1 Department of Gynecology and Obstetrics, Dongyang Hospital of Wenzhou Medical University, 322100 Jinhua, Zhejiang, China
Abstract
The incidence of endometrial cancer is steadily rising worldwide. Although surgical interventions, particularly comprehensive lymphadenectomy, have improved survival rates, postoperative lymphoceles formation remains a significant clinical complication adversely affecting patient recovery and quality of life. In recent years, the estimation of physiologic ability and surgical stress (E-PASS) scoring system has emerged as a valuable tool in perioperative risk assessment. This system provides a comprehensive evaluation by quantifying patients’ physiological reserves and surgical stress. Although this scoring method has demonstrated value across various surgical disciplines, its potential utility in gynecological oncology remains to be thoroughly explored.
In this retrospective cohort study conducted at Dongyang Hospital affiliated with Wenzhou Medical University, China, we analyzed 180 patients with endometrial cancer who underwent radical surgery between 2012 and 2023. We evaluated the predictive performance of E-PASS components: preoperative risk score (PRS), surgical stress score (SSS), and comprehensive risk score (CRS).
Lymphocele developed in 62 patients (34.4%). The CRS demonstrated superior predictive performance (area under the curve [AUC] = 0.930; 95% confidence interval [CI]: 0.893–0.966) with 0.806 sensitivity and 0.915 specificity. Multivariate analysis identified CRS (odds ratio [OR] = 1.161; 95% CI: 1.110–1.214) and advanced International Federation of Gynecology and Obstetrics stage (OR = 3.211, 95% CI: 1.262–8.172) as independent risk factors.
The E-PASS scoring system, particularly the CRS, effectively predicts postoperative lymphocele formation in patients with endometrial cancer undergoing radical surgery, potentially facilitating early risk assessment and guiding preventive interventions.
Keywords
- endometrial neoplasms
- lymphocele
- operative
- postoperative complications
- risk assessment
- surgical procedures
Endometrial cancer represents a growing threat in gynecologic oncology, with rising incidence worldwide and approximately 97,000 annual deaths [1]. This malignancy primarily spreads through lymphatic pathways, with lymph node metastasis present in 10%–15% of apparently early-stage cases [2]. Lymphadenectomy provides critical staging information for treatment planning, though its survival benefit remains debated. For selected cases where lymphadenectomy is performed, postoperative lymphocele formation represents a significant clinical challenge, affecting between 1%–58% of patients [3].
Lymphoceles significantly impair recovery and reduce quality of life [4, 5]. These fluid collections result from ineffective closure of lymphatic vessel stumps during surgery [6] and can lead to serious complications including infections and prolonged hospitalization [7, 8]. Key risk factors include extent of lymphatic dissection, obesity, and surgical drain management [9].
Current predictive models for surgical complications in endometrial cancer show important limitations. The american society of anesthesiologists (ASA) classification demonstrates value for general complications but lacks specificity for lymphatic complications [10]. While the frailty Index effectively predicts morbidity in elderly patients [11], its complexity limits routine clinical application. Similarly, the surgical complexity score has been primarily validated for ovarian cancer [12], with insufficient evidence for endometrial cancer complications.
The estimation of physiologic ability and surgical stress (E-PASS) system offers a promising alternative by uniquely integrating both patient factors and surgical parameters. Through its comprehensive risk score calculation [13], E-PASS potentially provides a more complete approach to predicting specific complications like lymphocele formation [14]. Though valuable across surgical specialties, its application in gynecologic oncology remains underexplored [15].
Within the evolving landscape of endometrial cancer management, where lymphadenectomy is increasingly applied selectively rather than routinely, this retrospective cohort study evaluated whether E-PASS scores can effectively predict lymphocele formation following endometrial cancer surgery. Our objective was to establish a clinically useful risk stratification tool to guide preventive strategies and support individualized surgical approaches.
In this single-center retrospective study conducted at Dongyang Hospital Affiliated to Wenzhou Medical University, Dongyang, China, 286 patients with endometrial cancer who underwent radical surgery between 2012 and 2023 were screened initially. Patient data were extracted through the Le9 medical record mining platform (Shanghai, China). We acknowledge that the single-center nature of this study from eastern China may limit the generalizability of our findings to different geographic regions, healthcare systems, and patient populations with varying demographic and clinical characteristics. This limitation highlights the need for future multicenter studies across diverse populations. Fig. 1 delineates the methodological framework employed in this investigation.
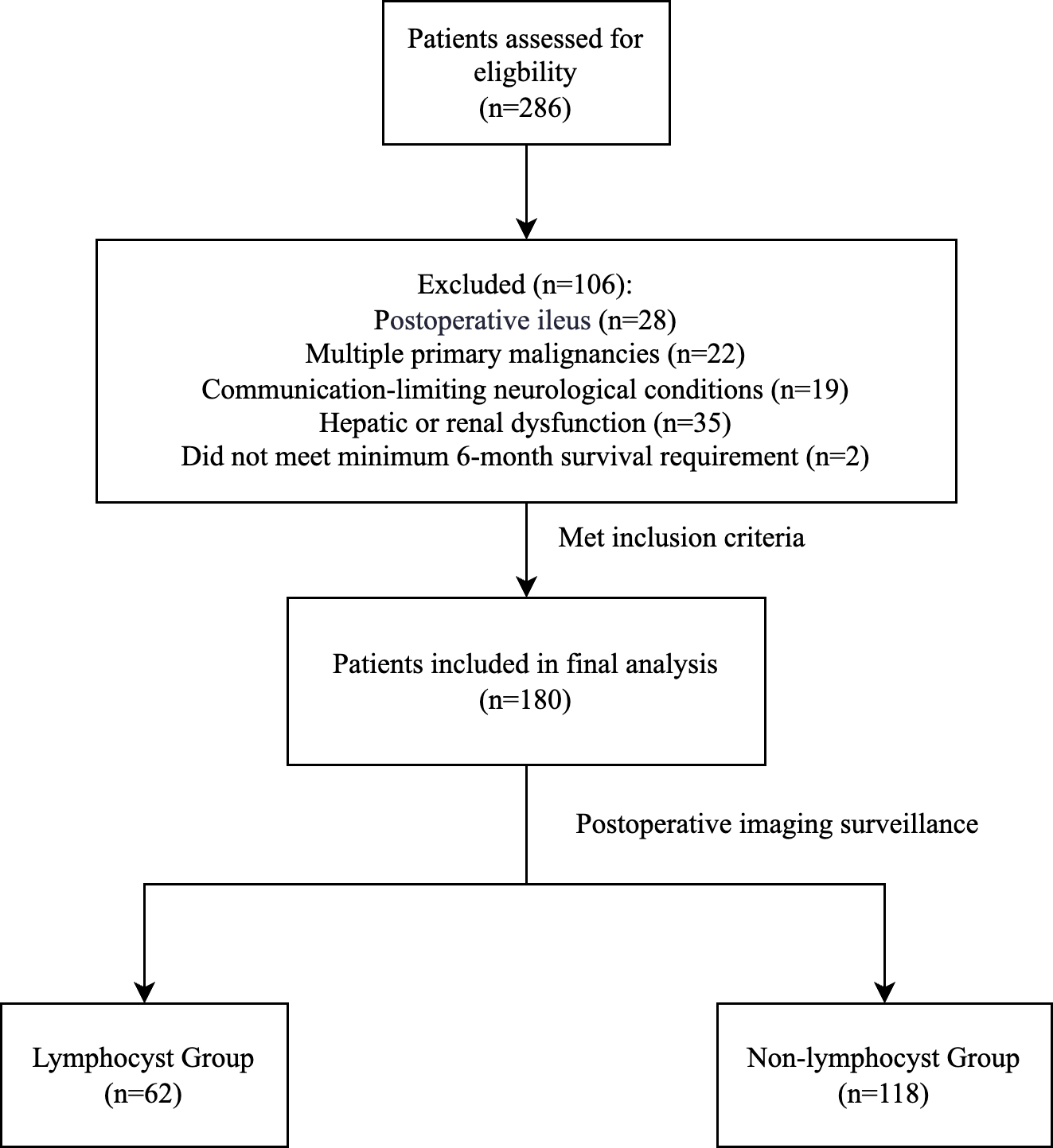 Fig. 1.
Fig. 1.
Patient selection flowchart for the analysis of postoperative lymphocele formation in patients with endometrial cancer.
• Radical surgery defined as total hysterectomy, bilateral salpingo-oophorectomy, and lymphadenectomy per International Federation of Gynecology and Obstetrics (FIGO) guidelines [16]. All patients underwent pelvic lymphadenectomy (external iliac, internal iliac, common iliac, and obturator nodes). High-risk cases (G3 grade, non-endometrioid histology, deep myometrial invasion, or cervical involvement) additionally received para-aortic lymphadenectomy from aortic bifurcation to renal vein level.
• Minimum 6-month post-surgical survival.
• Primary surgical treatment without previous therapy.
106 patients were excluded based on:
• Postoperative ileus (n = 28): bowel dysfunction requires interventions affecting fluid distribution and metabolic status, potentially interfering with lymphocele formation assessment and introducing confounding variables.
• Multiple primary malignancies (n = 22): may affect lymphadenectomy extent and technique.
• Communication-limiting neurological conditions (n = 19): prevents accurate symptom reporting.
• Hepatic or renal dysfunction (n = 35): these conditions alter protein metabolism and fluid balance, directly affecting lymphatic fluid dynamics and independently influencing lymphocele formation, which would confound E-PASS predictive assessment.
• Did not meet 6-month survival requirement (n = 2).
Lymphocele was defined as fluid collection
The final analysis included 180 patients, categorized into lymphocele (n = 62) and non-lymphocele (n = 118) groups.
Based on postoperative imaging, patients were categorized into Lymphocele Group
(n = 62) and Non-lymphocele Group (n = 118). Postoperative ultrasounds were
independently evaluated by two blinded ultrasonographers (
The E-PASS framework (preoperative risk score [PRS] for patient status, surgical stress score [SSS] for intraoperative burden, and comprehensive risk score [CRS] integrating both) was implemented to assess perioperative risks through its multidimensional evaluation system [18].
The PRS is calculated using the following equation:
Where:
• X1 represents age in years.
• X2 indicates severe heart disease (1 if present, 0 if absent).
o New York heart association (NYHA) functional class III or IV.
o Severe arrhythmia requiring mechanical support.
• X3 denotes severe pulmonary disease (1 if present, 0 if absent).
o Vital capacity (VC)
o Forced expiratory volume in 1 second (FEV 1.0%)
• X4 signifies diabetes mellitus according to the World Health Organization criteria (1 if present, 0 if absent).
• X5 reflects performance status index (0–4).
o 0: Fully active.
o 1: Restricted but ambulatory.
o 2: Ambulatory
o 3: Confined to bed
o 4: Completely disabled.
• X6 represents ASA classification (1–5).
The SSS is determined using the equation:
Where:
• X7 represents blood loss/body weight ratio (mL/kg).
• X8 indicates operation time (minutes).
• X9 denotes extent of surgical incision.
o 0: Minimally invasive (laparoscopic) approach.
o 1: Single cavity surgery (laparotomy or thoracotomy).
o 2: Combined cavity surgery (both laparotomy and thoracotomy).
The final CRS is calculated as:
Using R software version 4.4.1 (R Core Team, R Foundation for Statistical
Computing, Vienna, Austria), data distributions were assessed using the
Shapiro-Wilk normality test. Normally distributed continuous variables were
presented as mean
For categorical variables, standard Pearson chi-square tests using the direct
formula
Univariate analysis and multivariate binary logistic regression were performed
to identify independent risk factors for lymphocele formation. Variables with
p
The diagnostic performance of E-PASS components (PRS, SSS, CRS) for predicting
postoperative lymphocele was evaluated using receiver operating characteristic
(ROC) curve analysis. Diagnostic parameters including area under the curve (AUC),
sensitivity, specificity, and optimal cut-off values were calculated. Statistical
significance was set at p
Overall, 180 patients with endometrial cancer who underwent radical surgery were evaluated. Postoperative lymphocele developed in 62 patients (34.4%), while 118 patients (65.6%) remained lymphocele-free during follow-up.
Demographic and perioperative characteristics were comparable between groups.
Neither patient-specific variables (age, body mass index [BMI], comorbidity
burden, postoperative hemoglobin levels) nor surgical parameters (anesthetic
technique, drainage management, prior abdominal operations) exhibited significant
differences (all p
Notably, tumor staging analysis revealed that advanced-stage disease (FIGO
III–IV) occurred more frequently in patients who developed lymphoceles (40.3%
vs. 20.3%,
Analysis of E-PASS parameters revealed slight differences between groups that did not reach statistical significance. The lymphocele cohort showed marginally higher values in preoperative risk scores (0.35 [0.08] vs. 0.33 [0.05], t = 1.536, p = 0.128), surgical stress scores (0.36 [0.10] vs. 0.34 [0.06], t = 1.444, p = 0.152), and comprehensive risk scores (0.43 [0.15] vs. 0.40 [0.13], t = 1.419, p = 0.159). Of the examined variables, FIGO staging showed a significant association with lymphocele formation risk (p = 0.004) (Table 1).
| Characteristics | Lymphocele | Non-lymphocele | Difference | Statistics | p-value | |
| (n = 62) | (n = 118) | [95% CI] | (t/ | |||
| Demographic and clinical parameters | ||||||
| Age (years) | 0.939 | |||||
| 28 (45.2%) | 54 (45.8%) | |||||
| 34 (54.8%) | 64 (54.2%) | 0.6% [–30.0, 31.2] | ||||
| BMI (kg/m2) | 0.645 | |||||
| 47 (75.8%) | 93 (78.8%) | |||||
| 15 (24.2%) | 25 (21.2%) | 3.0% [–22.9, 28.9] | ||||
| Comorbidities | 0.675 | |||||
| 52 (83.9%) | 96 (81.4%) | |||||
| 10 (16.1%) | 22 (18.6%) | –2.5% [–25.6, 20.6] | ||||
| Disease-related parameters | ||||||
| FIGO stage | 0.004* | |||||
| I–II | 37 (59.7%) | 94 (79.7%) | ||||
| III–IV | 25 (40.3%) | 24 (20.3%) | 20.0% [–8.4, 48.4] | |||
| Histological type | 0.278 | |||||
| Non-endometrioid | 37 (59.7%) | 80 (67.8%) | ||||
| Endometrioid | 25 (40.3%) | 38 (32.2%) | 8.1% [–21.6, 37.8] | |||
| Surgical parameters | ||||||
| Previous abdominal surgery | 0.147 | |||||
| No | 44 (71.0%) | 95 (80.5%) | ||||
| Yes | 18 (29.0%) | 23 (19.5%) | 9.5% [–17.2, 36.3] | |||
| Postoperative management | ||||||
| Drainage placement | 0.869 | |||||
| No | 13 (21.0%) | 26 (22.0%) | ||||
| Yes | 49 (79.0%) | 92 (78.0%) | 1.1% [–24.1, 26.3] | |||
| Drainage duration (days) | 0.420 | |||||
| 25 (40.3%) | 55 (46.6%) | |||||
| 37 (59.7%) | 63 (53.4%) | 6.3% [–24.1, 36.6] | ||||
| Hemoglobin (g/L) | 0.645 | |||||
| 40 (64.5%) | 72 (61.0%) | |||||
| 22 (35.5%) | 46 (39.0%) | –3.5% [–33.1, 26.1] | ||||
| E-PASS scoring system | ||||||
| PRS score | 0.35 (0.08) | 0.33 (0.05) | 0.02 [–0.01, 0.04] | t = 1.536 | 0.128 | |
| SSS score | 0.36 (0.10) | 0.34 (0.06) | 0.02 [–0.01, 0.05] | t = 1.444 | 0.152 | |
| CRS score | 0.43 (0.15) | 0.40 (0.13) | 0.03 [–0.01, 0.08] | t = 1.419 | 0.159 | |
Note: Data are presented as n (%) for categorical variables and mean
Abbreviations: BMI, body mass index; FIGO, International Federation of Gynecology and Obstetrics; E-PASS, estimation of physiologic ability and surgical stress; PRS, preoperative risk score; SSS, surgical stress score; CRS, comprehensive risk score; CI, confidence interval.
We performed Spearman correlation analysis between FIGO staging and E-PASS
components due to the binary nature of FIGO stage. No significant correlations
were found between FIGO staging and PRS (Spearman correlation coefficients [rs] =
–0.027, p = 0.715), SSS (rs = 0.076, p = 0.310), or CRS (rs =
–0.009, p = 0.909), indicating cancer stage independence from
physiological risk assessment. Strong correlations existed among E-PASS
components: CRS correlated significantly with PRS (rs = 0.706, p
| Variables | Statistics | FIGO stage | PRS | SSS | CRS |
| FIGO stage | r | 1.000 | –0.027 | 0.076 | –0.009 |
| p | NA | 0.715 | 0.310 | 0.909 | |
| PRS | r | –0.027 | 1.000 | 0.256 | 0.706 |
| p | 0.715 | NA | 0.001 | ||
| SSS | r | 0.076 | 0.256 | 1.000 | 0.542 |
| p | 0.310 | 0.001 | NA | ||
| CRS | r | –0.009 | 0.706 | 0.542 | 1.000 |
| p | 0.909 | NA |
Note: Data presented are Spearman correlation coefficients (rs) with
corresponding p values. Spearman correlation was used due to the
inclusion of FIGO stage as a binary categorical variable. p
Logistic regression analysis was performed with CRS as a continuous variable,
FIGO stage (I–II = 0, III–IV = 1) as a categorical variable, and postoperative
lymphocele formation (yes = 1, no = 0) as the dependent variable (Table 3).
Binary logistic regression analysis was performed to evaluate the predictive
value of CRS and FIGO stage for postoperative lymphocele formation in patients
with endometrial cancer (Table 4). The results demonstrated that both CRS and
FIGO stage were independent risk factors for lymphocele formation. Specifically,
for each unit increase in CRS, the risk of developing lymphocele increased by
16.1% (OR = 1.161, 95% CI: 1.110–1.214, p
| Variables | Type | Coding/Definition | |
| Independent variables | |||
| CRS | Continuous | Original numerical value (range: 0–1) | |
| FIGO stage | Categorical | 0 = Stage I–II (early stage) | |
| 1 = Stage III–IV (advanced stage) | |||
| Dependent variable | |||
| Lymphocele formation | Binary | 0 = No | |
| 1 = Yes | |||
Note: CRS was calculated based on preoperative risk score and surgical stress score. FIGO staging was determined according to 2009 FIGO staging system for endometrial cancer.
| Variables | SE | OR (95% CI) | Wald | p-value | |
| CRS | 0.149 | 0.023 | 1.161 (1.110–1.214) | 42.516 | |
| FIGO stage | 1.167 | 0.477 | 3.211 (1.262–8.172) | 5.994 | 0.014 |
| Constant | –6.863 | 0.995 | 0.001 | 47.591 |
Note: CRS was included as a continuous variable. FIGO stage was coded as: 0 = stage I–II, 1 = stage III–IV. Dependent variable was coded as: 0 = no lymphocele, 1 = lymphocele formation.
Abbreviations:
The predictive performance of different E-PASS scoring systems for postoperative
lymphocele in patients with endometrial cancer was evaluated using ROC curve
analysis (Fig. 2). The CRS demonstrated superior discriminative ability with the
highest AUC of 0.930 (95% CI: 0.893–0.966), with optimal sensitivity of 80.60%
and specificity of 91.50%. The SSS showed good predictive performance with an
AUC of 0.873 (95% CI: 0.818–0.927), achieving 69.41% and 92.41% sensitivity
and specificity, respectively. In contrast, the PRS showed relatively poor
predictive capability, with AUC of 0.686 (95% CI: 0.600–0.772). The significant
difference in AUCs between CRS and other E-PASS components (all p
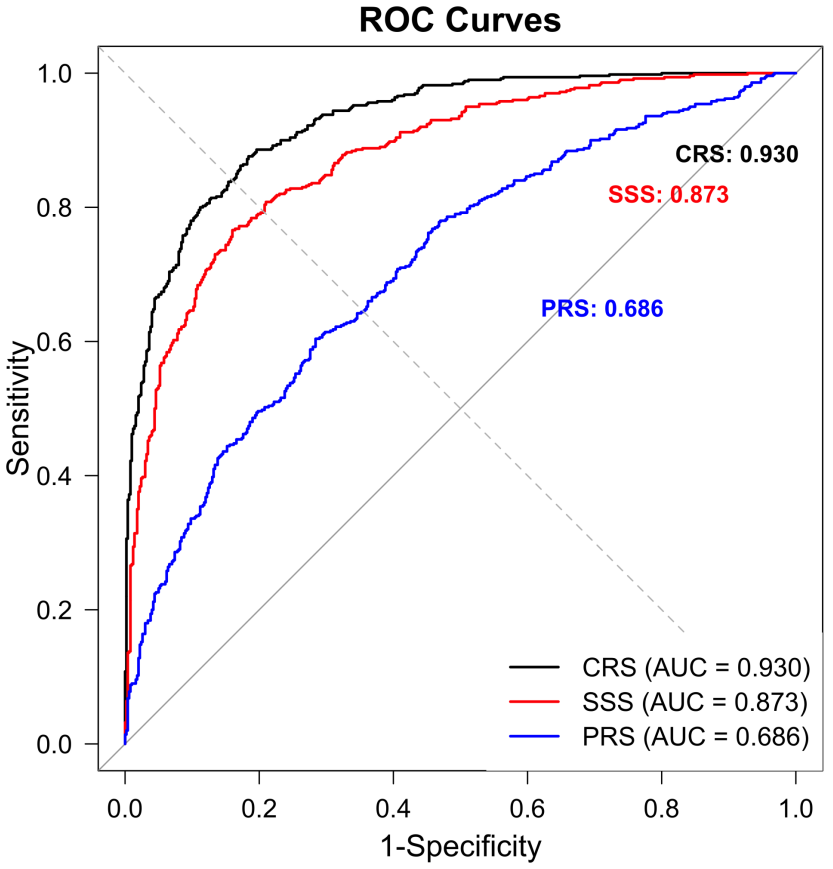 Fig. 2.
Fig. 2.
Receiver operating characteristic (ROC) curves comparing different predictive models for postoperative lymphocele in patients with endometrial cancer. ROC curves showing the discriminative ability of four different predictive models: comprehensive risk score (CRS, black line), surgical stress score (SSS, red line), physiological risk score (PRS, blue line). The area under the curve (AUC) values with 95% confidence intervals were: CRS 0.930 (0.893–0.966), SSS 0.873 (0.818–0.927), PRS 0.686 (0.600–0.772). The diagonal reference line (grey) represents random prediction (AUC = 0.500). CRS demonstrated superior predictive performance with optimal sensitivity of 80.60% and specificity of 91.50%. The curves were derived from a retrospective analysis of 180 patients who underwent radical surgery for endometrial cancer between 2012 and 2023.
| Predictive models | AUC (95% CI) | SE | Sensitivity | Specificity | Youden index |
| CRS | 0.930 (0.893–0.966) | 0.018 | 0.806 | 0.915 | 0.720 |
| SSS | 0.873 (0.818–0.927) | 0.028 | 0.694 | 0.924 | 0.620 |
| PRS | 0.686 (0.600–0.772) | 0.044 | 0.419 | 0.898 | 0.320 |
| FIGO stage | - | - | 0.403 | 0.797 | - |
Note: CRS combines both preoperative risk factors and surgical stress factors. SSS evaluates the extent of surgical intervention. PRS includes preoperative clinical risk factors. FIGO staging was determined according to 2009 FIGO staging system. FIGO stage: binary categorical variable (I–II vs. III–IV); AUC and Youden index not applicable for dichotomous variables.
Abbreviations: AUC, area under the curve.
This retrospective investigation spanning 11 years examined how E-PASS scoring components relate to lymphocele formation after endometrial cancer surgery. Our analysis revealed that the CRS offers the most robust predictive capability among the assessed parameters. This finding suggests that an integrated assessment combining both physiological capacity and surgical stress provides more accurate risk stratification than either component alone or traditional staging systems.
Physiological capacity reflects a patient’s overall health status and recovery potential, while surgical stress scoring quantifies both psychological and physiological impacts of surgery [14, 19]. The E-PASS scoring system, comprising PRS, SSS, and CRS, provides clinicians with precise patient assessment by comprehensively evaluating surgical risk and stress response [20]. Our findings align with several previous studies while also revealing some unique insights. Kayra et al. [19] demonstrated in their prospective study of 156 patients with endometrial cancer that E-PASS scoring components effectively predicted postoperative complications, with CRS showing the highest predictive value. Similarly, Chen et al. [14] reported that elevated CRS correlated with increased lymphatic complications in gynecologic surgery. However, our study demonstrated enhanced predictive accuracy for CRS compared to these previous reports, possibly due to our larger sample size and more comprehensive assessment protocol. Our findings regarding the correlation between FIGO staging and lymphocele formation corroborate Pan et al.’s [21] retrospective analysis, though with some variations that might be attributed to differences in surgical techniques and postoperative management. This difference might be attributed to variations in surgical techniques and postoperative management protocols. The mechanistic basis for our observations can be explained through several pathways. As demonstrated by Chen et al. [22] and Kitano et al. [23], elevated PRS often reflects compromised physiological reserves and reduced healing capacity, while Norimatsu et al. [24] and Dai et al. [25] established that higher SSS indicates more extensive surgical trauma and lymphatic disruption. The superior predictive performance of CRS in our study, consistent with Haga et al.’s [26] foundational work, likely stems from its comprehensive integration of both physiological and surgical stress parameters, particularly relevant in complex procedures like endometrial cancer surgery.
The clinical implications of our findings extend beyond mere risk prediction, offering a practical framework for personalized surgical planning in endometrial cancer treatment. The strong performance of CRS enables clinicians to identify high-risk patients and implement targeted interventions. Based on our ROC analysis, we propose CRS-based risk stratification for clinical decision-making, though specific thresholds require further validation in larger cohorts. For surgical approach, high-risk patients would benefit from sentinel lymph node mapping when oncologically appropriate and advanced vessel-sealing devices to minimize lymphatic disruption, while intermediate-risk patients should receive standard lymphadenectomy with careful attention to lymphatic preservation. Drainage strategy should be tailored to risk level: high-risk patients receiving extended closed-suction drainage (7–10 days) with daily output monitoring, intermediate-risk patients having standard drainage with removal upon output reduction below 50 mL/24 h, and low-risk patients potentially qualifying for early drain removal or selective placement. Postoperative monitoring should likewise be risk-stratified: high-risk patients undergoing enhanced ultrasound assessment on days 7, 14, and 21, intermediate-risk patients receiving evaluation on day 14, and low-risk patients following standard clinical protocols. Additional preventive measures for high-risk patients should include early mobilization with compression therapy, nutritional optimization (especially with low preoperative albumin), and thorough patient education regarding lymphocele symptoms. Notably, endometrial cancer lymphadenectomy guidelines have undergone significant changes in recent years. Both the European Society of Gynecological Oncology/European Society for Radiotherapy and Oncology/European Society of Pathology 2020 (ESGO/ESTRO/ESP 2020) consensus and the latest National Comprehensive Cancer Network (NCCN) guidelines recommend more individualized lymph node assessment strategies [27, 28]. Research from AC Camargo Cancer Center revealed that sentinel lymph node (SLN) mapping reduced lymphocele incidence from 14.1% with systematic lymphadenectomy to 3.4% with SLN alone (p = 0.009). Multivariate analysis identified systematic lymphadenectomy as an independent risk factor for lymphocele (OR 3.68) [29].
This study’s unique contribution lies in its comprehensive integration of both physiological and surgical parameters through the E-PASS system, providing a more nuanced risk assessment than traditional staging-based approaches. We recommend incorporating CRS calculation into routine preoperative assessment workflows, with particular attention to patients showing elevated values. Future research should pursue several key directions: multicenter prospective validation to confirm CRS threshold generalizability across diverse populations; integration of CRS with molecular biomarkers (such as vascular endothelial growth factor [VEGF]-C/D or inflammatory markers) to enhance prediction and reveal pathophysiological mechanisms; combination with radiological parameters like lymphatic mapping or ultrasound elastography to create stronger predictive models; development of artificial intelligence (AI) algorithms that integrate these clinical, molecular, and radiological factors; and interventional studies evaluating whether CRS-based preventive strategies actually reduce lymphocele incidence and improve outcomes.
Methodological rigor was ensured through: (1) 11-year cohort (n = 180) providing
temporal validity; (2) standardized lymphocele imaging protocols; (3)
multifactorial analysis integrating clinical/E-PASS parameters. Our statistical
framework combined correlation-multivariate modeling with ROC analysis and
confounder adjustment. Stage-inclusive enrollment (IA–IV) and
Study limitations include: (1) single-center Chinese cohort requiring multiethnic validation; (2) exclusion of high-risk subgroups (comorbidities/immunodeficiency/neoadjuvant); (3) observational design precludes causal inference. Residual confounding persists despite multivariate adjustment. Nevertheless, CRS maintained strong predictive validity (AUC = 0.930) supporting clinical translation for endometrial cancer lymphocele risk stratification.
Our study confirms E-PASS scoring, particularly CRS, as an effective predictor
of lymphocele formation after endometrial cancer surgery. We propose risk
stratification (low
E-PASS, estimation of physiologic ability and surgical stress; PRS, preoperative risk score; SSS, surgical stress score; CRS, comprehensive risk score; NYHA, New York heart association; VC, vital capacity; FEV1.0%, forced expiratory volume in 1 second; ASA, american society of anesthesiologists; IQR, interquartile range; SD, standard deviation; OR, odds ratios; CI, confidence intervals; ROC, receiver operating characteristic; AUC, area under the curve; BMI, body mass index; FIGO, International Federation of Gynecology and Obstetrics; SE, standard error; ESGO/ESTRO/ESP 2020, European Society of Gynecological Oncology/European Society for Radiotherapy and Oncology/European Society of Pathology 2020; NCCN, National Comprehensive Cancer Network; VEGF, vascular endothelial growth factor; AI, artificial intelligence.
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
HL and ZH conceived the research idea. HL, ZH, and JD jointly designed the research methodology and study framework. HL and ZH performed data collection, statistical analysis, and interpretation of results. HL and ZH drafted the initial manuscript. JD supervised the entire study and critically revised the manuscript for important intellectual content. All authors participated in manuscript revision and editing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work. All authors participated in manuscript revision and editing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki. This study was approved by the Ethics Committee of Dongyang People’s Hospital of Wenzhou Medical University (approval number: DRY-2024-YX-390). The requirement for informed consent was waived by the institutional review board due to the retrospective nature of this study.
We would like to thank Editage (https://www.editage.cn/) for English language editing.
This research received no external funding.
The authors declare no conflict of interest.
The authors acknowledge the use of language editing tools, including AI-assisted software, for grammar checking and linguistic refinement only; all scientific content, data analysis, results interpretation, and conclusions were exclusively generated and validated by the human authors. During the preparation of this work the authors used ChatGPT and DeepSeek in order to check spelling and grammar. After using these tools, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


