1 Department of Gynecology, First Affiliated Hospital, Zhejiang University School of Medicine, 310006 Hangzhou, Zhejiang, China
Abstract
Ovarian pregnancy (OP) is a rare type of ectopic pregnancy (EP) that can become life-threatening due to the risk of severe complications, such as pelvic hemorrhage and circulatory collapse. The diagnosis and treatment of OP are challenging. Thus, this study aimed to investigate the clinical characteristics of OP, explore current and potential diagnostic and therapeutic strategies, and summarize the experience with laparoscopic treatment for OP.
A retrospective analysis was performed on data from 16 consecutive patients with OP who underwent surgical treatment between March 2016 and May 2025. We collected clinical data from these patients, including sociodemographic information, suspected risk factors, clinical manifestations, preoperative examinations [e.g., serum beta human chorionic gonadotropin (β-hCG) test and ultrasonography], treatment methods, and outcomes.
The mean age of the patients was 32.44 ± 6.37 years, and the mean gestational age at diagnosis was 50.13 ± 9.91 days. Abdominal pain was the most common presenting symptom (reported in 15 patients, 93.75%), followed by vaginal bleeding (7 patients, 43.75%) and hemorrhagic shock (2 patients, 12.50%). Serum β-hCG (measured in 15 patients) ranged from 700.56 to 12,661.89 mIU/mL, with a median of 4756.03 mIU/mL [interquartile range (IQR), 1730.39–8174.70 mIU/mL]. The preoperative ultrasound diagnostic rate for OP was 6.25% (1/16). All patients underwent laparoscopic surgery for the excision of OP tissue. A ruptured OP was observed in 13 patients (81.25%). Hemoperitoneum ranged from 30 to 2000 mL, with a median of 900 mL (IQR, 350–1000 mL). The mean operative time was 1.23 ± 0.60 h, and the mean postoperative hospital stay was 2.50 ± 1.51 days. None of the patients required additional surgical or medical treatments after laparoscopy (LC).
Rupturing is a common complication of OP, highlighting the importance of early diagnosis and timely intervention. Laparoscopic techniques facilitate the early detection and accurate diagnosis of OP. Laparoscopic surgery is a safe, effective, and minimally invasive treatment option for OP.
Keywords
- ovarian pregnancy
- ectopic pregnancy
- laparoscopy
Primary ovarian pregnancy (OP) is a rare subtype of ectopic pregnancy (EP) in which a fertilized egg implant is placed in the ovary [1]. OP accounts for approximately 1.14–3.12% of all EPs and only 0.09% of all pregnancies [2]. The reported incidence of OP is growing because of the evolution of transvaginal sonography and careful histological examination of ovarian tissues [3].
However, the diagnosis and management of OP remain challenging. One of the main reasons is its rarity and high likelihood of misdiagnosis as more common conditions, such as tubal pregnancy (TP) or rupture of the corpus luteum (CL) [4, 5].
Despite its low incidence, OP can be life-threatening, similar to TP, because of the risk of severe complications, such as pelvic hemorrhage and circulatory collapse. Therefore, early diagnosis and timely intervention are critical for reducing morbidity and mortality.
Although laparoscopic surgery has increasingly become the preferred approach for managing OP, concerns remain regarding the potential for massive intra-abdominal bleeding following rupture, which may lead to a preference for laparotomy in some emergency situations [6].
This study aimed to analyze the clinical characteristics, ultrasound manifestations, and laparoscopic treatment of 16 patients with OP, explore the diagnosis and treatment strategies, and summarize the experience of applying laparoscopy (LC) for the treatment of OP.
Between March 2016 and May 2025, 16 patients diagnosed with OP were admitted to our department. A retrospective analysis was conducted by reviewing their electronic medical records and searching a computerized hospital database containing coded entries for all cases of EPs. All cases labeled as “ovarian pregnancy” or “nontubal ectopic pregnancy” were manually reviewed. Patients who did not undergo surgical treatment or had incomplete clinical records affecting accurate diagnosis of OP were excluded from the study. Ultimately, 16 histologically confirmed OP cases were identified. None of the 16 OP cases had severe underlying diseases, such as coagulation disorders, nor a history of anticoagulant use.
This study was approved by the ethics committee of the First Affiliated
Hospital, Zhejiang University School of Medicine, and all methods were conducted
in accordance with the relevant institutional guidelines and regulations. The
diagnostic gold standard for inclusion was intraoperative confirmation of OP
supported by histopathological evidence of the pregnancy tissue. The data were
entered and managed using WPS office (12.1.0.24034, Kingsoft Office Software Inc. Beijing, China). The normally
distributed variables were presented as mean
The following clinical and demographic data were extracted from patient records:
(1) sociodemographic information: age, body mass index (BMI), menstrual and
pregnancy histories; (2) Suspected risk factors: history of smoking, prior EP,
pelvic inflammatory disease, pelvic or abdominal surgery (including cesarean
section, adnexal surgery, appendectomy, etc.), intrauterine procedures (e.g.,
abortions, hysteroscopic surgeries), contraceptive use, infertility (defined as
the failure to achieve pregnancy after 12 months of regular unprotected sexual
intercourse), assisted reproductive technology, pelvic adhesions, and ovarian
endometrioma detected during surgery; (3) clinical presentation: gestational age
at admission and primary symptoms (abdominal pain, vaginal bleeding, and
hemorrhagic shock); and (4) preoperative investigations: initial serum
Treatment decisions were made by the attending gynecologist based on the
patient’s clinical history, physical examination, ultrasound findings,
The following intraoperative data were recorded: (1) the location of the OP; (2) presence of pelvic adhesions or ovarian endometriosis; (3) hemoperitoneum volume; (4) integrity of the ectopic gestational sac (EGS) (intact or ruptured); (5) duration of surgery; (6) Whether blood transfusion was required; and (7) postoperative length of hospital stay. The hemoperitoneum volume was calculated by subtracting the total volume of irrigation saline used from the total volume of fluid aspirated. After surgery, all patients were admitted for postoperative observation and recovery.
Between March 2016 and May 2025, 16 patients with OP were diagnosed and treated
at our institution. All patients underwent a physical examination, gynecologic
ultrasonography (TVS or TAS), and routine laboratory investigations, including
complete blood count. Detailed clinical data, including sociodemographic
information such as general condition, pregnancy history, gestational age at
admission, and suspected risk factors, are summarized in Table 1. The diagnostic
information, including preoperative
| Case | Patient age (years) | Gestational age (days) | Menstrual cycle (days) | BMI (kg/m2) | Gravidity | Parity | Suspected risk factors | |
| 1 | 21 | 43 | 30–31 | 31.96 | 0 | 0 | Pelvic adhesions (intestine and posterior uterine wall) | |
| 2 | 28 | 51 | 30 | 16.42 | 0 | 0 | Pelvic adhesions (intestine and pelvic peritoneum) | |
| 3 | 33 | 53 (36 days after IVF-ET) | 30–35 | 26.57 | 0 | 0 | Infertility, IVF-ET | |
| 4 | 42 | 61 | 30–60 | 26.71 | 4 | 2 | Cesarean section twice, SA/IA twice, tubal inflammation, pelvic adhesions (bilateral adnexa with intestinal, and pelvic peritoneum) | |
| 5 | 42 | 47 | 30 | 20.20 | 3 | 1 | SA/IA twice | |
| 6 | 25 | 50 | 45–60 | 21.37 | 0 | 0 | None | |
| 7 | 37 | 45 | 28–30 | 20.70 | 2 | 1 | SA/IA once | |
| 8 | 34 | 39 | 30 | 22.66 | 3 | 2 | SA/IA once, cesarean section once, pelvic adhesions (intestine and pelvic peritoneum) | |
| 9 | 23 | 48 | 30 | 21.83 | 0 | 0 | None | |
| 10 | 38 | 56 | 30 | 21.48 | 4 | 1 | SA/IA three times, downward IUD shifting | |
| 11 | 29 | 59 | 30 | 20.70 | 0 | 0 | None | |
| 12 | 35 | 33 | 28 | 20.31 | 4 | 1 | SA/IA three times | |
| 13 | 33 | 49 | 28–30 | 18.67 | 0 | 0 | Hysteroscopic endometrial polypectomy | |
| 14 | 35 | 49 | 30 | 21.83 | 2 | 1 | Oral emergency contraceptives (levonorgestrel), SA/IA once | |
| 15 | 27 | 43 | 30 | 19.03 | 0 | 0 | None | |
| 16 | 37 | 76 | 30–90 | 23.80 | 3 | 1 | SA/IA twice | |
| Mean | 32.44 |
50.13 |
\ | 22.14 |
\ | \ | \ | |
| Percentile | ||||||||
| Q1 (25%) | \ | \ | \ | \ | 0 | 0 | \ | |
| Median | 1 | 0.5 | ||||||
| Q3 (75%) | 3 | 1 | ||||||
OP, ovarian pregnancy; BMI, body mass index; IVF-ET, in vitro fertilization-embryo transfer; SA, spontaneous abortion; IA, induced abortion; IUD, intrauterine device; N, number of cases.
| Case | Pelvic ultrasound | Main clinical symptoms | |||||
| Type | Main outcome | Abdominal pain | Vaginal bleeding | Shock | |||
| 1 | Not measured | TAS | EP mass considered | + | – | – | |
| 2 | 7454.60 | TVS | EP mass considered | + | + | – | |
| 3 | 2370.00 | TVS | EP mass considered | + | + | – | |
| 4 | 1730.39 | TVS | EP mass considered | + | – | – | |
| 5 | 3143.10 | TVS | EP mass considered | + | – | – | |
| 6 | 2147.69 | TVS | Ruptured CL or EP mass indistinguishable | + | – | – | |
| 7 | 8912.95 | TVS | Ruptured CL considered | + | – | + | |
| 8 | 6902.21 | TVS | EP mass considered | + | + | – | |
| 9 | 6271.90 | TVS | EP mass considered | + | + | – | |
| 10 | 8174.70 | TVS | EP mass considered | + | + | – | |
| 11 | 4756.03 | TVS | EP mass considered | + | – | – | |
| 12 | 8370.91 | TVS | EP mass considered | + | – | – | |
| 13 | 12,661.89 | TVS | EP mass considered | – | + | – | |
| 14 | 828.10 | TAS | EP mass considered | + | – | + | |
| 15 | 700.56 | TVS | EP mass considered | + | + | – | |
| 16 | 1643.48 | TVS | OP mass considered | + | – | – | |
| Percentile | |||||||
| Q1 (25%) | 1730.39 | \ | \ | \ | \ | \ | |
| Median | 4756.03 | ||||||
| Q3 (75%) | 8174.70 | ||||||
TAS, transabdominal ultrasound; TVS, transvaginal ultrasound; EP, ectopic
pregnancy; CL, corpus luteum;
| Case | Surgical procedure | Duration of surgery (h) | Integrity of the EGS | Hemoperitoneum (mL) | Side | Blood transfusion | Postoperative hospital stay (days) | |
| 1 | LC/RGP | 0.97 | Ruptured | 500 | R | No | 3 | |
| 2 | LC/RGP | 1.65 | Ruptured | 1000 | L | No | 6 | |
| 3 | LC/RGP | 2.35 | Intact | 50 | L | No | 5 | |
| 4 | LC/RGP | 0.93 | Ruptured | 500 | R | No | 2 | |
| 5 | LC/RGP | 2.52 | Ruptured | 1000 | L | No | 2 | |
| 6 | LC/RGP | 1.33 | Ruptured | 1000 | L | No | 4 | |
| 7 | LC/RGP | 1.43 | Ruptured | 1500 | R | Yes | 2 | |
| 8 | LC/RGP | 1.13 | Ruptured | 800 | L | No | 4 | |
| 9 | LC/RGP | 1.60 | Ruptured | 500 | R | No | 2 | |
| 10 | LC/RGP + tubal sterilization + IUD removal | 1.48 | Ruptured | 1000 | R | No | 2 | |
| 11 | LC/RGP | 0.53 | Ruptured | 1000 | L | No | 1 | |
| 12 | LC/RGP | 0.60 | Intact | 100 | L | No | 1 | |
| 13 | LC/RGP | 0.67 | Ruptured | 200 | R | No | 1 | |
| 14 | LC/RGP | 1.07 | Ruptured | 2000 | R | Yes | 1 | |
| 15 | LC/RGP | 0.72 | Ruptured | 1000 | R | No | 2 | |
| 16 | LC/RGP | 0.63 | Intact | 30 | L | No | 2 | |
| Mean | \ | 1.23 |
\ | \ | \ | \ | 2.50 | |
| Percentile | ||||||||
| Q1 (25%) | \ | \ | \ | 350 | \ | \ | \ | |
| Median | 900 | |||||||
| Q3 (75%) | 1000 | |||||||
EGS, ectopic gestational sac; LC, laparoscopy; RGP, removal of gestational products; R, right; L, left.
The mean age of the patients was 32.44
All patients underwent emergency laparoscopic surgery. One patient, who
presented with life-threatening intraperitoneal bleeding, did not undergo a
preoperative
All patients were symptomatic at admission; 6 patients (37.50%) had symptoms of both abdominal pain and abnormal vaginal bleeding, 9 (56.25%) had abdominal pain without vaginal bleeding, and 1 (6.25%) presented with vaginal bleeding only. Abdominal pain was present in 15 patients (93.75%), and 7 (43.75%) experienced abnormal vaginal bleeding. Hemorrhagic shock occurred in 2 patients (12.50%, cases 7 and 14). All patients had a history of amenorrhea; however, three of them overlooked it due to previously irregular menstrual cycles. Preoperative ultrasonography identified abnormal adnexal or pelvic masses in all patients. EP was suspected in 14 patients (87.50%); one patient (case 16) showed a gestational sac-like structure with fetal cardiac activity within the ovary, leading to a preoperative diagnosis of OP. In the remaining 13 patients, although EP was suspected, its exact location could not be determined. One patient (case 7) was misdiagnosed as having a ruptured ovarian CL, and another (case 6) had indeterminate findings suggestive of either a ruptured CL or EP by ultrasound.
All patients (100%) underwent laparoscopic resection of the ectopic tissue. A typical intraoperative finding was the presence of an ovarian mass, as shown in Fig. 1. One patient (case 10) who no longer desired fertility underwent bilateral tubal sterilization and IUD removal during surgery. Postoperative pathology confirmed the diagnosis of OP in all the patients. The pregnancy was located in the left ovary in 8 patients (50%) and in the right ovary in the other 8 patients (50%). Obvious pelvic adhesions were present in 4 patients (25%). The main adhesion locations are shown in Table 1. Ovarian endometriosis was not observed.
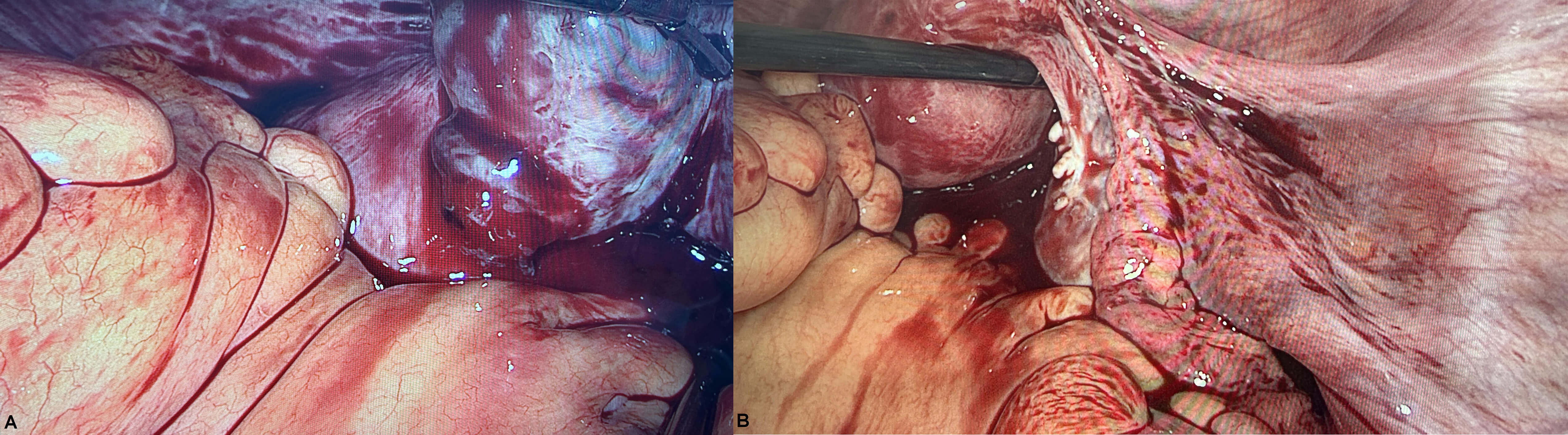 Fig. 1.
Fig. 1.
Typical presence of the OP. (A) LC revealed a rupture of OP on the right side. (B) The right fallopian tube appeared normal.
Ruptured OP was observed in 13 patients (81.25%). Hemoperitoneum volume ranged
from 30 to 2000 mL, with a median of 900 mL (IQR, 350–1000 mL) (Table 3). Two
patients (12.50%, cases 7 and 14) underwent blood transfusions. The mean
operative time was 1.23
OP is rare, and its underlying pathogenesis remains poorly understood. In this study, we explored the suspected risk factors for OP. One of the 16 patients with OP (6.25%) had an IUD in place, suggesting a potential association between IUD-induced aseptic uterine inflammation and OP through altered tubal peristalsis. Another patient (6.25%) conceived via IVF-ET because of infertility. Over half of the patients had a history of intrauterine procedures, whereas a few presented with obvious pelvic adhesions intraoperatively; however, no ovarian endometriosis was observed. Apart from the two patients with prior cesarean sections, most had no history of pelvic or abdominal surgery. Some individual patients had a history of salpingitis or emergency contraceptive use. Notably, 25% of the patients had no identifiable risk factors. Some researchers have suggested that the increased risk of OP may be associated with factors such as previous abdominal surgery, history of infectious diseases, infertility, IVF-ET, use of IUD and emergency oral contraceptives, and ovarian endometrioma [5, 6, 7, 8, 9, 10]. Among the various factors, the association between IUD use and IVF in OP is the most widely recognized or increasingly acknowledged [5, 8, 9, 11]. Seinera et al. [12] speculated that traditional risk factors of TP are unrelated to those of OP. The etiology of OP remains uncertain; traditional TP risk factors, such as IUD use, pelvic surgery, infection, and assisted reproduction, did not show a significant association with OP in a previous study [13]. Given the rarity of OP and limited availability of large-scale studies, identification of definitive risk factors remains challenging. Current evidence suggests a multifactorial etiology. Further research with larger sample sizes is necessary to clarify the underlying mechanisms and establish validated risk profiles for OP.
The typical clinical presentation of OP includes amenorrhea, abdominal pain, and
vaginal bleeding with or without associated symptoms, such as anal distension.
Rupture of OP may lead to more severe manifestations, including dizziness,
syncope, and potentially hemorrhagic shock. In our study, up to 93.75% of the
patients presented with abdominal pain, whereas less than half exhibited vaginal
bleeding. Interestingly, patients with OP are less likely to present with vaginal
bleeding, compared with those with TP, a difference attributed to the
well-maintained endometrium and higher
The rupture rate of OP in our cohort was high (81.25%). Owing to the fragility of the ovarian surface, all 13 cases of OP rupture occurred in the early stages of pregnancy, leading to intra-abdominal hemorrhage and, in two cases, hemorrhagic shock. These clinical features closely resemble those observed in TP or rupture of the CL, making the differential diagnosis challenging. Patients with OP tend to experience more severe clinical complications during the first trimester, such as hemorrhagic shock, ruptured gestational sacs, hemoperitoneum, and an increased need for emergency laparotomy, compared with those with TP [8]. For example, one study observed a rupture rate of 56.34% in patients with OP, with significantly greater peritoneal blood accumulation than in patients with TP [5]. Another study reported an even higher rupture rate of 86.61% in OP, markedly exceeding the 23% rupture rate previously documented in TP [9, 14]. Hemodynamic shock has been reported in 30–71.4% of the OP cases [1]. These findings collectively suggest a worse prognosis for OP relative to TP. Additionally, some patients with EP, particularly those with a history of irregular menstrual cycles, may mistakenly attribute vaginal bleeding for menstruation, leading to the clinical sign of amenorrhea being overlooked. Therefore, clinicians should pay close attention to menstrual history.
Although the clinical manifestations of OP lack specificity, a combination of
early quantitative serum
Although abnormally low and slowly increasing
Early ultrasound diagnosis facilitates appropriate treatment selection and monitoring, ultimately improving the clinical outcomes for patients with OP. The recommended ultrasound diagnostic criteria for OP include: (1) the presence of a wide echogenic ring on the surface of the ovary with an echo-transparent area inside; (2) ovarian cortex, CL or follicle around the mass; (3) The echo intensity of the ring is greater than that of the ovary itself [11]. However, differentiating ruptured OP from ruptured TP or CL hemorrhage using ultrasound remains challenging owing to overlapping sonographic features. In our study, all patients exhibited abnormal echoes or masses in the pelvic or adnexal regions on preoperative ultrasound. However, only one case revealed a gestational sac-like structure with fetal cardiac activity within the ovary, leading to the preoperative consideration of OP. One patient was diagnosed with ruptured CL hemorrhage using ultrasound, and the other’s imaging findings were inconclusive between CL rupture and EP. The remaining 13 patients were suspected to have EP; however, the exact implantation site could not be clearly identified. Overall, the preoperative ultrasound diagnostic rate for OP in this series was only 6.25%. Preoperative ultrasound diagnosis of OP is often inaccurate, with the reported accuracy rates being as low as 2.5–18% [17, 18, 19]. This diagnostic uncertainty with ultrasound may lead to unnecessary damage or removal of the fallopian tubes, often due to false-negative diagnoses of OP or misdiagnoses as TP during LC. Huang et al. [7] reported a case of concurrent OP and TP following IVF-ET, highlighting the need for clinicians to maintain a high index of suspicion in patients with suspected EP, especially when significant intra-abdominal hemorrhage is present, and to consider the possibility of OP to prevent missed diagnoses.
Surgery remains the primary treatment for patients with OP. Advances in
technology have led to LC becoming the standard approach for OP diagnosis and
management. In our series of 16 patients with OP, 81.25% presented with ruptured
OPs, and 50% had hemoperitoneum volume
The choice of the surgical technique for OP largely depends on the size and
extent of the lesions. Options include removal of gestational products (RGP) from
the ovary, ovarian wedge resection, or partial oophorectomy. Current surgical
trends favor conservative approaches such as RGP or ovarian wedge resection [20],
aiming to excise the intact gestational sac while preserving as much ovarian
tissue and function as possible, which is particularly important for patients
desiring future fertility. Resection of only the OP tissue itself is gaining
acceptance, as it is considered the least invasive surgery that maximally
preserves the ovarian cortex [22]. In our study, all patients with OP were
managed conservatively with the goal of preserving ovarian function. Complete
excision of the gestational sac was prioritized to prevent residual disease and
avoid secondary treatments, such as methotrexate (MTX) or repeat surgery. During
the procedure, meticulous resection of the gestational sac was performed with the
assistance of bipolar cautery, followed by precise electrocoagulation hemostasis
to minimize ovarian damage. For some patients, the ovarian wound surface was
sutured to secure hemostasis, which allowed us to limit the use of electrocautery
and thereby reduce thermal injury to the ovary. Other studies also recommend
local injection of argipressin between normal ovarian tissue and the lesion,
combined with ultrasound energy for hemostasis instead of electrocautery, have
been used to preserve ovarian tissue [23]. Ovarian wedge resection may be
necessary for larger or more advanced pregnancies. For patients in
life-threatening situations, such as uncontrollable bleeding, oophorectomy
remains a last-resort option. Previous studies reported oophorectomy rates of
14% and 21%, with blood transfusion rates ranging from 5% to 25% [6, 13, 24].
In our study, none of the patients underwent oophorectomy, and the blood
transfusion rate was 12.50%. Considering that 16 patients in this study
experienced OP during the first trimester and received timely intervention
treatment, conditions were created for ovarian preservation. The average surgical
duration and hospitalization period for the patients were within reasonable
ranges. Histopathological examination confirmed OP in all 16 patients.
Postoperative monitoring showed that the
OP is rare and the number of patients with OP in the present was limited. Large
sample and multicenter research is required to further investigation. One case
had a missing preoperative serum
Based on nearly a decade of research on OP, we have summarized some diagnostic and treatment experiences for OP. This study underscores the importance of recognizing the risk factors and key clinical features of OP, as well as understanding the limitations of ultrasound in its diagnosis. Patients with OP tend to experience more severe clinical outcomes. LC offers a superior option for both the diagnosis and treatment of OP, providing precise visualization with minimal invasiveness, even in cases of heavy pelvic and abdominal bleeding.
OP, ovarian pregnancy; EP, ectopic pregnancy; TP, tubal pregnancy; CL, corpus luteum; BMI, body mass index;
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
(I) Conception and design: LX; (II) Administrative support: JQ; (III) Collection of data: LX and TQ; (IV) Analysis of data: LX, TQ, LC and JQ. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki, and was approved by the Ethics Committee of the Institutional Review Board of the First Affiliated Hospital, Zhejiang University School of Medicine ([2025B] IIT Ethics Approval No. 0976). A written consent was signed by the patients or their families/legal guardia.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT-3.5 in order to check spelling and grammar. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

