1 Human Medicine Academic Program, Faculty of Health Sciences, Universidad Privada Norbert Wiener, 15046 Lima, Peru
Abstract
Platelet activation is central to preeclampsia (PE) pathobiology. We evaluated whether mean platelet volume (MPV) is independently associated with PE and quantified its individual discriminatory performance in a hospital-based case-control study in Peru.
We included 112 women with PE and 112 normotensive controls who delivered at the National Maternal Perinatal Institute (Lima) in 2024. MPV was obtained from the first complete blood count (CBC) performed at ≥20 gestational weeks. Logistic regression models estimated adjusted odds ratios (aORs) with 95% CIs. Receiver operating characteristic (ROC) analysis provided the area under the curve (AUC) with 95% CI and the optimal cutoff by Youden’s index.
Higher MPV was independently associated with PE (aOR: 2.16 per 1 fL; 95% CI: 1.55–3.01; p < 0.001). Gestational obesity (aOR: 2.43; 95% CI: 1.17–5.26; p = 0.039) and inadequate prenatal care (<6 visits; aOR: 3.71; 95% CI: 1.27–7.55; p = 0.006) were also associated with PE. MPV showed moderate discrimination (AUC: 0.686; 95% CI: 0.616–0.755). Using a cutoff of 8.80 fL, the sensitivity was 61.6%, the specificity was 71.4%, the positive likelihood ratio was 2.16, and the negative likelihood ratio was 0.54.
MPV is independently associated with PE and provides moderate individual discrimination. As MPV is routinely measured by hematology analyzers at no additional cost, incorporating it into trimester-specific, multivariable antenatal risk models may support cost-effective risk stratification in resource-limited settings.
Keywords
- preeclampsia
- mean platelet volume
- biomarkers
- maternal–fetal medicine
- SDG 3: good health and well-being
Hypertensive disorders of pregnancy, particularly preeclampsia (PE), are major
contributors to adverse outcomes, causing approximately 76,000 maternal deaths
and over 500,000 perinatal and infant deaths each year worldwide [1, 2, 3]. Platelet
activation, aggregation, and consumption at sites of endothelial injury are
central to PE pathobiology, supporting the exploration of platelet-based metrics
as accessible tools for risk stratification and screening [4]. A meta-analysis of
25 studies found lower platelet counts [pooled weighted mean difference (WMD): –41.45
Despite these advances, uncertainties remain regarding optimal timing,
thresholds, and clinical contexts for applying platelet indices [7]. In a large
population-based analysis, higher first-trimester MPV categories were associated
with gestational hypertension [9.8–10.3 fL: odds ratio (OR): 1.23;
Further heterogeneity is observed in cross-sectional data from Ethiopia, where
higher MPV (10.6
We conducted an analytical, hospital-based case-control study at the Instituto Nacional Materno Perinatal (INMP), a national tertiary referral center for obstetric care in Lima, Peru. The study was conducted in the Obstetrics Hospitalization Service during 2024. The INMP receives high-complexity maternal cases from across the metropolitan area and the national referral network, ensuring access to standardized clinical records and laboratory information relevant to the exposure and outcome under study.
Cases comprised pregnant women aged
Using Epidat 4.2 and INMP statistical yearbook parameters, assuming a 1:1 case-control ratio, we set: exposure among cases p1 = 0.46, exposure among controls p2 = 0.28, detectable OR = 3.06, 95% confidence, 80% power, and one control per case. This yielded a total sample of 224 participants (112 cases and 112 controls).
Data were abstracted from medical records and the standardized prenatal control
form. The MPV was taken from the first CBC performed at
All CBCs were processed in the INMP central laboratory using a single automated hematology analyzer (Sysmex XN-1000, Sysmex Corporation, Kobe, Japan), following the institution’s routine internal quality control program and participation in external proficiency testing. Peripheral blood samples were collected in K2-EDTA tubes (BD Vacutainer®, Cat. No. 367863; Becton, Dickinson and Company, Franklin Lakes, NJ, USA) and analyzed within a maximum of 2 hours after venipuncture, in accordance with the manufacturer’s recommendations. Thus, all MPV measurements were obtained under standardized analytical and pre-analytical conditions within the same platform.
The primary outcome was PE, recorded as a binary indicator (case vs. control)
based on the institutional protocol and aligned with international guidelines. PE
was defined as new-onset hypertension after 20 gestational weeks (systolic blood
pressure
The main exposure was MPV, expressed in fL and obtained from the first
CBC at
Covariates defined a priori were: maternal age (18–20, 21–34,
Pre-pregnancy obesity was defined as a body mass index (BMI)
Analyses were performed in Stata 17.0 (StataCorp, College Station, TX, USA).
Categorical variables were summarized as n (%) and continuous variables as
median (IQR) given their non-normal distributions. Group comparisons used
Pearson’s chi-square (or Fisher’s exact when appropriate) [15] for categorical
variables and the Wilcoxon rank-sum test for continuous variables [16]. Variables
with p
As a sensitivity analysis to address the potential impact of quasi-complete separation due to the low prevalence of prior PE, we fitted a penalized (Firth) logistic regression model including history of PE as an additional covariate. We report penalized aORs with 95% CIs to evaluate the robustness of the association between MPV and PE.
Given the potential time-dependent variation in MPV, we additionally adjusted the main multivariable model for gestational age at blood sampling. When available, we also evaluated models including systolic and diastolic blood pressure at admission as continuous covariates.
To assess internal validity, the AUC and Youden-derived MPV cutoff were evaluated using bootstrap resampling with 1000 iterations to obtain optimism-corrected estimates and 95% CIs. Model calibration was examined using the Hosmer–Lemeshow goodness-of-fit test and visually inspected with a calibration plot.
There were no missing data for MPV, PE status, or included covariates; all 224 women were retained in the analyses.
The study included 224 women (112 cases and 112 controls). Age distribution was
18–20 years in 12 (5.36%) cases, 21–34 years in 151 (67.41%) cases, and
| Characteristics | Total n (%) | |
| Age | ||
| 18–20 years | 12 (5.36) | |
| 21–34 years | 151 (67.41) | |
| 61 (27.23) | ||
| Parity | ||
| Nulliparous | 46 (20.54) | |
| Multiparous | 178 (79.46) | |
| Pre-pregnancy obesity | ||
| No | 143 (63.84) | |
| Yes | 81 (36.16) | |
| Gestational obesity | ||
| No | 83 (37.05) | |
| Yes | 141 (62.95) | |
| History of PE | ||
| No | 204 (91.07) | |
| Yes | 20 (8.93) | |
| Prenatal care visits | 7.00 (5.00–10.00) * | |
| Prenatal care adequacy (categories) | ||
| Inadequate ( |
59 (26.34) | |
| Adequate ( |
165 (73.66) | |
| MPV (fL) | 8.65 (8.10–9.40) * | |
| Gestational age at onset of PE† | ||
| 17 (15.20) | ||
| 95 (84.80) | ||
| PE (case/control status) | ||
| No (control) | 112 (50.00) | |
| Yes (case) | 112 (50.00) | |
* Median (IQR). † Among women with PE (n = 112). PE, preeclampsia; MPV, mean platelet volume; IQR, interquartile range.
In bivariate analysis, PE cases had higher MPV than controls (median 9.05 vs.
8.40 fL; Wilcoxon p
| Characteristics | Control (n = 112), n (%) | Case (n = 112), n (%) | p-value | |
| Age | 0.292 | |||
| 18–20 years | 5 (4.46) | 7 (6.25) | ||
| 21–34 years | 81 (72.32) | 70 (62.50) | ||
| 26 (23.22) | 35 (31.25) | |||
| Parity | 0.098 | |||
| Nulliparous | 18 (16.07) | 28 (25.00) | ||
| Multiparous | 94 (83.93) | 84 (75.00) | ||
| Pre-pregnancy obesity | 0.037 | |||
| No | 79 (70.54) | 64 (57.14) | ||
| Yes | 33 (29.46) | 48 (42.86) | ||
| Gestational obesity | 0.019 | |||
| No | 50 (44.64) | 33 (29.46) | ||
| Yes | 62 (55.36) | 79 (70.54) | ||
| History of PE † | ||||
| No | 112 (100.00) | 92 (82.14) | ||
| Yes | 0 (0.00) | 20 (17.86) | ||
| Prenatal care adequacy (categories) | ||||
| Inadequate ( |
17 (15.18) | 42 (37.50) | ||
| Adequate ( |
95 (84.82) | 70 (62.50) | ||
| MPV (fL) * | 8.40 (7.80–9.00) | 9.05 (8.30–9.85) | ||
* Median (IQR); p from Wilcoxon rank-sum for MPV.
† Column percentages;
Crude associations showed higher odds of PE per 1-fL increase in MPV (OR: 2.06;
95% CI: 1.51–2.82; p
| Characteristics | Crude analysis | Adjusted analysis* | |||||
| OR | 95% CI | p-value | aOR | 95% CI | p-value | ||
| MPV (per 1 fL) | 2.06 | 1.51–2.82 | 2.16 | 1.55–3.01 | |||
| Parity | |||||||
| Nulliparous | Reference | Reference | |||||
| Multiparous | 0.57 | 0.30–1.11 | 0.100 | 0.54 | 0.25–1.14 | 0.105 | |
| Pre-pregnancy obesity | |||||||
| No | Reference | Reference | |||||
| Yes | 1.80 | 1.03–3.12 | 0.038 | 1.45 | 0.78–2.98 | 0.315 | |
| Gestational obesity | |||||||
| No | Reference | Reference | |||||
| Yes | 1.93 | 1.11–3.35 | 0.019 | 2.43 | 1.17–5.26 | 0.039 | |
| Prenatal care adequacy | |||||||
| Adequate ( |
Reference | Reference | |||||
| Inadequate ( |
3.35 | 1.76–6.37 | 3.71 | 1.27–7.55 | 0.006 | ||
* Adjusted for variables with crude p
In a sensitivity analysis using penalized (Firth) logistic regression including
prior PE, the association between higher MPV and PE remained robust (aOR: 2.20
per 1 fL; 95% CI: 1.57–3.09; p
Further adjustment for gestational age at blood sampling did not materially modify the association between MPV and PE (aOR: 2.16 per 1 fL; 95% CI: 1.56–2.99). Associations for gestational obesity (aOR: 2.87; 95% CI: 1.50–5.49) and inadequate prenatal care (aOR: 4.00; 95% CI: 1.71–9.34) remained consistent, indicating that the observed effects were not driven by timing of sampling.
In ROC analysis, MPV showed fair ability to discriminate PE, with an AUC of
0.686 (95% CI: 0.616–0.755). Using the Youden-derived threshold of 8.80 fL
(classification rule: PE if MPV
| Metric | Value |
| AUC (95% CI) | 0.686 (0.616–0.755) |
| Optimal cutoff (fL) | 8.80 |
| Classification rule | PE if MPV |
| Sensitivity, % | 61.6 |
| Specificity, % | 71.4 |
| LR+ | 2.16 |
| LR– | 0.54 |
AUC from ROC analysis. Optimal cutoff selected by Youden’s index (sensitivity + specificity – 1). Likelihood ratios are calculated at the optimal cutoff. Positive/negative predictive values are not reported because this is a case-control design. AUC, area under the curve; ROC, receiver operating characteristic; LR, likelihood ratio.
Bootstrap internal validation with 1000 resamples yielded an optimism-corrected
AUC of 0.685 (95% CI: 0.616–0.755), confirming the stability of the
discriminatory performance. The multivariable model showed acceptable calibration
(Hosmer-Lemeshow
ROC analysis for MPV yielded an AUC of 0.686 (95% CI: 0.616–0.755). The
optimal threshold by Youden’s index was 8.80 fL, which classified cases as MPV
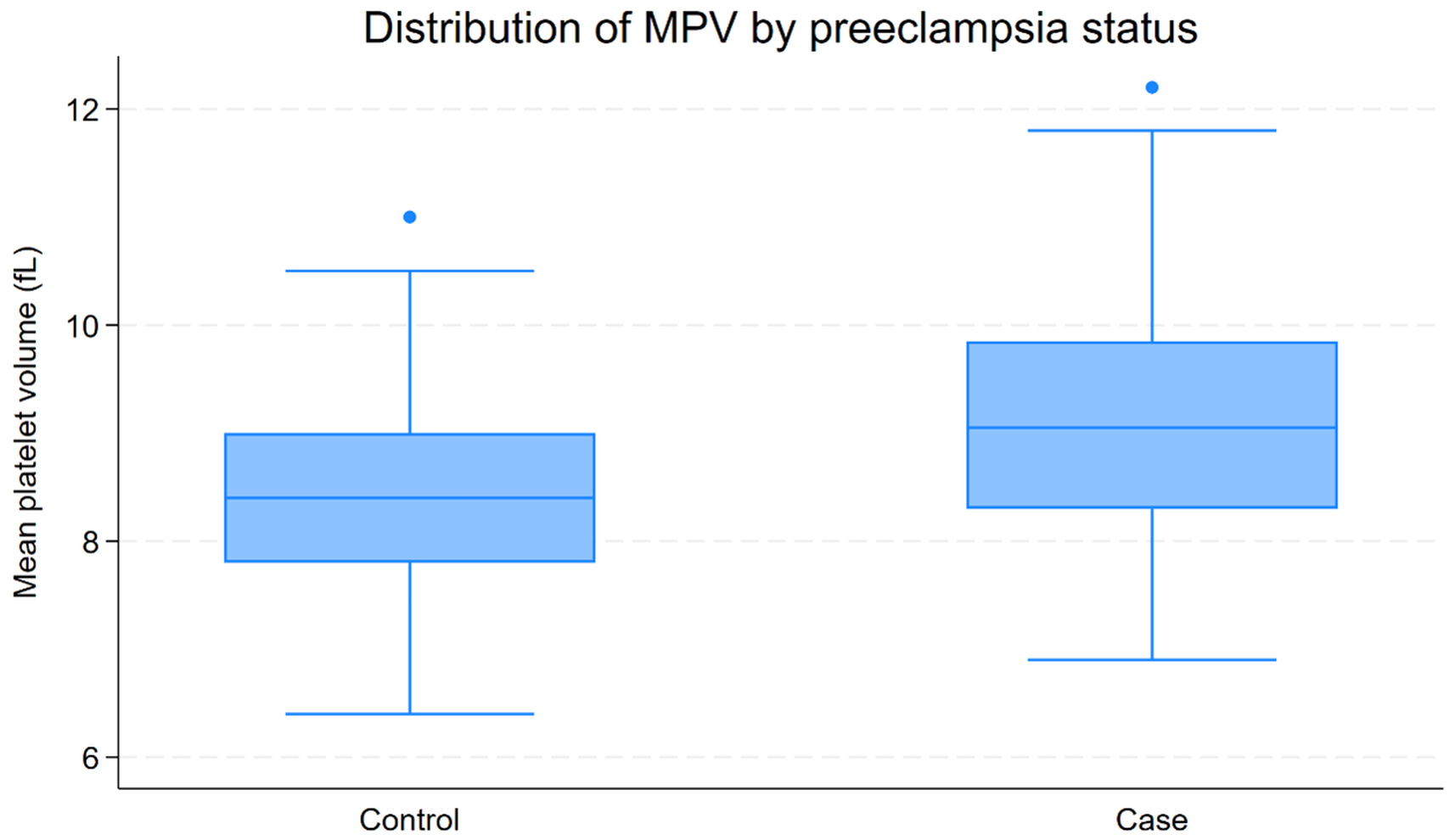 Fig. 1.
Fig. 1.
Distribution of MPV by PE status. Boxplots of MPV (fL) in controls (n = 112) and cases (n = 112). Boxes depict the median and IQR; whiskers denote adjacent values, and points beyond the whiskers (if any) indicate outliers. MPV values were higher in cases than controls, consistent with the bivariate comparison.
The ROC curve shows moderate discrimination of MPV to classify PE (AUC: 0.686; 95% CI: 0.616–0.755). The Youden-optimal cutoff of 8.80 fL corresponds to 61.6% sensitivity and 71.4% specificity (Fig. 2).
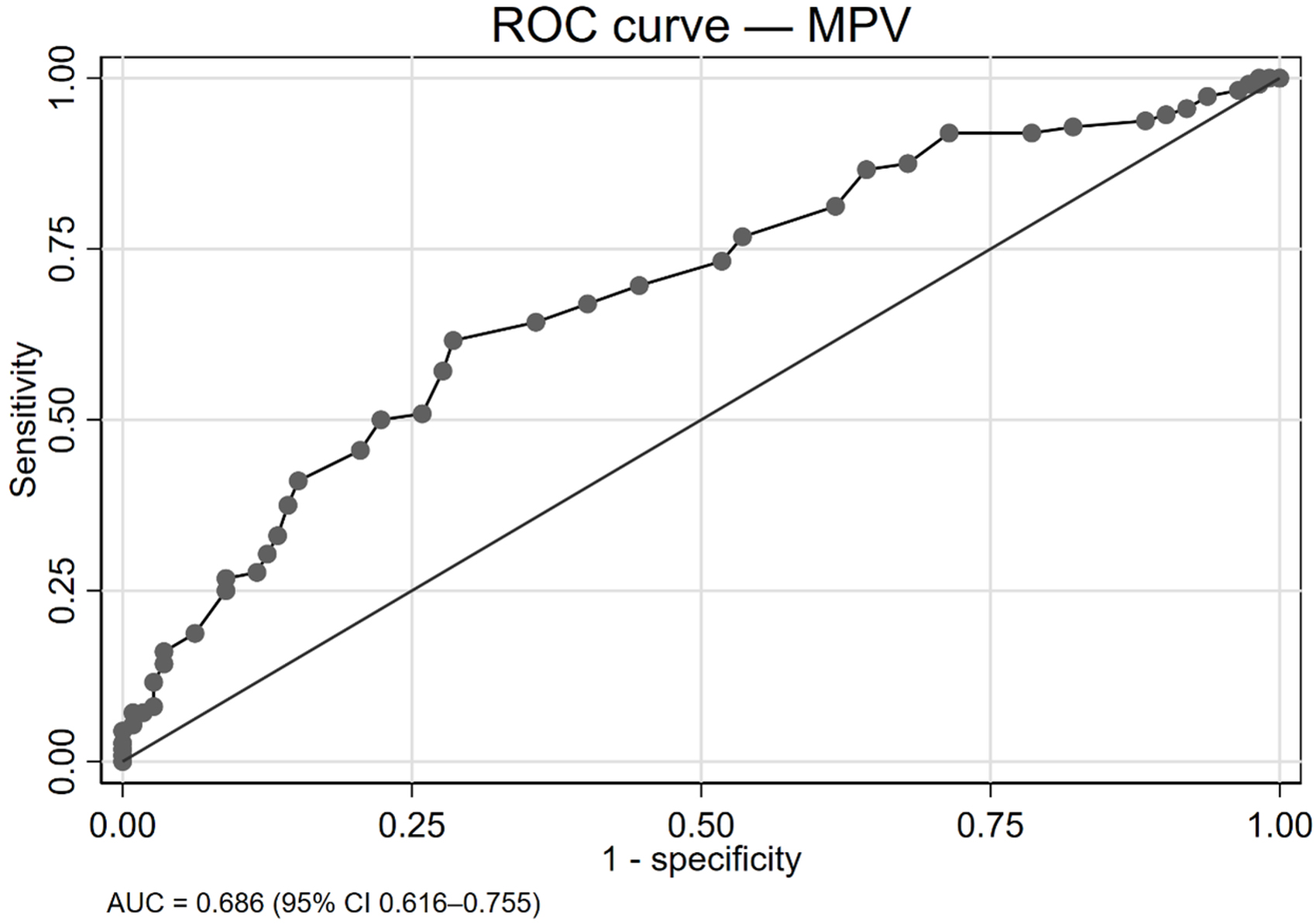 Fig. 2.
Fig. 2.
ROC curve analysis for MPV. ROC curve for MPV for classifying PE (case vs. control). The AUC was 0.686 (95% CI: 0.616–0.755). The optimal cutoff by Youden’s index was 8.80 fL, yielding 61.6% sensitivity and 71.4% specificity. Positive/negative predictive values are not reported because this is a case-control design.
This case-control analysis indicates that MPV is higher in women with PE and remains independently associated with the condition after adjustment for maternal factors. Furthermore, gestational obesity and inadequate prenatal care show independent associations, whereas parity and age do not. Prior PE differentiated groups in unadjusted comparisons but was excluded from multivariable modeling due to quasi-complete separation. As a single biomarker, MPV demonstrates moderate discriminatory capability with a clinically plausible threshold, supporting its value as part of a multivariable risk profile rather than an individual diagnostic test.
Our study indicates that higher MPV is independently associated with PE, in alignment with multiple reports. Li et al. [18] observed higher MPV in both mild and severe PE versus healthy pregnancies, as well as a significant univariate association with PE. Similarly, Bawore et al. [19] reported elevated MPV in PE with a clear severity gradient. Prospective data corroborate the pattern: Udeh et al. [20] found increased MPV among women who later developed PE at 14–18 gestational weeks and at end-of-follow-up, and Thakkar et al. [21] reported higher baseline MPV at 14–18 gestational weeks in early-onset PE. Severity-related increases were also confirmed in a prospective study with ROC verification [22]. However, Lin et al. [23] observed a partial divergence in timing, as found no group differences in the first trimester, with separation appearing later (e.g., 16–19 weeks), suggesting gestational-age dependence. Methodological factors (prospective vs. case-control design), PE phenotype (early/severe vs. mixed), and analytical variability in hematology platforms may underlie residual heterogeneity. Collectively, converging evidence supports MPV as a biologically plausible marker of platelet-activation in PE. MPV is most informative after the early first trimester and offers an accessible metric with potential to enhance risk stratification in antenatal care, particularly resource-limited settings.
In our settings, MPV alone offered only moderate discrimination for PE using a
single cut-point rule. Performance varied across studies, with early-onset
cohorts reporting strong accuracy. Udeh et al. [20] showed high AUC with
a 9.4 fL threshold and near-perfect accuracy in severe early-onset PE at
~10.7 fL, and Thakkar et al. [21] reported
similarly high AUC with a
We observed that gestational obesity was independently associated with PE. This aligns with studies showing higher mid-pregnancy adiposity among women with PE. Li et al. [18] reported a higher second-trimester BMI in PE alongside elevated systolic and diastolic pressures, and Thakkar et al. [21] found higher gestational BMI among PE cases while other baseline factors were comparable. Differences in effect sizes across reports likely reflect the timing and method of BMI assessment, PE phenotype mix (early vs. late onset), and residual confounding by hemodynamics or glycemic status that track with weight gain. From a policy perspective, these convergent findings support integrating routine mid-trimester weight surveillance and metabolic risk counseling into antenatal pathways to control preventable PE burden in resource-constrained settings.
In our analysis, the crude association between pregestational obesity and PE
lost significance after multivariable adjustment. External literature largely
documents higher baseline BMI among PE cases. Lin et al. [23] reported a
significant pregestational BMI difference, but does not explicitly test for
attenuation after adjustment. Moreover, one cohort restricted pre-pregnancy BMI
(
Parity and maternal age were not associated with PE in our study. The literature is mixed but broadly consistent with a limited role of these demographics in certain cohorts. Lin et al. [23] observed older age associated with PE cases but a similar parity distribution without significance. Udeh et al. [20] observed no differences in age or gravidity between women who developed PE and those who did not. Thakkar et al. [21] also found no group differences among these covariates. Variability likely reflects sampling frames, age ranges, and the dominance of metabolic or placental factors over demographic predictors. Public-health programs should therefore prioritize modifiable cardiometabolic risks over demographic screening alone to improve PE prevention yield.
A prior history of PE differentiated groups in bivariate analysis but could not be retained in multivariable modeling due to quasi-complete separation. The external evidence supports a similar direction of effect. In fact, Lin et al. [23] reported a higher proportion of multiparouss with prior pregnancy-induced hypertension in the PE group, whereas Bawore et al. [19] mitigated this issue by excluding women with prior PE, precluding its analysis as a predictor. Such handling decisions reflect the well-known strength of recurrence risk and its analytic challenges. In practice, meticulous obstetric history-taking remains a low-cost, high-value measure to identify elevated PE risk early, guiding intensified surveillance in settings with limited health-system resources.
Our case series predominantly comprised later-onset presentations. In contrast, prospective cohorts quantifying early-onset PE report relatively low prevalence but provide detailed distributions of onset timing. Thakkar et al. [21] estimated an overall early-onset PE of 5.3%, with most cases clustering between 28–32 weeks, and Udeh et al. [20] reported a comparable early-onset frequency of 5.9% with severity stratification. Differences between our hospital-based series and prospective cohorts likely reflect referral patterns, triage thresholds, and regional epidemiology. Recognizing a primarily late-onset profile in our setting argues for scalable second-trimester risk assessment and timely escalation pathways, aligning limited resources to the period of greatest preventable morbidity.
Taken together, our findings support MPV as a pragmatic, laboratory-ready biomarker that captures platelet activation relevant to PE pathophysiology and can be leveraged within routine antenatal workflows. Because MPV is generated automatically by standard hematology analyzers at no additional cost, it offers a scalable tool for risk stratification in settings with limited resources and high disease burden. Clinically, MPV is unlikely to serve as a stand-alone test; rather, its value lies in trimester-specific algorithms that integrate maternal characteristics and blood pressure with hematologic and metabolic indices to identify women who may benefit from closer surveillance or preventive measures. By enabling earlier identification of at-risk pregnancies, particularly beyond the earliest weeks when predictive accuracy improves, MPV-informed pathways could help reduce preventable maternal and perinatal complications and align with global efforts to strengthen low-cost, point-of-care diagnostics in maternal health programs.
This study was conducted at a national tertiary referral center with standardized clinical records and laboratory workflows, enhancing internal validity and consistency of MPV assessment. The case-control design enabled efficient capture of clinically adjudicated PE and appropriate normotensive controls from the same source population and time frame. We complemented group comparisons with multivariable modeling to isolate the independent contribution of MPV while accounting for key maternal factors. Diagnostic performance was reported transparently using ROC analysis and likelihood ratios at a pre-specified cut-point, with clear acknowledgment of the design-specific constraints (e.g., not estimating predictive values). Together, these features provide a coherent and clinically interpretable evaluation of MPV’s association with PE and its potential utility as an accessible component of antenatal risk assessment.
This study has several limitations. First, as a single-center hospital-based case-control study, it cannot estimate incidence or predictive values and may be subject to selection bias. Second, we did not perform stratified analyses according to PE severity (mild vs. severe) or timing of onset (early- vs. late-onset PE); and the number of severe and early-onset cases was insufficient to support stable subgroup models, which limits our ability to determine whether MPV behaves differently across phenotypes. Third, although all MPV values were generated in the same accredited laboratory using a single automated analyzer under standardized internal quality control procedures, MPV is inherently sensitive to instrument type, anticoagulant, and sample handling; therefore, the proposed 8.80 fL cutoff should be interpreted as platform-specific and requires calibration in other settings. Fourth, residual confounding cannot be excluded because we lacked inflammatory markers (e.g., C-reactive protein), additional platelet indices (e.g., platelet distribution width, platelet-large cell ratio), and socioeconomic indicators that might influence both MPV and PE risk. Moreover, because PE cases and controls were not matched on age, gestational age at sampling, or other baseline factors, residual confounding cannot be fully excluded despite multivariable adjustment. These constraints mean that our adjusted estimates may still partially capture unmeasured confounding. Future multicenter prospective cohorts with comprehensive phenotyping and extended biomarker and socioeconomic data are warranted to validate our findings and define robust, generalizable MPV-based risk thresholds.
In this hospital-based case-control study, higher MPV was independently associated with PE, and as a single biomarker, MPV showed moderate discrimination using a pragmatic cutoff. Gestational obesity and inadequate prenatal care were also associated with PE, whereas parity and age were not. Given that MPV is routinely generated by hematology analyzers at no additional cost, integrating MPV into trimester-specific, multivariable antenatal risk models could help prioritize surveillance and preventive measures, especially in resource-limited settings. Prospective, multicenter studies should establish analyzer- and trimester-specific thresholds and quantify absolute risk to guide implementation and decision-curve–based clinical use.
The datasets generated and analyzed during the current study are not publicly available due to institutional and patient confidentiality policies, but are available from the corresponding author upon reasonable request.
RJR-Y: Data Curation, Investigation, Methodology, Writing Original Draft, and Writing Review & Editing. GD-B: Data Curation, Investigation, Methodology, and Writing Review & Editing. MAA-H: Conceptualization, Methodology, Formal Analysis, Supervision, Validation, Writing Original Draft, and Writing Review & Editing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Declaration of Helsinki and approved by the Comité Institucional de Ética e Integridad Científica (CIEI) of Universidad Privada Norbert Wiener (Expediente 0995-2024; approval issued 12 November 2024). Informed consent was waived due to the retrospective nature of the study and the use of anonymized medical records without direct patient contact.
We thank the clinical and laboratory staff of the Obstetrics Hospitalization Service at the Instituto Nacional Materno Perinatal (INMP), Lima, for their support during data abstraction and verification.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT-3.5 in order to check spelling and grammar. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


