1 Division of Neonatology, Konya City Hospital, University of Health Science, 42020 Konya, Turkey
2 Department of Pediatrics, Konya City Hospital, University of Health Science, 42020 Konya, Turkey
3 Department of Biochemistry, Konya City Hospital, University of Health Science, 42020 Konya, Turkey
4 Clinic of Obstetrics and Gynecology, Konya City Hospital, University of Health Science, 42020 Konya, Turkey
Abstract
This study aimed to compare the cord blood phoenixin-14 (PNX-14) levels of newborns across various birth weight categories with those of infants born to mothers diagnosed with gestational diabetes mellitus, as well as those who were not classified by birth weight.
This prospective study was conducted between December 2022 and April 2024. Healthy term babies whose families agreed to participate in the study were divided into four groups: Three were according to their birth weight small for gestational age (SGA), appropriate for gestational age (AGA) and large for gestational age (LGA), with the fourth being according to the gestational diabetes mellitus (GDM) status of the mother. This study included a total of 160 patients, with 40 in each group.
The PNX-14 values for the AGA and LGA groups were statistically similar, while those between the PNX-14 values of the SGA and GDM groups were also statistically similar. However, the PNX-14 values in the AGA and LGA groups were statistically higher than those in the SGA and GDM groups (p < 0.001).
This study provides evidence that lower cord blood PNX-14 levels in infants are associated with low birth weight and maternal diabetes. A correlation was found between the umbilical cord PNX-14 level and birth weight.
The study has been registered on https://clinicaltrials.gov/ (registration number: NCT06656481; registration link: https://clinicaltrials.gov/study/NCT06656481?cond=NCT06656481&rank=1#collaborators-and-investigators).
Keywords
- phoenixin-14
- gestational diabetes mellitus
- small for gestational age
- umblical cord blood
- birth weight
In later life, birth weights by gestational week are linked to concomitant disorders. Newborn infant mortality and morbidity are determined by birth weight and week of pregnancy [1]. During the neonatal stage, large for gestational age (LGA) and small for gestational age (SGA) infants may experience issues such as respiratory support, hypoglycemia, hypoxia, and polycythemia. These babies may also develop conditions like metabolic syndrome, obesity, cardiovascular disease, and diabetes mellitus that impact their quality and length of life later in life [1, 2]. Infants are classified as SGA if their birth weight is less than the 10th percentile by gestational week, LGA if their birth weight is greater than the 90th percentile, and appropriate for gestational age (AGA) if their birth weight falls between the 10th and 90th percentiles [2]. The most prevalent pregnancy problem is called gestational diabetes mellitus (GDM), which is any level of glucose intolerance that develops or is initially identified during pregnancy [3]. Long linked to obstetric and neonatal problems, especially those involving high birth weight, GDM is now more well acknowledged as a risk factor for the child’s subsequent cardiometabolic disease. Since maternal sickness or changes in birth weight can be linked to major health complications, research on these topics is still ongoing [4].
In 2013, Yosten et al. [5] published the first description of the
neuropeptide phoenixin (PNX). The most common isoforms of PNX, phoenixin-14
(PNX-14) and phoenixin-20 (PNX-20), exhibit similar biological activities and are
peptides composed of 14 and 20 amino acids, respectively. As a member of the
phoenixin peptide family, PNX-14 exerts its effects through interaction with G
protein-coupled receptor 173 (GPR173), a G protein-coupled receptor found in the
central nervous system and peripheral tissues [5]. PNX-14 is a recently
discovered neuropeptide that plays critical roles in a range of physiological
processes, including reproductive function, inflammation regulation, energy
metabolism, and food intake [6, 7]. Emerging evidence suggests [6] that PNX-14
may be closely associated with metabolic homeostasis and inflammatory states; it
is a key element in pregnancy-related metabolic disorders, including GDM.
Research on animals has demonstrated that by lowering oxidative stress, PNX-14
therapies reduce pancreatic damage and
However, studies on PNX-14 in the context of pregnancy and neonatal health are still scarce. A few experimental studies [9, 10] have examined the expression of PNX-14 in metabolic disorders, but little is known about its levels in umbilical cord blood, particularly in the setting of term infants born to mothers with or without GDM. Furthermore, existing studies often lack a focus on neonatal outcomes such as birth weight, an important clinical marker of intrauterine health and placental function. Previous research [8, 9, 10, 11] has several limitations that this study aims to overcome: Most studies on PNX-14 are preclinical or based on small adult populations, with few focusing on pregnant women or neonates. Very few studies have analyzed PNX-14 levels in cord blood, despite its relevance in assessing the fetal metabolic environment. The potential influence of maternal GDM on fetal PNX-14 expression has not been systematically studied. Although fetal growth is a key outcome in obstetrics, previous studies have not directly compared PNX-14 levels across different birth weight groups.
This study aims to address these research gaps by evaluating PNX-14 levels in umbilical cord blood of term infants and examining their association with birth weight normal (AGA), low (SGA), high (LGA) and maternal GDM status regardless of birth weight. The rationale is twofold: to explore whether PNX-14 levels correlate with neonatal anthropometrics, particularly birth weight, and to determine whether a maternal metabolic condition as GDM influence fetal PNX-14 levels. Investigating these associations may provide insights into the intrauterine regulatory mechanisms of metabolic peptides and their potential as biomarkers for maternal-fetal health.
In this study, we examined the associations between phoenixin-14 levels in the umbilical cord blood of healthy infants of healthy mothers and infants of mothers with GDM, regardless of birth weight, and normal (AGA), low (SGA), and high (LGA) birth weight based on gestational week of pregnancy.
Newborns whose mothers suffered from any illness other than diabetes were not included. The study also eliminated infants who required intensive care, were born after early rupture of the membranes, had multiple congenital abnormalities, chorioamnionitis, or preeclampsia, or were otherwise premature or sick. The study comprised healthy term infants (born between 370/7 and 426/7 weeks) delivered at birth either cesarean section or spontaneous vaginal delivery. The PNX-14 levels of the patients who were part of the trial were documented, together with their demographic and neonatal information.
Based on earlier research [11], a two-way independent-samples t-test power analysis was used to establish the sample size. We intended to include 40 infants in each group in order to minimize the margin of error. Previous similar studies [8, 11] have shown that phoenixin-14 ratios with an error margin of 0.05, actual power level of 0.9554213, and effect size of 1.41 would require a minimum of 24 deliveries (12 vaginal deliveries and 12 cesarean sections) for each pairwise comparison. In this study, 4 different categories were taken into consideration:
(1) Low birth weight by gestational week (SGA) newborns: Two of the 42 infants in this group whose mothers consented to take part in the study were excluded because of either insufficient laboratory results or sepsis-related hospitalization in the neonatal intensive care unit, and 40 SGA infants (20 born by spontaneous vaginal delivery and 20 by cesarean delivery) were included in the study.
(2) Normal birth weight by gestational week (AGA) newborns: Of the 47 infants in this group, 7 were excluded due to incomplete laboratory data or mothers declining to participate, and 40 AGA infants (20 born by spontaneous vaginal delivery and 20 by cesarean delivery) were included in the study.
(3) High birth weight by gestational week (LGA) newborns: The study included 40 LGA infants (20 born by spontaneous vaginal delivery and 20 by cesarean delivery), out of the 44 babies in this group whose mothers consented to participate. Four of these babies were excluded because of suspected sepsis, incomplete laboratory data, or hospitalization in the neonatal intensive care unit.
(4) Newborns whose mothers had gestational diabetes mellitus (GDM): 40 infants (4 born by spontaneous vaginal delivery and 36 by cesarean delivery) were included in the study out of the 41 infants in this group whose mothers consented to participate; one was disqualified because prenatal demographic data was unavailable. In the GDM group, 7 infants were classified as LGA, and 33 were classified as AGA. There were no infants classified as SGA in this group.
All infants had their umbilical cord blood samples taken after their parents gave their informed consent. Within six hours, the collected blood samples were centrifuged, and the serum was extracted. Enzyme immunoassay studies of serum PNX-14 levels were conducted using commercial kits (BT Lab Bioassay Technology Laboratory Human ELISA Kits, Shanghai Korain Biotech, Shanghai, China) in accordance with the manufacturer’s instructions. The absorbance was measured at 450 nm using an ELx800 Absorbance Microplate Reader (Biotech, Winooski, VT, USA). The PNX-14 concentration measurements were given in ng/L. The standard curve range was 20–3800 ng/L, and the kit’s sensitivity was 8.19 ng/L. Differences within and between assays were less than 8% and 10%, respectively.
The data were analyzed using IBM SPSS Statistics 29 (IBM Corp., Armonk, NY,
USA). Descriptive statistics were presented as number of units (n), percentage
(%), mean
The study included a total of 160 patients, with 40 in each group. The gender
distributions of the groups were statistically different (p
| SGA | AGA | LGA | GDM | Test value | p-value | ||
| n = 40 | n = 40 | n = 40 | n = 40 | ||||
| Gender, n (%) | |||||||
| Male | 17 (42.5)ab | 15 (37.5)a | 26 (65.0)b | 25 (62.5)b | 9.288 | 0.026‡ | |
| Female | 23 (57.5)ab | 25 (62.5)a | 14 (35.0)b | 15 (37.5)b | |||
| Delivery, n (%) | |||||||
| Vaginal | 20 (50.0)a | 20 (50.0)a | 20 (50.0)a | 4 (10.0)b | 20.000 | ||
| Caesarean section | 20 (50.0)a | 20 (50.0)a | 20 (50.0)a | 36 (90.0)b | |||
| Gestational age (week) | 38.8 |
39.1 |
39.0 |
38.9 |
0.392 | 0.759† | |
| Birth weight (gram) | 2479.0 |
3288.9 |
4275.5 |
3575.5 |
187.762 | ||
| Birth length (cm) | 48.0 (46.0–49.0)a | 50.0 (49.0–52.0)b | 53.0 (52.0–54.0)c | 51.0 (49.0–52.0)b | 79.563 | ||
| Head circumference (cm) | 33.0 (32.0–34.0)a | 35.0 (34.0–35.0)b | 37.0 (36.0–38.0)c | 35.5 (35.0–36.7)b | 98.389 | ||
| Mother’s age (year) | 26.6 |
29.4 |
27.0 |
31.5 |
7.475 | ||
| The mother’s BMI before pregnancy (kg/m2) | 23.84 |
23.51 |
24.10 |
23.95 |
0.723 | 0.540† | |
| The mother’s BMI at delivery (kg/m2) | 26.97 |
26.81 |
27.08 |
26.70 |
0.388 | 0.762† | |
| Apgar (1 min score) | 8.0 (8.0–9.0) | 9.0 (8.0–9.0) | 8.0 (8.0–9.0) | 8.0 (8.0–9.0) | 7.108 | 0.069& | |
| Apgar (5 min score) | 9.5 (9.0–10.0) | 10.0 (9.0–10.0) | 9.0 (9.0–10.0) | 9.0 (9.0–10.0) | 7.435 | 0.059& | |
| PNX-14 (ng/L) | 1129.2 (1112.1)a | 2059.6 (1761.1)b | 2523.2 (1375.0)b | 1156.8 (1114.4)a | 39.261 | ||
n: Number of patients; %: Percentage value; Numerical variables are presented
as mean
There was a statistically significant difference between the PNX-14 values of
the groups (p
| SGA | AGA | LGA | GDM | Test value | p-value | |
| n = 40 | n = 40 | n = 40 | n = 40 | |||
| PNX-14 (ng/L)* | 1406.7 |
2173.9 |
2503.4 |
1514.5 |
4.181 | 0.007¥ |
The data are presented as mean
According to Table 2, there was a statistically significant difference between
the PNX-14 values of the groups (p
According to the ROC curve analysis presented in Table 3, all area under the curve (AUC) values were statistically significant. The cut-off values found for SGA & LGA and GDM & LGA completely separated the groups from each other (Fig. 1).
| AUC | p-value | Cut-off | Sens | Spec | |
| (95% CI) | (95% CI) | (95% CI) | |||
| SGA & AGA | 0.961 | 90.0 | 100.0 | ||
| (0.892–0.991) | (76.3–97.2) | (91.2–100.0) | |||
| SGA & LGA | 1.000 | 100.0 | 100.0 | ||
| (0.955–1.000) | (91.0–100.0) | (91.2–100.0) | |||
| GDM & AGA | 0.962 | 97.5 | 90.0 | ||
| (0.893–0.992) | (86.8–99.9) | (76.3–97.2) | |||
| GDM & LGA | 1.000 | 100.0 | 100.0 | ||
| (0.955–1.000) | (91.2–100.0) | (91.2–100.0) |
Analyses were conducted on PNX-14 values adjusted for gender, maternal age, and mode of delivery. Cut-off, cut-off point; AUC, area under the curve; 95% CI, 95% confidence interval; Sens, sensitivity; Spec, specificity; ROC, receiver operating characteristic.
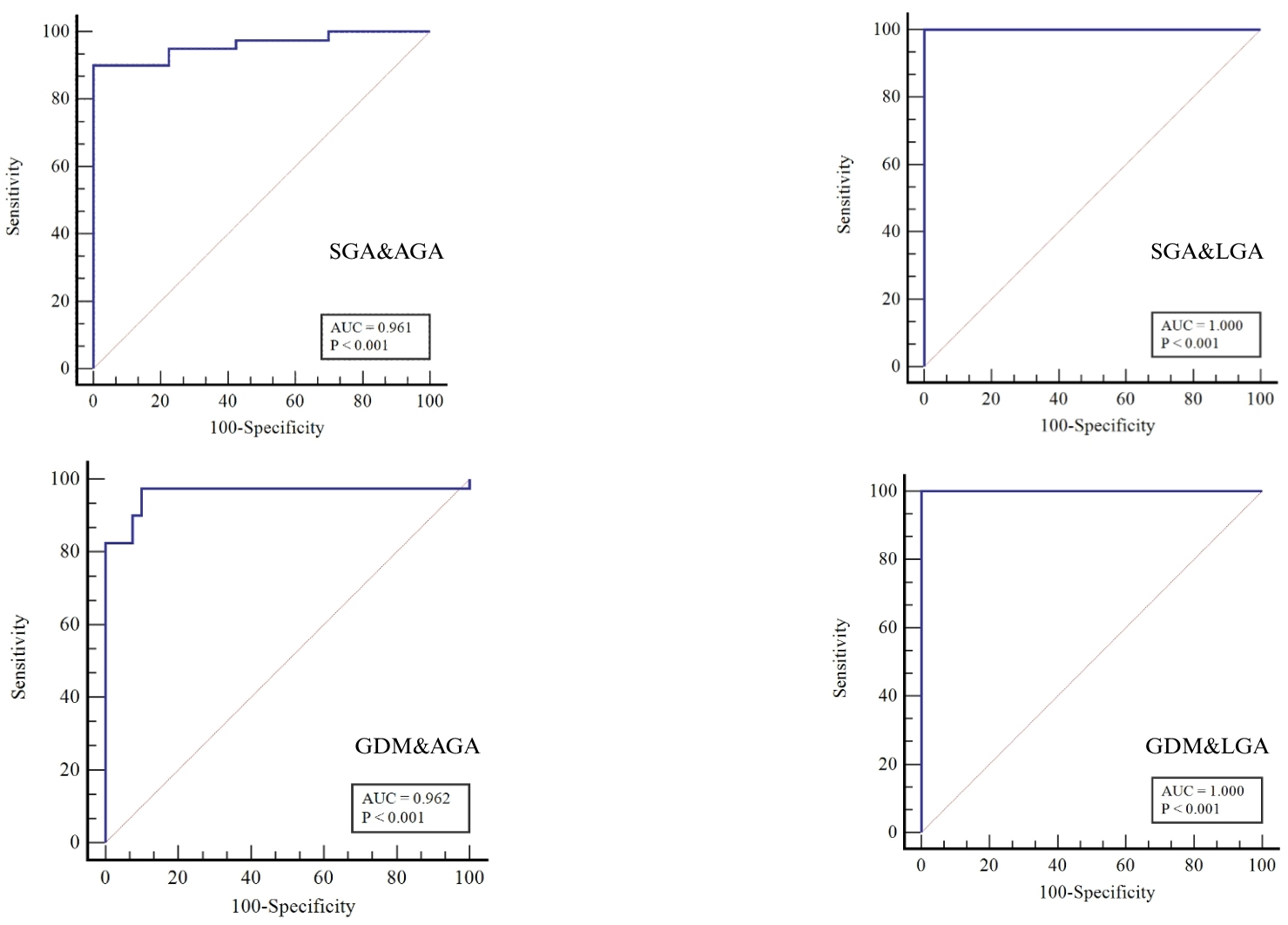 Fig. 1.
Fig. 1.
PNX-14 ROC curves. ROC, receiver operating characteristic; SGA, small for gestational age; AGA, appropriate for gestational age; LGA, large for gestational age; GDM, gestational diabetes mellitus; AUC, area under the curve; PNX-14, phoenixin-14.
We assessed the association between PNX-14 and birth weight regardless of the
group distinction. The relationship between PNX-14 and birth weight is minor but
statistically significant (r = 0.286; p
The birth weight of a newborn is the most important anthropometric measure used in the assessment of pregnancy. Low or high birth weight based on gestational age can cause serious health problems leading to morbidity and mortality both in the perinatal period and later in life [12]. Therefore, recognition of the factors affecting fetal growth and birth weight is important [13]. Fetal growth is controlled by many factors, primarily genetic factors. İt was found that the height of fathers played a significant role in determining the size of the fetus and the weight of the newborn [14]. However, fetal growth is a complex process influenced not only by genetic factors but also by maternal nutrition, placental functions, and hormonal balances. Intrauterine growth restriction or overgrowth can be associated with deficiencies or excesses in the intrauterine environment. These conditions may impact health not only during the perinatal period but also throughout an individual’s lifetime. Particularly, the roles of neuropeptides affecting fetal growth have been gaining increasing attention in recent years.
This study aimed to investigate the relationship between PNX-14 levels in
umbilical cord blood, and birth weight. There are similar studies in the
literature investigating factors that may affect fetal growth and birth weight.
Compared to AGA and LGA newborns, higher alarin levels were found in the
umbilical cord blood of SGA babies and babies born to mothers with GDM,
regardless of birth weight [15]. Umbilical cord blood spexin and leptin levels
showed positive correlations with birth weight, length, and head circumference in
the study conducted by Pekal et al. [16]. Han et al. [17]
linked the underdevelopment of fetal adipose tissue and gastrointestinal tract
with changes in umbilical cord blood adiponectin, leptin, and ghrelin levels. In
another study, SGA newborns showed significantly higher visfatin concentration
and lower levels of ghrelin, leptin, resistin, and adiponectin than AGA and LGA
newborns [18]. Studies have also shown that umbilical cord ghrelin concentrations
correlate negatively with birth weight and BMI [19, 20]. Studies associating
omentin-1 and insulin-like growth factor 1 levels in neonatal umbilical cord
blood with birth weight are also available in the literature [21, 22]. In our
study, we assessed the association between umblical cord blood PNX-14 levels and
birth weight regardless of the group distinction. The relationship between PNX-14
and birth weight is minor but statistically significant (r = 0.286; p
In our study, the umbilical cord blood PNX-14 levels of newborn term infants were evaluated according to birth weight and it was found that the PNX-14 values of the SGA group were statistically significantly lower than those of the AGA and LGA groups. The PNX-14 values of the AGA and LGA groups were statistically similar. Parker and Bloom [26] emphasized that food intake and body weight are regulated by orexigenic and anorexigenic neuropeptides expressed in the hypothalamic arcuate nucleus. Studies showing that PNX-14 levels are related to the nutritional status of the animal and that externally administered peptide increases the ability of PNX-14 to affect nutrition and play a role in the control of BMI regulation in humans suggest that PNX-14 is an orexigenic neuropeptide [7, 23, 24, 25]. The hypothesis that PNX-14 affects fetal nutrition behaviors and consequently influences fetal growth may not be entirely accurate. However, the study suggests that PNX enhances mitochondrial biogenesis and protects cells from inflammation. It is possible that in SGA infants, the lower levels of PNX-14 could be due to its antioxidant effects serving as an anti-inflammatory agent [27]. Additionally, the anti-inflammatory and antioxidant properties of PNX-14 may modulate cellular damage and stress responses. These features call for a broader evaluation of the potential effects of PNX-14 on fetal growth and placental functions.
In our study, PNX-14 levels were low in the GDM group regardless of birth
weight, similar to the SGA group. In mouse studies, PNX-14 treatments reduced
oxidative stress and prevented pancreatic damage and
According to the ROC curve analysis presented in Table 3, the cut-off values found for SGA & LGA and GDM & LGA completely separated the groups from each other. The AUC values for SGA & LGA and GDM & LGA reported in Fig. 2 appear unusually high, which may suggest overfitting or other methodological concerns. Considering that the sample size of each group is relatively small, only 40 cases, the results may have potential biases. Future research should expand the sample size for verification.
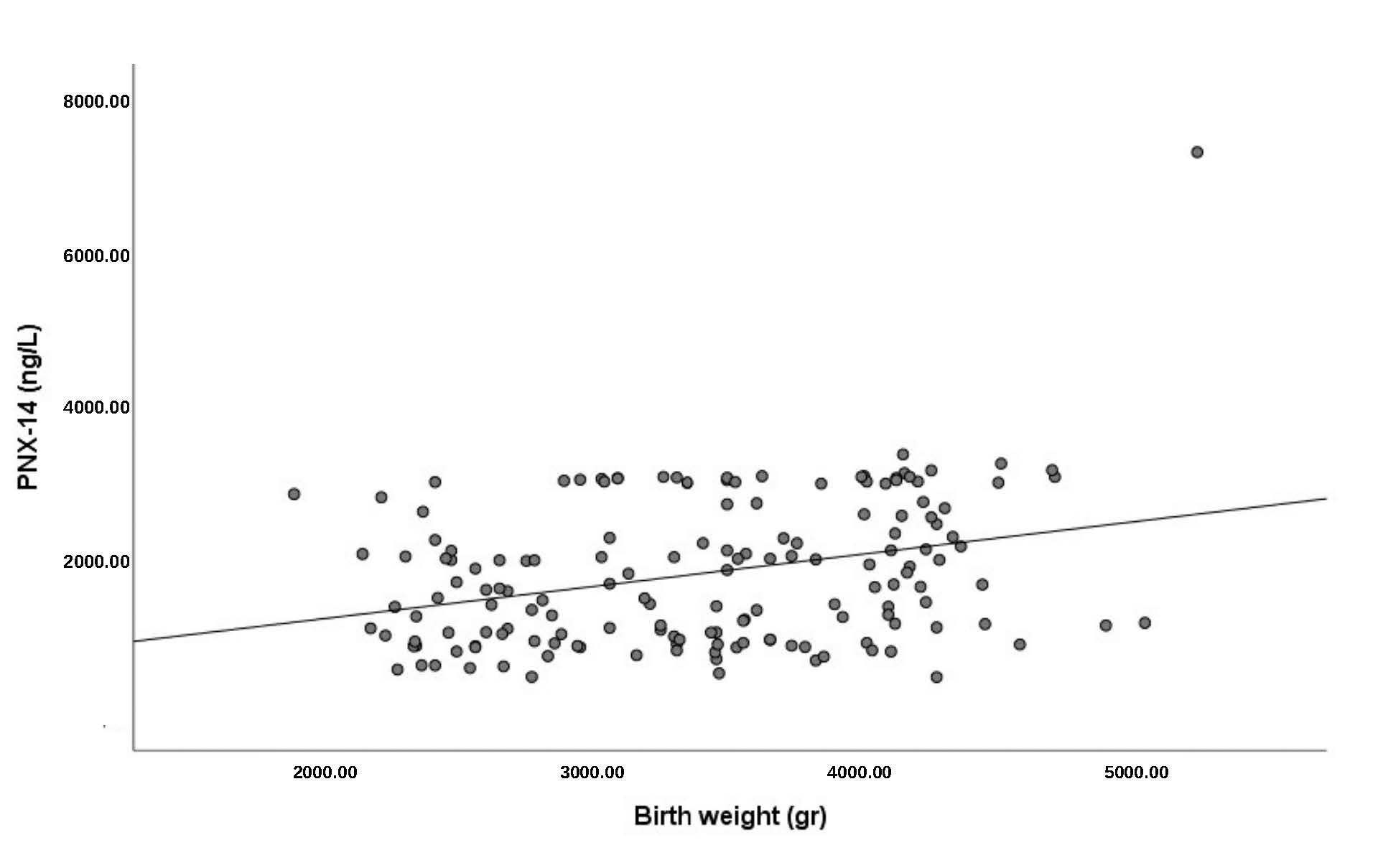 Fig. 2.
Fig. 2.
Scatterplot for umbilical cord PNX-14 levels and birth weight. PNX-14, phoenixin-14.
While our study provides valuable insights, our study has some limitations. The cross-sectional design limits our ability to establish causality between PNX-14 levels and birth outcomes. Additionally, the study focused on term infants, and the findings may not be generalizable to preterm populations. Future longitudinal studies are needed to further elucidate the role of PNX-14 in fetal development and its potential as a biomarker for assessing fetal health in the context of maternal metabolic conditions. Only the infant PNX-14 levels were examined so simultaneous maternal PNX-14 levels were not assessed. Additionally, in the GDM group, the numbers of vaginal and cesarean births, as well as the numbers of SGA, AGA, and LGA, were not evenly distributed. Distinguishing the independent effects of birth weight and GDM on PNX-14 levels proves to be challenging. It had a small sample size and was conducted in a single center. More comprehensive multi-center studies with larger numbers of patients should be conducted to confirm the effects of PNX-14 on fetal growth and maternal diabetes and to allow for clearer conclusions.
Low birth weight and maternal diabetes have been associated with many diseases in the perinatal period, and some processes that start in the intrauterine period may have effects until the adult period, causing morbidity and mortality in adulthood. Our findings support the need for broader studies investigating the effects of PNX-14 on fetal growth and maternal metabolic health. Specifically, large-scale, multi-center studies involving simultaneous maternal and fetal PNX-14 measurements across diverse populations and maternal metabolic conditions are essential to better understand the biological functions of this neuropeptide. Furthermore, the potential therapeutic implications of PNX-14 for complications related to both gestational diabetes and fetal growth require further exploration.
The authors confirm that data supporting the findings of this study are available within the article and its supplementary material. Raw data that support the findings of this study are available from the corresponding author.
RK, MB and ÜC designed the study and wrote the manuscript. ÜC, MB and FKY provided help and advice on the ELISA studies. AK, FHY and BÖ analyzed the data. RK, BÖ, FHY and FKY collected data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Both Konya City Hospital and the Karatay University Ethics Committee gave their approval for this cross-sectional study (Ethics Committee Decision No: 2022/005). The study was carried out in accordance with the guidelines of the Declaration of Helsinki in the Department of Obstetrics and Gynecology, the Neonatal Intensive Care Unit, and the Department of Medical Biochemistry at Konya City Hospital from December 2022 to April 2024 (Registered on https://clinicaltrials.gov/ (registration number: NCT06656481; registration link: https://clinicaltrials.gov/study/NCT06656481?cond=NCT06656481&rank=1#collaborators-and-investigators). All patients or their families/legal guardians gave their informed consent for inclusion before they participated in the study.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


