1 Department of Nosocomial Infection Management, West China Second University Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
2 Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, 610041 Chengdu, Sichuan, China
3 Department of Medical Affairs, West China Second University Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
4 Department of Medical and Education, WCSUH-Tianfu·Sichuan Provincial Children’s Hospital, 610000 Meishan, Sichuan, China
5 Department of Administrative, Chengdu High-tech Zone Center for Disease Control and Prevention, 610041 Chengdu, Sichuan, China
†These authors contributed equally.
Abstract
Surgical site infection (SSI) following breast surgery remain a significant clinical challenge, with reported incidence rates ranging from 1% to 35%. Despite advancements in surgical techniques, SSIs contribute to prolonged hospitalization, increased mortality, and substantial healthcare costs. This research applied a quantitative systematic review and meta-analysis to identify and summarize risk factors for SSIs following breast surgery.
Relevant literature from PubMed, Medline, Embase, Web of Science, and the Cochrane Central Register of Controlled Trials published between January 1, 2004, and December 25, 2023, was searched and screened using the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) procedure. The effect sizes for each identified risk factor were calculated using STATA v18 and RevMan 5.3. Heterogeneity was tested using the Q-test, and sensitivity analysis was performed using the leave-one-out method, in which one dataset was removed at a time to evaluate changes in the pooled effect sizes. A funnel plot was employed to evaluate potential publication bias.
12 studies were identified, including 2412 SSI-positive and 166,794 SSI-negative cases undergoing breast surgery. 22 potential risk factors were identified, and those reported in ≥3 studies were analyzed. Mastectomy emerged as the strongest risk factor (odds ratio [OR] = 2.61, p < 0.001), followed by diabetes (OR = 2.49, p < 0.001), body mass index (BMI) ≥25 kg/m2 (OR = 2.08, p < 0.001), American Society of Anesthesiologists (ASA) score ≥3 (OR = 1.99, p < 0.001), and smoking (OR = 1.38, p < 0.001).
Patients who underwent mastectomy demonstrated 2.61 times higher odds of developing SSI post-breast surgery (OR = 2.61). Similarly, diabetes was associated with more than twice the odds of developing SSIs (OR = 2.49), BMI ≥25 kg/m2 with twofold increase in odds (OR = 2.08), ASA score ≥3 with 99% higher odds (OR = 1.99), and smoking with a 38% increased odds (OR = 1.38). This study highlights the importance of closely monitoring surgical incisions in patients with a history of smoking, high ASA scores, or those who have undergone mastectomy. BMI and diabetes may affect each other; therefore, future studies should provide detailed reporting on the number of patients with these correlated factors.
The study has been registered on https://www.crd.york.ac.uk/prospero/ (registration number: CRD42023492359).
Keywords
- breast surgery
- SSI
- risk factors
- meta-analysis
Breast surgery is a broad concept that encompasses various surgical procedures. Common breast surgeries include mastectomy, breast reconstruction, breast reduction, and breast augmentation, among others. The frequency of mammoplasty surgeries has increased significantly in recent years, driven by medical advancements and a growing desire for aesthetic enhancement, particularly in the United States [1]. Breast surgery is generally considered a safe procedure [2]; however, the incidence of surgical site infections (SSIs) remains high, ranging between 1% to 35% [3]. SSI is one of the most common complications of breast surgery. It has been shown that SSIs prolong hospital stays, lead to reoperations and readmissions, and can even increase mortality. SSIs also result in a substantial financial burden, with additional costs arising from medical staff, investigations, and treatment [4].
The risk factors for SSIs following breast surgery have been reported in various studies; however, the results differ across these studies. There are various risk factors for SSI following breast surgery, including age [5], tobacco use [6], diabetes [7], obesity [8], flap necrosis [9], medical staff [10], malignancy [5], mastectomy [11], prior radiotherapy or chemotherapy [12, 13], inappropriate use of antibiotics [14], prolonged postoperative drainage [15], and immediate reconstructive surgery [16].
The existing literature primarily relies on observational studies with small sample sizes, characterized by wide methodological variations and varying conclusions. In addition, many risk factors lack reliable quantitative syntheses. This meta-analysis provides updated and more comprehensive data to support the prediction and prevention of SSIs after breast surgery by pooling high-quality evidence, utilizing standardized definitions, and quantifying pooled effect sizes of risk factors in 169,206 patients. The aim is to provide clinicians actionable insights for risk stratification and prevention.
The data was extracted from published empirical studies retrieved the databases, including PubMed (Central), Medline (Ovid), Embase (Ovid), Web of Science, and the Cochrane Library. The search terms followed the standard PICO (population, intervention, comparison, outcome) guideline and were adapted according to Medical Subject Headings (MeSH) terms [17]. A detailed search methodology is provided in Supplementary File 1. The search was conducted upon the completion of study registration and was finalized on December 25, 2023.
The inclusion criteria were as follows: (1) Population: female participants who developed SSI after various breast surgeries; (2) Intervention: breast surgeries; (3) Comparators: participants who did or did not develop SSI after various breast surgeries; (4) Outcomes: occurrence of SSI. The exclusion criteria were as follows: (1) Non-English studies; (2) Studies that did not provide the odds ratios (ORs) or relative risks (RRs) with 95% confidence intervals (CIs); (3) Studies that did not meet the National Healthcare Safety Network’s definition of Breast Surgery SSI Criteria.
The studies were retrieved in RIS format (.ris) and managed with EndNote X9.3.3 (Clarivate Analytics, Philadelphia, PA, USA). The screening process followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. Two independent reviewers conducted the screening process. Initially, they removed all duplicate articles. Then, articles that did not meet the inclusion criteria were excluded. For studies that met the criteria, full-text papers were obtained. Any discrepancies between the reviewers were resolved through discussion and consensus. Data extraction was done independently by two reviewers. The extraction included patient demographic characteristics, type of surgery, SSI diagnostic criteria, risk factors, OR or RR, and 95% CIs.
Two reviewers independently scored the studies using the Cochrane Risk of Bias Tool (ROB-2) and Newcastle-Ottawa Scale (NOS) for quality assessment. NOS was used for observational studies, and the ROB-2 was used for randomized controlled trials (RCTs). Any disagreements between reviewers were resolved through discussion, and if necessary, third-party adjudication was employed.
The effect size of each identified risk factor was calculated using STATA v18
(Stata Corporation, College Station, TX, USA) and RevMan 5.3 (The Nordic Cochrane
Centre, Copenhagen, Denmark). Only risk factors reported in at least 3 datasets
were synthesized for the meta-analysis. A significance level of
A search of keywords in PubMed, Medline (Ovid), Embase (Ovid), Web of Science, and the Cochrane Library yielded 6947 records. After screening based on the PRISMA guidelines, 12 studies met the inclusion criteria. The PRISMA flowchart is shown in Fig. 1.
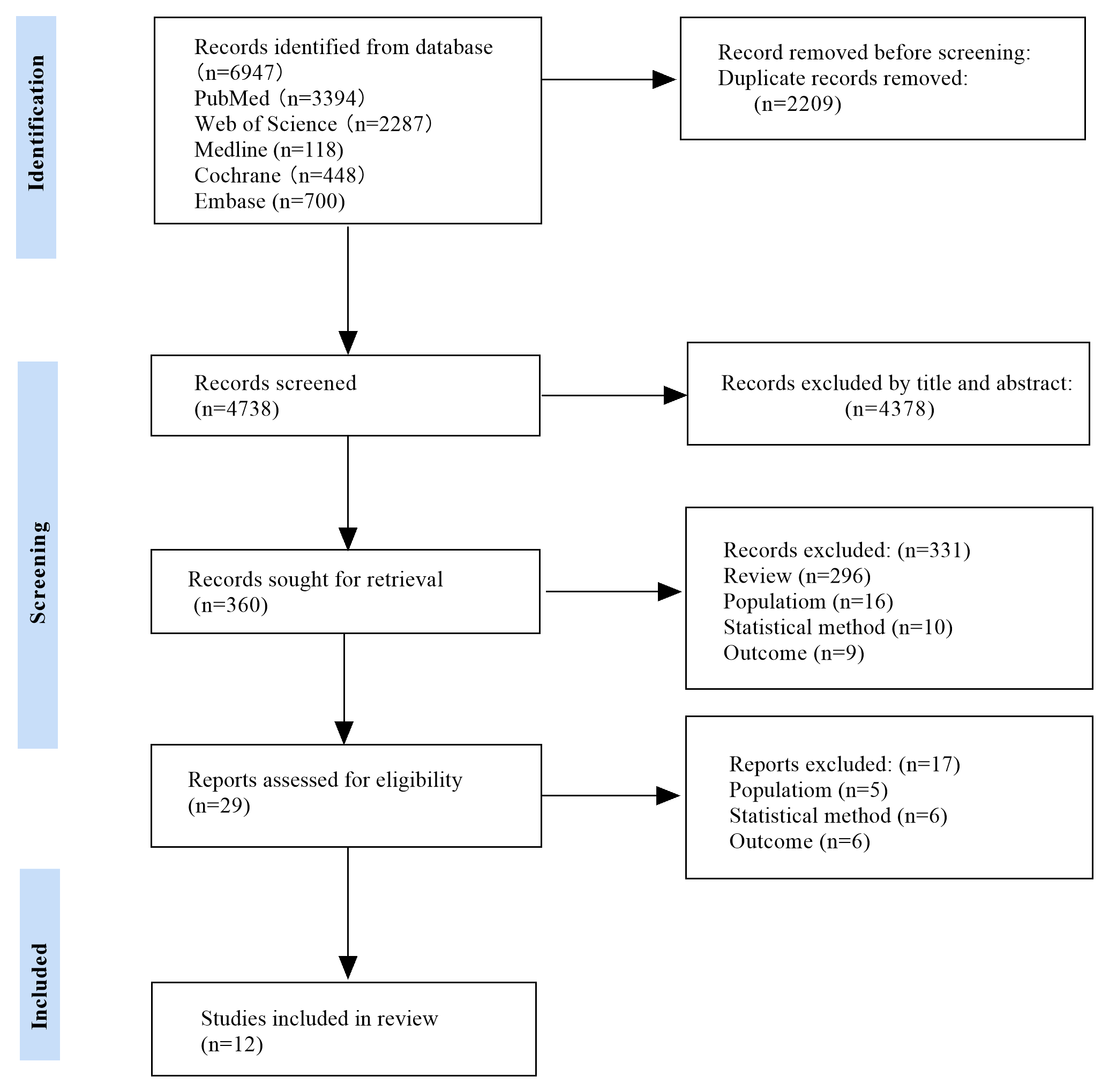 Fig. 1.
Fig. 1.
PRISMA flowchart. Note: Population: studies that included male
patients or patients undergoing non-breast surgery were excluded. Outcomes: the
primary outcome of the study was surgery site infections (SSIs). Studies that did
not provide effect estimates (RR/OR) with 95% CIs were excluded. Statistical
methods: studies that did not perform multivariate analysis after identifying
significant indicators in univariate analysis (p
A total of 12 articles were included, including 5 case-control study [18, 19, 20, 21, 22], 6 retrospective cohort studies [23, 24, 25, 26, 27, 28], and 1 RCT [29]. A total of 169,206 female patients who underwent breast surgery were included in the current analysis across 12 studies. Among them, 2412 had SSI-incisions of various types, and 166,794 with no SSI-incisions were included as controls. The details of all studies are described in Table 1 (Ref. [18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29]).
| Author, year | Vilar-Compte et al., 2004 [18] | Olsen et al., 2008 [19] | Nguyen et al., 2012 [20] | Davis et al., 2013 [21] | Chung et al., 2015 [22] | Olsen et al., 2016 [23] | Parikh et al., 2016 [24] | Struik et al., 2018 [29] | Kraenzlin et al., 2022 [25] | Rothe et al., 2022 [26] | Zhang et al., 2022 [27] | Chin et al., 2023 [28] |
| Sample origin | 2000 INCan, USA | 1998–2002 Barnes-Jewish Hospital, USA | 2005–2009 ACS-NSQIP, USA | 2005–2009 ACS-NSQIP, USA | 2005–2012 ACS-NSQIP, USA | 2004–2011 Commercial claims data, USA | 2010–2014 NHSN, USA | 2013–2016 Franciscus Gasthuis, NL | 2016–2018 Medical center, USA | 2011–2018 University hospital, Germany | 2012–2021 CAMS, China | 2016–2020 Sahlgrenska University Hospital, Sweden |
| N | 230 | 325 | 9315 | 38,739 | 2899 | 7115 | 110,987 | 230 | 490 | 240 | 4793 | 958 |
| SSI | 76 | 57 | 330 | 891 | 143 | 595 | 630 | 23 | 66 | 62 | 34 | 100 |
| BMI (kg/m2) | NR | NR | NR | NR | NR | NR | ||||||
| Previous RT/CH | Y/N | Y/N | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Mastectomy | Y/N | Y/N | NR | NR | NR | NR | NR | NR | Y/N | NR | Y/N | |
| Length of drain stay (days) | NR | NR | NR | NR | NR | NR | Mean | NR | NR | NR | ||
| 2nd drain insertion | Y/N | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Implant | NR | Y/N | NR | NR | NR | Y/N | NR | NR | NR | NR | NR | NR |
| Antibiotic | NR | dosing | NR | NR | NR | NR | NR | NR | NR | NR | Y/N | NR |
| Transfusion | NR | Y/N | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Smoking | NR | Y/N | NR | Y/N | Y/N | Y/N | NR | NR | NR | NR | NR | NR |
| Anesthetic | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Alcohol consumption | NR | NR | Y/N | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| ASA | NR | NR | NR | NR | NR | NR | NR | NR | NR | |||
| Flap failure | NR | NR | Y/N | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Operative time | NR | NR | NR | NR | NR | NR | NR | NR | NR | |||
| Diabetes | NR | NR | NR | Y/N | NR | Y/N | NR | NR | NR | NR | NR | Y/N |
| Hypertension | NR | NR | NR | NR | Y/N | Y/N | NR | NR | NR | NR | NR | NR |
| Age | NR | NR | NR | NR | NR | NR | Y/N | NR | Mean | NR | Mean | NR |
| ASC | NR | NR | NR | NR | NR | NR | Y/N | NR | NR | NR | NR | NR |
| Dressing change | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
| Seroma | NR | NR | NR | NR | NR | NR | NR | NR | Y/N | NR | NR | Y/N |
| Axillary incision | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | Y/N | NR |
Note: The content under each risk factor reflects how the data were presented in the original studies. The detailed case numbers are provided in Table 3. “Sample Origin” refers to the geographic or institutional source of the study population. “N” refers to the total sample size. “SSI” refers to the number of patients with surgical site infections. BMI, body mass index; RT/CH, radiotherapy/chemotherapy; ASA, American Society of Anesthesiologists; ASC, ambulatory surgery center; NR, not reported; NA, not applicable; Y/N, reported in Yes/No; NSQIP, National Surgical Quality Improvement Program; NHSN, National Healthcare Safety Network; CAMS, Collaborative Assessment and Management of Suicidality; INCan, Instituto Nacional de Cancerología.
Among these studies, only 1 reported each incision type separately [28]. The remaining 11 studies reported infection cases as a combination of three incision types (superficial, deep, or organ space). The studies reporting cases with independent incision types were combined into the mixed incision category to align with those reporting mixed incision types for synthesis. As shown in Table 2 (Ref. [18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28]), the NOS risk of bias assessment rated 2 studies with a score of 6 [20, 23], 7 studies with a score of 7 [18, 19, 21, 22, 24, 25, 28], and the remaining 2 with a score of 8 [26, 27]. The only RCT article was assessed using ROB-2 and was considered to have a low-risk of bias [29]. The process is detailed in Supplementary File 1. All 12 studies were included in the following review.
| Author, year | Selection | Comparability | Exposure | Total score | |||||
| Vilar-Compte et al., 2004* [18] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 7 |
| Olsen, 2008 et al.* [19] | 1 | 1 | 1 | 0 | 2 | 1 | 1 | 0 | 7 |
| Nguyen et al., 2012* [20] | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 6 |
| Davis et al., 2013* [21] | 1 | 1 | 0 | 0 | 2 | 1 | 1 | 1 | 7 |
| Chung et al., 2015* [22] | 1 | 1 | 1 | 0 | 2 | 1 | 1 | 0 | 7 |
| Olsen et al., 2016# [23] | 0 | 1 | 1 | 0 | 2 | 1 | 1 | 0 | 6 |
| Parikh et al., 2016# [24] | 1 | 1 | 1 | 0 | 2 | 1 | 0 | 1 | 7 |
| Kraenzlin et al., 2022# [25] | 1 | 1 | 1 | 0 | 2 | 1 | 1 | 0 | 7 |
| Rothe et al., 2022# [26] | 1 | 1 | 1 | 0 | 2 | 1 | 1 | 1 | 8 |
| Zhang et al., 2022# [27] | 1 | 1 | 1 | 0 | 2 | 1 | 1 | 1 | 8 |
| Chin et al., 2023# [28] | 1 | 1 | 1 | 0 | 2 | 1 | 1 | 0 | 7 |
Note. * indicates the study was assessed using the criteria for case-control studies. #indicates the study was assessed using the criteria for cohort studies. NOS, Newcastle-Ottawa Scale.
Among the 12 studies, a total of 22 risk factors were identified, including body mass index (BMI), previous radiotherapy/chemotherapy (RT/CH), mastectomy, length of drain stay, second drain insertion, implant, antibiotic use, transfusion, smoking, anesthetic type, alcohol consumption, American Society of Anesthesiologists (ASA) classification, flap failure, operative time, diabetes, hypertension, age, ambulatory surgery center (ASC), dressing change, seroma, and axillary incision. However, only 2 factors, BMI and mastectomy, were reported in 5 datasets. 4 factors-length of drain stay, ASA, operation time, and age were reported in 3 datasets. The remaining factors were reported in only 1 or 2 datasets. Only factors reported in 3 or more datasets were selected for analysis in this study.
A total of 8 risk factors had sufficient data subsets. Age, length of drain
stay, and operative time reported continuous data and could not be directly
synthesized. Lastly, mastectomy, diabetes, ASA
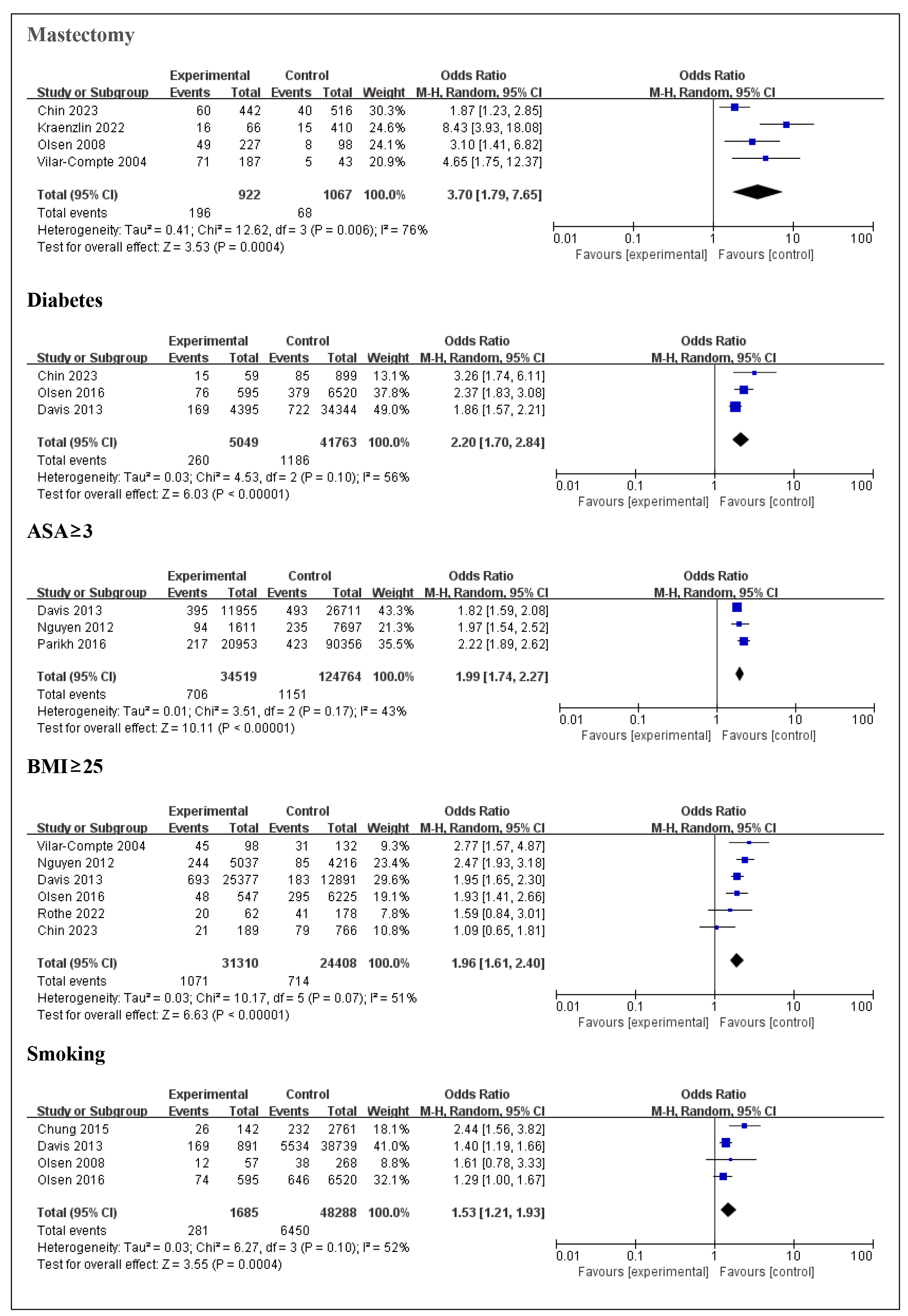 Fig. 2.
Fig. 2.
Forest plots for each risk factor. This figure displays the forest plots for the identified risk factors, showing the pooled effect sizes for each factor associated with SSIs after breast surgery. ASA, American Society of Anesthesiologists; BMI, body mass index; OR, odds ratio; CI, confidence interval; SSI, surgical site infection; M-H, Mantel-Haenszel.
| Case number | Meta-analysis | Heterogeneity test | Egger’s publication bias | ||||||||
| Risk factor | RF+ SSI+ | RF+ SSI– | RF– SSI+ | RF– SSI– | OR (95% CI) | Z | p | I2 | df | p | p |
| BMI |
1071 | 30,239 | 714 | 23,694 | 1.96 (1.61, 2.40) | 6.63 | 51% | 5 | 0.07 | 0.20 | |
| Mastectomy | 196 | 726 | 88 | 979 | 3.70 (1.79, 7.65) | 3.53 | 0.0004 | 76% | 3 | 0.006 | 0.04 |
| Smoking | 281 | 1404 | 6450 | 41,838 | 1.53 (1.21, 1.93) | 3.55 | 0.0004 | 52% | 3 | 0.10 | 0.15 |
| ASA |
706 | 33,813 | 1151 | 123,613 | 1.99 (1.74, 2.27) | 10.11 | 43% | 2 | 0.17 | 0.30 | |
| Diabetes | 260 | 4789 | 1186 | 40,577 | 2.20 (1.70, 2.84) | 6.03 | 56% | 2 | 0.10 | 0.12 | |
Note. RF+ refers to exposure to the risk factor, RF– refers to no exposure to the risk factor, SSI+ refers to patients with a positive SSI incision, and SSI– refers to patients with a negative SSI incision.
However, BMI
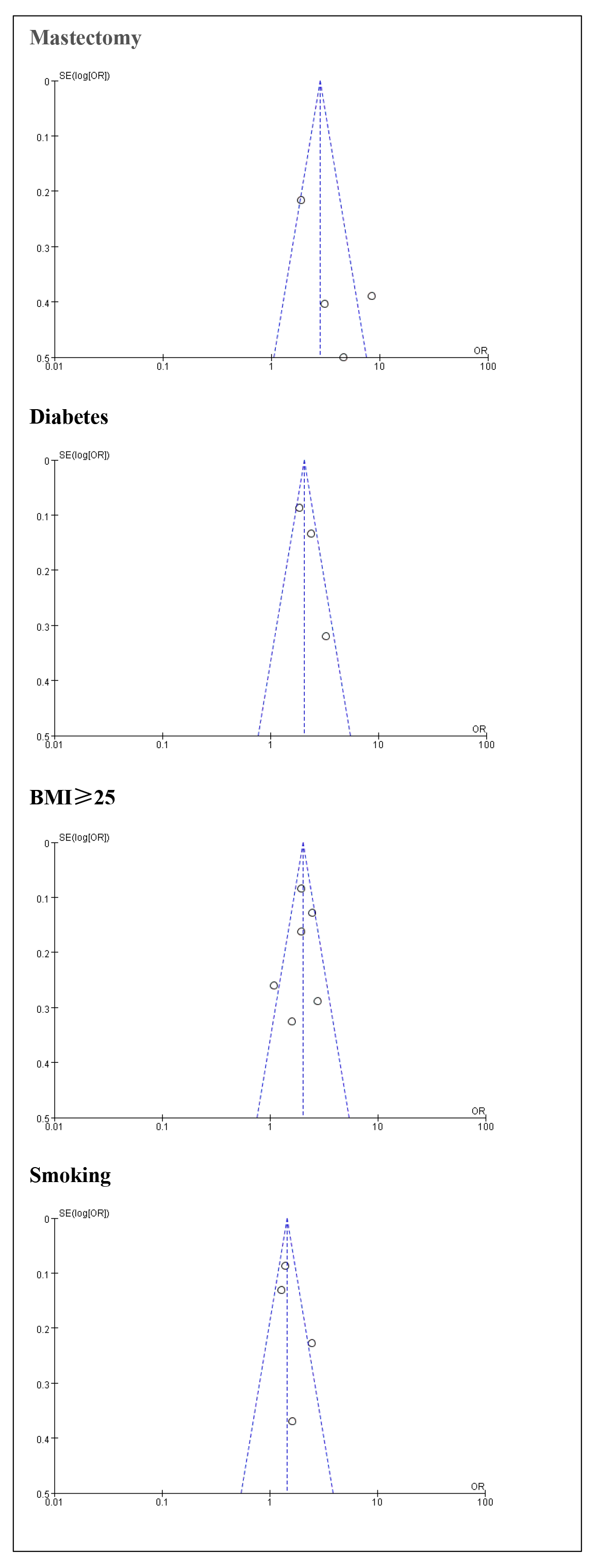 Fig. 3.
Fig. 3.
Funnel plots for each risk factor.
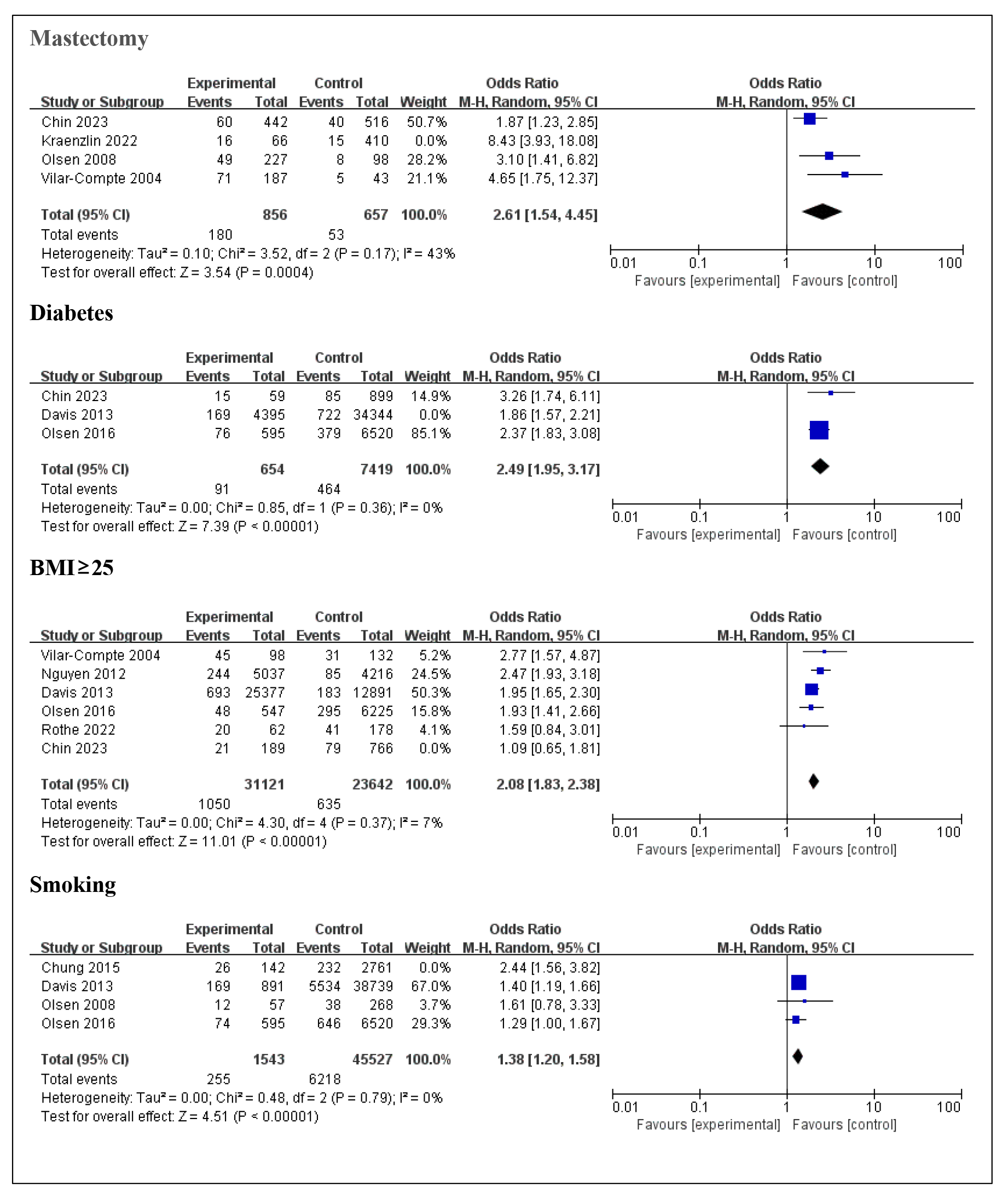 Fig. 4.
Fig. 4.
Forest plot for each risk factor after reducing heterogeneity.
The current study used a quantitative systematic review and meta-analysis to
summarize the data for risk variables for SSI incisions following breast surgery.
In total, 12 studies were identified with 2412 SSI-positive cases and 166,794
SSI-negative cases, under 22 identified risk factors. These factors included BMI,
previous RT/CH, mastectomy, length of drain stay, second drain insertion,
implant, antibiotic use, transfusion, smoking, anesthetic use, alcohol
consumption, ASA score, flap failure, operative time, diabetes, hypertension,
age, ASC, dressing change, seroma, and axillary incision. However, only 5 could
be synthesized for the meta-analysis. Of these, mastectomy (OR = 2.61, p
The largest risk factor for SSI incisions is mastectomy (OR = 2.61), which demonstrates 2.61 times higher odds of developing SSI [30]. Some articles have shown that mastectomy is one of the risk factors for postoperative SSI when compared to breast-conserving surgery (BCS) [31, 32]. Compared to BCS, mastectomy involves extensive tissue removal and a larger surgical wound, which can disrupt the vascular supply, create a more hypoxic environment in the tissue, and increase the risk of bacterial colonization. However, this does not imply that BCS should always be prioritized in clinical practice. Clinicians should focus on the patient’s individual condition and requirements. After mastectomy, the patient’s surgical incision should be closely monitored, and measures to prevent incision infection should be implemented.
Diabetes is the second significant risk factor for SSI. Elevated blood glucose
levels promote bacterial adhesion and biofilm formation, exacerbating infection
risks. It could be argued that BMI and diabetes are linked, as obesity-induced
insulin resistance is one of the leading causes of type 2 diabetes [33]. This may
explain the variability of the ORs in BMI
To further address this issue, the current study conducted a meta-regression analysis to investigate whether BMI predicts the ORs of diabetes. The analysis found no significant relationship between continuous BMI values and the ORs of diabetes. Notably, the absence of a significant predictive relationship between BMI and the OR for diabetes does not imply that these two factors were unrelated or that obesity does not influence the estimation of OR for diabetes. On the one hand, the absence of a significant predictive relationship might arise from the inclusion of both type I and type II diabetes in the studies, with type I diabetes having less direct relevance to obesity. On the other hand, the estimation of ORs may have been inflated due to the repeated counting of cases exposed to multiple risk factors. Instead, this result might suggest that the pathological mechanisms of obesity and diabetes operate relatively independently in the context of SSI risk, rather than one being a direct predictor of the other.
Apart from diabetes and BMI
Smoking was the last risk factor (OR = 1.38, p
To account for multiple comparisons across risk factors, we applied the
Benjamini-Hochberg false discovery rate (FDR) correction. After adjusting for
multiple comparisons using the FDR correction, the significance threshold for
each test was adjusted to q = 0.05/5 = 0.01 and all identified risk factors
remained statistically significant, confirming that mastectomy, diabetes, BMI
This meta-analysis identified 5 significant risk factors for SSI after breast
surgery, with mastectomy being most strongly associated with SSI. The factors
that significantly increased the risk of SSI included mastectomy, diabetes, BMI
All relevant data are within the manuscript and its Supporting information files.
YL and ZY designed the research study, and drafted the protocol documents for registration. YL, ZY and HZ were the independent investigators to conducted the study’s review, quality assessment, and data extraction. YL and ZY completed the data analysis, DW interpretation of data for the work, and YL drafted the manuscript. Finally, ZY and DW revised the draft. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Thanks to all the researchers who contributed to this paper and provided advice.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG37161.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




