1 Department of Obstetrics, Puer People’s Hospital, 665000 Puer, Yunnan, China
2 Department of Imaging Medicine and Nuclear Medicine, Puer People’s Hospital, 665000 Puer, Yunnan, China
3 Department of Gynecology, Puer People’s Hospital, 665000 Puer, Yunnan, China
†These authors contributed equally.
Abstract
Type A2 gestational diabetes mellitus (A2GDM) is a challenging subtype of gestational diabetes, requiring insulin therapy in addition to exercise and dietary interventions. While multiple daily subcutaneous injections of regular insulin (RI) and insulin pumps are commonly used, their clinical efficacy and impact on maternal and infant metabolic indices remain unclear.
This retrospective study included 98 patients with A2GDM admitted to Puer People’s Hospital between March 2021 and September 2023. Patients were divided into a control group (n = 41) and an experimental group (n = 57) receiving insulin via an insulin pump. Both groups received treatment until delivery, and treatment efficacy, glucose metabolism, lipid metabolism, and maternal and infant complications were compared.
Pre-pregnancy, the experimental group had a lower body mass index (BMI) and fewer cases with a family history of diabetes. The experimental group achieved blood glucose targets faster, required a lower insulin dose, and had fewer hypoglycemic events (p < 0.05). Post-treatment, the experimental group showed greater improvements in fasting plasma glucose (FPG), 2-hour postprandial blood glucose (2hPBG), glycosylated hemoglobin (HbAlc), and homeostasis model assessment of insulin resistance (HOMA-IR), and lipid profile (total cholesterol [TC], high-density lipoprotein cholesterol [HDL-C], and low-density lipoprotein cholesterol [LDL-C]), with significantly better maternal and neonatal outcomes (p < 0.05). Complication rates were lower in the experimental group, including pregnancy-induced hypertension, cesarean section, polyhydramnios, and neonatal complications like macrosomia, hypoglycemia, and jaundice.
Multiple subcutaneous injections via insulin pump, that simulates physiological insulin secretion, requires a lower insulin dose for managing A2GDM and demonstrates superior efficacy in blood glucose control, improvement of glucose and lipid metabolism, as well as enhancement of both maternal and neonatal outcomes.
Keywords
- insulin
- type A2 gestational diabetes mellitus
- efficacy
- glucose metabolism
- lipid metabolism
- maternal and infant outcomes
- effect
Gestational diabetes mellitus (GDM) is an abnormal glucose metabolism syndrome occurring during pregnancy. Based on the effect of blood glucose management and control, this special type of diabetes mellitus (DM) involved in pregnancy can be divided into two subtypes: A1 and A2 [1]. Type A1, more commonly encountered in clinical settings, refers to cases in which optimal blood glucose control can be achieved through exercise guidance and nutrition management. A2 denotes that routine management cannot maintain blood glucose at a normal level, necessitating the use of metformin (MET), glibenclamide (Gli), and other hypoglycemic agents or insulin for auxiliary control [2, 3]. Auvinen et al. [4] conducted a 23-year cohort study and found that type A1 gestational diabetes mellitus (A1GDM) often develops within seven years prior to pregnancy, while type A2 gestational diabetes mellitus (A2GDM) shows a consistently higher incidence after delivery, with its risk linearly increasing to 50% by the end of the follow-up period, bringing about a life-long impact on the patients. A2GDM represents a prevalent complication in pregnant and lying-in women. Although the condition is diagnosed as DM associated with the gestation period, it is distinct from DM concurrently occurring during pregnancy. A2GDM carries a higher risk for both mothers and infants. If the blood glucose level cannot be stabilized, pregnant women are more prone to miscarriage, hypertensive disorder complicating pregnancy (HDCP) (including preeclampsia), infection, polyhydramnios, macrosomia, diabetic ketoacidosis (DKA), and type 2 diabetes mellitus (T2DM). Additionally, fetal growth restriction (FGR), neonatal hypoglycemia, and respiratory distress syndrome (RDS) may occur, impacting offspring health [5, 6, 7]. Literature suggests that A2GDM possibly leads to changes in the fetal heart structure. In contrast with fetuses under normal delivery conditions, fetuses of A2GDM patients display a thickened interventricular septum, thickened left and right ventricular walls, as well as a bigger impact on cardiac morphology and structural integrity [8]. Based on this, early diagnosis and adoption of appropriate blood glucose management are critical for improving maternal and neonatal outcomes. In addition to routine diet control, exercise, and other lifestyle interventions, insulin administration remains the cornerstone of treatment for A2GDM [9, 10]. A study has demonstrated that different administration methods of insulin may contribute to different therapeutic outcomes [11]. Therefore, it is particularly pivotal to select a more efficacious intensive insulin therapy regimen. Multiple subcutaneous injections and continuous subcutaneous insulin injection are two frequently employed methods at the current stage. In this study, we investigated the efficacy of different intensive insulin therapy regimens for the management of A2GDM, with the aim of identifying an effective strategy to support favorable delivery outcomes and improve fetal health, thereby enhancing overall maternal and neonatal well-being.
This retrospective study incorporated 98 patients diagnosed with A2GDM and
treated at Puer People’s Hospital between March 2021 and September 2023. The
patients’ ages ranged from 20 to 37 years, with a mean age of (27.02
The selected subjects shall conform to the diagnostic criteria for GDM published
in July 2022 by the National Medical Service Standards Committee of the Ministry
of Health of the People’s Republic of China. According to the internationally
recognized International Association of Diabetes and Pregnancy Study
Groups (IADPSG)/World Health Organization (WHO) diagnostic criteria [12] in terms
of the 75 g oral glucose tolerance test (OGTT), GDM can be diagnosed if any of
the following indicators is met: fasting plasma glucose (FPG)
All enrolled subjects met the diagnostic criteria for A2GDM and had singleton pregnancies, with complete clinical data available. Individuals receiving other drug therapies or other treatment regimens were excluded from our study. Moreover, patients with contraindications to the medications in this protocol, malignant tumors, immune system diseases, cognitive impairments, or hepatic/renal dysfunction were excluded. See Supplementary Fig. 1.
All patients received routine health education and individualized interventions, including diet control and exercise guidance tailored to their condition. Based on this, the experimental and control groups were assigned different intensive insulin therapy regimens.
The control group received multiple daily subcutaneous injections of RI (MPA approval number: J20100116; Novo Nordisk [China] Pharmaceuticals
Co., Ltd., Tianjin, China), administered 0.5 h before each meal with a daily dose
distribution of 40% for breakfast, 20% for lunch, and 40% for dinner. Isophane
protamine biosynthetic human insulin was injected subcutaneously every night
before sleep. Blood glucose levels were monitored using an Accu-Chek Active
glucometer (20182401730; Roche Diagnostics GmbH, Mannheim, Germany), with results
recorded in detail, and the insulin dose was maintained between 0.5–1.0
U/(kg
For the experimental group, continuous subcutaneous insulin infusion (CSII) was
implemented using an insulin pump Fornia IP-101 (20182401730; Fornia Medical
Equipment Co., Ltd., Shenzhen, Guangdong, China) with Novolin insulin (NMPA
approval number: J20100116; Novo Nordisk [China] Pharmaceuticals Co., Ltd.,
Tianjin, China). A continuous catheter and needle were used to deliver insulin
via the pump, programmed to provide a basal infusion rate and preprandial bolus
doses. The total daily insulin dose was evenly divided between basal and bolus
components (50% each), with the preprandial bolus doses distributed as 50% for
breakfast, 25% for lunch, and 25% for dinner. The pump was programmed to
control the infusion rate, maintaining the total insulin dose between 0.5–1.0
U/(kg
To ensure uniformity and comparability between the experimental and control
groups, this study implemented a standardized monitoring protocol. The
experimental group, receiving insulin pump therapy simulating physiological
insulin secretion, and the control group, receiving conventional multiple daily
subcutaneous insulin injections, were randomized to ensure no significant
differences in baseline characteristics, including age, gestational age, body
mass index (BMI), and diabetes duration (p
To ensure the scientific rigor and reproducibility of maternal and neonatal
outcomes related to A2GDM, this study clearly
defines the diagnostic criteria and measurement methods for all clinical
outcomes. Pregnancy-induced hypertension is defined as a systolic blood pressure
All data were analyzed using SPSS Statistics Version 24.0 (IBM, Chicago, IL,
USA). Measurement data were expressed as mean
In the experimental group (n = 57): The mean age was (27.35
| Group | n | Age (years) | WG (weeks) | Body mass (kg) | Pre-pregnancy BMI (kg/m2) | Parity | Education level | Family history of DM | |||
| Primipara | Multipara | Junior high school education or below | High school education or above | Presence | Absence | ||||||
| Experimental group | 57 | 27.35 |
30.09 |
68.41 |
30.28 |
33 (57.89%) | 24 (42.11%) | 28 (49.12%) | 29 (50.88%) | 4 (7.02%) | 53 (92.98%)* |
| Control group | 41 | 26.56 |
30.23 |
69.20 |
32.47 |
25 (60.98%) | 16 (39.02%) | 19 (46.34%) | 22 (53.66%) | 9 (21.95%) | 32 (78.05%) |
| 1.022 | 0.324 | 0.646 | 2.597 | 0.094 | 0.074 | 4.622 | |||||
| p | 0.309 | 0.747 | 0.520 | 0.010 | 0.760 | 0.786 | 0.032 | ||||
Note: * suggests compared with the control group, p
n, number of patients; WG, weeks of gestation; BMI, body mass index; DM, diabetes mellitus; SD, standard deviation.
As shown in Section 3.1, the proportions of patients with different
pre-pregnancy BMI and a family history of diabetes differed significantly between
the two groups. Therefore, these two factors were included in a multivariate
logistic regression equation to analyze their impact on insulin application. The
results confirmed that high BMI pre-pregnancy and a family history of DM were
both independent influencing factors for A2GDM patients requiring insulin
treatment (p
| Factor | S.E. | Wald | OR | 95% CI | p | |
| Pre-pregnancy BMI | 1.326 | 0.577 | 5.281 | 3.766 | 1.215–11.669 | 0.022* |
| Family history of DM | 1.156 | 0.563 | 4.216 | 3.177 | 1.054–9.578 | 0.041* |
Note: * suggests the factor is an independent influencing factor for A2GDM
patients requiring insulin therapy, p
S.E., standard error; OR, odds ratio; A2GDM, type A2 gestational diabetes mellitus.
The experimental group displayed a time of (4.15
| Group | n | Time of blood glucose reaching the standard (d) | Insulin dose (U/kg·d) | Incidence of hypoglycemia (times) |
| Experimental group | 57 | 4.15 |
32.78 |
2 (3.51%)* |
| Control group | 41 | 7.62 |
51.49 |
7 (17.07%) |
| 24.205 | 12.852 | - | ||
| p | 0.039a |
Note: a Fisher’s exact test was used due to expected frequency
Prior to treatment, FPG, 2 hours postprandial blood glucose (2hPBG), fasting
insulin, glycosylated hemoglobin (HbAlc), and homeostasis model assessment of
insulin resistance (HOMA-IR) displayed no statistically significant variances
between the two groups (p
| Group | n | FPG (mmol/L) | 2hPBG (mmol/L) | HbAlc (%) | HOMA-IR | Fasting insulin (µIU/mL) | ||||
| Pre-treatment | Post-treatment | Pre-treatment | Post-treatment | Pre-treatment | Post-treatment | Pre-treatment | Post-treatment | Pre-treatment | ||
| Experimental group | 57 | 7.45 |
4.01 |
12.36 |
7.12 |
7.91 |
5.13 |
4.25 |
2.08 |
12.85 |
| Control group | 41 | 7.48 |
5.19 |
12.31 |
8.25 |
7.89 |
6.60 |
4.23 |
3.11 |
12.72 |
| t | 0.246 | 11.171 | 0.187 | 6.427 | 0.126 | 15.328 | 0.294 | 16.190 | 0.305 | |
| p | 0.806 | 0.852 | 0.9 | 0.769 | 0.761 | |||||
Note: * means compared with pre-treatment levels in the same group,
p
FPG, fasting plasma glucose; 2hPBG, 2 hours postprandial blood glucose; HbAlc, glycosylated hemoglobin; HOMA-IR, homeostasis model assessment of insulin resistance.
Prior to treatment, no statistically significant variances were found in total
cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), or low-density
lipoprotein cholesterol (LDL-C) between the two groups (p
| Group | n | TC (mmol/L) | HDL-C (mmol/L) | LDL-C (mmol/L) | |||
| Pre-treatment | Post-treatment | Pre-treatment | Post-treatment | Pre-treatment | Post-treatment | ||
| Experimental group | 57 | 8.35 |
4.18 |
2.45 |
4.51 |
8.32 |
4.21 |
| Control group | 41 | 8.39 |
6.35 |
2.47 |
3.96 |
8.33 |
5.68 |
| t | 0.238 | 22.345 | 0.440 | 6.429 | 0.057 | 14.157 | |
| p | 0.812 | 0.661 | 0.955 | ||||
Note: * suggests compared with pre-treatment levels in the same group,
p
TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
Pregnancy-induced hypertension (PIH), cesarean section, polyhydramnios,
ketoacidosis, preeclampsia, premature delivery, and premature rupture of fetal
membranes are prevalent adverse pregnancy outcomes in A2GDM women. In our
research, the experimental group encompassed 3 cases of PIH, 4 cases with
cesarean section, 3 cases of polyhydramnios, 1 case of ketoacidosis, 3 cases of
preeclampsia, 2 cases of premature delivery, and no premature rupture of fetal
membranes. The incidence of the above outcomes was substantially lower than that
observed in the control group, and the variances showed statistical significance
(p
| Group | n | PIH | Cesarean section | Polyhydramnios | Ketoacidosis | Preeclampsia | Premature delivery | Premature rupture of fetal membranes |
| Experimental group | 57 | 3 (5.26%)* | 4 (7.02%) * | 3 (5.26%)* | 1 (1.75%) * | 3 (5.26%)* | 2 (3.51%)* | 0 (0.00%)* |
| Control group | 41 | 11 (26.83%) | 9 (21.95%) | 9 (21.95%) | 5 (12.20%) | 8 (19.51%) | 7 (17.07%) | 3 (7.32%) |
| 9.058 | 4.622 | 6.180 | - | 4.859 | - | - | ||
| p | 0.003 | 0.032 | 0.013 | 0.042a | 0.028 | 0.034 | 0.049 |
Note: a Fisher’s exact test was used due to expected frequency
PIH, pregnancy-induced hypertension.
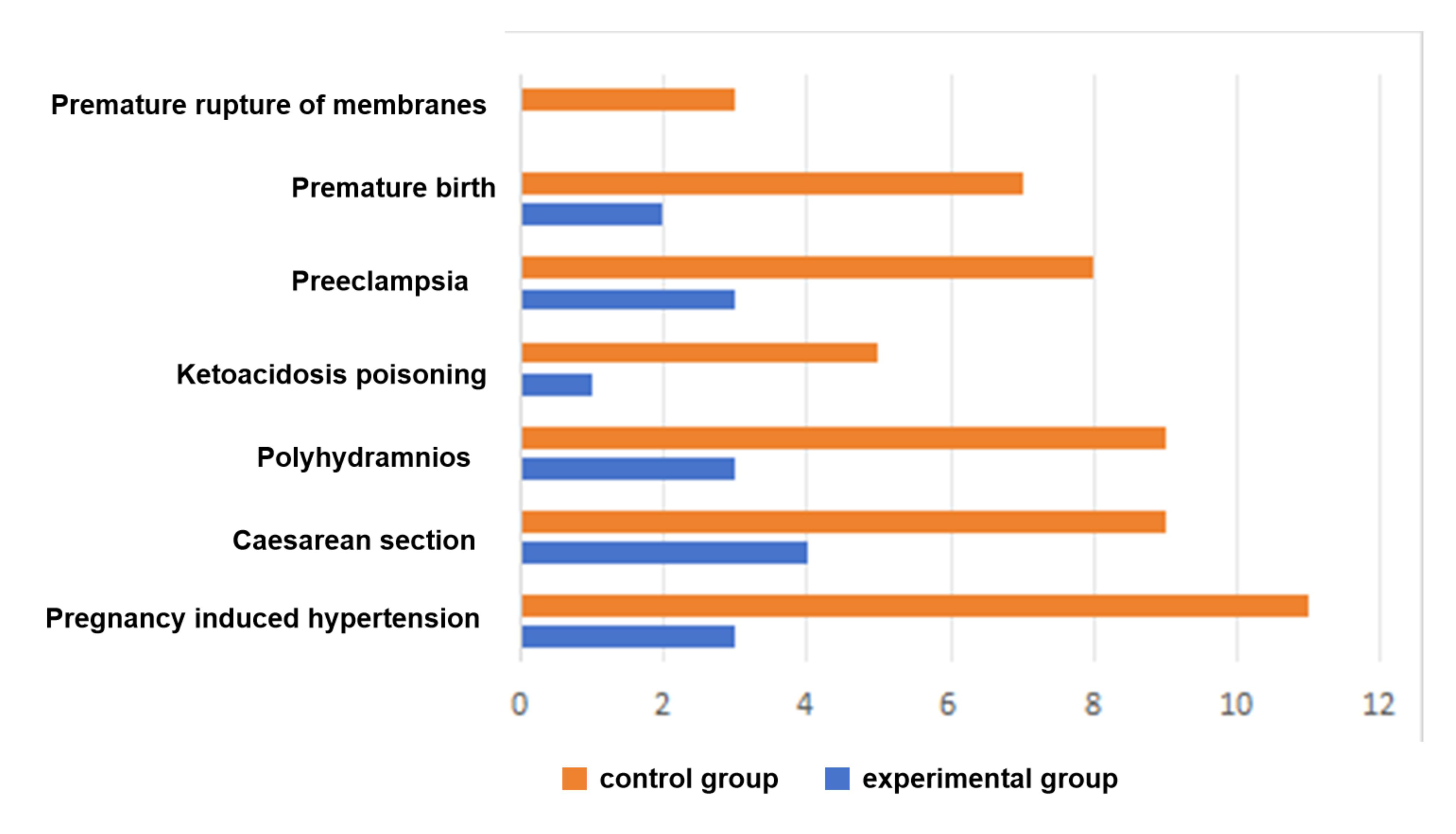 Fig. 1.
Fig. 1.
Differences in pregnancy outcomes between the two groups.
Newborns delivered by A2GDM patients are at increased risk for complications
such as macrosomia, neonatal hypoglycemia, low birth weight, neonatal respiratory
distress, and neonatal jaundice. In this study, neonatal outcomes were
statistically analyzed between the two groups. As displayed in Table 7 and Fig. 2, there were 8.77% newborns with macrosomia, 1.75% with hypoglycemia, 5.26%
with low birth weight, 3.51% with respiratory distress, and 10.53% with
jaundice. These proportions were all noticeably lower than those observed in the
control group, and the differences were statistically significant (p
| Group | n | Macrosomia | Neonatal hypoglycemia | Low birth weight | Respiratory distress | Neonatal jaundice |
| Experimental group | 57 | 5 (8.77%)* | 1 (1.75%)* | 3 (5.26%)* | 2 (3.51%)* | 6 (10.53%)* |
| Control group | 41 | 10 (24.39%) | 5 (12.20%) | 8 (19.51%) | 7 (17.07%) | 11 (26.83%) |
| 4.487 | - | - | - | 4.421 | ||
| p | 0.034 | 0.042a | 0.040 | 0.039a | 0.036 |
Note: a Fisher’s exact test was used due to expected frequency
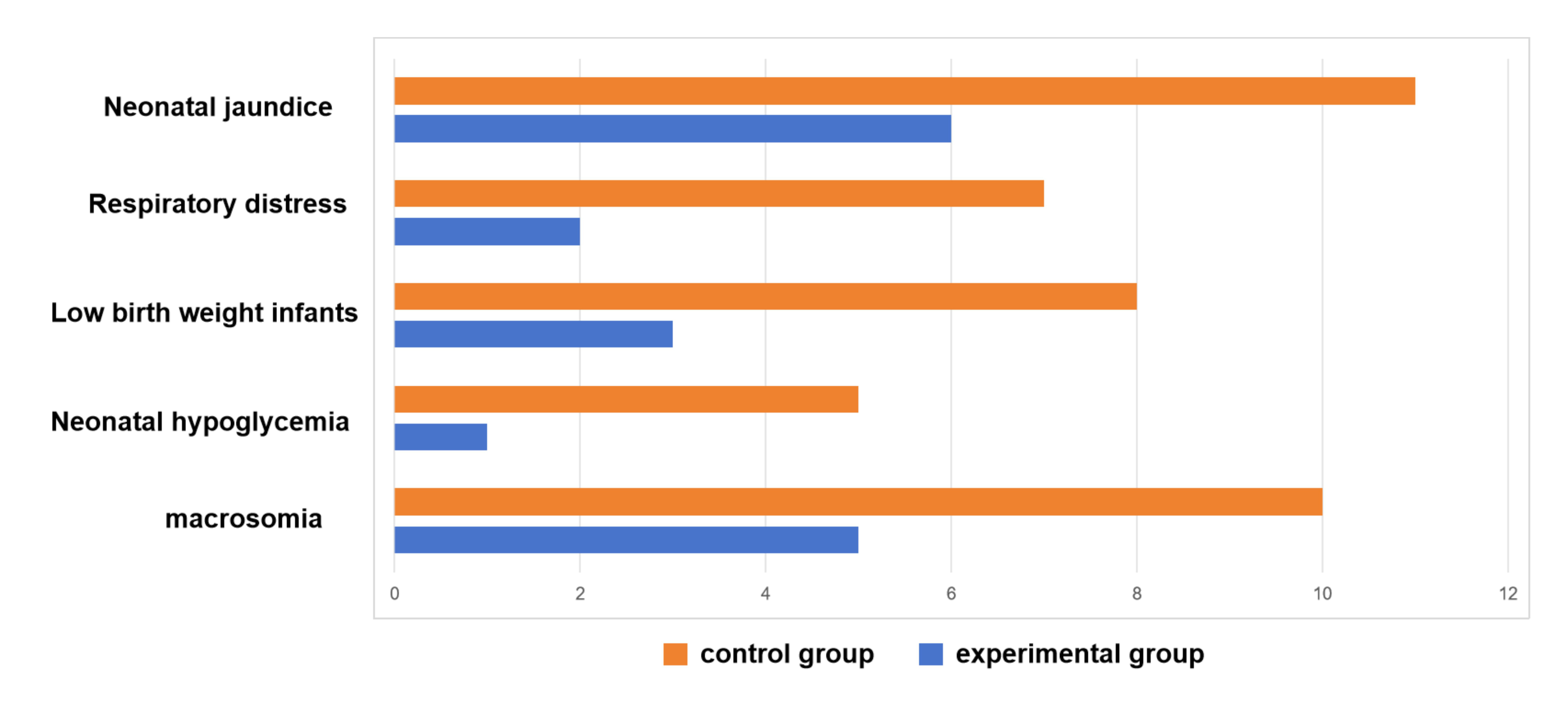 Fig. 2.
Fig. 2.
Differences in neonatal outcomes between the two groups.
Pregnancy represents a unique physiological state characterized by significant
increases in growth hormones, glucocorticoids, and sex hormones, which elevate
insulin requirements [6, 13]. These hormones antagonize insulin action,
exacerbating IR during gestation [7]. Consequently, some pregnant women
experience marked blood glucose fluctuations, particularly with excessive
nutritional supplementation, leading to GDM [14, 15]. With its incidence rising
annually, A2GDM, which accounts for over 50% of GDM cases, cannot be adequately
managed through diet and exercise alone. Uncontrolled A2GDM disrupts glucose,
lipid, and protein metabolism, causing vascular pathology under chronic
inflammatory conditions. This restricts tissue and organ blood supply, increasing
risks of premature delivery, preeclampsia, premature rupture of membranes, and
cesarean section, while also complicating delivery. For fetuses, uncontrolled
A2GDM heightens the likelihood of hypoglycemia, macrosomia, respiratory distress,
and other adverse outcomes [5, 16, 17], posing significant threats to maternal
and neonatal health. Therefore, A2GDM requires heightened clinical attention, and
effective blood glucose control strategies are essential to improve outcomes. Our
study identified baseline differences in pre-pregnancy BMI (30.28 vs. 32.47
kg/m2, p = 0.010) and family history of diabetes (7.02% vs.
21.95%, p = 0.032) between the CSII and multiple daily injections (MDI)
groups (Table 1). To address their potential confounding effects, we conducted
multivariate logistic regression analysis, confirming these factors as
independent predictors of insulin treatment requirements (Table 2, OR = 3.766 and
3.177, p
Insulin remains the cornerstone of clinical blood glucose management. Extensive research confirms its dual role in promoting glycogenolysis and suppressing gluconeogenesis, achieving sustained glycemic control through multiple pathways [18, 19, 20]. MDI of RI effectively manage A2GDM but have practical limitations, including operational inconvenience, susceptibility to subjective factors, and suboptimal glycemic control precision, which may hinder efficacy in some patients. In contrast, CSII offers an advanced insulin delivery method that mimics physiological insulin secretion [21]. Our study compared the efficacy of CSII and MDI in A2GDM, focusing on their impact on glucose and lipid metabolism and maternal and neonatal outcomes. The CSII group achieved target blood glucose levels faster, required lower insulin doses, and experienced fewer hypoglycemic events compared with the MDI group. Post-treatment, the CSII group exhibited significantly lower FPG, 2hPBG, HbA1c, and HOMA-IR levels, alongside improved lipid profiles (TC, HDL-C, and LDL-C). These findings indicate that while both insulin regimens improve glycemic and lipid metabolism in A2GDM, CSII provides superior glycemic control, reduced insulin requirements, and enhanced maintenance of glucose and lipid homeostasis. Additionally, CSII reduces hypoglycemia risk, offering a safer profile. Combined with adequate nutrition, exercise, and dietary interventions, CSII modulates insulin polypeptide levels and glucose tolerance, further improving metabolic outcomes [22, 23]. Notably, CSII alleviates the burden of multiple daily injections, reducing patient discomfort and enhancing convenience, efficiency, and safety for A2GDM management.
This study also evaluated maternal and neonatal outcomes for both insulin
regimens. The CSII group demonstrated significantly lower rates of maternal
complications, including pregnancy-induced hypertension (PIH), cesarean section,
polyhydramnios, ketoacidosis, preeclampsia, premature delivery, and premature
rupture of membranes, compared to the MDI group. Similarly, neonates born to
mothers in the CSII group had reduced incidences of hypoglycemia, low birth
weight, respiratory distress, and jaundice. These results underscore the superior
efficacy of CSII in improving maternal and neonatal outcomes. This advantage
likely stems from two key mechanisms: First, CSII mimics physiological insulin
secretion through continuous basal infusion and adjustable preprandial bolus
doses, allowing precise glycemic control tailored to meal timing and fasting
periods. This precision reduces postprandial glucose spikes and prevents morning
or nocturnal hypoglycemia and ketoacidosis [23, 24]. Second, intensive short-term
glycemic control with CSII rapidly achieves target glucose levels, minimizing the
adverse effects of sustained hyperglycemia, preserving residual pancreatic
Despite these findings, prior studies, including those by Gong et al. [23] and Kjölhede et al. [25], highlight limitations in A2GDM research. For instance, Gong et al. (2024) [23] compared CSII and MDI in pregnant women with type 1 diabetes but was constrained by a small sample size and single-center design, limiting generalizability. Similarly, Kjölhede et al. (2021) [25], a secondary analysis of an observational cohort, lacked randomization and long-term follow-up, restricting causal inferences and insights into sustained outcomes. These limitations underscore the need for larger, multicenter randomized controlled trials to validate CSII’s benefits in A2GDM.
Advancements in insulin delivery technology offer promising prospects for A2GDM management. Modern insulin pumps integrated with continuous glucose monitoring (CGM) systems provide real-time glucose feedback and automated insulin delivery, enhancing glycemic control precision and reducing hypoglycemic events. Emerging closed-loop systems, often termed artificial pancreas systems, dynamically adjust insulin doses based on real-time glucose levels, potentially offering superior outcomes for A2GDM patients. However, these technologies require further evaluation in pregnant populations to confirm their safety and efficacy.
Implementing CSII in low-resource settings presents significant challenges. High costs of insulin pumps and consumables, limited access to trained healthcare professionals, and inadequate infrastructure for device maintenance restrict adoption in rural or developing regions. Low health literacy and socioeconomic constraints further hinder patient adherence. To address these barriers, cost-effective pump designs, simplified training programs for healthcare providers, and telemedicine support could improve accessibility. Future research should explore scalable strategies to integrate advanced insulin delivery systems into diverse healthcare settings, ensuring equitable access for A2GDM patients.
Our study has limitations. As a retrospective analysis with a small sample size and limited case diversity, its generalizability is constrained. Prospective, multicenter, large-scale studies are needed to further validate our findings.
To sum up, multiple subcutaneous injections of RI and multiple subcutaneous injections via an insulin pump that simulates insulin secretion are both effective in managing A2GDM. However, in comparison, the insulin pump approach, which requires a lower dose of insulin, is more efficacious in controlling blood glucose levels, improving glucose and lipid metabolism, and enhancing both maternal and neonatal outcomes, making it suitable for clinical implementation and wider adoption.
The datasets used during the current study are available from the corresponding author upon reasonable request.
XY, YL designed the research study; XY, YL and YG performed the research; XY, YL and YG collected and analyzed the data. XY, YL and YG have been involved in drafting the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was reviewed and approved by the Puer People’s Hospital (2025-009-01), complies with the Declaration of Helsinki. Written informed consent was obtained from all participants before their inclusion in the study.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this manuscript, authors used ChatGPT to assist in checking spelling and grammar. The authors reviewed and edited the content as necessary and take full responsibility for the final version of the manuscript.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG41004.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


