction, enable objective characterization of microstructural patterns beyond human visual perception [9]. Nevertheless, current AI models predominantly focus on isolated pulmonary parameters, overlooking the integral role of uteroplacental hemodynamics in fetal lung development, a critical gap given the established correlation between GDM-induced placental vascular dysfunction and adverse respiratory outcomes.
Uterine artery PI serves as a robust hemodynamic marker of placental vascular resistance, frequently elevated in GDM cohorts due to endothelial dysfunction [10] and impaired trophoblastic invasion. Elevated PI values correlate significantly with fetal hypoxia and growth perturbations [11], yet their standalone predictive value for NRDS remains suboptimal. This limitation may stem from the complex, multifactorial nature of NRDS pathogenesis, wherein pulmonary maturational delays and placental insufficiency likely operate synergistically [12]. Thus, integrating quantifiable AI-based pulmonary texture signatures with dynamic uteroplacental flow parameters offers a novel systems-biology approach to risk stratification, potentially transcending the diagnostic ceiling [13] of unimodal assessments.
Our study pioneers this integrative methodology by developing a dual-parameter predictive framework that synthesizes TensorFlow-processed fetal lung texture analysis with transvaginally acquired uterine artery PI metrics [14]. This approach addresses the technical limitations of existing FLM evaluation tools. Furthermore, it establishes a physiological basis for understanding the interplay between metabolic dysregulation, vascular compromise, and pulmonary immaturity in GDM. Through this investigation, we establish a comprehensive evidence base for optimizing perinatal decision-making [15]—balancing the imperative for timely intervention against the risks of iatrogenic harm in this vulnerable population.
Approved by the Institutional Ethics Committee of Wuxi People’s Hospital
Affiliated to Nanjing Medical University (Approval No. 2021-1624011), this
prospective cohort enrolled 50 GDM patients between February 2021 and January
2023. Participants met diagnostic criteria per the 2022 Guidelines for Diagnosis
and Management of Hyperglycemia in Pregnancy [16], with gestational ages of
28+0 to 38+6 weeks scheduled for elective delivery. Inclusion criteria
comprised: (1) completion of AI-based FLM assessment and uterine artery Doppler
within 48 hours before delivery; (2) written informed consent. Exclusion criteria
included: (1) suboptimal ultrasound image quality; (2) concurrent preeclampsia,
fetal anomalies, or major complications; (3) requirement for emergency cesarean
delivery; (4) incomplete glucose monitoring records. After applying these
criteria, all 50 initially enrolled patients were included in the final
analytical cohort, which consisted of 22 NRDS cases and 28 controls. The cohort
(age range: 24–38 years; mean gestational age: 35.2
The detailed baseline clinical and biochemical characteristics of the study cohort are provided in Supplementary Material 1.
Two ISUOG-certified sonographers performed blinded examinations with strict masking of glycemic status and neonatal outcomes, using a Toshiba Aplio500 system with a convex probe (C251, 2–5 MHz). Specifically, sonographers were blinded to maternal glycemic control classification (optimal/suboptimal) and subsequent NRDS diagnosis during all image acquisition and PI measurements. Patients were positioned supine, with standardized four-chamber views ensuring complete bilateral lung fields within rib boundaries. Image acquisition disabled smoothing, frame correlation, and adaptive gain functions while activating tissue harmonic imaging (THI) to minimize artifacts. Three consecutive cardiac cycle cine-loops were stored in uncompressed DICOM format.
Quality control mandated: (1) image validation by two associate professors
excluding studies with
Qualified images were processed using TensorFlow 2.15 LTS (Google LLC, Mountain
View, CA, USA) with CUDA 11.8 (NVIDIA Corp., Santa Clara, CA, USA) acceleration. The framework was migrated from an initial
TensorFlow 2.10 implementation with validated output consistency (mean absolute
error
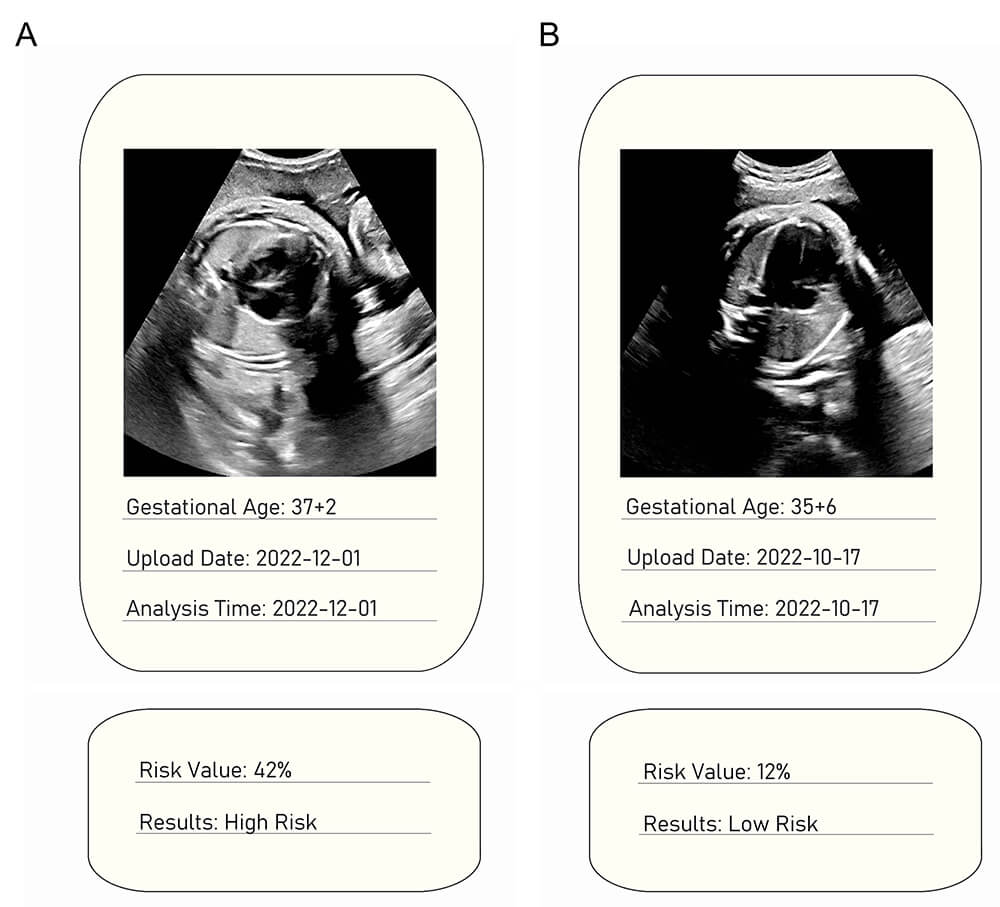 Fig. 1.
Fig. 1.
Representative ultrasound images with AI-based NRDS risk stratification. (A) Fetal ultrasound at 37+2 weeks gestation assessed as high risk (42.0% risk value). (B) Fetal ultrasound at 35+6 weeks gestation assessed as low risk (12.0% risk value).
Transvaginal measurements (PLT-705BT probe, 5–9 MHz) acquired uterine artery
spectra at the cervical internal os level. Pulse Doppler settings maintained a
2-mm sample volume and
The sample size was calculated a priori using GPower 3.1 software (version
3.1.9.7; Heinrich Heine University Düsseldorf, Düsseldorf, Germany) based
on expected group differences in uterine artery PI. Assuming a Cohen’s d effect
size of 0.8 (derived from prior GDM studies reporting median PI differences
Diagnostic performance metrics including sensitivity, specificity, and κ were calculated using contingency tables derived from 10-fold cross-validation. It is important to note the distinct purposes of the statistical corrections applied. The Benjamini-Hochberg procedure was employed during the initial, high-dimensional feature screening phase (as described in Section 2.2.2) to control the false discovery rate when evaluating 342 individual GLCM features. In contrast, the subsequent hypothesis tests (e.g., Mann-Whitney U tests for group comparisons) and model performance evaluations were conducted on a consolidated set of variables (the final model output or established clinical parameters). These latter analyses do not involve the same multiplicity issue as the initial feature screening, and therefore report uncorrected p-values to avoid excessive reduction in statistical power for these confirmatory tests. For the key primary outcome (comparison of uterine artery PI between NRDS and control groups), a post-hoc Bonferroni correction was also applied to confirm the robustness of the finding, given its central role in our hypothesis. Analyses used SPSS 26.0, R 4.3.1, and TensorFlow 2.15 LTS with CUDA 11.8 acceleration.
Following a comprehensive retrospective review of neonatal medical records applying the strict clinical, radiological, and laboratory criteria for NRDS (as defined in Methods), we confirmed the final cohort stratification of 22 NRDS cases and 28 controls. Consequently, the cohort stratification (22 NRDS cases vs. 28 controls) remained identical to the initial analysis.
The study cohort comprised 50 GDM patients with mean maternal age 28.6
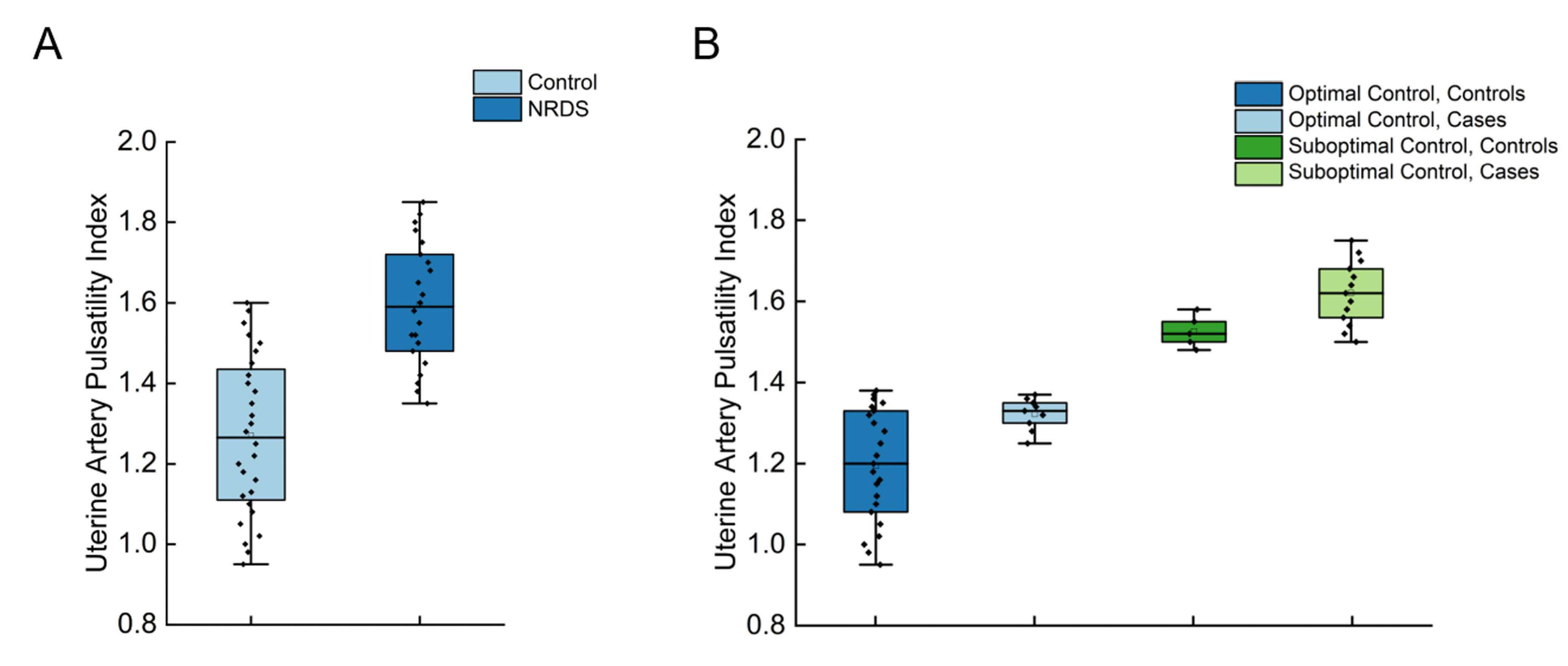 Fig. 2.
Fig. 2.
Uterine artery pulsatility index (UtA-PI) distribution
stratified by NRDS status and glycemic control. (A) Significantly elevated
UtA-PI in neonates with NRDS (n = 22) vs. controls (n = 28) (p
| Characteristics | Total (n = 50) | NRDS Group (n = 22) | Control Group (n = 28) | p-value | |
| Maternal age (years) | 28.6 |
28.9 |
28.3 |
0.512 | |
| Gestational age (weeks) | 35.2 |
34.8 |
35.5 |
0.226 | |
| Glycemic control, n (%) | 0.004 | ||||
| Optimal (FPG |
32 (64.0%) | 9 (40.9%) | 23 (82.1%) | ||
| Suboptimal | 18 (36.0%) | 13 (59.1%) | 5 (17.9%) | ||
| Delivery mode, n (%) | 0.569 | ||||
| Vaginal | 21 (42.0%) | 8 (36.4%) | 13 (46.4%) | ||
| Cesarean | 29 (58.0%) | 14 (63.6%) | 15 (53.6%) | ||
| Uterine artery PI | 1.32 [1.08–1.58] | 1.52 [1.35–1.70] | 1.16 [0.95–1.30] | ||
Notes: Data presented as mean
| Parameter | Optimal Control (n = 32) | Suboptimal Control (n = 18) | Effect size | p-value |
| Uterine artery PI | 1.21 [1.05–1.30] | 1.61 [1.50–1.75] | r = 0.62* | |
| NRDS incidence, n (%) | 9 (28.1%) | 13 (72.2%) | RR = 2.57 (1.35–4.88) | 0.004 |
| False-negative AI cases | 1 (3.1%) | 2 (11.1%) | - | 0.429 |
Notes: Data as median (IQR) or n (%). Uterine artery PI comparison: U = 63.5,
p
Uterine artery PI was significantly elevated in the NRDS group compared to
controls (median 1.52 vs. 1.16, Table 1; U = 155.0, p
Post hoc power analysis confirmed that with the observed PI effect size (r = 0.62, Cohen’s d = 1.59) and n = 50, the achieved statistical power exceeded 99.0% (GPower 3.1), substantially surpassing the initial 80.0% target.
The AI-derived pulmonary texture signatures identified NRDS cases with 86.4% sensitivity (19/22) and 78.6% specificity (22/28). Model-clinical diagnosis concordance was substantial (κ = 0.67, 95% CI: 0.61–0.89), validating algorithmic reliability. The standalone AI classification achieved an overall accuracy of 82.0%. The corresponding confusion matrix, delineating true positives, false negatives, false positives, and true negatives, is provided in Fig. 3.
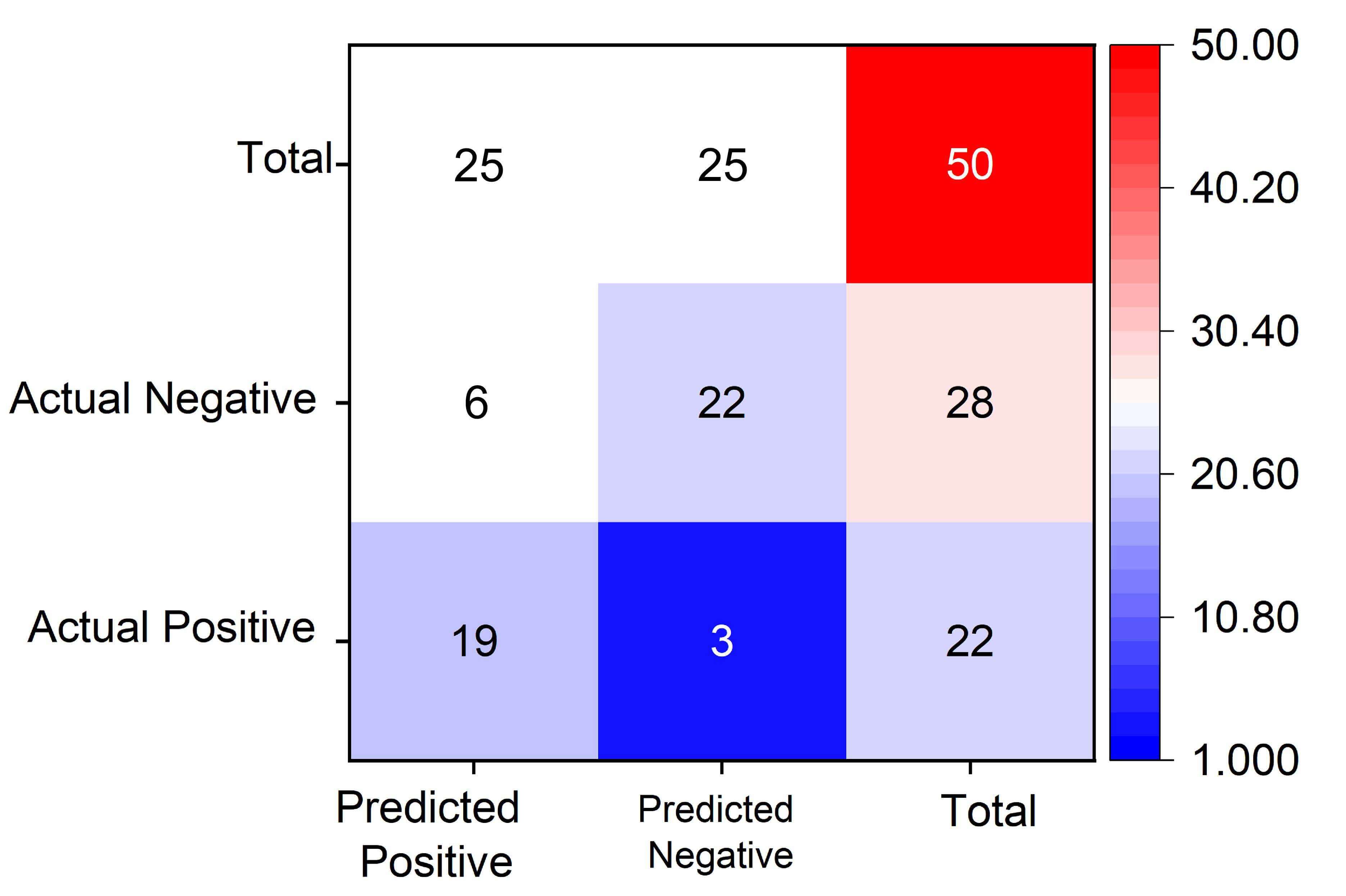 Fig. 3.
Fig. 3.
Confusion matrix evaluating AI model performance for neonatal respiratory distress syndrome (NRDS) prediction. The matrix compares actual clinical diagnoses (rows) against model predictions (columns) in the test cohort (n = 50 neonates), demonstrating 19 true positives (NRDS correctly predicted), 22 true negatives (controls correctly identified), 3 false negatives (NRDS cases missed), and 6 false positives (controls misclassified as NRDS). Key performance metrics derived include: sensitivity = 19/(19+3) = 86.4% (ability to detect NRDS cases), specificity = 22/(22+6) = 78.6% (ability to exclude controls), and overall accuracy = (19+22)/50 = 82.0%. The model shows stronger precision in NRDS identification than control group discrimination. TP, true positives; FN, False Negative; FP, False Positive;TN, True Negative.
The composite diagnostic criterion (concurrent AI high-risk classification and
PI
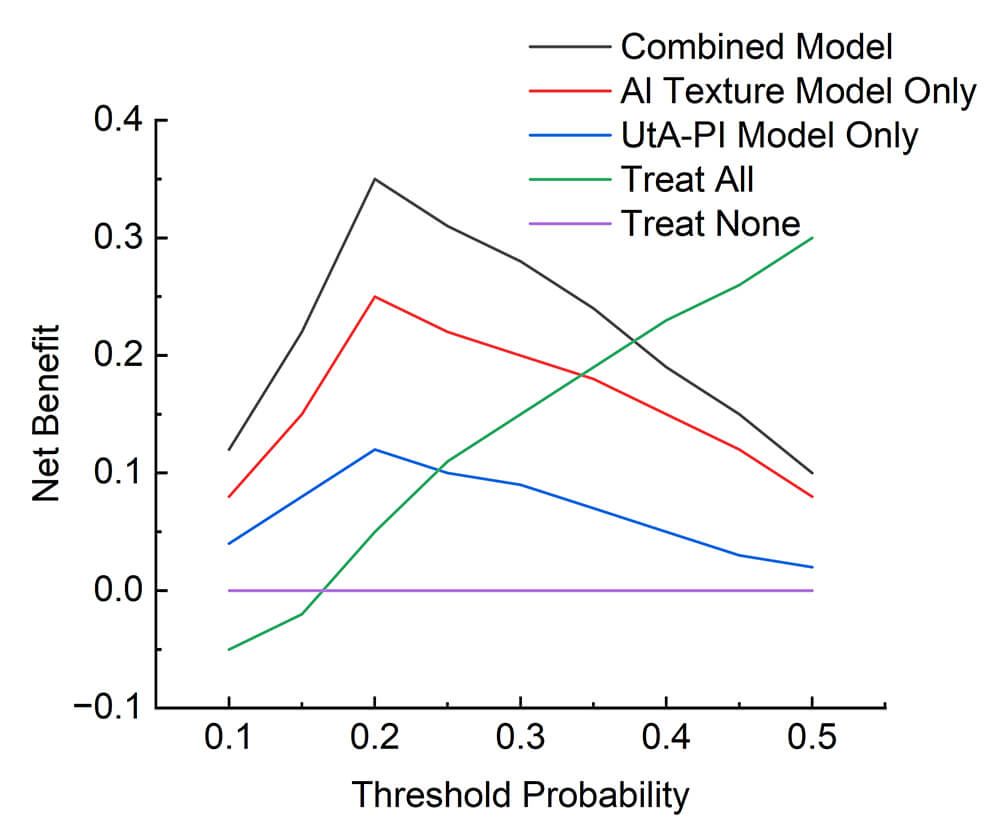 Fig. 4.
Fig. 4.
Decision curve analysis (DCA) evaluating the clinical utility of predictive models for neonatal respiratory distress syndrome (NRDS). The net benefit (y-axis) of intervention strategies derived from the combined model (black), AI texture model only (red), UtA-PI model only (blue), treat-all approach (green), and treat-none strategy (purple) is plotted against threshold probabilities ranging from 10.0% to 50.0% (x-axis). The integrated (combined) model demonstrates superior net benefit compared to both standalone models across the entire threshold range (10.0%–50.0%). Crucially, at clinically actionable intervention thresholds exceeding 15.0%, the net benefit of the combined model is significantly higher than that of the best unimodal alternative (either AI or PI model). The combined model achieves a peak net benefit of 0.35 at the 20.0% threshold probability.
| Model | Sensitivity (95% CI) | Specificity (95% CI) | Accuracy (95% CI) | κ (95% CI) |
| Composite (AI + PI) | 90.9% (71.0–98.9) | 92.9% (82.0–98.0) | 92.0% (81.0–98.0) | 0.82 (0.70–0.94) |
| AI texture only | 86.4% (65.1–97.1) | 78.6% (59.0–91.7) | 82.0% (68.6–91.4) | 0.67 (0.61–0.89) |
| Uterine artery PI only | 82.0% (68.9–91.0) | 75.0% (55.1–89.3) | 76.0% (61.8–87.0) | 0.52 (0.38–0.66) |
Thresholds: PI cutoff = 1.28; AI high-risk defined by SVM classification. McNemar test: composite vs. PI (p = 0.002), composite vs. AI (p = 0.021). SVM, support vector machine.
Decision curve analysis (DCA) quantified the net benefit profile across therapeutically relevant threshold probabilities (10.0%–50.0%). The integrated model consistently demonstrated superior net benefit compared to both standalone AI and standalone PI prediction methods across this range. Crucially, at clinically actionable intervention thresholds exceeding 15.0%, the net benefit of the combined model was significantly higher than that of the best unimodal alternative. Fig. 4 graphically validates these results, illustrating the net benefit superiority of the combined model across threshold probabilities (10.0%–50.0%), with peak net benefit of 0.35 at 20.0% threshold. This analysis confirms the enhanced clinical utility of the integrated diagnostic paradigm for guiding perinatal management decisions in GDM pregnancies at risk for NRDS.
This investigation proposes a novel dual-parameter diagnostic framework, integrating AI-quantified fetal lung texture signatures with uterine artery Doppler hemodynamics, to substantially enhance the prediction of NRDS in GDM. This approach effectively captures the synergistic pathophysiological interplay between placental vascular insufficiency and pulmonary immaturity, with the former quantified by elevated uterine artery PI (median: 1.52 vs. 1.16, Cohen’s r = 0.62) and the latter evidenced by AI-derived lung texture analysis (sensitivity: 86.4%, specificity: 78.6%). The foundational role of hyperglycemia was further confirmed by the strong association between suboptimal glycemic control and NRDS incidence (p = 0.004). The composite model achieved exceptional diagnostic accuracy (92.0%) by synergistically integrating placental hemodynamics and pulmonary immaturity markers. This mechanistic alignment with established GDM pathophysiology provides compelling biological plausibility for the model’s predictive efficacy [3], by demonstrating how maternal hyperglycemia concurrently impairs uteroplacental perfusion and disrupts fetal surfactant synthesis.
Our framework connects elevated uterine artery PI and AI-assessed pulmonary immaturity to predict NRDS in GDM. This association is context-specific: while in preeclampsia high PI often accelerates lung maturation via cortisol-mediated surfactant release, in GDM, hyperglycemia-induced fetal hyperinsulinemia suppresses surfactant synthesis. Thus, in GDM, placental dysfunction coincides with, rather than counteracts, metabolically driven pulmonary delay. This mechanistic distinction underscores the specificity of our dual-pathway model, which captures the synergistic detriment of both hemodynamic and metabolic impairment, justifying the combined use of Doppler and pulmonary texture analysis for GDM-specific risk assessment.
The AI-driven pulmonary phenotyping exhibited substantial concordance with
clinical NRDS diagnosis (
Decision curve analysis (DCA) quantitatively validated the superior clinical utility of the integrated model across therapeutically relevant threshold probabilities (10.0%–50.0%) [23]. The model demonstrated a peak NB of 0.35 at a 20.0% intervention probability, translating to the prevention of approximately 8 unnecessary antenatal corticosteroid courses per 100 patients while concurrently avoiding 2 missed high-risk cases [24]. This represents a decisive advantage over invasive diagnostic benchmarks such as amniocentesis [25]. The model’s sustained 1.4-fold higher net benefit compared to the best unimodal predictor at thresholds exceeding 15.0% underscores its pivotal role in guiding evidence-based perinatal interventions [26].
The transition to TensorFlow 2.15 LTS during manuscript preparation addressed end-of-life concerns for the original 2.10 version. Rigorous validation confirmed no impact on feature extraction or classification outcomes (Bland-Altman 95% limits of agreement: –0.03 to 0.02 for prediction probabilities).
Several methodological limitations warrant consideration. The single-center design and modest cohort size (n = 50) constrain immediate generalizability and necessitate external validation in larger, multiethnic populations [27]. Furthermore, potential confounding factors were not fully adjusted for in our analysis. These include not only established variables such as maternal body mass index [28], insulin therapy intensity, and fetal growth trajectories, but also the specific indications for elective premature delivery. Although major comorbidities were excluded, residual confounding from unreported pathologies associated with early delivery could independently influence NRDS risk. Additionally, the explanation of false-negative cases via a “metabolic threshold” remains speculative due to a lack of quantitative metabolic profiling, such as glycated hemoglobin (HbA1c), glucose variability, or insulin dosage, which limits our ability to precisely define the level of dysregulation in misclassified cases. Future studies should incorporate detailed metabolic, clinical, and delivery indication data to improve model specificity, explanatory power, and population-specific risk calibration [29].
This study successfully establishes and validates a novel, non-invasive framework that integrates AI-based fetal lung texture analysis with uterine artery PI for stratifying NRDS risk in GDM pregnancies. The model demonstrated high diagnostic accuracy, a robust net benefit profile across clinical decision thresholds, and substantial agreement with clinical diagnosis, confirming its potential as a physiologically grounded tool. This paradigm offers a compelling strategy to optimize perinatal management by balancing the imperative for timely intervention against the risks of iatrogenic harm [30] in this vulnerable population.
All data associated with this study and the custom code developed for the AI model are available upon reasonable request from the corresponding author.
JZ and YD designed the research study. JS and BL performed the research, including patient recruitment and ultrasound examinations. BL developed the AI model and performed feature extraction. JZ and JS analyzed the data and interpreted the results. YD supervised the project and provided critical revision of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Ethical oversight for this research was managed by the Medical Ethics Committee at Wuxi People’s Hospital Affiliated to Nanjing Medical University (Approval No. 2021-1624011). Written informed consent was obtained from all participants before their participation. Blood collection and data handling followed ethical standards set by the Declaration of Helsinki.
The authors would like to thank the clinical and nursing staff of the Department of Ultrasound Medicine at Wuxi People’s Hospital for their assistance in patient recruitment and data collection. We also extend our gratitude to our colleagues in the IT and technical support departments for their assistance with computational infrastructure. Finally, we thank the peer reviewers and editors for their valuable comments and suggestions which have significantly improved this manuscript.
This study was supported by the Wuxi Municipal Double Hundred Young and Middle-Aged Reserve Top Talents Program in Medical and Health Fields (Grant No. HB2023001) and the Jiangsu Provincial Health Commission Scientific Research Fund Project (Grant No. X202336).
The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGpt-3.5 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG46350.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




