1 Clinical Science Department, College of Medicine, Princess Nourah bint Abdulrahman University, 13412 Riyadh, Saudi Arabia
2 Department of Medicine, King Fahad Armed Forces Hospital, 21159 Jeddah, Saudi Arabia
3 Faculty of Medicine, King Abdulaziz University, 21589 Jeddah, Saudi Arabia
4 College of Medicine, King Saud University, 11451 Riyadh, Saudi Arabia
5 College of Medicine, Bishah University, 67714 Bishah, Saudi Arabia
6 College of Medicine and Surgery, Batterjee Medical College, 21442 Jeddah, Saudi Arabia
7 College of Medicine, Umm Al-Qura University, 28821 Al-Qunfudhah, Saudi Arabia
8 College of Medicine, King Saud Bin Abdulaziz University for Health Sciences College of Medicine, 11481 Riyadh, Saudi Arabia
Abstract
Gefitinib is a tyrosine kinase inhibitor of epidermal growth factor receptor (EGFR), mainly used for non-small cell lung cancer. Because EGFR is also highly expressed in placental tissue, its use has been explored in the treatment of ectopic pregnancy. This review examines the available evidence on the safety and effectiveness of combining gefitinib with methotrexate for the treatment of ectopic pregnancy.
Following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a comprehensive search was conducted across PubMed, Web of Science, ProQuest, and Google Scholar for English-language studies published between 2013 and 2023 comparing gefitinib plus methotrexate to methotrexate alone. Study quality was assessed using Joanna Briggs Institute (JBI) tools for quasi-experimental studies and case reports. Meta-analysis was performed using OpenMeta-Analyst with a random-effects model at a 0.05 significance level and 95% confidence intervals (CI).
Of 162 identified studies, five met the inclusion criteria, and three were included in the meta-analysis, comprising 526 participants. The pooled analysis revealed no statistically significant difference in complete resolution rates between the combination therapy group (69.3%) and the methotrexate-alone group (75.5%) (relative risks (RR): 1.004, 95% CI: 0.802–1.257; p = 0.973; I2 = 63.96%). Adverse events were generally mild and self-limiting, with rash (60.8%) and diarrhea (46.5%) being the most common. Serious adverse events were rare (<4%) and occurred at similar rates in both groups.
These findings suggest that while the combination of gefitinib and methotrexate is safe, it does not significantly enhance treatment outcomes compared to methotrexate alone in managing ectopic pregnancy.
The study has been registered on https://www.crd.york.ac.uk/prospero/ (registration number: CRD42024500567; registration link: https://www.crd.york.ac.uk/PROSPERO/view/CRD42024500567).
Keywords
- gefitinib
- methotrexate
- ectopic pregnancy
- resolution
- systematic review
Ectopic pregnancies (EPs), characterized by implantation of the blastocyst
exterior to the uterine endometrium, predominantly occur within the fallopian
tubes. However, implantation can also occur in the ovaries, cervix, or at prior
cesarean section scar sites [1]. Ectopic pregnancies occur in about 1–2% of all
conceptions and are considered a medical emergency because of the risk of tubal
rupture and severe maternal bleeding [2, 3]. Management of tubal EPs encompasses
a spectrum of treatment modalities, including medical therapy, surgical
intervention, and expectant management. Selection of the optimal approach hinges
on several critical factors, such as initial
A 2013 study reported faster declines in serum hCG levels and earlier resolution of tubal ectopic pregnancies in women treated with a combination of gefitinib and methotrexate compared with methotrexate alone [8]. Gefitinib is a tyrosine kinase inhibitor that specifically influences the epidermal growth factor receptor (EGFR) pathway and is predominantly utilized in the treatment of non-small cell lung cancer. It is noteworthy that EGFR is present in much higher levels in placental tissue compared to other types of non-malignant human tissue [9]. Furthermore, preclinical studies have demonstrated that gefitinib enhances the methotrexate-induced regression of pregnancy-like tissue. Notably, the combination of these two agents acts synergistically in trophoblast cells, effectively inhibiting cell growth, blocking EGFR signaling pathways, and promoting apoptosis [10]. Subsequently, in 2015, a follow-up study reported shortened recovery times for patients treated with this pharmacological regimen [11]. Overall, these studies suggest that combining gefitinib with methotrexate may be an effective treatment option.
Nevertheless, the authors of both studies highlight the necessity for additional empirical studies and in-depth research to solidify these preliminary findings. Other researchers have also emphasized the need for larger, well-designed trials to confirm these findings [12]. While studies have examined the combination therapy of gefitinib and methotrexate, to date, no systematic reviews or meta-analyses have been conducted to synthesize the available evidence and directly compare this combination therapy with methotrexate monotherapy. A meta-analysis is crucial for this topic, as it provides a comprehensive and conclusive evaluation of the combined efficacy and safety of gefitinib and methotrexate in treating tubal EPs. This analysis addresses inconsistencies and gaps in current individual studies, informing clinical decision-making and future research.
In response to these gaps, this systematic review and meta-analysis were conducted to evaluate the current evidence. This scholarly effort intends not only to fill the existing research gap but also to inform future research trajectories and refine clinical management strategies. The primary objective of this systematic review and meta-analysis is to thoroughly evaluate the existing research, assessing the safety and efficacy of concurrent gefitinib and methotrexate use in the management of tubal ectopic pregnancies. The inquiry’s primary focus is to determine whether this combination therapy provides superior efficacy to methotrexate monotherapy in resolving the condition, as confirmed through ultrasonographic and hCG monitoring.
If proven more effective, this combination therapy could influence future treatment protocols. A more efficacious medical regimen might lessen the reliance on surgical interventions, which is advantageous given the inherent risks, such as potential injury to internal organs. Developing effective nonsurgical options is especially important in settings with limited surgical resources or for patients who prefer less invasive treatments [4]. Additionally, this approach could help preserve reproductive structures, thereby enhancing or safeguarding fertility outcomes. Furthermore, by reducing the need for additional treatments, such as supplementary methotrexate injections, and potentially decreasing hospitalization periods, healthcare expenditures could be significantly reduced [11]. In light of these potential benefits, this review aims to systematically evaluate and synthesize the existing evidence on the safety and effectiveness of combining gefitinib with methotrexate for the treatment of ectopic pregnancy.
This systematic review and meta-analysis adhered to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting standards [13], and methodological procedures were guided by the Cochrane Handbook throughout the review process [14]. This review is registered on the prospective register of systematic reviews (PROSPERO) database under the ID CRD42024500567.
Studies written in the English language were included based on the following population, intervention, comparison, outcome pico (PICO) criteria: Population (P): ectopic pregnancy patients; Intervention (I): gefitinib and methotrexate; Control (C): methotrexate alone; Outcomes (O): resolution of ectopic pregnancy and adverse events; Study design (S): clinical trials and observational studies. Studies involving additional interventions beyond gefitinib, methotrexate, or standard supportive care were excluded from the analysis. Editorials, letters, or conference abstracts were also excluded.
A comprehensive search was conducted across PubMed, Web of Science, ProQuest, and Google Scholar using the following keywords: (“Ectopic Pregnancy”) AND (“Gefitinib” OR “Iressa” OR “N-(3-Chloro-4-fluorophenyl)-7-methoxy-6-(3-(4-morpholinyl) propoxy)-4-quinazolinamide” OR “ZD1839” OR “ZD 1839”) AND (“Methotrexate” OR “Amethopterin” OR “Mexate”). The search was limited to studies published in English from January 2013 to January 2023. Detailed search strategies and database-specific adaptations are provided in Supplementary File 1.
Study selection was performed in two phases to ensure thorough screening and to reduce the risk of bias. In the first phase, two independent authors screened the titles and abstracts of the retrieved studies using Rayyan [15], a web-based application developed to assist with systematic review screening. Rayyan also facilitated duplicate removal and blinded decision-making. Studies that met the initial inclusion criteria were then subjected to a second phase of full-text screening, which was conducted independently by two authors using Excel. Any discrepancies or conflicts that arose during either phase were resolved through discussion and consensus. If consensus could not be reached, a third author was consulted to make the final decision. A PRISMA flow diagram was used to document the selection process, including reasons for exclusion at each stage.
Data extraction was performed by the authors using formatted Excel sheets. The extracted data from the included studies were categorized into general characteristics, baseline characteristics, and outcomes. General characteristics included study design, country, intervention dosage, inclusion and exclusion criteria, treatment duration, and outcomes. Baseline patient characteristics included age, body mass index (BMI), smoking status, number of previous ectopic pregnancies, conception by Assisted reproductive technologies (ART), and baseline hCG level. The primary outcome was the complete resolution of ectopic pregnancy without surgical intervention, confirmed by ultrasound and hCG monitoring. Secondary outcomes included the need for an additional methotrexate dose, serious adverse events, diarrhea, and rash. For dichotomous outcomes, the number of participants with the event and the total number of participants in each group were recorded. Pilot testing of the data extraction form was conducted to ensure consistency and accuracy of the results.
The quality of the included randomized clinical trials (RCTs) was assessed using the Cochrane Revised Cochrane risk-of-bias tool for randomized trials (RoB 2) tool [16], which evaluates domains such as bias from randomization, deviations from intended interventions, missing outcome data, outcome measurement, and selection of reported results. Judgments were classified as low risk, some concerns, or high risk of bias. For quasi-experimental studies, case series, and case reports, the Joanna Briggs Institute (JBI) critical appraisal tools were used. These tools include a series of questions with responses of ‘yes’, ‘no’, ‘unclear’, and ‘not applicable’, leading to an overall quality score based on the number of ‘yes’ responses. The checklist for Quasi-Experimental studies involves nine items [17]; the checklist for case series involves 10 items [18]; and the checklist for case reports includes eight items.
The analysis was conducted using OpenMeta-Analyst, version 12.11.14 (Tufts
Medical Center, Boston, MA, USA) [19], employing a random-effects model [20] with
a significance level of 0.05 and 95% confidence intervals (CI). For the primary
outcome, risk ratios (RR) were calculated, while untransformed proportions were
calculated for secondary outcomes in a single-arm meta-analysis. Heterogeneity
was assessed using the I2 statistic, with values over 50% and
p
The database search identified 162 articles, 46 of which were duplicates. Title and abstract screening was conducted for 116 articles, and six articles were retrieved for full-text screening. Ultimately, five studies were included in the systematic review [8, 21, 22, 23, 24], and three studies were included in the meta-analysis [8, 22, 24]. The PRISMA flow diagram is shown in Fig. 1.
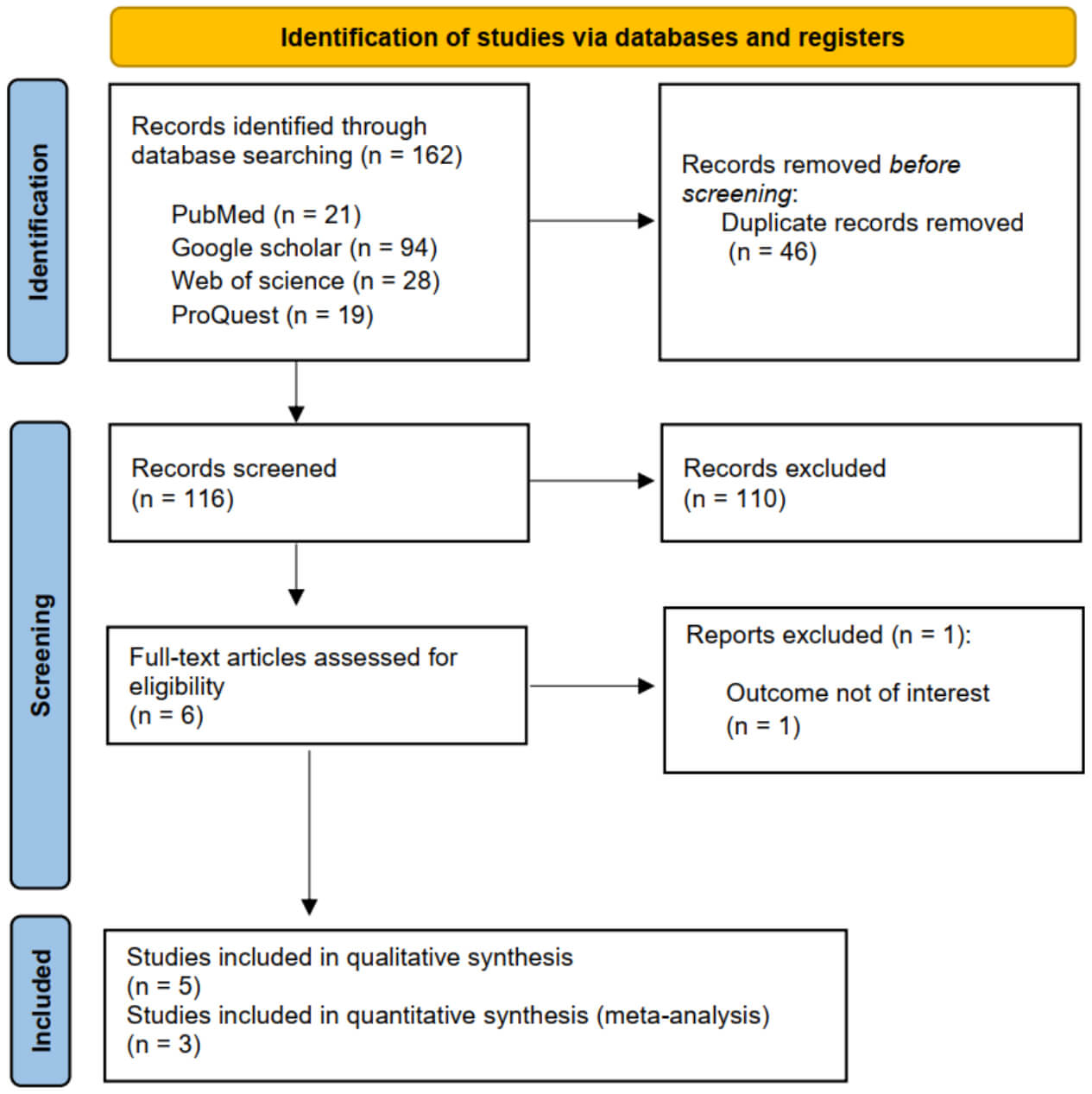 Fig. 1.
Fig. 1.
PRISMA flow diagram. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
The included studies comprised various designs: one randomized controlled trial, two single-arm clinical trials with historical and contemporaneous controls, one case series, and one case report. These studies were conducted in Australia and the United Kingdom, with a combined sample size of 526 participants. Three studies focused on women with tubal EPs, while two addressed non-tubal ectopic cases. Participant mean age ranged from 29.9 to 35.27 years. The proportion of women with a history of EP ranged from 6.5% to 31%, and the median pre-treatment hCG levels varied from 1593 to 7990 IU/L. The characteristics of the included studies are summarized in Supplementary File 2, and baseline participant characteristics are provided in Supplementary File 3.
All included studies were assessed for risk of bias or methodological quality. The randomized controlled trial by Horne et al. (2023) [22] demonstrated a low risk of bias across all domains, as shown in Fig. 2. The two quasi-experimental studies (Skubisz et al., 2013 [8] and Skubisz et al., 2018 [24]) were appraised using the JBI Critical Appraisal Checklist, with results presented in Table 1 (Ref. [8, 24]). Both studies received “Yes” ratings in domains 1, 2, 4, 5, 7, and 8, yielding overall quality scores of seven out of nine, indicating good methodological quality. The case report by Italiano et al. (2020) [23], assessed using the JBI tool for case reports, achieved a perfect score of eight with “Yes” in all domains, as shown in Table 2 (Ref. [23]). The case series by Horne et al. (2014) [21], evaluated with the JBI checklist for case series, received “Yes” in six of ten applicable domains (1, 2, 3, 6, 7, and 8), “Unclear” in domains 4 and 5, and “Not applicable” in domain ten, resulting in a total score of 6, as presented in Table 3 (Ref. [21]).
 Fig. 2.
Fig. 2.
Risk of bias summary and graph.
| Skubisz et al., 2013 [8] | Skubisz et al., 2018 [24] | |
| 1. Is it clear in the study what the ‘cause’ is and what the ‘effect’ is (i.e., there is no confusion about which variable comes first)? | Yes | Yes |
| 2. Were the participants included in any similar comparisons? | Yes | Yes |
| 3. Were the participants included in any comparisons receiving similar treatment/care, other than the exposure or intervention of interest? | Unclear | Yes |
| 4. Was there a control group? | Yes | Yes |
| 5. Were there multiple measurements of the outcome, both pre- and post-intervention/exposure? | Yes | Yes |
| 6. Was the follow-up complete, and if not, were the differences between groups in terms of their follow-up adequately described and analyzed? | Unclear | Unclear |
| 7. Were the outcomes of participants included in any comparisons measured in the same way? | Yes | Yes |
| 8. Were outcomes measured in a reliable way? | Yes | Yes |
| 9. Was an appropriate statistical analysis used? | Yes | Unclear |
| Overall score | 7 | 7 |
JBI, Joanna Briggs Institute.
| Italiano et al., 2020 [23] | |
| 1. Were the patient’s demographic characteristics clearly described? | Yes |
| 2. Was the patient’s history clearly described and presented as a timeline? | Yes |
| 3. Was the current clinical condition of the patient on presentation clearly described? | Yes |
| 4. Were diagnostic tests or assessment methods and the results clearly described? | Yes |
| 5. Was the intervention(s) or treatment procedure(s) clearly described? | Yes |
| 6. Was the post-intervention clinical condition clearly described? | Yes |
| 7. Were adverse events (harms) or unanticipated events identified and described? | Yes |
| 8. Does the case report provide takeaway lessons? | Yes |
| Overall score | 8 |
| Horne et al., 2014 [21] | |
| 1. Were there clear criteria for inclusion in the case series? | Yes |
| 2. Was the condition measured in a standard, reliable way for all participants included in the case series? | Yes |
| 3. Were valid methods used for the identification of the condition for all participants included in the case series? | Yes |
| 4. Did the case series have consecutive inclusion of participants? | Unclear |
| 5. Did the case series have complete inclusion of participants? | Unclear |
| 6. Was there clear reporting of the demographics of the participants in the study? | Yes |
| 7. Was there clear reporting of clinical information of the participants? | Yes |
| 8. Were the outcomes or follow-up results of cases clearly reported? | Yes |
| 9.Was there clear reporting of the presenting site(s)/clinic(s) demographic information? | No |
| 10. Was statistical analysis appropriate? | Not applicable |
| Overall score | 6 |
The syntheses included three studies: one randomized controlled trial and two quasi-experimental designs. Risk of bias among these studies was generally low, as described in Section 3.3. Table 4 (Ref. [8, 21, 22, 23, 24]) summarizes the outcomes of the included studies. Adverse events were reported in all studies involving gefitinib. Common side effects included rash, diarrhea, nausea, dizziness, lethargy, and pruritus, which were generally mild and self-limiting. Serious adverse events were rare, with Horne et al. (2023) [22] reporting 3% in the gefitinib group and 4% in the placebo group. Return to menses was observed in multiple studies. In Horne et al. (2023) [22], the median time was 24 days in both groups. Skubisz et al. (2013) [8] reported prompt return in all participants, while Horne et al. (2014) [21] noted resumption within six weeks. Subsequent pregnancies occurred in three studies. Skubisz et al. (2018) [24] documented seven spontaneous pregnancies, including five intrauterine. Horne et al. (2014) [21] reported three intrauterine pregnancies, and Skubisz et al. (2013) [8] noted one term delivery. Serum hCG levels at days 4 and 7 showed consistent declines with gefitinib plus methotrexate. Skubisz et al. (2013) [8] reported significantly lower levels in the treatment group compared to controls at both time points. The time to resolution of EP varied. The shortest duration was 21 days (Interquartile range (IQR) 17–27) in the gefitinib group (Skubisz et al., 2013 [8]), compared to 32 days (IQR 25–49) in the control group. Other studies reported median times ranging from 28 to 50.5 days.
| Author and year | Adverse events | Return to menses | Subsequent pregnancies | Serum hCG at day four (IU/L) | Serum HCG at day seven (IU/L) | Time to resolution median (IQR) |
| Horne et al., 2023 [22] | Serious adverse events occurred in 3% of participants receiving gefitinib and 4% of those given a placebo. Diarrhea and rash were reported more frequently among participants in the gefitinib group than in the placebo group. | The median time to resumption of menses was identical in both groups: 24.0 days (IQR 24.0–38.0, n = 132) in the gefitinib arm and 24.0 days (IQR 24.0–38.0, n = 134) in the placebo arm. | NR. | NR. | NR. | The median (IQR) time to resolution of the ectopic pregnancy was 28.0 (23.5–36.0, n = 108) days in the gefitinib group and 28.0 (21.0–36.5, n = 108) days in the placebo group. |
| Skubisz et al., 2018 [24] | No severe adverse events were reported, and laboratory assessments showed no abnormalities in renal or hematologic function. Six participants experienced mild, temporary increases in liver enzymes (ALT or AST). Reported side effects included nausea in 15 cases (with vomiting in 2), lethargy in 10, dizziness in eight, and pruritus in seven. | NR. | Seven spontaneous pregnancies were recorded: five resulted in successful intrauterine pregnancies, one ended in miscarriage, and one was a tubal ectopic pregnancy requiring surgical management. | The median (IQR) of hCG level at day four in women receiving gefitinib and methotrexate was: 1627.3 (1200.7–2318.8). | The median (IQR) of HCG level at day seven in women receiving gefitinib and methotrexate was 1307.3 (832.6–1586). | The median ( |
| Skubisz et al., 2013 [8] | Participants most frequently reported mild, short-lived adverse effects such as rash and diarrhea, while no significant pulmonary, renal, hepatic, or hematologic toxicity was observed either clinically or biochemically. | Menstrual cycles resumed promptly in all enrolled participants. | One participant had a spontaneous pregnancy and delivered a healthy neonate at term. | On day 4, women in the gefitinib arm had lower median hCG concentrations 785 (204–2047) compared with those in the control arm 1838 (1500–2649), a difference that reached statistical significance. | By day 7, the median (IQR) hCG levels in combination group 261 (55–1445) were less than one-fifth of the levels observed among participants in the control group 1426 (940–2573). | The median (IQR) time for resolution was 11 days shorter (34%) among trial participants, 21 days (17–27 days) compared with participants in the control group, 32 days (25–49). |
| Horne et al., 2014 [21] | The most common adverse events were rash, diarrhea, and dizziness; these adverse events were either mild or moderate and resolved spontaneously upon completion of treatment. | All participants returned to their menstrual cycles within 6 weeks. | Among the participants, three conceived spontaneously; two carried pregnancies to term, while one delivered preterm. | The median (IQR) of hCG level at day 4 in women receiving gefitinib and methotrexate was 6144 (4821.5–12,901). | The median (IQR) of hCG level at day 7 in women receiving gefitinib and methotrexate was 5656 (3269–11,195). | For women treated with the combination of gefitinib and methotrexate, the median resolution time was 50.5 days (34.5–65.0). |
| Italiano et al., 2020 [23] | The patient treated with gefitinib experienced mild lower abdominal pain and vaginal spotting on day 4, developed diarrhea on day 5, and a facial rash on day 9. | NR. | NR. | NR. | The serum hCG dropped from 19,510 to 15,970 IU/L in the patient who received the combination therapy. | In the combination therapy group, the single treated patient required 42 days for resolution, while in the methotrexate group, the median time to resolution was also 42 days. |
ALT, alanine transaminase; AST, aspartate aminotransferase; IQR, interquartile range; hCG, human chorionic gonadotropin; NR, mot reported.
The pooled data from three studies [8, 22, 24] showed no statistically significant difference in the rate of complete resolution between women receiving a combination of methotrexate and gefitinib (69.3%) compared to women receiving methotrexate alone (75.5%) (RR: 1.004, 95% CI: 0.802–1.257; p = 0.973). The pooled results were heterogeneous (I2 = 63.96%, p = 0.062). After excluding the study by Skubisz et al. [24]. The remaining studies demonstrated homogeneity (I2 = 30.31%, p = 0.231) and continued to show no significant difference between the two groups (RR: 0.923, 95% CI: 0.776–1.098; p = 0.364) (Fig. 3, Ref. [24]).
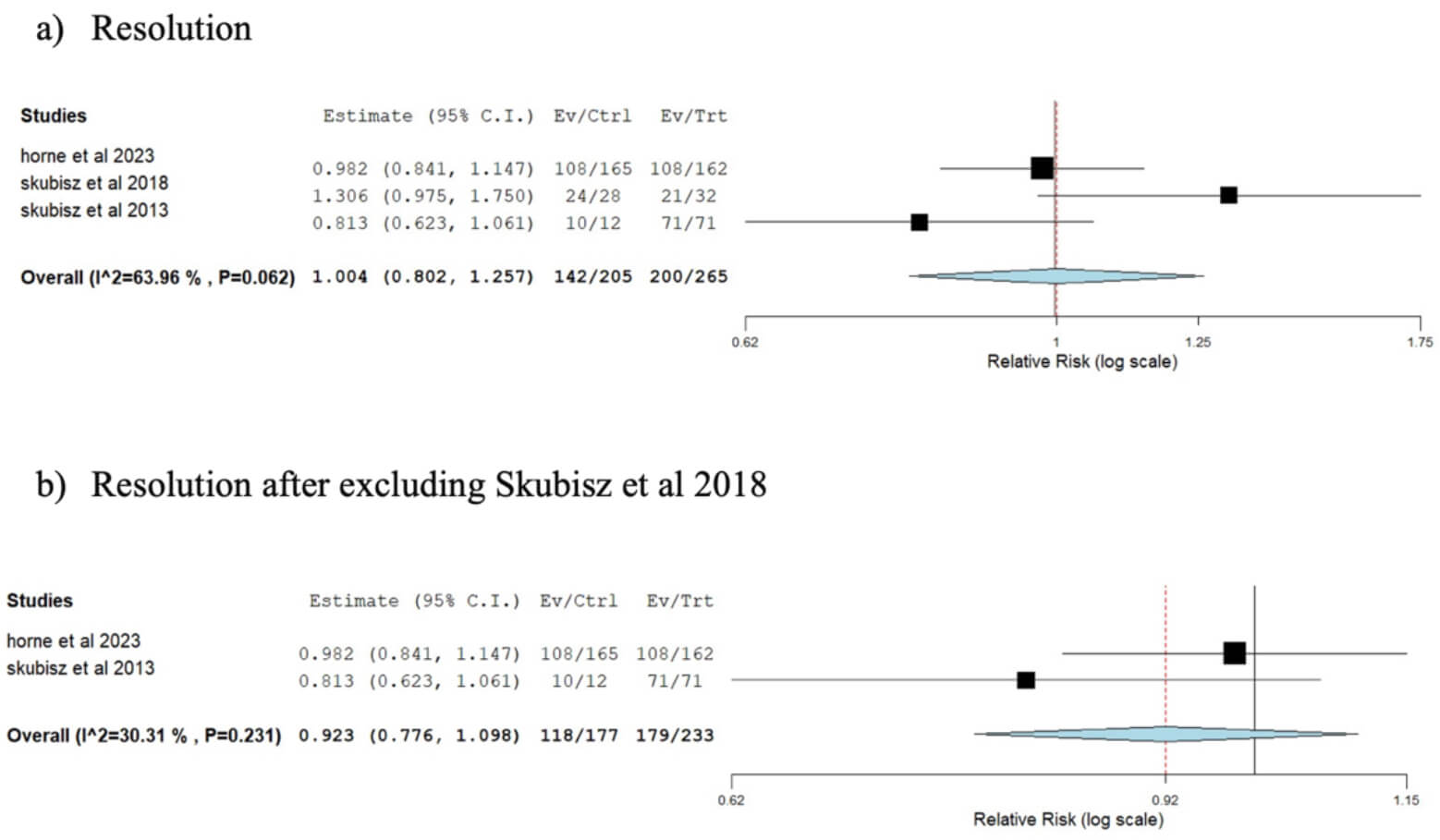 Fig. 3.
Fig. 3.
Forest plot comparing the rate of resolution between the group receiving a combination of gefitinib and methotrexate and the methotrexate-only group. (a) Pooled analysis of all three included studies. (b) Sensitivity analysis after excluding the study by Skubisz et al. [24]. The p-value represents heterogeneity. CI, confidence interval; Ev, Event; Ctrl, control group; Trt, treatment group.
Two studies [8, 24] reported the need for an additional dose of methotrexate
after administering methotrexate and gefitinib. The overall effect estimate
indicated that 11.8% of patients needed an additional dose (95% CI:
7%–16.5%, p
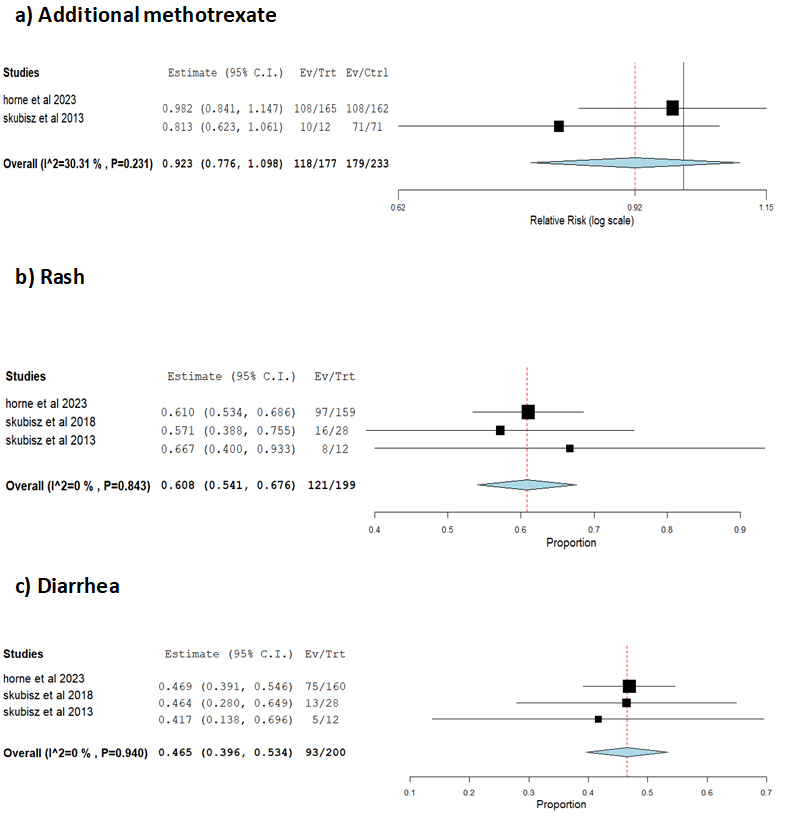 Fig. 4.
Fig. 4.
Forest plot showing the rates of additional methotrexate, rash, and diarrhea following methotrexate and gefitinib administration. (a) Patients requiring an additional methotrexate dose. (b) Incidence of rash. (c) Incidence of diarrhea. The p-value represents heterogeneity.
To our knowledge, this is the first systematic review and meta-analysis investigating the combined effect of methotrexate and gefitinib in EP. In this study, there was no statistically significant difference in the rate of complete resolution between the methotrexate-only group and the methotrexate plus gefitinib group.
Skubisz et al. [8, 24] reported a higher resolution rate in the methotrexate plus gefitinib group compared to the methotrexate only group. A planned subgroup analysis revealed that, in comparison to a historical cohort receiving methotrexate alone, trial participants with pre-treatment serum hCG levels between 1000 and 3000 IU/L experienced a significantly more rapid decline in hCG levels and achieved resolution 34% faster. On the other hand, the study done by Horne et al. [22] observed that the addition of oral gefitinib to parenteral methotrexate did not yield a demonstrable clinical benefit compared to methotrexate alone. Furthermore, their study reported an increase in minor adverse reactions associated with the combination therapy. It is important to note that their study assessed a single dosing regimen, which may have influenced the observed outcomes. This raises the possibility that gefitinib could demonstrate efficacy under an alternative dosing protocol, such as extended-duration administration. Skubisz et al. [24] identified a significant positive correlation between pre-treatment beta-hCG levels and the complete resolution of the condition. Patients who required surgery due to incomplete resolution with medical treatment exhibited significantly higher median hCG levels (3500 IU/L) compared to those achieving complete resolution with medication alone (1922 IU/L). In contrast to the meta-analysis by Solangon et al. [25], which did not identify a statistically significant advantage for methotrexate compared to expectant management in achieving complete resolution of tubal ectopic pregnancy among women with baseline hCG levels below 2000 IU/L. A larger sample size could help determine the accuracy of the Solangon et al. [25] study. However, the limited sample size may result in no discernible change.
A network meta-analysis has compared the efficacy of several medical and surgical management plans in expectant management. They found that no management plan has shown a significant effect in increasing complete resolution rates when compared to expectant management. In addition, they reported higher failure rates for all the methods when compared to salpingectomy [26]. Another meta-analysis has reported that methotrexate is effective in enhancing subsequent pregnancy outcomes when compared to salpingectomy in hemodynamically stable patients. However, no significant difference was reported between methotrexate and salpingostomy or expectant management [27].
This study observed an 11.86% rate of additional methotrexate administration in the methotrexate plus gefitinib group. Interestingly, the meta-analysis by Solangon et al. [25] reported a higher rate of additional methotrexate administration (21.95%) within the methotrexate-only group across two randomized controlled trials. They claimed that the use of additional doses of methotrexate might be associated with a higher efficacy rate for methotrexate as in the van Mello et al. [28] study since they permitted the use of additional doses of methotrexate. On the other hand, Jurkovic et al. [29] did not permit the use of additional methotrexate doses and consequently, their efficacy rates were lower. On the contrary, Skubisz et al. [8] reported a lower rate of additional methotrexate dose administration than Horne et al. [22] (8.33% and 12.12%, respectively). Despite this, Skubisz et al. [8] showed a higher rate of complete resolution than Horne et al. [22] (83.3% vs. 65.45%, respectively).
Regarding the safety of methotrexate and gefitinib in combination, we found that the rate of adverse events was 2.8%. Horne et al. [22] claimed that the serious adverse event was primarily due to methotrexate, and the patient was discharged from the hospital after 24 hours. The most frequently reported adverse effects were rash (60.8%) and diarrhea (46.5%). Diarrhea and acneiform eruption rash are frequent side effects usually associated with gefitinib administration [30].
Several limitations should be considered when interpreting the findings of this systematic review and meta-analysis. First, the number of eligible studies was small, with only five studies meeting the inclusion criteria and three contributing to the meta-analysis, which limits the statistical power and precision of the pooled estimates. Second, substantial heterogeneity was observed for the primary outcome (I2 = 63.96%), suggesting variability in treatment effects across studies. This heterogeneity may be attributed to several factors, including differences in baseline serum hCG levels, which ranged from less than 3000 IU/L in some studies to more than 7000 IU/L in others, a factor known to influence methotrexate efficacy; variation in the type of EP studied, with most studies focusing on tubal cases while others included non-tubal locations; differences in dosing regimens for gefitinib and methotrexate, such as single-dose versus extended administration; and methodological variability arising from the inclusion of both randomized controlled trials and quasi-experimental studies with historical or contemporaneous controls. Finally, although the risk of bias was generally low or moderate, non-randomized designs remain inherently more susceptible to confounding factors. The relatively small patient population analyzed may have also limited the ability to detect clinically meaningful differences; it remains possible that specific subgroups, such as those with high EGFR expression or other baseline characteristics, may derive a greater benefit from the combination regimen. Collectively, these factors necessitate cautious interpretation of the results and underscore the need for larger, well-designed randomized controlled trials, ideally incorporating biomarker profiling, to confirm the efficacy and safety of gefitinib combined with methotrexate in diverse clinical populations.
In conclusion, combining gefitinib with methotrexate shows promise as an alternative medical approach for managing EP. Adverse effects such as rash and diarrhea were more commonly reported with combination therapy but were generally mild and self-limiting. However, based on the current limited evidence, the combination does not demonstrate a statistically significant advantage over methotrexate monotherapy in terms of resolution rates or the need for additional interventions. Overall, the available evidence indicates that although the combination therapy is tolerable, its clinical advantage over methotrexate alone remains unconfirmed. Further large-scale, well-designed randomized controlled trials are necessary to determine whether specific patient subgroups may benefit from this regimen and to confirm its efficacy, cost-effectiveness, and impact on fertility outcomes.
This study is a systematic review and meta-analysis of previously published studies. No new data were generated. All data sources are cited in the reference list.
NA conceived and designed the study. NA, MS, and YA conducted the investigation and MA contributed to data acquisition. HA, RAlh, SA, and RAlj curated the data. NA performed the formal analysis. Validation was carried out by NA, YA, and RS. NA and YA drafted the initial version of the manuscript with contributions from HA, FY, and MS. Critical review and editing were performed by NA, YA, RS, SA, FY, and MA. NA and RS contributed to visualization. MA supervised the project. FY and MA made substantial contributions to the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors extend their sincere gratitude to Dr. Marwa Afify for her invaluable contributions to this study through her expertise in data analysis. Her insightful guidance and analytical prowess were instrumental to the successful execution of this research project.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG43664.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




