1 Epidemiology Department, Kahn-Sagol-Maccabi Research and Innovation Institute, 6801296 Tel Aviv, Israel
2 Janssen Pharmaceuticals NV, 2340 Beerse, Belgium
3 Departments of Neonatology, Dana Dwek Children’s Hospital, Tel Aviv Medical Center, 6423906 Tel Aviv, Israel
4 Gray Faculty of Medical & Health Sciences, Tel Aviv University, 6997801 Tel Aviv, Israel
5 Department of Obstetrics and Gynecology, The Chaim Sheba Medical Center, 5265601 Ramat-Gan, Israel
Abstract
Hemolytic disease of the fetus and newborn (HDFN) can cause severe prenatal and postnatal outcomes. The main objective of this study is to characterize the clinical impact of HDFN on pregnant women and their newborns.
A retrospective cohort study was performed on de-identified data extracted from Maccabi Healthcare Services (MHS), a large nationwide health organization. The cohort included women and newborns diagnosed with HDFN between January 1998 and December 2021. Cohort characteristics and outcomes are described.
Over the 24-year study period, the incidence rate of HDFN among pregnant women remained stable, while the incidence rate among newborns declined. Severe HDFN was diagnosed in 22 (30.1%) of 73 HDFN-affected pregnancies. Among 450 HDFN newborns, one-third were diagnosed with anemia or jaundice, and 5 cases of kernicterus were observed.
Severe prenatal and postnatal outcomes were observed in cases of HDFN. Further studies are needed to evaluate treatment regimens and to assess the association between treatment management and both short- and long-term outcomes.
Keywords
- HDFN
- prenatal outcomes
- postnatal outcomes
Hemolytic disease of the fetus and newborn (HDFN) occurs when maternal immunoglobulin G (IgG) alloantibodies cross the placenta and bind to fetal red blood cells (RBC) carrying the corresponding antigen, thereby leading to RBC destruction and anemia in the fetus or newborn [1]. The clinical presentation of HDFN ranges from mild anemia to severe cases that may result in fetal demise, hydrops, preterm labor induction, or the need for intrauterine transfusion (IUT) [1, 2].
The epidemiology of HDFN is poorly characterized in the medical literature. The global HDFN incidence has been estimated at 27.6 per 10,000 live births, whereas in developed countries it is reduced to 2.5 per 10,000 live births, likely reflecting higher-quality perinatal and neonatal care [3]. Similarly, in the United States (US), the estimated incidence was 0.3–8 HDFN cases per 10,000 live births per year [4].
In Israel, current antenatal screening recommendations state that blood typing and a Coombs test should be performed at the first prenatal visit [5]. These practices are consistent with guidelines in England and the US [6, 7]. After the initial screening, Israeli guidelines recommend that Rh-negative women undergo antibody testing at week 26–28 weeks to determine the need for anti-D prophylaxis. A 300-µg dose of anti-D should be administered at 28 weeks, and refusal of treatment should be documented in the medical record. An additional dose of anti-D should be given within 72 hours after birth. In some cases, a 600-µg dose of anti-D is indicated [5].
There is no approved drug therapy for the treatment of HDFN. The standard of care for pregnancies at risk of severe fetal anemia includes monitoring middle cerebral artery peak systolic velocity (MCA-PSV) by Doppler ultrasound to detect fetal anemia, followed by IUTs once fetal anemia is confirmed by cordocentesis. Intravenous immunoglobulin G (IVIG) or plasmapheresis may be administered in selected pregnancies to delay the onset of fetal anemia and reduce the need for IUT [8].
Neonatal complications of HDFN include severe hyperbilirubinemia, neonatal jaundice, kernicterus, thrombocytopenia, iron overload, and cholestasis [9, 10]. Close monitoring for abnormal laboratory parameters in neonates is recommended to allow early detection of late anemia and other complications [11]. Long-term consequences of kernicterus may include auditory dysfunction and development disorders [12].
The main objective of this study was to better understand the clinical impact of non-ABO HDFN on pregnant women, fetuses and newborns. Secondary objectives included: (1) the evaluation of the incidence and trends of HDFN in Israel, (2) assessing the frequency and outcomes of severe HDFN, and (3) describing treatment patterns and complications of HDFN among affected pregnant women, fetuses, and newborns.
De-identified data from all pregnancies and births between January 1998 and
December 2021 were extracted from the Maccabi Healthcare Services (MHS) database.
MHS is a nationwide health plan (payer-provider) and covers more than one quarter
of the population in Israel. The MHS database contains longitudinal data on a
stable population of
The study population consisted of two main complementary cohorts, identified through an antenatal or postnatal non-ABO HDFN diagnosis codes during the period 1998–2021. The ICD-9 diagnosis codes used are described in Supplementary Table 2. Women diagnosed with HDFN during pregnancy were included in the pregnancy cohort. Women with multiple HDFN pregnancies were included once for each affected pregnancy. The newborn cohort included offspring from these pregnancies, as well as children diagnosed with HDFN during their first year of life.
Severe HDFN was defined as the occurrence of at least one of the following during pregnancy: IUT, fetal hydrops, preterm labor induction, or intrauterine death [1]. Fetal hydrops was identified based on ICD-9 codes in the Electronic Health Record (EHR) (Supplementary Table 1), and IUT was identified using procedure codes. HDFN diagnosed at 24 weeks of gestation or later was classified as late-onset HDFN.
Sensitivity analyses were conducted on subgroups of the pregnancy and newborn
cohorts in which the HDFN diagnosis was confirmed by a maternal positive
alloantibody test. Alloantibody data have been available in the MHS database
since 2005. Women with an HDFN diagnosis during pregnancy who had an antibody
titer of
Children born from these pregnancies were included in the alloantibody-confirmed
newborn cohort, as were children diagnosed with HDFN during their first year of
life whose mothers had an antibody titer of
HDFN incidence rates were calculated as the total number of diagnosed cases
divided by the corresponding total number of MHS members (pregnant women or
newborns). Gamma regression models were used to assess incidence trends over
time. Descriptive statistics are presented as mean
The incidence of HDFN during pregnancy in Israel remained stable, ranging
between 0.4–1 per 10,000 pregnancies for most of the study period. The incidence
of HDFN diagnosed in newborns was higher compared to the incidence of HDFN
diagnosed during pregnancy and declined from 1.8 per 10,000 newborns (95%
confidence interval [CI]: 0.9–3.4) in 1998 to 1.1 per 10,000 newborns (95% CI:
0.4–2.5) in 2021 (p
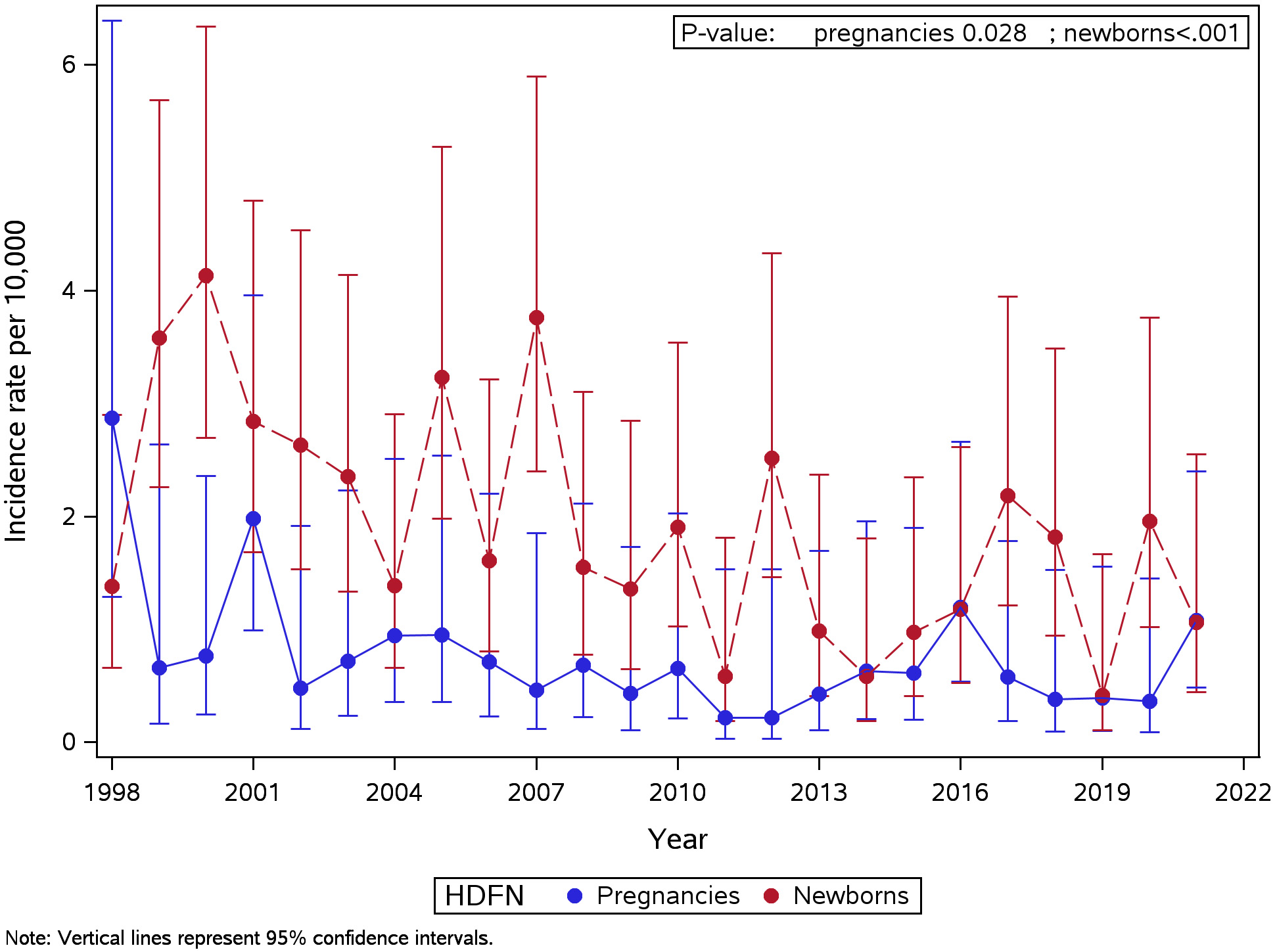 Fig. 1.
Fig. 1.
Incidence rate of HDFN diagnosed during pregnancy and in newborns. p-value of Gamma regression for trend over time; error bars represent the 95% CI. CI, confidence interval; HDFN, hemolytic disease of the fetus and newborn.
A total of 76 pregnancies with an HDFN diagnosis were identified in 70 women (6 women had 2 HDFN pregnancies). Medical information regarding pregnancy course and outcomes was available for 73 pregnancies (96%). Alloantibodies were confirmed in 27 women during or before 28 pregnancies (1 women had 2 HDFN pregnancies), representing 38.4% of all HDFN pregnancies (Fig. 2).
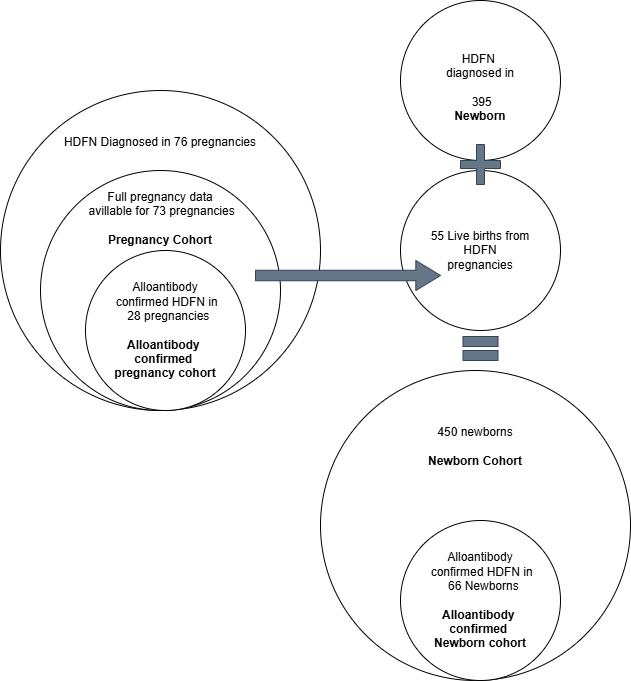 Fig. 2.
Fig. 2.
Cohort description.
During the 24-year study period, 395 additional newborns were diagnosed with HDFN postnatally; these cases were not identified during pregnancy because their mothers were not diagnosed during pregnancy (72%) or were not included in the MHS database. Out of the 57 newborns born to mothers diagnosed with HDFN, 55 were found in the MHS database and included in the HDFN newborn cohort. Therefore, a total of 450 HDFN newborns were identified and confirmed during their first year of life. Among these, 66 (14.7%) were confirmed to have HDFN during their first year of life based on maternal alloimmunization (Fig. 2).
The mean maternal age of 73 women diagnosed with HDFN at the time of diagnosis
was 31.4
The mean gravidity at which HDFN was diagnosed among the 73 pregnancies was 3.4
| Parameter | Pregnancy cohort | Alloantibody confirmed pregnancy cohort | |||
| Total (N = 73) | Severe (N = 22) | Total (N = 28) | Severe (N = 11) | ||
| Pregnancy characteristics | |||||
| GA at HDFN diagnosis (Mean |
23.4 |
24.5 |
23.9 |
24.5 | |
| Gravidity (Mean |
3.4 |
4.1 |
4.1 |
4.7 | |
| Pregnancy number category at diagnosis (N [%]) | |||||
| 1 | 12 (16.4) | 2 (9.1) | 1 (3.6) | 0 | |
| 2 | 9 (12.3) | 2 (9.1) | 1 (3.6) | 0 | |
| 3 | 12 (16.4) | 2 (9.1) | 8 (28.6) | 2 (18.2) | |
| 4+ | 30 (41.1) | 12 (54.5) | 17 (60.7) | 9 (81.8) | |
| Unknown | 10 (13.7) | 4 (18.2) | 1 (9.1) | 0 | |
| Twins (N [%]) | 2 (2.7) | 0 | 0 | 0 | |
| Late onset HDFN at 24 GA or later (N [%]) | 38 (52.1) | 12 (54.5) | 17 (60.7) | 6 (54.5) | |
| Any IVIG (N [%]) | 3 (4.1) | 2 (9.1) | 2 (7.1) | 2 (18.2) | |
| Any IUT (N [%]) | 14 (19.2) | 14 (63.6) | 9 (32.1) | 9 (81.8) | |
| Hydrops (N [%]) | 8 (11.0) | 8 (36.4) | 2 (7.1) | 2 (18.2) | |
| Pregnancy Outcomes | |||||
| Outcome (N [%]) | |||||
| Live birth | 57 (78.1) | 16 (72.7) | 24 (85.7) | 9 (81.8) | |
| Miscarriage | 9 (12.3) | 6 (27.3) | 3 (10.7) | 2 (18.2) | |
| Unknown | 7 (9.6) | 0 | 1 (3.6) | 0 | |
| GA at delivery (Mean |
37.6 |
34.9 |
36.2 |
34.9 | |
| GA at miscarriage (Mean |
18.2 |
19.9 |
13.9 |
20.7 | |
| Birthweight (g [Mean |
2964.2 |
2464.8 |
2750.9 |
2702.3 | |
| Type of miscarriage (N [%] among miscarriages) | |||||
| IUFD | 1 (11.1) | 1 (16.7) | 0 | 0 | |
| Spontaneous | 2 (22.2) | 0 | 1 (33.3) | 0 | |
| Induced | 6 (66.7) | 5 (83.3) | 2 (66.7) | 2 (100.0) | |
| Induced preterm labor (N [%] among preterm births) | 6 (31.6) | 6 (46.2) | 5 (33.3) | 5 (71.4) | |
| Preterm labor (N [%]) | 19 (33.3) | 13 (81.3) | 15 (62.5) | 7 (77.8) | |
| Preterm Category (N [%] among miscarriages) | |||||
| Extreme preterm ( |
0 | 0 | 0 | 0 | |
| Very preterm (28–31+6 GA) | 3 (15.8) | 3 (23.1) | 2 (13.3) | 2 (28.6) | |
| Moderate preterm (32–33+6 GA) | 3 (15.8) | 1 (7.7) | 2 (13.3) | 0 | |
| Late preterm (34–36+6 GA) | 13 (68.4) | 9 (69.2) | 11 (73.3) | 5 (71.4) | |
GA, gestational age; IVIG, intravenous immunoglobulin G; IUFD, intrauterine fetal death; IUT, intrauterine transfusion; SD, standard deviation; N, number of samples.
Among the alloantibody-confirmed cohort (N = 28), the mean gravidity at HDFN
diagnosis was 4.1
Among the 73 pregnancies with HDFN, 57 (78.1%) resulted in live births, 9 in
miscarriages, and 7 had unknown outcomes. Of the 57 live births, 19 (33%) were
preterm, of which 6 were induced. The majority of these were late preterm
(68.4%). Overall, the mean birthweight was 2964.2
Pregnancy outcomes in 22 severe HDFN pregnancies included 16 (72.7%) live births, of which 13 (81.3%) were preterm. Among the preterm births, 3 were very preterm (28–32 weeks of gestation), 1 was moderate preterm (32–34 weeks of gestation), and 9 were late preterm (34–37 weeks of gestation). Of the remaining 6 cases of severe HDFN, 5 were induced miscarriages and 1 was an intrauterine fetal death (IUFD). Among the 7 preterm deliveries in the severe HDFN alloantibody-confirmed pregnancies, 5 were induced labors (Table 1). Characteristics and outcomes of early- and late-onset HDFN pregnancies are presented in Supplementary Tables 4,5.
Among the 28 alloantibody-confirmed pregnancies, 24 (85.7%) resulted in live
births, 2 were induced abortions (1 at 20.7 weeks gestation and 1 at unknown GA),
1 was a spontaneous miscarriage at 7.1 weeks of gestation, and 1 had an unknown
outcome. Of the 24 live births, 15 (62.5%) were preterm, most of which were late
preterm (73.3%), with a mean birthweight of 2750.9
The pregnancy cohort included 3 women, each with 2 HDFN pregnancies during the study period. Pregnancy characteristics and outcomes for both pregnancies are presented in Supplementary Table 6. 1 woman delivered in both pregnancies, 1 woman delivered in the first pregnancy and had an induced miscarriage in the second pregnancy, and 1 woman spontaneously miscarried in the first pregnancy and delivered very preterm in the second. No cases of hydrops or IUFD were reported.
The median age at postnatal diagnosis among the 395 newborns was 2.7 weeks (IQR 0.7–5.7). The majority (58.2%) of all 450 HDFN newborns, including 55 born from HDFN pregnancies, were male.
Anemia and jaundice were the most common complications of HDFN during the first year of life, each occurring in more than 30% of the full newborn cohort. 12% of the newborns (n = 54) had developmental disorders at the one-year follow-up. Other complications included edema (5.1%), auditory dysfunction (3.3%), blood disorders (2.7%) and kernicterus (1.1%) as shown in Table 2. Among the alloantibody-confirmed newborns, anemia was diagnosed in 63.6% and jaundice in 27.3%; kernicterus was observed in 4 newborns (6.1%), edema in 4.5%, and developmental disorders in 3.0%. Almost one-third of alloantibody-confirmed newborns underwent blood transfusions (31.8%) and 10.6% received phototherapy.
| Complication | Newborn cohort (N = 450) | Alloantibody confirmed newborn cohort (N = 66) | ||
| N (%) | Age at diagnosis (months)/Median [IQR] | N (%) | Age at diagnosis (months)/Median [IQR] | |
| Anemia | 158 (35.1) | 1.2 [0.6–1.9] | 42 (63.6) | 0.8 [0.5–1.6] |
| Auditory dysfunction/deafness | 15 (3.3) | 3.3 [0.0–6.8] | 0 | NA |
| Development disorder | 54 (12.0) | 6.3 [4.2–9.3] | 2 (3.0) | 3.6 [2.9–4.2] |
| Edema | 23 (5.1) | 7.3 [3.5–9.2] | 3 (4.5) | 9.4 [8.2–9.9] |
| Jaundice | 154 (34.2) | 0.3 [0.1–1.0] | 18 (27.3) | 0.5 [0.2–0.9] |
| Kernicterus | 5 (1.1) | 0.2 [0.2–0.3] | 4 (6.1) | 0.3 [0.2–0.3] |
| Movement disorder | 1 (0.2) | 7.3 (NA) | 0 | NA |
| Oculomotor impairments | 4 (0.9) | 5.2 [2.5–9.3] | 0 | NA |
| Blood disorder (acidosis, calcemia) | 12 (2.7) | 1.5 [0.7–2.2] | 0 | NA |
| Other blood disorder (transient neonatal neutropenia) | 13 (2.9) | 2.7 [0.2–4.7] | 1 (1.5) | 0.2 [0.2–0.2] |
IQR, interquartile range; NA, not applicable; IQR not applicable for categories with n = 1.
Of the 450 newborns, 29% had complete blood count (CBC) results at 2–3 months of age and 37% had results at 10–12 months of age. Among the alloantibody-confirmed newborns, CBC data were available for 40% up to 1 month of age as well as in 10–12 month. Laboratory test results were similar between the full newborn cohort and the alloantibody-confirmed cohort (Supplementary Tables 7,8). Mean hemoglobin and hematocrit values were low during the first 3 months of life; however, both increased to normal levels by one year of age (Supplementary Fig. 1).
This study covered a 24-year timeframe which allowed the identification of long-term trends in HDFN. Based on MHS data, prenatal HDFN diagnosis has remained stable in Israel, whereas postnatal HDFN diagnosis has declined from 1.8 to 1.1 per 10,000 newborns since 1998. These results present a lower incidence compared to a Canadian single-center from 2010 to 2017, which reported an HDFN incidence rate of 6 per 10,000 newborns, and a national US study from 1996 to 2010, which found an incidence rate of 4 to 10 per 10,000 newborns [15, 16]. Consistent with our findings, the US study reported a decline in HDFN incidence attributed to reduced alloimmunization rates over time [16].
During the study period, 76 women were diagnosed with HDFN during pregnancy, compared to 395 newborns diagnosed during their first year of life. This discrepancy may indicate insufficient screening and surveillance in pregnancies at risk. Timely perinatal diagnosis of HDFN is essential to initiate optimal management and thus reduce the risk of hydrops fetalis or fetal death [17]. A European study showed that when IVIG was initiated before 13 weeks of gestation, the onset of fetal anemia was delayed, and anemia was less likely to occur prior to 20 weeks of gestation [8]. The finding that the majority of patients in the present study received a postnatal diagnosis of HDFN may help explain the rates of severe cases observed.
Severe HDFN was defined as the occurrence of at least one of the following during pregnancy: IUT, fetal hydrops, preterm labor induction, or intrauterine death. In our study, more than one quarter (28.8%) of all HDFN pregnancies and nearly 40% of alloantibody-confirmed pregnancies were classified as severe. Hydrops occurred in 7.1% of alloantibody-confirmed cases, a rate comparable to the 7.3% reported in a recent systematic review [18]. IVIG was administered in 7.1% of alloantibody-confirmed cases, and both individuals required at least 1 IUT. In addition, the only cases of very preterm delivery, which increases the risk of neonatal complications, occurred in severe HDFN pregnancies [19]. Most miscarriages were induced during the first trimester, which may indicate that the fetuses were in critical condition. A 10-year observational study from Finland reported 3.8% IUFD and 82% live births among women treated with IUTs for HDFN, comparable to the findings in the present study [20].
One third of all newborns in our study were diagnosed with anemia (mean age
Kernicterus, a severe complication of hyperbilirubinemia, causing chronic neurologic impairments, including hearing loss, visual abnormalities, dental abnormalities, and involuntary movements, was reported in 5 (over 1%) newborns, 4 of whom had alloantibody-confirmed HDFN. These complications occurred at rates comparable to or higher than those previously reported, indicating a more severe patient cohort in this database [17, 21].
Over 10% of newborns were reported to have a developmental disorder; however, among the alloantibody-confirmed HDFN cases, the rate was reduced to 3%. Lindenburg and colleagues [21] previously reported moderate and severe developmental delay in 14.4% and 3.1%, respectively, of a severe population exposed to IUT. Given the lack of prenatal data available for this newborn cohort (severity in pregnancy, incidence of hydrops) it is difficult to identify clear factors contributing to the high incidence observed in this study. However, these data reiterate the importance of continued investigation into risk factors for developmental disorders to ensure early identification and the implementation of prenatal interventions, which may improve short- and long-term outcomes. Measures that allow early detection of severe fetal anemia prior to development of fetal hydrops, as well as medical interventions that can postpone onset of severe fetal anemia, may prove to be beneficial for improving both neonatal and longer-term developmental outcomes.
The reason for the decrease in neonatal index cases, despite no change in prenatal diagnosis rates, is unknown. One possible explanation is that improved and earlier management of high-risk pregnancies has contributed to fewer HDFN events among neonates.
The main strength of this study is the longitudinal follow-up over a large database covering a quarter of Israel’s population for more than 20 years. This database includes comprehensive medical information recorded continuously, including treatments, diagnoses, laboratory results, and procedures for all members. In addition, Israel has a high birth rate, with a stable total fertility rate of 3 children per woman during the study period [22]. As the risk of HDFN rises with each pregnancy, this high birth rate allowed the capture of more HDFN cases, as well as more severe cases than expected in countries with lower birth rates [4]. Lastly, this study included sensitivity analyses on alloantibody-confirmed cases, which validated the study findings.
Several limitations should be noted. First, as a retrospective observational study, not all clinical and demographic information was fully captured in the database. Missing data included information needed to match the pregnant women with the child from the same pregnancy. As a result, the study cohorts did not capture all HDFN cases, and the approach involved two initial sets of cohorts. Second, alloantibody test results were not recorded in the MHS database until 2005, therefore the inclusion of HDFN cases in the main cohort of this study was based solely on diagnoses captured in the database. Third, HDFN management and treatment administered in inpatient settings are not always reported back to MHS (a community-based organization), and therefore may not captured in this database. As a result, treatment rates may be underestimated in this report. Therefore, this study cannot provide evidence of associations between HDFN management during pregnancy with perinatal outcomes. Further studies including larger cohorts of HDFN pregnancies are needed to assess fetal and neonatal outcomes following specific HDFN treatment protocols.
Although the incidence of HDFN diagnoses in Israel declined between 1998 and 2021, we observed cases with severe outcomes, including pregnancy termination, hydrops, preterm labor, anemia, jaundice, and kernicterus. Routine maternal blood testing for alloantibodies during early gestation can enable early diagnosis and management of HDFN pregnancies at risk for development of fetal anemia. Further studies are needed to understand and evaluate current treatment regimens for HDFN and to assess the association between perinatal management and both short- and long-term outcomes.
Upon request, and subject to review, the study Sponsor, Kahn-Sagol-Maccabi Research and Innovation Institute, will provide data that support the findings of this study. Subject to certain criteria, conditions and exceptions, Kahn-Sagol-Maccabi Research and Innovation Institute may also provide access to the related individual de-identified participant data. Contact Kahn-Sagol-Maccabi Research and Innovation Institute for more information.
NF, MF, WK, NB, SI, SG, AB and WN have made substantial contributions to the conception and design of the work. MH, YS, DM, and YY contributed to the discussion and critical revision of the manuscript, contributed to the interpretation of the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by Maccabi Healthcare Services Ethics Committee (approval #0021-22-MHS). As this non-interventional administrative study involves de-identified structured data, which according to applicable legal requirements does not contain data subject to privacy laws, obtaining informed consent from patients was waived by MHS IRB, as in accordance with the Declaration of Helsinki.
Not applicable.
This study was sponsored by Kahn-Sagol-Maccabi Research and Innovation Institute and funded by Janssen Pharmaceuticals.
All authors declare no conflicts of interest. Although some authors (MF, WK, NB, SI, AB, WN) are employees of Janssen Pharmaceuticals and the study received sponsorship from Janssen Pharmaceuticals, the interpretation of data and the writing of the manuscript were not influenced by this relationship.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG39932.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


