1 Department of Obstetrics, Jinhua People’s Hospital, 321000 Jinhua, Zhejiang, China
Abstract
Proper invasion and growth of trophoblast cells are crucial for the prevention of preeclampsia. This pilot study, conducted with a small sample size, focused on the regulatory effects of interferon-inducible protein 10 (IP-10) and lipoxin A4 (LXA4) on HTR-8-SVneo trophoblast cells.
A total of 5 patients with preeclampsia and 5 normotensive pregnant women were enrolled in this study. Serum and placental levels of IP-10, LXA4, and the LXA4 receptor (ALX) were measured. Both IP-10 and ALX were silenced in HTR-8-SVneo cells. The effects on HTR-8-SVneo cell behaviors were assessed using wound healing assay, transwell assay, colony formation assay, Cell Counting Kit-8 (CCK8) assay, flow cytometry, and terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick-end labeling (TUNEL) staining. Inflammatory and oxidative stress markers were measured using biochemical assay. Apoptosis-related proteins were measured via western blotting.
High levels of IP-10 and low levels of LXA4, accompanied by decreased ALX expression, were observed in patients with preeclampsia. IP-10 knockdown significantly stimulated the invasion, migration and proliferation of HTR-8-SVneo cells while inhibiting apoptosis, whereas ALX silencing exerted the opposite effects. Silencing of IP-10 or ALX also produced opposing effects on the regulation of oxidative stress and inflammation in HTR-8-SVneo cells.
Although this pilot study has a small sample size (n = 5 per group) and inherent limitations, it provides noteworthy preliminary evidence of an antagonistic relationship between IP-10 and LXA4 in HTR-8-SVneo cells.
Keywords
- preeclampsia
- interferon-inducible protein 10
- lipoxin A4
- HTR-8-SVneo cell
Preeclampsia is a pregnancy-specific disorder that typically develops after the 20th week of gestation. It is clinically characterized by proteinuria and hypertension [1]. It causes multi-organ impairment, being a leading cause of maternal mortality, and may also result in placental abruption, fetal growth restriction, and perinatal deaths [2, 3]. While the exact etiology of preeclampsia remains unclear, defective spiral artery remodeling is widely recognized as a key pathogenic factor [4]. Appropriate trophoblast invasion and growth are crucial for spiral artery remodeling and placenta development [5]. Consequently, factors inducing trophoblast dysfunction may trigger abnormal placental formation, providing new insights into preeclampsia pathogenesis.
Interferon-inducible protein 10 (IP-10) is a Cys-X-Cys (CXC; X means any amino
acid) chemokine produced by activated fibroblasts, monocytes, endothelial cells,
and lymphocytes [6], playing a significant biological role in mediating Th1-type
inflammatory responses and inhibiting angiogenesis. Clinical studies have
identified elevated levels of IP-10 in patients with various diseases
characterized by Th1-dominant inflammatory reactions, such as pulmonary
tuberculosis, systemic lupus erythematosus, rheumatoid arthritis, and
atherosclerosis [7, 8, 9, 10]. These conditions elicit the corresponding immune
responses. Notably, serum IP-10 levels are significantly higher in pregnant women
with preeclampsia than in those without [11, 12], suggesting a potential
association between IP-10 and preeclampsia onset. Lipoxin A4 (LXA4), produced via
dual lipoxygenase-mediated lipoxygenation of arachidonic acid, serves as a ‘stop
signal’ in inflammation-related diseases [13]. Its primary receptor is formyl
peptide receptor 2, also referred to as the LXA4 receptor (ALX). Studies have
demonstrated that LXA4 effectively reduces the production of tumor necrosis
factor-alpha (TNF-
This pilot study investigated the role of IP-10 and LXA4 in preeclampsia progression using in vitro models. They exhibited antagonistic effects on the proliferation, migration, and invasion capabilities of HTR-8-SVneo cells. Our findings suggest that molecular-targeted agents enhancing LXA4 levels or inhibiting IP-10 levels may facilitate proper trophoblast invasion and growth, thereby reducing preeclampsia risk.
Five pregnant women with preeclampsia who underwent cesarean section at Jinhua
People’s Hospital were enrolled. Venous blood samples (5 mL) were obtained via
venipuncture and centrifuged at 3000 revolutions per minute for 10 min to obtain
serum. Placental tissues were collected within 60 min after cesarean delivery,
immediately frozen in liquid nitrogen, and kept at –80 °C for subsequent
analysis. Preeclampsia was diagnosed based on the following criteria: (i) after
20 weeks of gestation, the diastolic (
| Baseline characteristics | Normal (n = 5) | Preeclampsia (n = 5) | p-value |
| Maternal age (years) | 30.8 |
31.2 |
0.78 |
| Gestational age (weeks) | 37.1 |
36.5 |
0.42 |
| Diastolic blood pressure (mmHg) | 72.0 |
95.0 |
|
| Systolic blood pressure (mmHg) | 118.0 |
152.0 |
|
| 24-hour urine protein (g) | 0.12 |
1.80 |
The human immortalized HTR-8-SVneo trophoblast cell line (cat.no. CL-0765), obtained from Pricella Biotechnology (Wuhan, Hubei, China), was maintained in a specialized medium (cat.no. CM-0765; Pricella Biotechnology). The cells tested negative for the presence of mycoplasma and were authenticated by short tandem repeat profiling. Cells in the control group were cultured under standard normoxic conditions (37 °C, 5% CO2). For hypoxia treatment, cells were exposed to an atmosphere of 93% N2, 2% O2, and 5% CO2 [17, 18].
HTR-8-SVneo cells (1
HTR-8-SVneo cells and placental tissues were collected for RNA isolation via a
Total RNA extraction Kit (cat.no. RE-03011; Foregene Biotechnology, Chengdu, Sichuan,
China). cDNA was then synthesized with the SeqHunt® First Strand
cDNA Synthesis Kit (cat.no. CA01; Seq-Hunt Biotechnology, Beijing, China). PCR
analysis was performed on a Real-time PCR System (version 1.4.1; Applied
Biosystems, Foster City, CA, USA) using 2
| Gene | Sequences | |
| IP-10 | Forward | 5′- TCTGAGCCTACAGCAGAGGA -3′ |
| Reverse | 5′- ATGCAGGTACAGCGTACAGTT -3′ | |
| ALX | Forward | 5′- AAGCCAAGAAGCACACAGGA -3′ |
| Reverse | 5′- CAATGGGAGGATCCGCAGAA -3′ | |
| Bcl-2 | Forward | 5′- AAAAATACAACATCACAGAGGAAGT -3′ |
| Reverse | 5′- GTTTCCCCCTTGGCATGAGA -3′ | |
| Bax | Forward | 5′- GAGCAGCCCAGAGGCG -3′ |
| Reverse | 5′- TGAGACACTCGCTCAGCTTC -3′ | |
| Caspase-3 | Forward | 5′- TCCTAGCGGATGGGTGCTAT -3′ |
| Reverse | 5′- CTCACGGCCTGGGATTTCAA -3′ | |
| TNF- |
Forward | 5′- GACAAGCCTGTAGCCCATGT -3′ |
| Reverse | 5′- GGAGGTTGACCTTGGTCTGG -3′ | |
| IL-1 |
Forward | 5′- AACCTCTTCGAGGCACAAGG -3′ |
| Reverse | 5′- AGATTCGTAGCTGGATGCCG -3′ | |
| GAPDH | Forward | 5′- AAGGTCATCCCTGAGCTGAAC -3′ |
| Reverse | 5′- ACGCCTGCTTCACCACCTTCT -3′ | |
IP-10, interferon-inducible protein 10; ALX, LXA4 receptor; TNF-
Protein extracts from HTR-8-SVneo cells and placental tissues were extracted using radioimmunoprecipitation assay (RIPA) lysis buffer (cat.no. BL504A; Biosharp Life Sciences, Hefei, Anhui, China). Subsequently, the protein samples were separated by SDS-PAGE and transferred onto a Polyvinylidene Fluoride (PVDF) membrane. Subsequently, the membrane was incubated with 5% non-fat milk (cat.no. BS102; Biosharp Life Sciences) at room temperature for 1 h to block any non-specific binding sites. The membrane was probed with primary antibodies against IP-10 (cat.no. 10937-1-AP; 1:200; Proteintech, Wuhan, Hubei, China), ALX (cat.no. bs-23765R; 1:1000; Bioss Biotechnology, Beijing, China), B-cell lymphoma 2 (Bcl-2) (cat.no. ABP50759; 1:1000; Abbkine Scientific, Wuhan, Hubei, China), B-cell lymphoma 2-associated X protein (Bax) (cat.no. 60267-1-Ig; 1:10,000; Proteintech), Caspase-3 (cat.no. ab32351; 1:5000; Abcam, Cambridge, UK) and GAPDH (cat.no. 60004-1-Ig; 1:50,000; Proteintech) at 4 °C overnight. After three washes with Tris-buffered saline with Tween 20 (TBST, 0.1% Tween 20) for 10 min each, the membrane was further incubated with secondary antibodies (cat.no. A21020 & A21010; 1:10,000; Abbkine Scientific) at 25 °C for 2 h. Protein signals were detected using a SuperKine™ ECL Kit (cat.no. BMU101-CN; Abbkine Scientific), and analyzed with Image Tool software (version 3.0; UTHSCSA, San Antonio, TX, USA). Relative protein expression levels were normalized to GAPDH. Experiments were conducted in triplicate and repeated three times. Clinical results were performed with three human samples per group.
Placental tissues were fixed, paraffin-embedded, deparaffinized, and rehydrated. After antigen retrieval, tissue sections were incubated overnight at 4 °C with primary antibodies IP-10 (1:200; Proteintech), CXC motif chemokine receptor 3 (CXCR3) (1:200; Proteintech) and ALX (1:1000; Bioss Biotechnology). This was followed by a subsequent incubation step using horseradish peroxidase-conjugated secondary antibody (1:3000; Abbkine Scientific) for 1 h at 37 °C. After that, each slice was stained with 3,3’-diaminobenzidine (DAB), and then counterstained with hematoxylin for 1 min. The sections were differentiated with 1% hydrochloric acid ethanol for 30 sec, followed by bluing in 0.5% ammonia water for 1 min. After dehydration through a graded ethanol series and clearing with xylene, the slices were mounted. Microscopic imaging was performed under a light microscope (YONGXIN OPTICS, Ningbo, Zhejiang, China). Clinical results were performed with three human samples per group.
Colony formation assay was performed to assess the proliferative potential.
Briefly, HTR-8-SVneo cells, with a density of 1
For the measurement of cell viability, HTR-8-SVneo cells with the density of 3
Briefly, 100 µL of Matrigel (1:8 diluted in serum-free medium) was
uniformly spread onto the polycarbonate membrane surface in upper chamber, and
then incubated at 37 °C for 30 min to allow gelation. Complete medium
(containing 10% fetal bovine serum) was added to the lower chamber of the
24-well plates, and the transwell chamber was placed in the 24-well plates with
tweezers. In the upper chamber, a total of 2
Horizontal lines were drawn on the back of 6-well plates with a marker pen.
HTR-8-SVneo cells (5
Cell apoptosis was assessed using an Annexin V-/Propidium Iodide (PI) Apoptosis Detection kit
(cat.no. KTA0002; Abbkine Scientific). HTR-8-SVneo cells were trypsinized, washed
three times with PBS, and resuspended in 1
ROS levels in HTR-8-SVneo cells were measured using a ROS assay Kit (Beyotime,
Shanghai, China). Briefly, HTR-8-SVneo cells (1
One-step TUNEL Apoptosis Assay Kit (cat.no. KTA2011; Abbkine Scientific) was used to measure the TUNEL-positive cells. HTR-8-SVneo cells were fixed with 4% paraformaldehyde for 15 min and permeabilized with 0.25% Triton-X 100 for 20 min. Then, 50 µL of TUNEL reaction agent were added and incubated at 37 °C for 1 h. Following washing with PBS, cells were stained with DAPI (5 µg/mL) for 5 min in the dark. Images were captured under a fluorescence microscope (Olympus). Experiments were conducted in triplicate and repeated three times.
Following the manufacturer’s guidelines, malondialdehyde (MDA; cat.no. KTB1050)
and superoxide dismutase (SOD; cat.no. KTB1030) concentrations were assessed
using kits from Abbkine Scientific. Meanwhile, the levels of IL-1
Data were presented as mean
A total of 5 patients with preeclampsia and 5 normal pregnancies were enrolled
in this study. As illustrated in Fig. 1A, the preeclampsia group exhibited
significantly decreased SOD levels (p
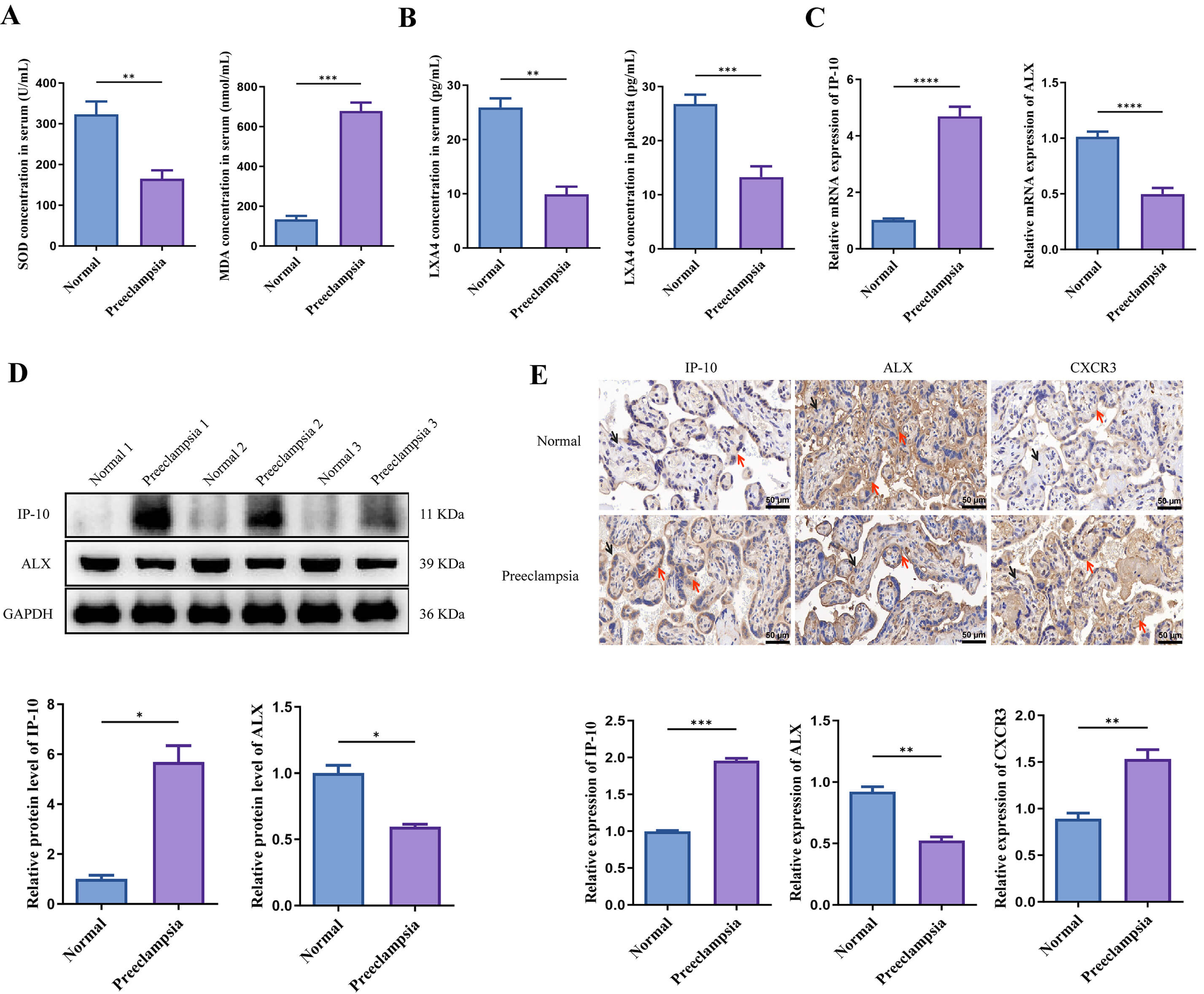 Fig. 1.
Fig. 1.
IP-10 expression is upregulated, while LXA4 level is
downregulated in patients with preeclampsia. (A,B) SOD and MDA levels as well as
LXA4 concentration in patients with preeclampsia were measured via biochemical
assay. (C) The mRNA expression of IP-10 and ALX in patients with preeclampsia was
examined via qRT-PCR. (D,E) The protein levels of IP-10, ALX and/or CXCR3 in
patients with preeclampsia were measured by western blot and
immunohistochemistry. Scale bar = 50 µm. The hematoxylin-stained nuclei
appear blue (black arrow), and the positive DAB staining is characterized by a
brown color (red arrow). *p
We subsequently silenced IP-10 and ALX in hypoxia-induced HTR-8-Svneo cells to
investigate their effects on cellular behaviors. IP-10 (Fig. 2A, p
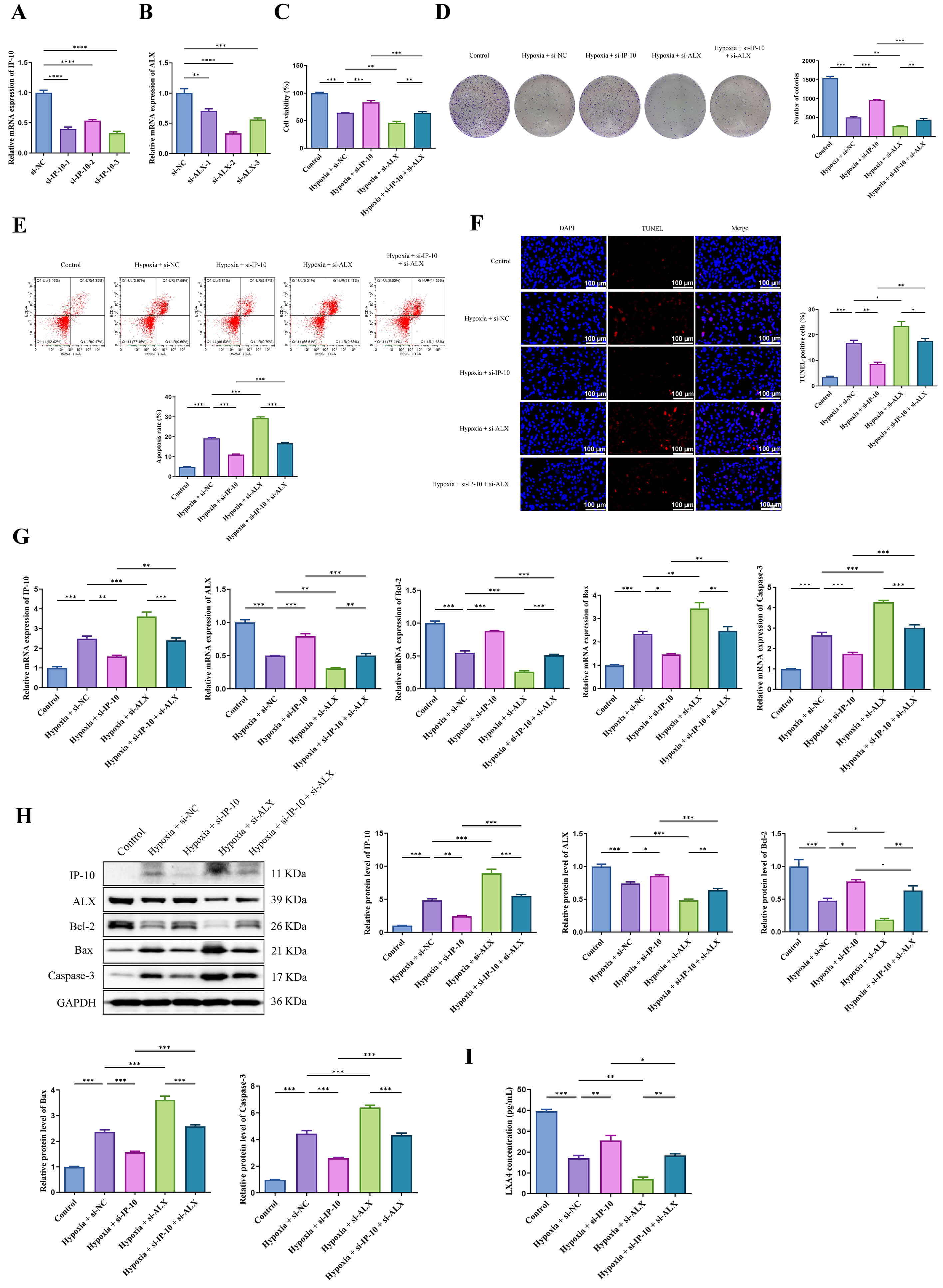 Fig. 2.
Fig. 2.
The antagonistic relationship between IP-10 and LXA4 in the
proliferation and apoptosis of hypoxia-induced HTR-8-SVneo cells. (A) IP-10 mRNA
expression in hypoxia-induced HTR-8-Svneo cells after transfection of
si-IP-10-1/-2/-3 or si-NC. (B) ALX mRNA expression in hypoxia-induced HTR-8-SVneo
cells after transfection of si-ALX-1/-2/-3 or si-NC. (C,D) Following
transfection, HTR-8-SVneo cell proliferation was assessed by CCK-8 and colony
formation assays. (E,F) Flow cytometer and TUNEL staining were conducted to
measure the apoptosis of HTR-8-SVneo cells. Scale bar = 100 µm. (G) The
mRNA expression of IP-10, ALX, Bcl-2, Bax and Caspase-3 in hypoxia-induced
HTR-8-Svneo cells was detected via qRT-PCR. (H) The protein levels of IP-10, ALX,
Bcl-2, Bax and Caspase-3 in hypoxia-induced HTR-8-SVneo cells were measured by
western blot. (I) LXA4 concentrations in hypoxia-induced HTR-8-SVneo cells were
measured via biochemical assay. *p
Subsequently, the effects of IP-10 and LXA4 on the invasion, migration,
oxidative stress and inflammation of HTR-8-SVneo cells were further determined.
si-IP-10 transfection significantly enhanced the invasion and migration of
HTR-8-SVneo cells (Fig. 3A,B, p
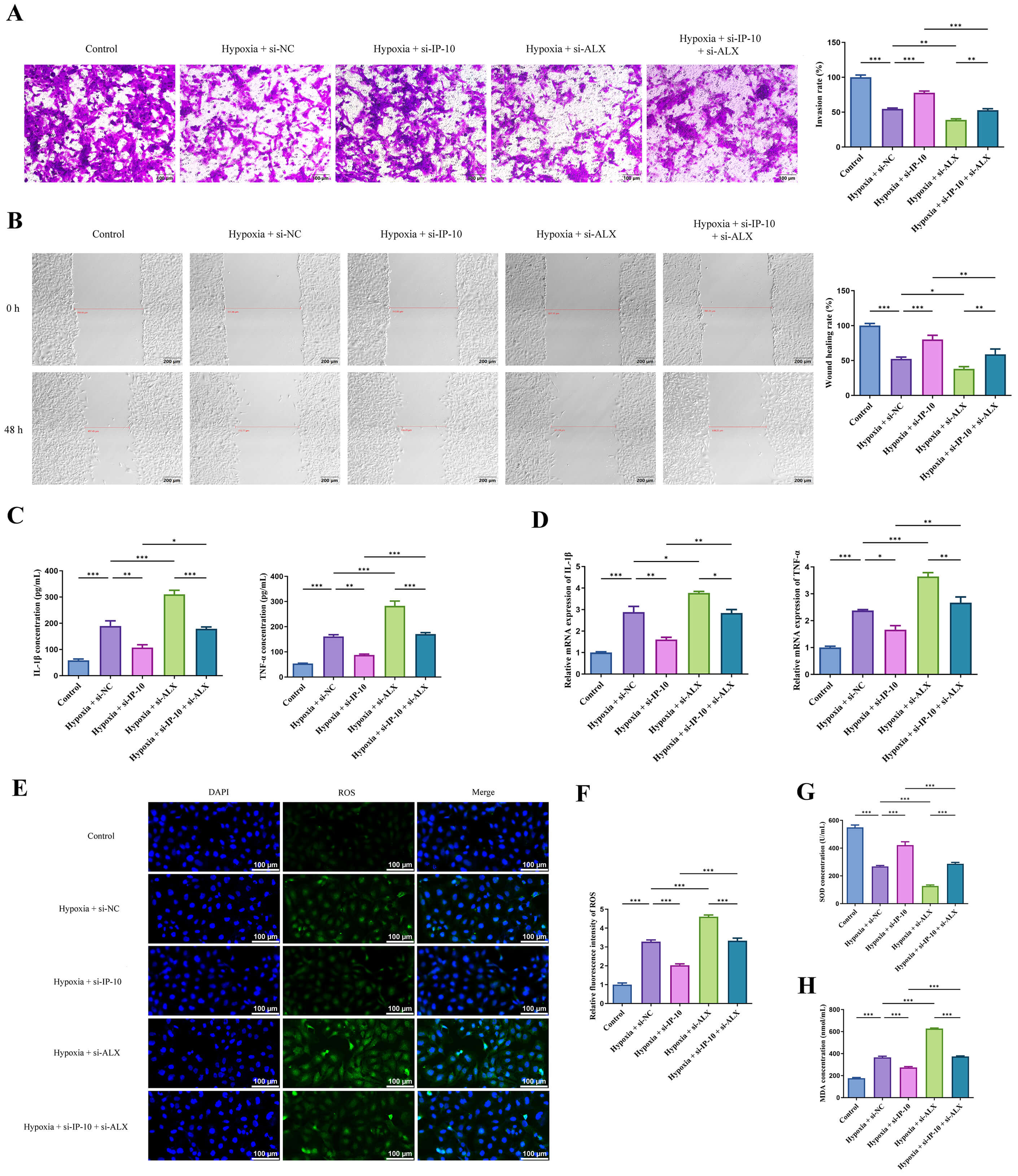 Fig. 3.
Fig. 3.
The antagonistic relationship between IP-10 and LXA4 in the
invasion, migration, oxidative stress and inflammation of HTR-8-Svneo cells.
(A,B) The invasion (scale bar = 100 µm) and migration (scale bar = 200
µm) of HTR-8-SVneo cells were evaluated through transwell and wound healing
assays. (C,D) The concentrations and mRNA expression of IL-1
The prevalence of preeclampsia in China ranges from 2.4% to 4.2%, with a
higher burden in economically underdeveloped regions where preventive and
therapeutic resources are limited [21]. Currently, the most effective strategy
for the management of preeclampsia remains pregnancy termination and placental
delivery. However, determining the optimal timing for delivery remains a
challenging study [22]. While studies indicate that fish oil, vitamin D, and
aspirin may play key roles in the prevention of preeclampsia, conclusive
scientific evidence supporting their efficacy is still lacking [23, 24].
Furthermore, the healthcare burden of preeclampsia is substantial, with average
maternal and infant medical costs reaching
Preeclamptic patients showed characteristic expression patterns in serum and placental tissues: elevated IP-10 levels and reduced LXA4 (and its receptor ALX) levels. Compared with single biomarkers, the combined detection of serum IP-10 and LXA4 can improve diagnostic accuracy, especially for early screening in the second trimester. Since serum samples are obtained through minimally invasive procedures, this combined biomarker panel is feasible for routine prenatal screening, even in resource-limited settings. Future studies with larger cohorts are needed to validate threshold values for IP-10 and LXA4 that distinguish preeclamptic from normal pregnancies, and to assess their predictive value for early-onset or severe preeclampsia.
Numerous studies have reported that elevated IP-10 levels may act as a
pathogenic factor in cancer development [26, 27, 28]. These findings collectively
indicate that IP-10 may serve as a promising therapeutic target for cancer
treatment. Given the shared importance of cell proliferation and invasion in both
cancer pathogenesis and preeclampsia development, we hypothesized that abnormal
IP-10 levels may also be associated with preeclampsia development. In this study,
significantly upregulated expression levels of IP-10 were observed in placental
tissues of patients with preeclampsia. This finding aligns with the conclusions
of previous study [20], further validating the pathogenic role of IP-10 in
preeclampsia progression. Notably, a seemingly paradoxical finding emerged when
comparing IP-10’s function in cancer and trophoblast cells: knocking down IP-10
effectively promoted the metastasis and growth of HTR-8-SVneo cells and inhibited
their apoptosis, while silencing of IP-10 typically exerts anti-tumor effects in
cancer development, such as inhibiting cancer cell proliferation and migration.
This discrepancy stems from fundamental differences in the regulatory mechanisms
governing cell proliferation and migration in cancer cells versus trophoblast
cells. Cancer cells exhibit unregulated, dysplastic proliferation and invasion
driven by disrupted signaling networks, where IP-10 may act as an “oncogenic
driver” by activating pathways such as CXCR3-NF-
In contrast to IP-10’s pathogenic role, LXA4 activation has been shown to exert anti-cancer effects [32, 33]. Building on these findings, we hypothesized that LXA4 may exert effects antagonistic to IP-10 in preeclampsia. Given that LXA4 is a non-protein mediator and ALX is its primary receptor, we used ALX expression as an indirect surrogate for LXA4 activity. In this study, we indicated that the deficiency of ALX markedly induced HTR-8-SVneo cell apoptosis, while concurrently suppressing the metastasis and growth. These findings supported our hypothesis. Meanwhile, numerous studies have illustrated the inhibitory effects of LXA4 overexpression on the progression of preeclampsia [34, 35], thereby further corroborating our experimental results. Oxidative stress and inflammation are well-recognized pathogenic factors in preeclampsia [36, 37]. Our data showed that IP-10 and LXA4 also exert opposing regulatory effects on these processes. Specifically, silencing IP-10 inhibited oxidative stress and inflammation, while knocking down LXA4 increased both. Collectively, these results confirmed that IP-10 and LXA4 exhibit antagonistic roles in regulating HTR-8-Svneo cell proliferation, migration, and invasion, as well as oxidative stress and inflammation, during preeclampsia progression.
This study has several notable limitations that should be acknowledged. First,
as a pilot study, this research enrolled only 5 patients per group. The small
sample size may limit the generalizability of our clinical findings, and larger
cohorts are needed in future studies to validate these results. Second, the
regulatory mechanisms of IP-10 and LXA4 in HTR-8-Svneo cells—including
potential downstream signaling pathways—remain to be fully elucidated. For
example, IP-10 activates NF-
In a word, this pilot study presents preliminary findings. We have preliminarily characterized the antagonistic effects of IP-10 and LXA4 on HTR-8-SVneo trophoblast cells during preeclampsia progression using in vitro models. Our in vitro data may provide theoretical support for future animal model experiments and clinical data analysis.
The data are available from the corresponding author on reasonable request.
JW made substantial contributions to the conception and design of the work. JW, CF, LQ and YZ made substantial contributions to the acquisition, analysis and interpretation of data for the work. JW drafted the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Ethical approvals were granted by the Ethics Committee of Jinhua People’s Hospital (approval number: 2023-007), and this study complies with the principles set forth in the Declaration of Helsinki. All participants gave their written informed consent.
Not applicable.
This work was supported by Key Science and Technology Projects of Jinhua City in 2023 (Project No: 2023-3-137).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



