1 Postgraduate Training Base of Shenyang Women’s and Children’s Hospital of Jinzhou Medical University, 110011 Shenyang, Liaoning, China
2 Department of Obstetrics and Gynecology, Shenyang 245 Hospital, 110041 Shenyang, Liaoning, China
3 Shenyang Clinical Medical Research Center for Obstetrics and Gynecology, Shenyang Women’s and Children’s Hospital, 110011 Shenyang, Liaoning, China
4 Department of Obstetrics, Shenyang Women’s and Children’s Hospital, 110011 Shenyang, Liaoning, China
5 Department of Anesthesiology, Shenyang Women’s and Children’s Hospital, 110011 Shenyang, Liaoning, China
Abstract
Background: Postpartum hemorrhage (PPH) is a major cause of maternal mortality, and uterine fibroids are a common condition that may contribute to this risk. This study aimed to determine the risk factors for PPH following cesarean myomectomy and to evaluate their predictive value.
Methods: We conducted a retrospective analysis of 1002 women with fibroids ≥4 cm in diameter who underwent cesarean myomectomy at the Shenyang Women’s and Children’s Hospital from January 2014 to November 2022. The PPH and non-PPH groups consisted of 109 and 893 patients, respectively. Patient, clinical, and surgical data were collected from medical records. Factors associated with PPH were identified through univariate and multivariable logistic regression analyses. A receiver operating characteristic (ROC) curve analysis was used to assess the predictive value of independent risk factors for PPH. Propensity score matching analysis was used to determine whether bilateral uterine artery ligation before myomectomy and the use of potent uterotonic drugs during this procedure protected against PPH.
Results: Independent risk factors for PPH included multiple fibroids, specific sites of fibroids (located in the lower segment of the uterus, cervix, uterine cornu, or adjacent to the uterine vessels or the interstitial part of the fallopian tube), a longer surgery duration, and a larger fibroid size (diameter >7.5 cm for single fibroids and total diameter >14.5 cm for multiple fibroids). The results of the propensity score matching analysis indicated that bilateral uterine artery ligation and the administration of potent uterotonic drugs before myomectomy were protective factors against PPH. ROC curve analysis showed that both the maximum diameter of a single uterine fibroid and the sum of the maximum diameters of multiple uterine fibroids are predictive of PPH risk.
Conclusions: Cesarean myomectomy is relatively safe for fibroids located at non-specific sites with a diameter ≤7.5 cm (single) or a total diameter ≤14.5 cm (multiple). The duration of surgery should be minimized. In high-risk cases, bilateral uterine artery ligation and the administration of potent uterotonic drugs before myomectomy should be considered.
Keywords
- cesarean section
- uterine fibroids
- postpartum hemorrhage
- uterine artery ligation
Postpartum hemorrhage (PPH), which is defined as blood loss
Nonetheless, PPH remains a serious complication because cesarean myomectomy may increase the risk of PPH [12, 13]. A recent Cochrane review of cesarean myomectomy concluded that the available evidence for all critical outcomes is of very low certainty [14]. Therefore, drawing definitive conclusions regarding the effects of cesarean myomectomy on the risk of requiring blood transfusion, risk of hemorrhage, length of hospitalization, duration of surgery, and risk of major surgery at the time of the procedure is not possible.
This study aimed to identify the risk factors for PPH following cesarean myomectomy and to evaluate their predictive value. Hopefully, our findings will assist in reducing the incidence of PPH and developing targeted interventions for high-risk pregnancies.
This retrospective cohort study included women who underwent cesarean myomectomy
at Shenyang Women’s and Children’s Hospital from January 2014 to November 2022.
The inclusion criteria were as follows: (1) elective cesarean delivery with
concurrent myomectomy; (2) uterine fibroids
The following data were collected from medical records: maternal age, height, weight, gestational age at delivery, parity, newborn weight, duration of surgery, number and location of fibroids, maximum diameter of individual uterine fibroids, sum of the maximum diameters (in those with multiple fibroids), presence of fibroid degeneration, bilateral uterine artery ligation before myomectomy, and use of potent uterotonic drugs during surgery.
(1) PPH [1] was defined as blood loss
(2) Specific sites of uterine fibroids [16] were defined as those located in the lower segment of the uterus, cervix, or uterine cornu, or adjacent to the uterine vessels or the interstitial part of the fallopian tube. The prevalence of uterine fibroids was lower in these sites than in the fundus and body of the uterus. We mainly focused on the more common locations because of their higher prevalence and greater clinical significance.
(3) Potent uterotonic agents [17] included carboprost tromethamine or carbetocin. Some patients required these agents in addition to the standard dose of oxytocin.
(4) The duration of surgery was defined as the time elapsed from the start of the procedure to the end of the procedure.
Statistical analyses were performed using SPSS software version 27.0 (IBM Corp.,
Armonk, NY, USA) and R software version 4.4.2 (R Foundation for Statistical
Computing, Vienna, Austria, https://www.R-project.org), and p
| Characteristic | Non-PPH group (n = 893) | PPH group (n = 109) | U/ |
p-value | |
| Age (years) | 32 (29, 36) | 33 (30, 37) | 44,391.500 | 0.133 | |
| BMI (kg/m2) | 0.237 | 0.888 | |||
| Normal weight (18.50–23.99) | 44 (4.93) | 5 (4.59) | |||
| Overweight (24.00–27.99) | 272 (30.46) | 31 (28.44) | |||
| Obesity ( |
577 (64.61) | 73 (66.97) | |||
| Gestational weeks at delivery | 39 (38.40, 39.40) | 39 (38.60, 39.60) | 48,048 | 0.828 | |
| Parity | 0.119 | 0.730 | |||
| Primipara | 759 (84.99) | 94 (86.24) | |||
| Multipara | 134 (15.01) | 15 (13.76) | |||
| Newborn weight (g) | 3370 (3100, 3690) | 3430 (3195, 3770) | 44,076.500 | 0.107 | |
| Operative duration (min) | 52 (41, 64) | 65 (50, 88) | 31,289.500 | ||
| Number of fibroids | 16.229 | ||||
| Multiple fibroids | 337 (37.74) | 63 (57.80) | |||
| Solitary fibroid | 556 (62.26) | 46 (42.20) | |||
| Location of uterine fibroids | 109.423 | ||||
| Uterine fibroids in special locations | 62 (6.94) | 43 (39.45) | |||
| Uterine fibroids in non-special locations | 831 (93.06) | 66 (60.55) | |||
| Fibroid degeneration | 2.052 | 0.152 | |||
| No | 802 (89.81) | 93 (85.32) | |||
| Yes | 91 (10.19) | 16 (14.68) | |||
| Diameter of a single uterine fibroid (cm) | 5 (4, 6) | 8 (6, 9) | 5932 | ||
| Sum of the maximum diameters of multiple uterine fibroids (cm) | 9 (7, 12) | 15 (9, 20) | 5657 | ||
| Administration of potent uterine contraction-inducing medication prior to myomectomy | 2.691 | 0.101 | |||
| No | 789 (88.35) | 102 (93.58) | |||
| Yes | 104 (11.65) | 7 (6.42) | |||
| Bilateral uterine artery ligation prior to myomectomy | 0.082 | 0.775 | |||
| No | 829 (92.83) | 102 (93.58) | |||
| Yes | 64 (7.17) | 7 (6.42) | |||
PPH, postpartum hemorrhage; BMI, body mass index.
A total of 1002 patients met the study criteria and were included in the analysis. Among these, 109 experienced PPH (PPH group) and 893 did not (non-PPH group). The patients’ characteristics according to the groups are shown in Table 1. The duration of surgery, individual uterine fibroid diameter, sum of the maximum diameters of multiple uterine fibroids, number of uterine fibroids, and fibroid location were significantly different between the groups.
Although age was not statistically significant in the univariate analysis
(p = 0.133), it was included in the multivariate analysis due to its
clinical importance. A longer duration of surgery (OR, 1.025; 95% CI,
1.015–1.035; p
| Characteristics | SE | Z | p-value | OR | 95% CI | |
| Operative duration | 0.025 | 0.005 | 4.833 | 1.025 | 1.015–1.035 | |
| Number of fibroids (multiple/single) | 0.654 | 0.238 | 2.750 | 0.006 | 1.923 | 1.209–3.078 |
| Location of uterine fibroids (special/non-special) | 2.092 | 0.255 | 8.202 | 8.099 | 4.911–13.376 | |
| Age | –0.020 | 0.027 | –0.727 | 0.467 | 0.981 | 0.930–1.033 |
SE, standard error; OR, odds ratio; 95% CI, 95% confidence interval.
To better analyze the subgroup differences between patients with a single
uterine fibroid and those with multiple uterine fibroids, we performed subgroup
analyses and conducted univariate and multivariate regression analyses for each
group. The specific details can be found in Table 3 (Univariate analysis of the
subgroup with a single uterine fibroid), Table 4 (Logistic regression analysis in
patients with a single uterine fibroid), Table 5 (Univariate analysis of the
subgroup with multiple uterine fibroids), and Table 6 (Logistic regression
analysis in patients with multiple uterine fibroids). In patients with a single
uterine fibroid, a greater fibroid diameter was an independent risk factor for
PPH. The odds of PPH increased as the diameter increased (OR, 1.705; 95% CI,
1.446–2.011; p
| Characteristic | Non-PPH group (n = 556) | PPH group (n = 46) | U/ |
p-value | |
| Age (years) | 32 (29, 35) | 32.5 (29, 37) | 11,644.500 | 0.312 | |
| BMI (kg/m2) | 1.552 | 0.460 | |||
| Normal weight (18.50–23.99) | 35 (6.29) | 1 (2.17) | |||
| Overweight (24.00–27.99) | 166 (29.86) | 16 (34.78) | |||
| Obesity ( |
355 (63.85) | 29 (63.05) | |||
| Gestational weeks at delivery | 39 (38.43, 39.43) | 39 (38.57, 39.57) | 12,083 | 0.534 | |
| Parity | 0.851 | 0.356 | |||
| Primipara | 467 (83.99) | 41 (89.13) | |||
| Multipara | 89 (16.01) | 5 (10.87) | |||
| Newborn weight (g) | 3375 (3110.00, 3682.50) | 3437.50 (3212.50, 3787.50) | 11,458.500 | 0.241 | |
| Operative duration (min) | 47 (39.00, 58.00) | 65 (48.50, 73.75) | 7670.500 | ||
| Bilateral uterine artery ligation prior to myomectomy | Fisher | 0.386 | |||
| No | 538 (96.76) | 46 (100.00) | |||
| Yes | 18 (3.24) | 0 (0.00) | |||
| Administration of potent uterine contraction-inducing medication prior to myomectomy | 0.065 | 0.799 | |||
| No | 520 (93.53) | 44 (95.65) | |||
| Yes | 36 (6.47) | 2 (4.35) | |||
| Fibroid degeneration | Fisher | 0.039 | |||
| No | 504 (90.65) | 37 (80.43) | |||
| Yes | 52 (9.35) | 9 (19.57) | |||
| Diameter of a single uterine fibroid (cm) | 5 (4, 6) | 8 (6, 9) | 5932 | ||
| Location of uterine fibroids | 76.824 | ||||
| Uterine fibroids in non-special locations | 515 (92.63) | 23 (50.00) | |||
| Uterine fibroids in special locations | 41 (7.37) | 23 (50.00) | |||
| Characteristics | SE | Z | p | OR | 95% CI | |
|---|---|---|---|---|---|---|
| Diameter of a single uterine fibroid | 0.534 | 0.084 | 40.25 | 1.705 | 1.446–2.011 | |
| Duration of surgery | 0.016 | 0.010 | 2.650 | 0.104 | 1.016 | 0.997–1.035 |
| Location of uterine fibroids (special/non-special) | 2.806 | 0.413 | 46.25 | 16.543 | 7.369–37.138 | |
| Age | 0.003 | 0.043 | 0.004 | 0.947 | 1.003 | 0.922–1.090 |
| Myoma degeneration (No/Yes) | 0.586 | 0.500 | 1.373 | 0.241 | 1.797 | 0.674–4.791 |
| Characteristic | Non-PPH group (n = 337) | PPH group (n = 63) | U/ |
p-value | |
| Age (years) | 33 (30.00, 37.00) | 34 (31.00, 36.50) | 10,601 | 0.987 | |
| Body mass index (BMI) (kg/m2) | 3.361 | 0.186 | |||
| Normal weight (18.50–23.99) | 9 (2.67) | 4 (6.35) | |||
| Overweight (24.00–27.99) | 106 (31.45) | 15 (23.81) | |||
| Obesity ( |
222 (65.88) | 44 (69.84) | |||
| Gestational weeks at delivery | 39 (38.43, 39.57) | 39 (38.57, 39.43) | 10,817.500 | 0.811 | |
| Parity | 0.284 | 0.594 | |||
| Primipara | 292 (86.65) | 53 (84.13) | |||
| Multipara | 45 (13.35) | 10 (15.87) | |||
| Newborn weight (g) | 3360 (3040.00, 3690.00) | 3410 (3187.50, 3760.00) | 9566.500 | 0.213 | |
| Operative duration (min) | 58 (48.00, 70.00) | 70 (50.00, 93.50) | 7923 | 0.001 | |
| Bilateral uterine artery ligation prior to myomectomy | 0.298 | 0.585 | |||
| No | 291 (86.35) | 56 (88.89) | |||
| Yes | 46 (13.65) | 7 (11.11) | |||
| Administration of potent uterine contraction-inducing medication prior to myomectomy | 5.331 | 0.021 | |||
| No | 269 (79.82) | 58 (92.06) | |||
| Yes | 68 (20.18) | 5 (7.94) | |||
| Fibroid degeneration | 0.011 | 0.916 | |||
| No | 298 (88.43) | 56 (88.89) | |||
| Yes | 39 (11.57) | 7 (11.11) | |||
| Diameter of a single uterine fibroid (cm) | 9 (7, 12) | 15 (9, 20) | 5657 | ||
| Location of uterine fibroids | 37.560 | ||||
| Uterine fibroids in non-special locations | 316 (93.77) | 43 (68.25) | |||
| Uterine fibroids in special locations | 21 (6.23) | 20 (31.75) | |||
| Characteristics | SE | Z | p-value | OR | 95% CI | |
|---|---|---|---|---|---|---|
| Sum of the maximum diameters of multiple uterine fibroids | 0.163 | 0.029 | 32.049 | 1.177 | 1.112–1.245 | |
| Duration of surgery | 0.014 | 0.007 | 4.448 | 0.035 | 1.014 | 1.001–1.027 |
| Location of uterine fibroids (special/non-special) | 2.209 | 0.432 | 26.086 | 9.102 | 3.900–21.244 | |
| Age | –0.092 | 0.041 | 4.968 | 0.026 | 0.913 | 0.842–0.989 |
| Administration of potent uterine contraction-inducing medication prior to myomectomy (No/Yes) | –2.094 | 0.543 | 14.856 | 0.123 | 0.042–0.357 |
The sensitivity of a single uterine fibroid diameter in predicting PPH was 0.587, with a specificity of 0.872 and an area under the receiver operating characteristic curve of 0.768. The optimal cutoff value was 7.5 cm, with an accuracy of 0.850. These findings suggested that the diameter of a single uterine fibroid was a reliable predictor of PPH (Table 7 and Fig. 1).
| Characteristics | Cutoff value | Sensitivity | Specificity | AUC (95% CI) | Accuracy | p-value |
| Diameter of a single uterine fibroid (cm) | 7.5 | 0.587 | 0.872 | 0.768 (0.689–0.847) | 0.850 |
AUC, area under the receiver operating characteristic curve.
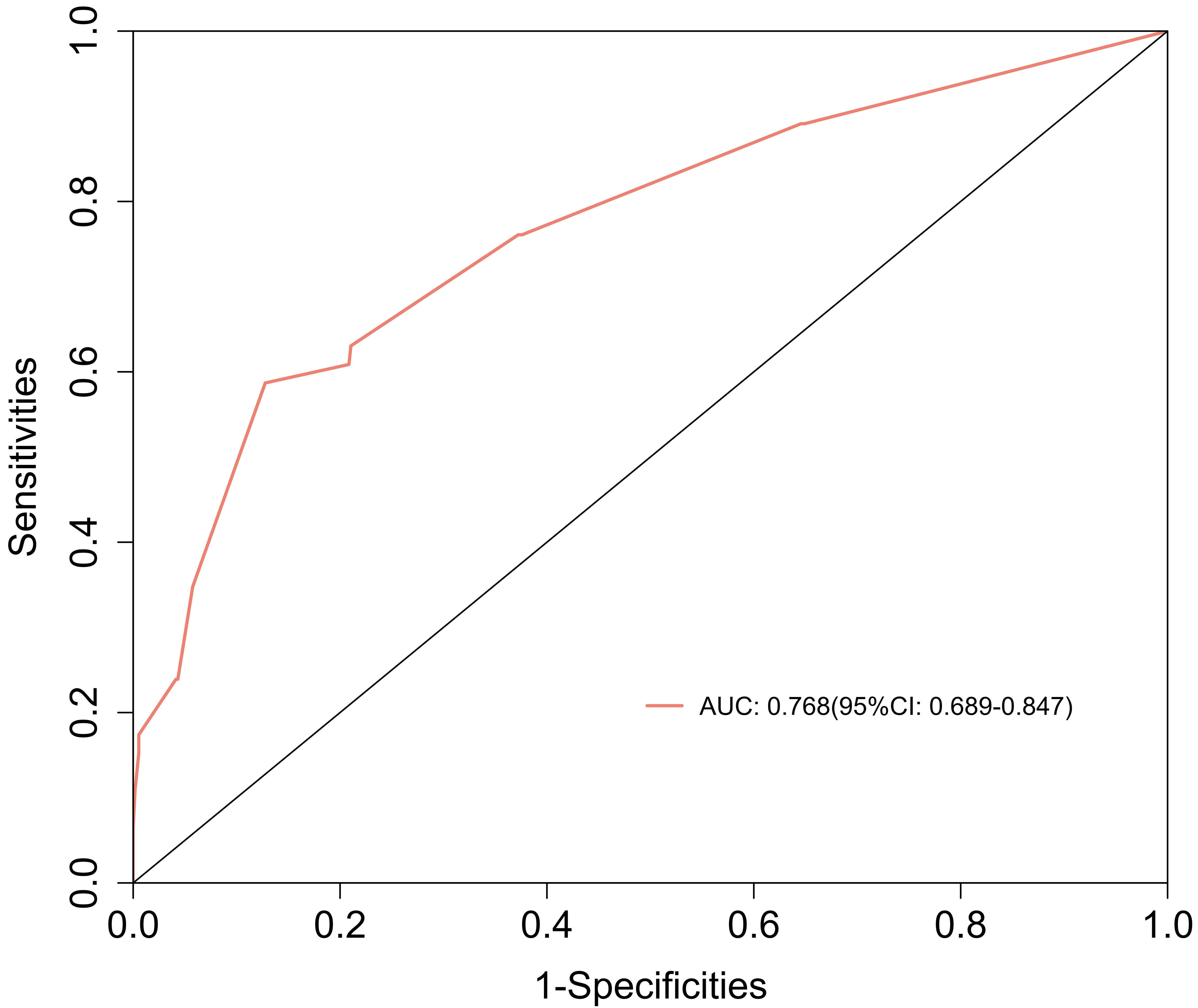 Fig. 1.
Fig. 1.
Receiver operating characteristic curve showing the performance of a single uterine fibroid diameter for predicting postpartum hemorrhage. Note: The X-axis shows the proportion of actual negative samples incorrectly predicted as positive. The Y-axis shows the proportion of actual positive samples correctly predicted. The red curve indicates the current model (AUC = 0.768). Optimal threshold: sensitivity = 0.587, specificity = 0.872.
The sensitivity of the sum of the maximum diameters of multiple uterine fibroids in predicting PPH was 0.556, with a specificity of 0.846 and an area under the receiver operating characteristic curve of 0.734. The optimal cutoff value was 14.5 cm, with an accuracy of 0.800. These findings indicated that the combined maximum diameters of multiple uterine fibroids also reliably predicted PPH (Table 8 and Fig. 2).
| Characteristics | Cutoff value | Sensitivity | Specificity | AUC (95% CI) | Accuracy | p-value |
| Sum of the maximum diameters of multiple uterine fibroids (cm) | 14.5 | 0.556 | 0.846 | 0.734 (0.662–0.805) | 0.800 |
AUC, area under the receiver operating characteristic curve.
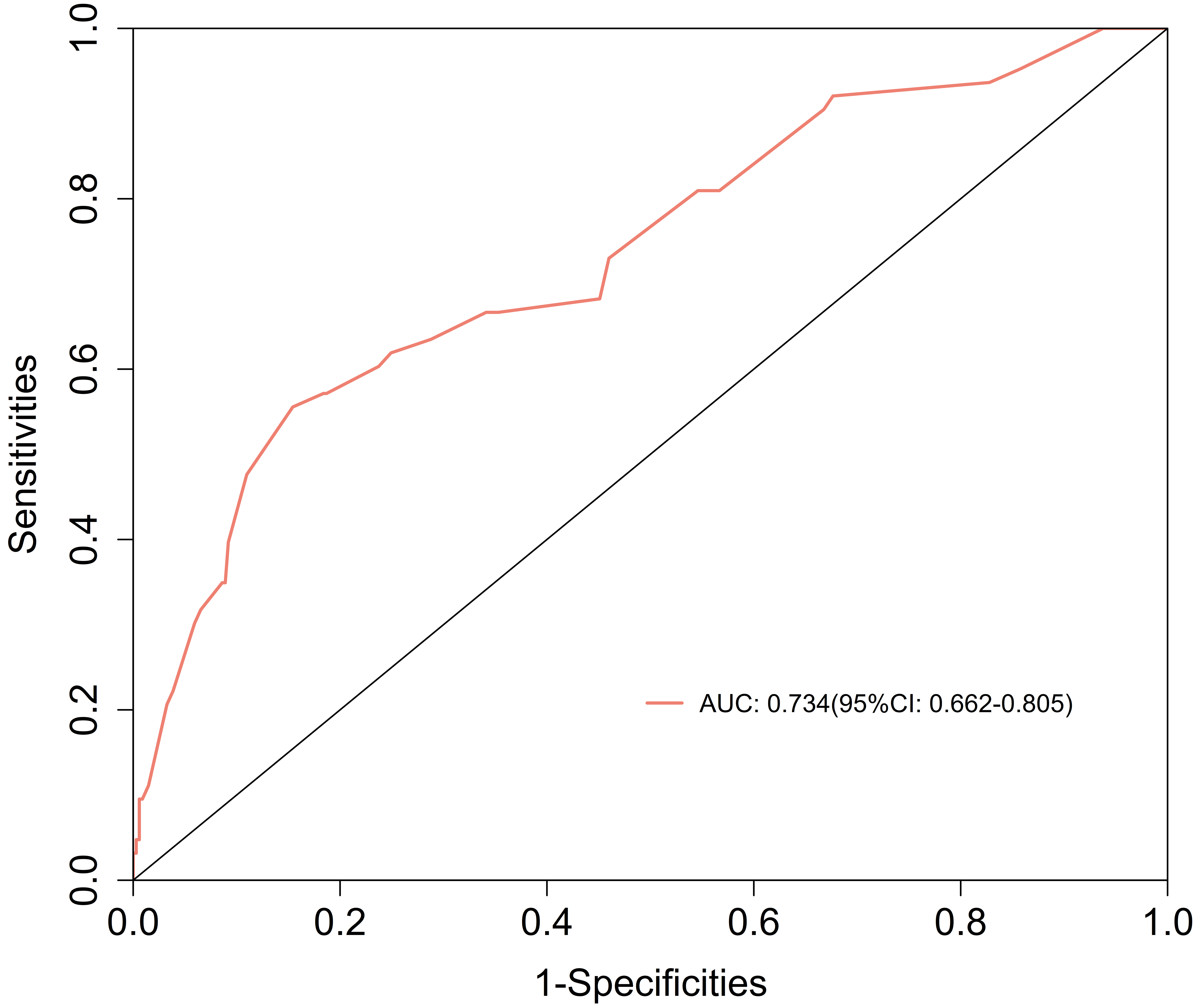 Fig. 2.
Fig. 2.
Receiver operating characteristic curve showing the performance of the sum of the maximum diameters of multiple uterine fibroids for predicting postpartum hemorrhage. Note: The X-axis shows 1-specificity (false positive rate), which indicates the proportion of actual negative samples incorrectly predicted as positive. The Y-axis shows sensitivity (true positive rate), which indicates the proportion of actual positive samples correctly predicted. The red curve indicates the current model (AUC = 0.734). Optimal threshold: sensitivity = 0.556, specificity = 0.846.
Significant confounding variables were present because of selection bias arising from the performance of bilateral uterine artery ligation before myomectomy and the use of potent uterotonic drugs during cesarean myomectomy. PSM was used to effectively mitigate these and investigate the potential effect of pre-myomectomy interventions on PPH. A 1:3 matching ratio was used to match patients in the PPH group with those in the non-PPH group using a caliper value of 0.03 to ensure the precision and reliability of the matching process. However, due to some extreme propensity scores in the PPH group, it was not possible to find three sufficiently close matched samples in the non-PPH group. As a result, the actual matching ratio was lower than 1:3, as shown in Table 9.
| Characteristics | Non-PPH group (n = 213) | PPH group (n = 88) | U/ |
p-value | |
| Age (years) | 32 (31, 36) | 33 (30, 36) | 9676 | 0.658 | |
| Body mass index (BMI) (kg/m2) | Fisher | 0.921 | |||
| Normal weight (18.50–23.99) | 9 (4.23) | 4 (4.55) | |||
| Overweight (24.00–27.99) | 59 (27.70) | 26 (29.55) | |||
| Obesity ( |
145 (68.08) | 58 (65.91) | |||
| Gestational weeks at delivery | 39.1 (38.60, 39.60) | 39 (38.60, 39.40) | 10,119 | 0.276 | |
| Parity | 0.099 | 0.752 | |||
| Primipara | 186 (87.32) | 78 (88.64) | |||
| Multipara | 27 (12.68) | 10 (11.36) | |||
| Newborn weight (g) | 3200 (3110, 3800) | 3410 (3180, 3800) | 0.129 | 0.898 | |
| Operative duration (min) | 60 (48.00, 75.00) | 65 (47.00, 75.50) | 9136 | 0.732 | |
| Number of fibroids | 0.039 | 0.843 | |||
| Multiple fibroids | 99 (46.48) | 42 (47.73) | |||
| Solitary fibroid | 114 (53.52) | 46 (52.27) | |||
| Location of uterine fibroids | 4.645 | 0.031 | |||
| Uterine fibroids in special locations | 39 (18.31) | 26 (29.55) | |||
| Uterine fibroids in non-special locations | 174 (81.69) | 62 (70.45) | |||
| Fibroid degeneration | 0.268 | 0.605 | |||
| No | 184 (86.38) | 74 (84.09) | |||
| Yes | 29 (13.62) | 14 (15.91) | |||
| Diameter of a single uterine fibroid (cm) | 5 (4.00, 6.00) | 8 (6.00, 9.75) | 860.500 | ||
| Sum of the maximum diameters of multiple uterine fibroids (cm) | 10 (8.00, 13.38) | 15.5 (9.12, 20.00) | 1656.500 | ||
| Administration of potent uterine contraction-inducing medication prior to myomectomy | 11.897 | ||||
| No | 162 (76.06) | 82 (93.18) | |||
| Yes | 51 (23.94) | 6 (6.82) | |||
| Bilateral uterine artery ligation prior to myomectomy | 4.848 | 0.028 | |||
| No | 185 (86.85) | 84 (95.45) | |||
| Yes | 28 (13.15) | 4 (4.55) | |||
Prior to matching, significant differences in the surgery duration, number of fibroids, and fibroid location were observed between the two groups. Propensity scores were derived using a logistic regression model that included the following variables: surgery duration, number of fibroids, and fibroid location. These variables were chosen because they may have a significant effect on the risk of PPH. After matching, the proportions of patients who had undergone bilateral uterine artery ligation prior to myomectomy and had received potent uterotonic drugs were significantly different (Table 9). These results suggested a notable protective effect of pre-myomectomy interventions on PPH.
To assess the quality of PSM, the distribution of propensity scores was compared between the PPH and non-PPH groups before and after matching using a distribution histogram. As shown in Fig. 3, there were notable differences in the distributions between the groups before matching. However, the distributions were nearly identical after matching, which indicated successful matching.
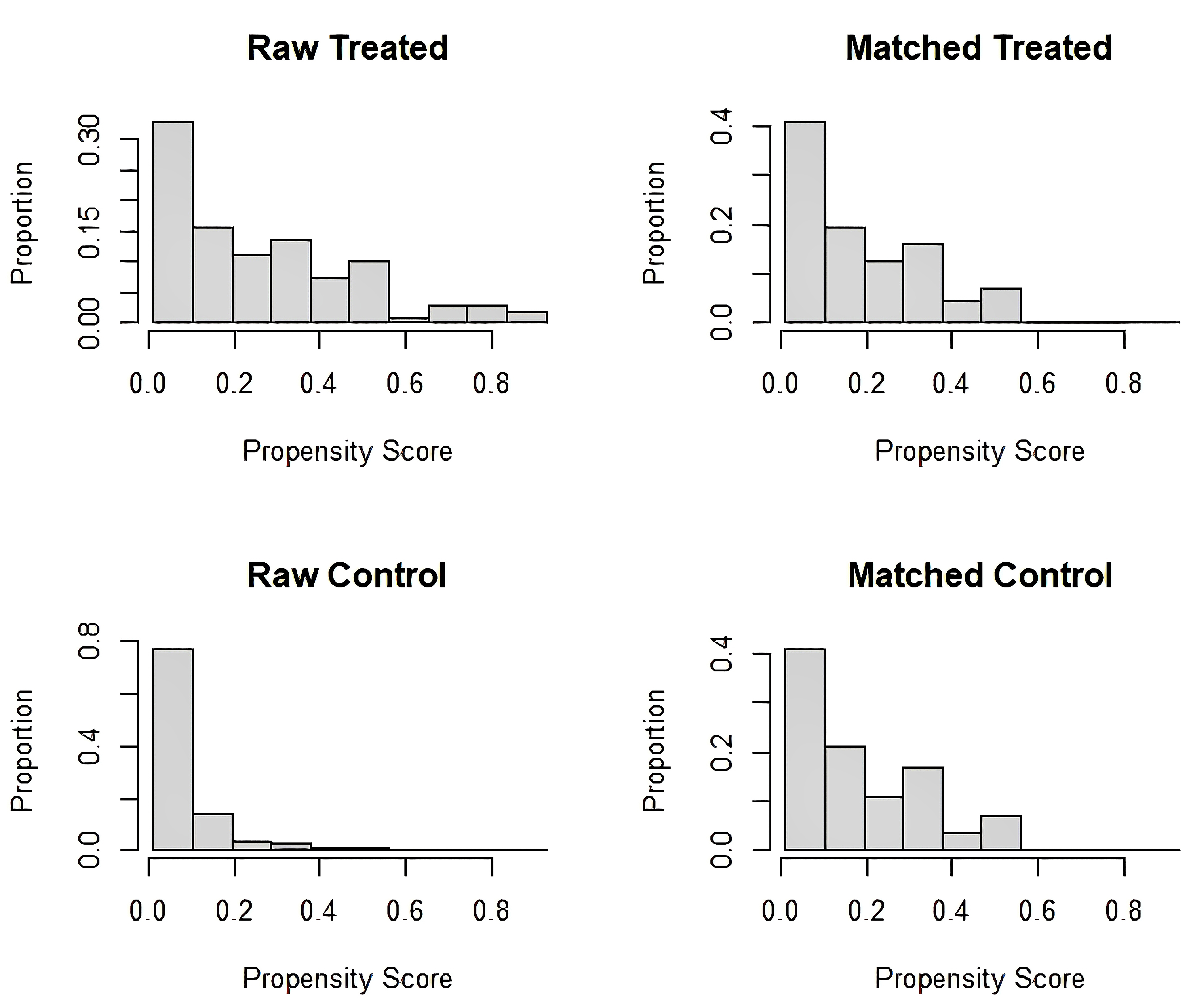 Fig. 3.
Fig. 3.
Distribution of propensity scores in the postpartum hemorrhage and non-postpartum hemorrhage groups before and after propensity score matching. Note: “Raw treated” and “raw control” refer to the data before matching. The substantial overlap deviation indicates baseline covariate imbalances. “Matched treated” and “matched control” refer to the data after matching. Post-matching distributions are shown after 1:3 propensity score matching (caliper = 0.03). “Matched treated” and “matched control” show improved distributional alignment, reflecting effective mitigation of confounding variables through the matching process.
Performing bilateral uterine artery ligation before myomectomy (OR, 0.175; 95%
CI, 0.052–0.591; p
| Characteristics | SE | Z | p-value | OR | 95% CI | |
|---|---|---|---|---|---|---|
| Pre-myomectomy bilateral uterine artery ligation (Yes/No) | –1.745 | 0.622 | –2.805 | 0.175 | 0.052–0.591 | |
| Pre-myomectomy use of potent uterotonic drugs (Yes/No) | –1.744 | 0.471 | –3.703 | 0.175 | 0.069–0.440 |
The limited research on the association between cesarean myomectomy and PPH has
added an extra burden to obstetricians in clinical management. This study showed
that the diameter of individual uterine fibroids was a risk factor for PPH,
particularly those with a diameter
Multiple uterine fibroids were an independent risk factor for PPH in this study.
Multifocal fibroids destroy the gap junctions between uterine smooth muscle
cells, weakening their ability to contract synchronously. Specifically, in our
study, a sum of the maximum diameters of the fibroids exceeding 14.5 cm had good
predictive value for PPH, which is slightly different from a finding by Li
et al. [18]. They found that the number of fibroids has no effect on the
risk of PPH. Lee et al. [20] found that larger (
We also found that particular fibroid locations were an independent risk factor
for PPH, possibly because of differences in uterine contraction and vascularity.
The lower uterine segment is predominantly composed of longitudinal muscle fibers
and it shows weaker contractility than the circular myometrial layers of the
fundal region. This architectural disparity predisposes this area to localized
uterine atony after fibroid resection. Furthermore, the dual blood supply of the
uterine segment from the cervical-vaginal arteries creates rich anastomotic
vasculature, posing challenges in achieving complete hemostasis during surgery,
which is in contrast to the fundal region’s singular uterine arterial supply.
This partially aligns with the findings of Kwon et al. [13], who studied
cesarean myomectomy for intramural fibroids. They reported that lower segment
fibroids and those with a diameter
Akbaba and Kilicci [22] demonstrated that bilateral uterine artery ligation reduced intraoperative blood loss in patients who underwent laparoscopic and open myomectomy. Furthermore, Hiratsuka et al. [23] reported that temporary uterine artery occlusion decreased hemorrhage during laparoscopic myomectomy. Although these studies confirmed the value of uterine artery ligation in reducing hemorrhage from non-pregnant uteri, the underlying mechanism (reducing uterine artery perfusion pressure) may also apply to cesarean myomectomy. The propensity score-matched analysis in our study indicated that bilateral uterine artery ligation before myomectomy protected against PPH in cesarean myomectomy. Several studies have shown that using potent uterotonic agents during abdominal myomectomy can reduce intraoperative blood loss [24, 25]. However, research on whether the use of these agents before cesarean myomectomy truly protects against PPH is relatively limited. Our study suggested that using uterotonic agents before cesarean myomectomy protected against PPH. In patients undergoing cesarean myomectomy who have a high risk of PPH, bilateral uterine artery ligation or the administration of potent uterotonic agents before myomectomy can considerably reduce intraoperative blood loss, lower the incidence of PPH, and decrease the occurrence of severe complications.
Our findings provide critical guidance for the prevention and management of PPH during cesarean myomectomy. When investigating PPH in this context, preventative strategies are important, particularly for patients with multiple fibroids or fibroids in certain locations, and for those whose operation time is expected to be prolonged. Meticulous surgical management is crucial in these cases, and procedures should ideally be performed by experienced obstetricians to ensure rapid suturing and to minimize the length of the operation.
To further mitigate the risk of PPH, bilateral uterine artery ligation should be strongly considered, especially for patients at high risk. This intervention reduces blood flow to the uterus, thereby decreasing the likelihood of excessive intraoperative bleeding. Additionally, the use of potent uterotonic drugs is highly recommended to enhance uterine contractions and promote hemostasis after delivery. These preventive measures should be considered critical components of the surgical plan because they greatly contribute to reducing the incidence of PPH and improving patients’ outcomes.
This study has several limitations. As a retrospective, single-center, non-randomized analysis, it was susceptible to selection and information biases. Although we attempted to control for confounders using PSM and other statistical methods, confounding may still have been present. Therefore, large-scale, multicenter, prospective studies with diverse patient populations are required. A multi-indicator predictive model should be a key focus of future prospective studies to further refine risk stratification.
Independent risk factors for PPH following cesarean myomectomy include multiple
fibroids, fibroids in certain locations, and a prolonged duration of surgery. A
fibroid diameter
The datasets used and analyzed during this study are available from the corresponding author on reasonable request.
XW: methodology, validation, formal analysis, investigation, data curation, writing-original draft; DZ: conceptualization, resources, writing-original draft, project administration, writing-review & editing, supervision; QS: validation, resources, formal analysis, investigation, data curation; XY: writing-review & editing,reviewing it critically for important intellectual content, supervision; HC: Conceptualization (retrospective study design), methodology (outcome definition and interpretation), writing – review & editing, supervision (data validation and methodological oversight). All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Shenyang Women’s and Children’s Hospital (Approval Number: 202358). All data were fully anonymized. The requirement for informed consent was waived owing to the study’s retrospective design.
The authors would like to thank all the journal’s anonymous reviewers for their opinions and suggestions, and Liwen Bianji (Edanz) (https://www.liwenbianji.cn) for editing the English text of a draft of this manuscript.
This work was supported by Shenyang Young and middle-aged science and technology innovation talents support program project Fund, Shenyang Science and Technology Bureau (grant number RC210043).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



