1 Department of Obstetrics and Gynecology, Catholic University of Daegu School of Medicine, 42472 Daegu, Republic of Korea
Abstract
Minimally invasive cosmetic procedures, including soft tissue filler injections, are increasingly performed worldwide. While complications such as inflammation, migration, and granuloma formation have been reported, pregnancy-related complications resulting from dermal filler migration have not yet been documented.
We describe the first known case of a 45-year-old primigravida with extensive breast filler migration, originally injected 17 years before conception, that extended to the lower abdomen and complicated both prenatal ultrasound evaluation and cesarean delivery. By 36 weeks of gestation, the migration had extended to the vulvar region. At 36 weeks and 2 days, an elective cesarean section was performed. Pelvic magnetic resonance imaging (MRI) and intraoperative findings demonstrated filler deposits between the subcutaneous tissue and the fascial layer, which restricted fascial mobility and hindered fetal extraction. Although additional fascial incisions were required to facilitate delivery, the neonate experienced no postnatal complications, and the maternal postoperative course was uneventful.
This case highlights the importance of thorough cosmetic history-taking in pregnant women and emphasizes the need for clinical awareness and guidelines that address the long-term obstetric implications of dermal filler migration.
Keywords
- cesarean section
- dermal fillers
- pregnancy complications
- tissue adhesions
- ultrasonography
- prenatal
Minimally invasive cosmetic procedures have become increasingly prevalent worldwide, with soft tissue filler injections among the most frequently performed interventions. These fillers are commonly administered to areas such as the face, lips, breasts, and buttocks. However, a significant proportion of these procedures and the materials used lack official approval, which limits the availability of objective data on safety and long-term outcomes. Complications following filler injections are well reported in the literature. Reported adverse events include localized inflammation, infection, filler migration, subsequent degradation (degeneration) of migrated material, and persistent pain [1, 2, 3].
Complications associated with breast implants inserted prior to pregnancy have been reported during gestation, likely due to hormonal changes and physiological transformations. These complications include lactation difficulties, seroma formation, and breast deformation. Rare associations with intrauterine growth restriction have also been described [1, 2, 4, 5, 6, 7]. However, to date, no reports have described pregnancy-related complications arising from dermal filler injections administered prior to conception. Unlike breast implants, which remain encapsulated within mammary tissue, dermal fillers can infiltrate soft tissue planes, migrate to distant sites, and pose risks of vascular occlusion or embolization.
Herein, we report the first documented case of extensive breast filler migration into the lower abdomen, which significantly interfered with prenatal evaluation and cesarean delivery. This case highlights the importance of obtaining a detailed cosmetic procedure history in pregnant patients and emphasizes the need for careful clinical awareness of the potential long-term sequelae associated with dermal filler use during pregnancy.
A 45-year-old primigravida at 17+2 weeks of gestation was referred from a local medical center for detailed evaluation. Antenatal screening conducted at the referring center revealed an elevated alpha-fetoprotein level of 2.5 multiples of the median, which warranted a detailed ultrasound examination. However, clinicians at the referring center reported that adequate fetal ultrasonography could not be performed for reasons that were not initially identified.
At our institution, repeat ultrasound failed to clearly visualize the fetus, as the maternal lower abdomen was obscured by a large, ill-defined hypoechoic, mass-like area, suggestive of a non-native soft tissue component interfering with acoustic penetration. The mass appeared to occupy the subcutaneous and potentially deeper fascial planes, although precise delineation was limited by the imaging modality (Fig. 1A,B). After conducting fetal ultrasound examinations over an extended period and from multiple angles, no structural abnormalities were detected in the fetus, and no abnormal findings were observed in the amniotic fluid, although placenta previa was noted.
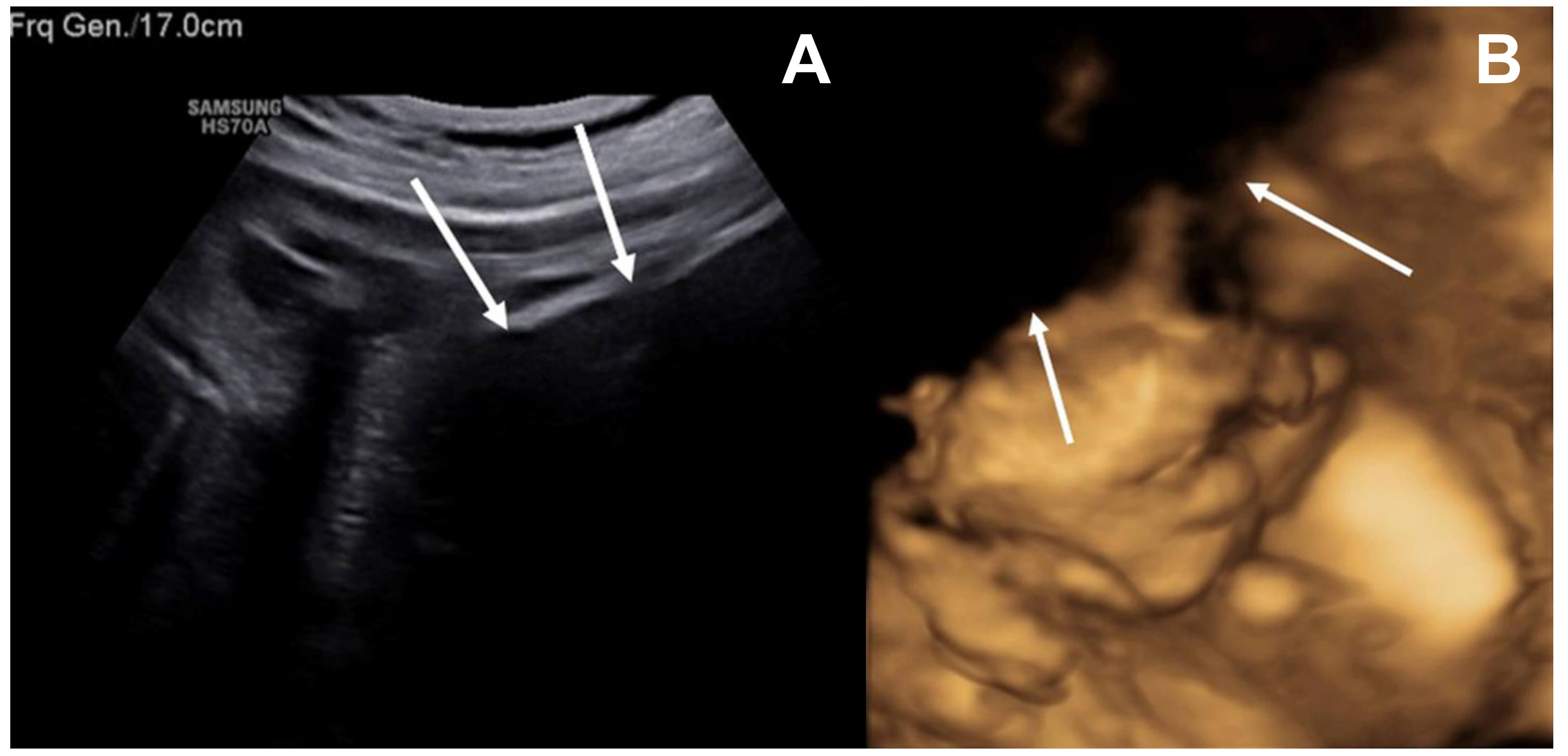 Fig. 1.
Fig. 1.
Maternal lower abdominal hypoechoic mass limiting acoustic penetration on fetal ultrasonography. (A) Ultrasound imaging was limited by a large hypoechoic mass-like lesion in the maternal lower abdomen, which impeded adequate acoustic penetration. (B) A portion of the fetal face was not visualized on the three-dimensional ultrasound due to the presence of a maternal lower abdominal hypoechoic mass-like opacity. White arrows highlight the hypoechoic mass-like region interfering with acoustic penetration.
Detailed history revealed no underlying medical conditions or prior surgical history, but the patient reported receiving breast filler injections 17 years prior. She first noticed filler migration approximately 1–2 years after the procedure. On abdominal palpation, firm, irregular masses—presumed to be migrated filler—were palpable in the lower abdomen. No signs of infection, such as redness, tenderness, or edema, were observed.
The patient underwent routine prenatal care, and placenta previa persisted, prompting a planned cesarean delivery. From 34 weeks’ gestation, she developed elevated blood pressure and proteinuria, consistent with preeclampsia. Cesarean delivery was scheduled to be performed at approximately 36 weeks of gestation. To assess filler distribution, its relationship with the abdominal wall, and the extent of placental invasion, pelvic magnetic resonance imaging (MRI) was performed. MRI revealed hyperechoic fluid collections containing small, round, bubble-like particles located between the subcutaneous tissue and the fascial layer of the abdominal wall (Fig. 2).
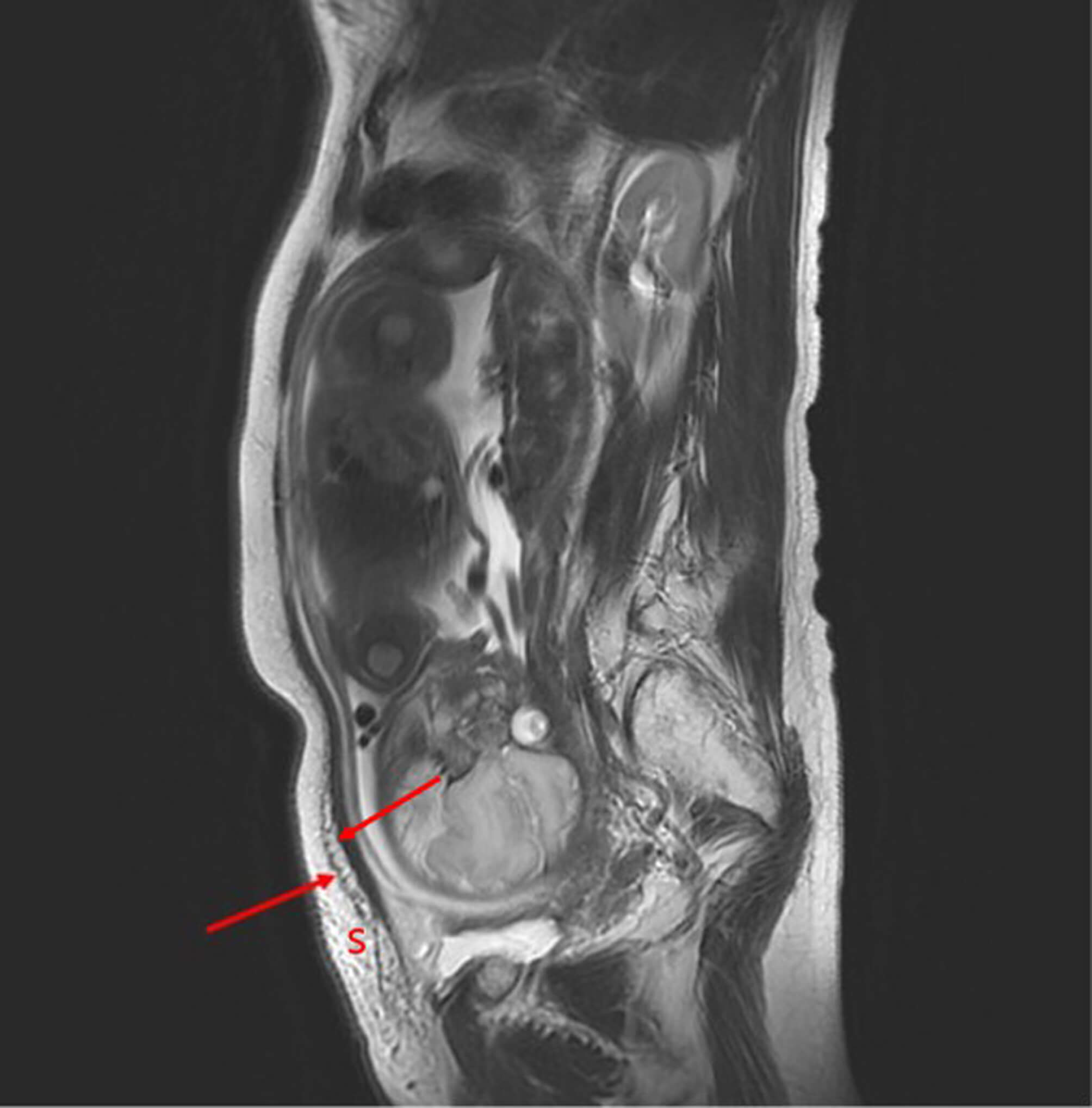 Fig. 2.
Fig. 2.
Sagittal non-enhanced pelvic MRI demonstrating abdominal wall fluid collection. Sagittal non-enhanced pelvic MRI revealed hyperintense fluid collections containing small, round, bubble-like particles located between the subcutaneous tissue and fascial layer of the abdominal wall (red arrow). S, subcutaneous tissue; MRI, magnetic resonance imaging.
At 36 weeks and 2 days, an elective cesarean section was performed under spinal anesthesia. A Pfannenstiel incision was made, and upon dissection through the subcutaneous layer, multiple translucent globules approximately 5 mm in diameter, suspected to be filler nodules, were observed. These nodules were diffusely distributed along the undersurface of the subcutaneous tissue, adherent to the fascial layer, and extended along its surface (Fig. 3). Some ruptured globules released a sticky, gelatinous fluid. During fetal extraction, multiple granules adhered to the maternal abdominal wall, significantly restricting fascial relaxation and complicating fetal delivery. Therefore, delivery was achieved only after performing additional incisions on both sides of the maternal abdominal wall, involving the fascia, subcutaneous tissue, and peritoneum.
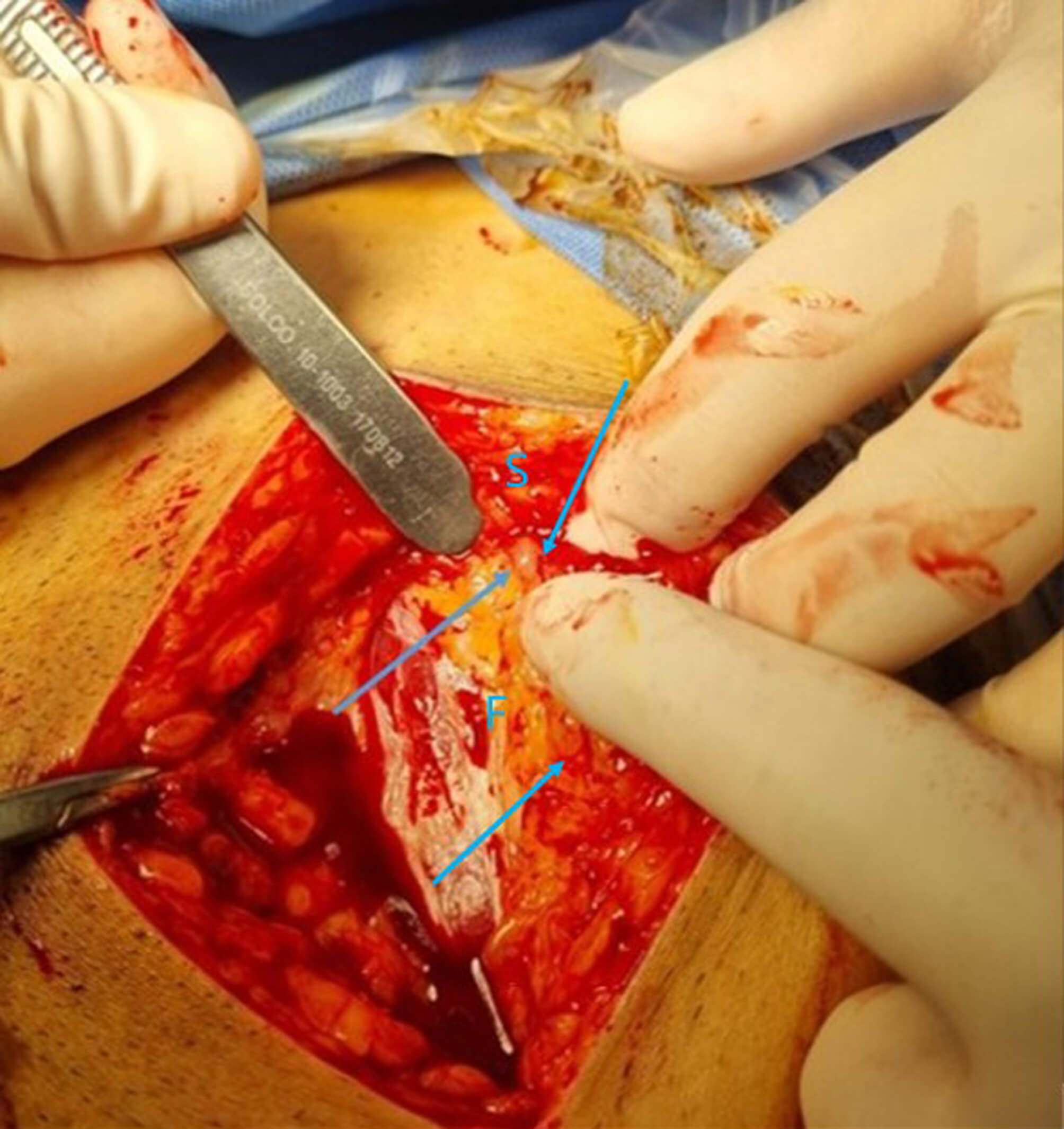 Fig. 3.
Fig. 3.
Gross appearance of subcutaneous filler nodules. Small, translucent filler nodules, approximately 5 mm in diameter, were observed beneath the subcutaneous tissue, adjacent to the fascial layer (blue arrow). S, subcutaneous tissue; F, fascia.
A male infant weighing 3400 grams was delivered with Apgar scores of 8 and 10 at 1 and 5 minutes, respectively. Placental removal and uterine closure were performed using standard cesarean techniques. The globular foreign material in the abdominal wall showed no signs of inflammation and was densely adherent to the subcutaneous tissue; therefore, it was left in situ, and fascial closure was completed. Postoperatively, the maternal wound healed without complications, and sutures were removed on postoperative day 10. Postpartum breast examination revealed no palpable residual filler or local inflammatory changes. Breastfeeding was not initiated at the patient’s request. One-month outpatient follow-up showed no postoperative complications.
In this case, we report a patient who experienced significant difficulties in prenatal evaluation and delivery due to long-term migration of a breast filler—originally administered 17 years prior to conception—that had descended to the lower abdomen. Although complications associated with dermal fillers have been well documented, this case is, to the best of our knowledge, the first to describe obstetric complications of migrated filler material, specifically its impact on prenatal ultrasound imaging and the delivery process.
In many countries, women undergo filler injections for cosmetic purposes, and this trend appears to be increasing. Adverse effects following filler administration range from mild to severe and can be classified as early-onset (within one week of injection) or mid-to-late-onset complications. Compared with absorbable fillers, such as collagen, hyaluronic acid, and poly-L-lactic acid, non-absorbable fillers, including polyalkylimide, silicone, and paraffin, are associated with cause more prolonged adverse reactions [8, 9]. Among these, filler migration is recognized as a late-onset complication, occurring weeks to years after injection [2, 8]. Filler migration is influenced by multiple factors, including injection technique, filler volume, gravitational effects, massage or external pressure, and lymphatic spread [1, 3, 10]. Accordingly, breast fillers—often administered in relatively large volumes and subject to greater gravitational and mechanical forces—may be more prone to migration. Furthermore, unapproved or improperly performed procedures may significantly increase the risk of such complications.
Prior studies have described local and distant migration of filler material, particularly in cosmetic and radiologic contexts [2, 3, 8]; however, no previous reports have evaluated its clinical significance during pregnancy. While breast implant complications during pregnancy have been reported, they typically involve encapsulated devices within the mammary tissue and rarely affect abdominal wall compliance [4, 5, 6, 7]. In contrast, filler materials—especially non-absorbable types—can infiltrate soft tissue planes and migrate extensively, posing risks during abdominal procedures.
In this case, the patient experienced considerable difficulty during prenatal ultrasound examinations. During cesarean delivery, granulated filler nodules were found firmly adherent between the subcutaneous tissue and fascial layer, restricting abdominal wall expansion and complicating fetal delivery. Although individual filler particles may not exert significant compressive effects, chronic granulomatous inflammation and fibrosis surrounding the filler nodules can result in fascial stiffness and reduced abdominal wall compliance. In this case, diffuse adhesion of filler granules along the fascial plane likely reduced fascial elasticity during abdominal wall stretching, thereby hindering fetal extraction. This represents a novel obstetric complication stemming from a minimally invasive cosmetic procedure performed years before pregnancy.
Chiang et al. [8] describe how dermal fillers may induce granulomatous reactions and fibrosis, which can result in tissue stiffness and altered surgical planes, especially when fillers are located near fascial structures. Similarly, Carella et al. [11] report that permanent fillers embedded in deep soft tissue layers often require surgical removal due to functional impairment and adhesion. In the case we report, the filler deposits were adherent to the fascial layer, and their consistency and location directly hindered fascial exposure and fetal extraction.
Abdominal wall stiffness resulting from prior filler injections presents a distinct surgical challenge, differing from adhesions caused by previous abdominal surgeries. Unlike postoperative adhesions, filler material may migrate under the influence of gravity and, when present in large volumes, may produce extensive adhesion in dependent regions. Furthermore, adhesions from prior abdominal surgeries typically involve the peritoneum or intra-abdominal structures, most commonly between the anterior uterine wall and the bladder, where careful dissection often permits fetal delivery without major difficulty. In addition to these mechanical effects, vascular complications represent one of the most serious risks associated with injectable fillers. Inadvertent intravascular injection or vascular compression can cause ischemia, necrosis, or embolization. Although such events are rare in the breast or abdominal regions, these mechanisms emphasize the potential for systemic and obstetric implications when filler material migrates extensively [12, 13].
With the increasing prevalence of cosmetic procedures across all age groups, obstetricians are more likely to encounter filler-related complications during prenatal care. Gravitational forces can cause filler migration not only to the lower abdomen but also to the vulvar and perineal regions, potentially interfering with vaginal delivery. In this patient, palpation at 36 weeks of gestation, shortly before delivery, revealed that the migrated filler had extended to the vulvar region. These findings highlight an unrecognized interaction between prior aesthetic procedures and obstetric outcomes, warranting careful medical surveillance and multidisciplinary management.
Therefore, thorough history-taking during prenatal care, including documentation of minimally invasive cosmetic procedures, is important. In pregnant patients with a history of dermal filler procedures, it is essential to determine the type of filler used and whether it is absorbable. In cases involving unknown dermal filler materials, high-resolution (5–17 MHz) color Doppler ultrasound can serve as a useful diagnostic tool for characterizing filler deposits and assessing their distribution [14]. The patient should be queried about any changes in the area where the filler was injected, and if necessary, a physical examination should be performed. If unexplained areas of increased opacity are observed during prenatal ultrasound, filler migration should be considered. When the affected region could influence critical obstetric evaluation, intervention, or delivery planning, further imaging, such as MRI, may be warranted. Subsequent careful consultation and appropriate planning of the delivery method and surgical approach are strongly recommended. In cases of cesarean delivery, clinicians should be aware of associated risks, including inflammatory reactions, postoperative wound infection, and granuloma formation [9, 15]. These factors warrant careful postoperative monitoring and management.
Awareness and interdisciplinary collaboration with dermatologists or plastic surgeons may be necessary to optimize patient outcomes. Additionally, establishing clinical guidelines for evaluating prior filler use and its potential impact on pregnancy could help standardize care. These considerations extend beyond cesarean sections and should also apply to other abdominal surgical procedures in patients with a history of dermal filler injections. Further research is necessary to determine the prevalence and long-term consequences of filler migration in pregnant patients.
Histopathologic confirmation of the filler material was not performed, and the absence of specimen collection constitutes a limitation of this report.
This case highlights the importance for obstetricians of recognizing the potential obstetric implications of prior minimally invasive cosmetic procedures. Migrated dermal filler material can interfere with prenatal evaluation and delivery mechanics, even years after injection. Thorough documentation of cosmetic history, awareness of potential filler migration, and multidisciplinary planning are essential for ensuring maternal and fetal safety. Establishing clinical guidelines for evaluating pregnant women with a history of filler injection may help prevent unexpected surgical complications and improve obstetric outcomes.
All datasets on which the conclusions of a manuscript are available to readers.
HJL: Data Curation, Visualization; JYB: Conceptualization, Investigation, Methodology, Supervision, Writing—Original Draft, Preparation, Writing—Review & Editing. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This retrospective case report is exempt from deliberation by the Institutional Ethics Committee of Daegu Catholic University Hospital Ethical Committee policy. We received informed consent from the patient. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration, including its later amendments or comparable ethical standards.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG47219.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



