1 Reproductive Medicine Department, Zhongda Hospital, Southeast University, 210009 Nanjing, Jiangsu, China
Abstract
Atypical recurrent implantation failure (RIF) poses a challenge for freeze-thawed embryo transfer (FET) as current interventions showing limited efficacy. Autologous platelet-rich plasma (PRP) may improve the pregnancy outcomes, but its value before FET in such patients remains unclear.
A retrospective analysis was conducted on the medical records of patients with atypical RIF (a history of one or two prior failed embryo transfers) who underwent another FET in Reproductive Medicine Center of Zhongda Hospital between January 1, 2022, and June 1, 2024. Patients who received autologous PRP intrauterine infusion before FET were designated as the PRP group (n = 59), while matched patients from the same period who did not receive PRP served as the control group (n = 79). The two groups were compared for endometrial thickness on the day of embryo transfer, biochemical pregnancy rate, embryo implantation rate, clinical pregnancy rate, and early miscarriage rate.
No statistically significant differences were observed in the baseline characteristics between the control and PRP groups (all p > 0.05). The PRP group had a significantly higher biochemical pregnancy rate (66.10% vs. 45.57%), embryo implantation rate (43.75% vs. 30.83%), and clinical pregnancy rate (57.63% vs. 39.24%) compared to the control group (p < 0.05).
For patients with atypical RIF, intrauterine infusion with autologous PRP can increase the embryo implantation and clinical pregnancy rates in subsequent FET cycles.
Keywords
- autologous platelet-rich plasma
- freeze-thawed embryo transfer cycle
- intrauterine infusion
- atypical recurrent implantation failure
- embryo implantation rate
- clinical pregnancy rate
Embryo implantation failure is an important factor leading to pregnancy failure in assisted reproductive technology (ART) [1]. Many factors can contribute to the failure of embryo implantation, such as embryo quality, endometrial receptivity, immune factors, and thrombophilia [2]. Among these, impaired endometrial receptivity accounts for approximately two-thirds of embryo implantation failures [3]. Recurrent implantation failure (RIF) refers to the failure of at least three embryo implantations, involving the transfer of a total of four high-quality embryos, and including both fresh embryo transfer and freeze-thawed embryo transfer (FET) [4]. However, previous research in ART has revealed that the proportion of patients with atypical RIF (history of one or two prior implantation failures) is much higher than that of patients with standard RIF [5]. Due to factors such as insufficient embryo quantity and the financial burden associated with multiple embryo transfers, in clinical practice these patients may not have the opportunity to attempt additional transfers after experiencing three implantation failures. Consequently, a key focus of our research has been to identify effective strategies in atypical RIF patients that enable intervention in endometrial receptivity at an early stage.
Endometrial receptivity has been regarded as a pivotal influencing factor in previous clinical studies on embryo implantation failure, garnering significant attention [6]. Approaches to enhance endometrial receptivity include testing for endometrial receptivity [7], endometrial scratching [8], and intrauterine infusion. Among these methods, intrauterine infusion is widely applied in clinical settings as an effective means of improving endometrial receptivity [9]. A study has demonstrated that intrauterine infusion of substances such as human chorionic gonadotropin (HCG) [10], autologous peripheral blood mononuclear cells (PBMC) [11], granulocyte colony-stimulating factor (G-CSF) [12], and autologous platelet-rich plasma (PRP) [13] can improve embryo implantation and pregnancy outcomes.
Notably, the beneficial effects of autologous PRP intrauterine infusion in RIF
patients have been validated in a study [14]. Autologous PRP is derived
from the patient’s own peripheral blood and is a plasma product enriched with
platelets, proteins, and growth factors. It can promote cell proliferation,
tissue repair, and regeneration, thereby playing a vital role in the regeneration
of various tissues [15]. In the context of the endometrial microenvironment,
intrauterine infusion of autologous PRP can facilitate endometrial development
[16]. For RIF patients complicated by chronic endometritis, PRP can significantly
improve clinical outcomes [17]. For RIF patients with a thin endometrium, PRP can
effectively increase endometrial thickness and increase the rate of clinical
pregnancy [18]. Meanwhile, a study has indicated that PRP is rich in a variety
of growth factors, including platelet-derived growth factor (PDGF), transforming
growth factor-
Therefore, in this study we conducted a retrospective cohort study on atypical RIF patients who received intrauterine infusion of autologous PRP at the Reproductive Center of Zhongda Hospital. Our aim was to investigate whether intrauterine infusion with autologous PRP can improve the clinical outcomes of atypical RIF patients undergoing FET.
We retrospectively analyzed patients (n = 726) receiving FET at the Reproductive Medicine Center of Zhongda Hospital, Affiliated with Southeast University, from January 1, 2022 to June 1, 2024. Among them, 186 cycles received intrauterine infusion with autologous PRP before embryo transfer, and 540 cycles had no intrauterine intervention.
The inclusion criteria were: (1) a history of one or two failed embryo
transfers; (2) age
A total of 138 atypical RIF patients were included: 39 with a history of one
failed embryo transfer, and 99 with a history of two failed embryo transfers.
Among them, 59 patients had received PRP prior to FET (the PRP group), while 79
matched patients received no infusion (no placebo) prior to FET (control group).
Additionally, we reviewed FET cycles of RIF patients who received autologous PRP
infusion during the same period. After applying the above criteria and excluding
patients with
Depending on the patient’s clinical scenario, endometrial preparation for FET was performed via one of the following protocols:
• GnRHa (Gonadotropin-releasing hormone agonist) downregulation hormone
replacement therapy (HRT) cycle: On days 2–4 of the menstrual cycle, 3.75 mg of
leuprorelin acetate (Livzon, H20090299, Shanghai, China) was administered via
subcutaneous injection for pituitary downregulation. After 28–30 days, and once
downregulation was confirmed to be satisfactory, oral estradiol valerate
(Femoston red tablets) was started at 4 mg/day (Abbott Healthcare Products BV,
H20110208, Weesp, Netherlands). One week after estrogen administration,
endometrial thickness was measured by ultrasound, and the estradiol dose was
adjusted accordingly. The maximum duration of estrogen administration was 20
days. When ultrasound indicated the endometrial thickness was • HRT cycle (no downregulation): Femoston (red tablets, 4 mg/day) was started
directly from D2–4 of the menstrual cycle. One week later, endometrial thickness
was measured by ultrasound, and the dose of red tablets was adjusted according to
the endometrial response. Estrogen administration continued for a maximum of 20
days. When ultrasound revealed an endometrial thickness of • Controlled ovarian stimulation (COS) cycle: 2.5–5 mg of letrozole (Hengrui, H19991001, Lianyungang, Jiangsu,
China) was taken orally for 5 days from D4 of the menstrual cycle. Ultrasound was
used to monitor the follicular size, and 75–150 U HMG (Livzon, H10940097,
Shanghai, China) was administered to support follicle growth when necessary. When
the serum estradiol level reached 200–300 pg/mL, or the follicle diameter was
The quality of the embryo was assessed before transplantation. A high-quality,
cleavage-stage embryo was defined as an embryo derived from a normal fertilized
egg, with an embryonic cell count of 7–9 on the 3rd day after fertilization, and
All patients signed the informed consent form for intrauterine infusion of
autologous PRP before receiving treatment. A two-step centrifugation method was
used for PRP preparation as described in a previous report [22]. One or two
rounds of PRP intrauterine infusion were applied depending on the patient’s
individual condition. Two days before embryo transfer (or 4 days if possible), 15
mL of peripheral venous blood from the patient was drawn into a syringe preloaded
with 5 mL of 3.2% sodium citrate anticoagulant solution (NIGALE, H20058913,
Jianyang, Sichuan, China). This was immediately centrifuged at 300
Baseline characteristics for both groups were collected by review of medical
records. These included baseline follicle-stimulating hormone (basal FSH), age,
baseline luteinizing hormone (basal LH), years of infertility, baseline estradiol
2 (basal E2), BMI, baseline progesterone (basal P), AMH, and number of embryos transferred. The clinical outcomes were:
endometrial thickness on the day of embryo transfer; embryo implantation rate =
(number of implanted embryos/number of embryos transferred)
SPSS (version 26.0, IBM, Armonk, NY, USA) was used for data analysis.
Comparisons between two groups were made using Student’s t-test for data
with a normal distribution and homogeneous variance, or the nonparametric
Wilcoxon rank-sum test for data that was not normally distributed. Data that did
not show a normal distribution were represented by the median and interquartile
range [M (P25, P75)]. Categorical data were analyzed using the Chi-square test or
Fisher’s exact test. Logistic regression analysis was performed with clinical
pregnancy as the dependent variable, and potential influencing factors as
covariates. This was used to calculate odds ratios (ORs) with 95% CIs and
p-values. Considering interaction effects, the variables of PRP,
duration of infertility, AMH, and endometrial thickness on transfer day were
included in the final model using the forward selection method. To quantify the
clinical impact of PRP on primary outcomes, the effect size (Cohen’s d) was
calculated using the arcsin transformation method to standardize and present the
actual magnitude of differences between groups. To assess the limitations of
sample size in subgroup analysis, a post-hoc power analysis was conducted on key
outcome indicators (biochemical pregnancy rate and clinical pregnancy rate) using
G*Power (version 3.1.9, Heinrich-Heine-Universität Düsseldorf,
Düsseldorf, North Rhine-Westphalia, Germany). A two-tailed p value
The total number of FET cycles at our center from January 1, 2022 to June 1, 2024 was 726. Of these, 186 cycles included autologous PRP intrauterine infusion before FET, and 540 cycles had no intrauterine intervention. After filtering based on the inclusion and exclusion criteria, 59 cycles remained in the PRP group, and 79 cycles in the control group (Fig. 1).
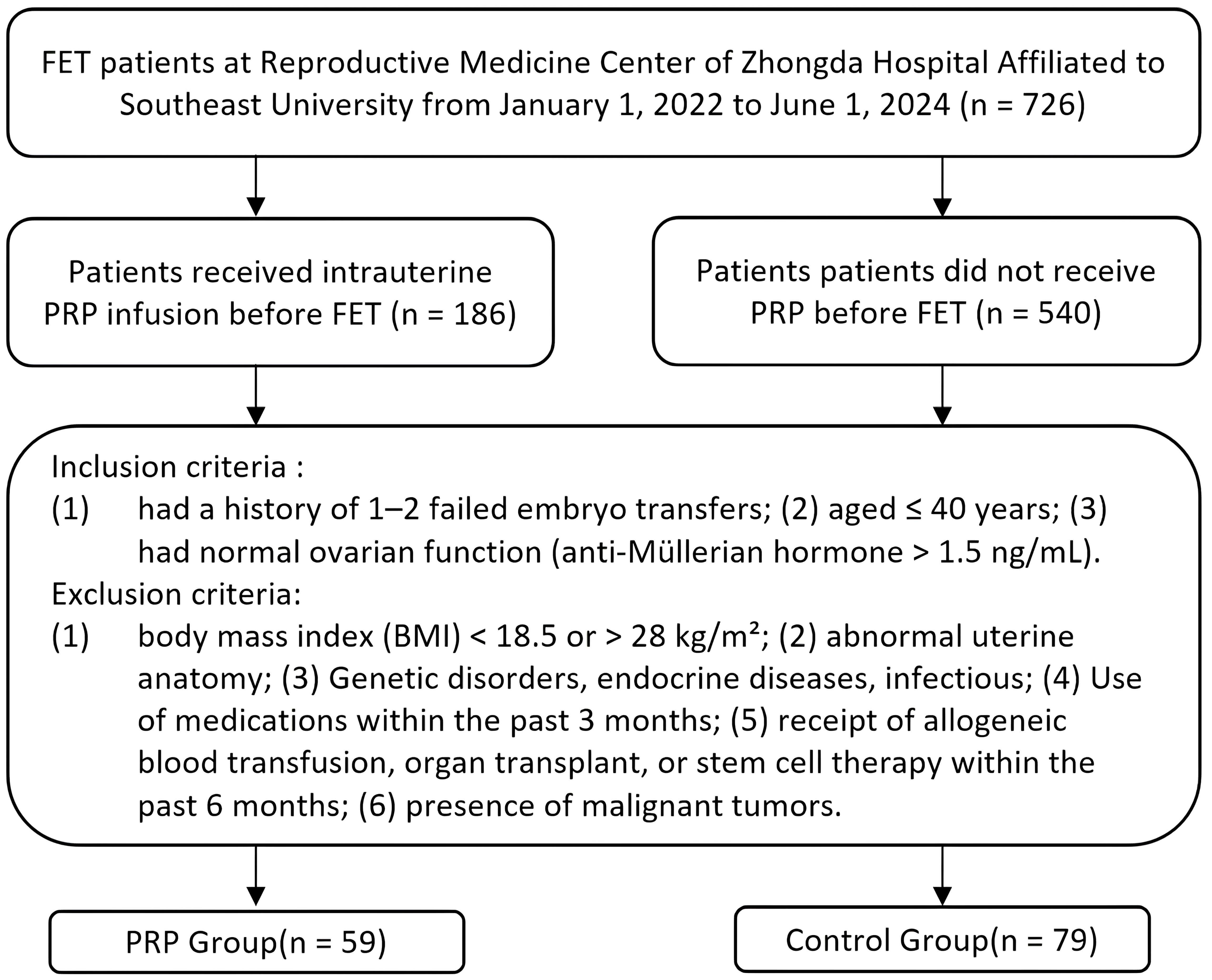 Fig. 1.
Fig. 1.
Flowchart for screening and grouping of patients in freeze-thawed embryo transfer (FET) cycles. PRP, platelet-rich plasma.
No significant differences were detected between the PRP and control groups in
terms of age, duration of infertility, BMI, AMH, basal FSH, basal LH, basal E2,
basal P, embryo transfer number, or high-quality embryo rate (p
| Characteristic | PRP group (59 cycles) | Control group (79 cycles) | p value |
| Age (years) | 32.19 |
31.23 |
0.151 |
| Duration of infertility (years) | 3.90 |
3.95 |
0.925 |
| BMI (kg/m2) | 23.47 |
23.49 |
0.952 |
| AMH (ng/mL) | 5.2 (3.1, 7.5) | 4.6 (2.6, 7.6) | 0.637 |
| Basal FSH (mIU/mL) | 7.0 (5.8, 8.5) | 6.9 (6.1, 8.3) | 0.752 |
| Basal LH (IU/L) | 4.1 (3.2, 6.2) | 4.0 (3.1, 6.0) | 0.836 |
| Basal E2 (pg/mL) | 29.3 (23.0, 35.6) | 32.2 (23.3, 37.3) | 0.541 |
| Basal P (ng/mL) | 0.5 (0.3, 0.7) | 0.5 (0.3, 0.7) | 0.812 |
| Average number of embryos transferred | 1.63 |
1.52 |
0.207 |
| High-quality embryos rate | 57.29% | 65.00% | 0.247 |
AMH, anti-Müllerian hormone; basal FSH, baseline follicle-stimulating hormone; basal LH, baseline luteinizing hormone; basal E2, baseline estradiol 2; basal P, baseline progesterone; BMI, body mass index.
As shown in Table 2, the biochemical pregnancy rate in the PRP group was
significantly higher than in the control group (66.10% vs. 45.57%, p
| Outcome measure | PRP group (59 cycles) | Control group (79 cycles) | p value |
| Endometrial thickness on transfer day (mm) | 10.75 |
11.44 |
0.343 |
| E2 on transfer day (pg/mL) | 456.9 [206.0, 1117.3] | 398.0 [190.3, 802.5] | 0.416 |
| P on transfer day (ng/mL) | 8.5 [5.0, 10.8] | 7.0 [3.4, 10.8] | 0.332 |
| Biochemical pregnancy rate (%) | 66.10% | 45.57% | 0.017 |
| Embryo implantation rate (%) | 43.75% | 30.83% | 0.050 |
| Clinical pregnancy rate (%) | 57.63% | 39.24% | 0.032 |
| Early miscarriage rate (%) | 26.47% | 16.13% | 0.311 |
| Live birth rate | 42.37% | 32.91% | 0.255 |
Embryo implantation rate = (number of implanted embryos/number of embryos
transferred)
Biochemical pregnancy rate = (number of HCG-positive cycles/number of transfer
cycles)
Clinical pregnancy rate = (number of clinical pregnancy cycles/number of
transfer cycles)
Early miscarriage rate = (number of miscarriage cycles/number of clinical
pregnancy cycles)
Live birth rate = (number of successful live births/number of transfer cycles)
The results of logistic regression analysis are shown in Table 3. In univariate analysis, the use of PRP had a positive impact on clinical pregnancy outcomes (OR = 2.25, 95% CI: 1.11–4.60, p = 0.025). Other variables such as the number of embryos transferred and patient age showed no significant effect in the univariate analysis. In multivariate analysis, the OR for PRP showed very wide CIs and this factor was no longer significant for pregnancy outcomes (OR = 92.82, 95% CI: 0.04–243,922.60, p = 0.259). Most variables, such as BMI and basal FSH, were not significantly associated with pregnancy outcomes. In the final model, the adjusted OR for PRP was 2.76 (95% CI: 1.29–5.90, p = 0.009), thus indicating a significant impact.
| Variable | OR univariable | OR multivariable | OR final |
| No PRP | |||
| PRP | 2.25 (1.11–4.60, p = 0.025) | 92.82 (0.04–243,922.60, p = 0.259) | 2.76 (1.29–5.90, p = 0.009) |
| Number of embryos transferred | 1.33 (0.61–2.88, p = 0.469) | 0.88 (0.34–2.31, p = 0.800) | |
| Age | 0.97 (0.88–1.06, p = 0.444) | 1.04 (0.88–1.24, p = 0.639) | |
| Duration of infertility | 0.91 (0.80–1.02, p = 0.115) | 0.89 (0.77–1.03, p = 0.120) | 0.90 (0.79–1.02, p = 0.093) |
| BMI | 0.95 (0.82–1.09, p = 0.462) | 0.95 (0.81–1.11, p = 0.507) | |
| Basal FSH | 0.93 (0.78–1.11, p = 0.403) | 0.96 (0.78–1.17, p = 0.660) | |
| Basal E2 | 1.01 (0.99–1.03, p = 0.460) | 1.01 (0.99–1.03, p = 0.525) | |
| Basal P | 1.06 (0.42–2.71, p = 0.897) | 1.32 (0.44–3.95, p = 0.626) | |
| Basal LH | 1.04 (0.93–1.16, p = 0.517) | 1.02 (0.89–1.17, p = 0.765) | |
| AMH | 1.06 (0.98–1.15, p = 0.154) | 1.16 (0.66–2.06, p = 0.601) | 1.07 (0.98–1.16, p = 0.146) |
| Endometrial thickness on transfer day | 1.11 (0.95–1.30, p = 0.188) | 1.26 (0.99–1.59, p = 0.059) | 1.16 (0.99–1.38, p = 0.074) |
| E2 on transfer day | 1.00 (1.00–1.00, p = 0.843) | 1.00 (1.00–1.00, p = 0.929) | |
| P on transfer day | 1.01 (0.97–1.05, p = 0.628) | 1.01 (0.96–1.06, p = 0.644) | |
| No PRP: Age | 0.94 (0.86–1.04, p = 0.218) | ||
| PRP: Age | 0.97 (0.88–1.06, p = 0.484) | 0.95 (0.77–1.17, p = 0.618) | |
| No PRP: Endometrial thickness | 1.12 (0.95–1.31, p = 0.168) | ||
| PRP: Endometrial thickness | 1.21 (1.01–1.44, p = 0.036) | 0.85 (0.60–1.21, p = 0.376) | |
| Age: AMH | 1.00 (1.00–1.00, p = 0.192) | 1.00 (0.98–1.02, p = 0.713) |
To further analyze the effect of autologous PRP on pregnancy outcomes in
patients with a history of embryo implantation failure, patients were subdivided
into those with a history of one failed embryo transfer (subgroup 1), and those
with a history of two failed embryo transfers (subgroup 2). We then compared
subgroup 1 after PRP treatment (PRP-1) to subgroup 1 with no intervention
(control-1), and subgroup 2 after PRP treatment (PRP-2) to subgroup 2 with no
intervention (control-2). No significant differences were detected between
subgroups 1 and 2 in terms of BMI, age, basal FSH, duration of infertility, basal
LH, basal E2, AMH, basal P, or average number of embryos transferred (p
| Characteristic | Subgroup 1 | Subgroup 2 | ||||
| PRP 1 (29 cycles) | Control 1 (70 cycles) | p value | PRP 2 (30 cycles) | Control 2 (9 cycles) | p value | |
| Age (years) | 32.45 |
31.16 |
0.144 | 31.93 |
31.78 |
0.912 |
| Duration of infertility (years) | 3.86 |
3.91 |
0.942 | 3.93 |
4.22 |
0.807 |
| BMI (kg/m2) | 23.35 |
23.61 |
0.653 | 23.58 |
22.57 |
0.246 |
| AMH (ng/mL) | 5.0 (3.2, 6.6) | 4.8 (2.5, 8.3) | 0.848 | 5.3 (3.1, 9.2) | 4.6 (2.8, 5.0) | 0.243 |
| Basal FSH (mIU/mL) | 7.1 (5.7, 8.3) | 7.0 (6.1, 8.2) | 0.619 | 7.0 (5.9, 8.5) | 6.2 (6.0, 8.6) | 0.671 |
| Basal LH (IU/L) | 4.0 (3.2, 6.2) | 4.1 (3.2, 6.6) | 0.781 | 4.1 (3.2, 6.2) | 3.2 (1.9, 3.5) | 0.147 |
| Basal E2 (pg/mL) | 28.3 (22.8, 36.2) | 32.5 (24.9, 37.8) | 0.314 | 30.8 (23.5, 35.6) | 23.2 (17.2, 34.1) | 0.190 |
| Basal P (ng/mL) | 0.5 (0.3, 0.6) | 0.5 (0.3, 0.7) | 0.229 | 0.5 (0.4, 0.8) | 0.5 (0.4, 0.6) | 0.505 |
| Number of embryos transferred | 1.59 |
1.50 |
0.440 | 1.67 |
1.67 |
1.000 |
Subgroup 1: patients with a history of one failed embryo transfer cycle.
Subgroup 2: patients with a history of two failed embryo transfer cycles.
As shown in Table 5, the biochemical pregnancy rate in PRP-1 of subgroup 1 was
significantly higher than in control-1 (68.97% vs. 47.14%, respectively,
p
| Outcome Measure | Subgroup 1 | Subgroup 2 | ||||
| PRP 1 (29 cycles) | Control 1 (70 cycles) | p value | PRP 2 (30 cycles) | Control 2 (9 cycles) | p value | |
| Endometrial thickness on transfer day (mm) | 10.66 |
11.51 |
0.123 | 10.85 |
10.91 |
0.934 |
| E2 on transfer day (pg/mL) | 353.5 (190.5, 846.0) | 367.9 (190.2, 760.5) | 0.717 | 367.9 (190.2, 760.5) | 1233.4 (209.1, 1329.7) | 0.484 |
| P on transfer day (ng/mL) | 7.3 (4.6, 10.8) | 6.5 (3.0, 9.6) | 0.329 | 6.5 (3.0, 9.6) | 13.6 (7.4, 16.3) | 0.129 |
| Biochemical pregnancy rate (%) | 68.97% | 47.14% | 0.048 | 63.33% | 33.33% | 0.142 |
| Embryo implantation rate (%) | 43.90% | 25.83% | 0.357 | 53.33% | 25.83% | 0.084 |
| Clinical pregnancy rate (%) | 55.17% | 40.00% | 0.167 | 60.00% | 33.33% | 0.255 |
| Early miscarriage rate (%) | 37.50% | 14.29% | 0.133 | 16.67% | 33.33% | 0.489 |
The biochemical pregnancy rate, embryo implantation rate and clinical pregnancy rate in subgroup 1 were analyzed using the chi-square test.
The biochemical pregnancy rate, embryo implantation rate and clinical pregnancy rate in subgroup 2, and the early miscarriage rate in both subgroups, were analyzed using Fisher’s exact test.
The development of ART has assisted many infertile patients. However, the
failure of embryo transfer remains a major issue for medical professionals and
patients. A previous study has mostly focused on improving the clinical outcomes
of RIF patients. Nevertheless, previous data indicate that embryo implantation
rates and clinical outcomes decrease significantly according to the number of
previous transfer failures [23]. Therefore, in the current study we implemented
early intervention for atypical RIF patients to minimize the number of repeated
cycles as much as possible. PRP has recently found several applications in the
field of reproductive medicine. Due to its cost-effectiveness and easy
accessibility, it is used to treat reproductive system disorders such as
Diminished Ovarian Reserve (DOR), Premature Ovarian Insufficiency (POI), and thin
endometrium [24]. When the number of previous implantation failures in patients
is
The embryo implantation rate, biochemical pregnancy rate and clinical pregnancy rate of atypical RIF patients in the PRP group were found to be significantly higher than in the control group. The effect size for biochemical pregnancy rate and clinical pregnancy rate both showed moderate impact (Cohen’s d of 0.45 and 0.40, respectively), and both outcomes were significantly better with PRP (p = 0.024 and p = 0.032, respectively). These findings indicate that PRP can lead to clinically meaningful improvements in the positive rate of early pregnancy biochemical markers, suggesting potential value in the initial stage of embryo implantation. Additionally, we also observed a 10% absolute increase in the live birth rate in the PRP group (35% vs. 25%). Although this did not reach statistical significance due to the relatively small sample size, the magnitude of the improvement suggests that PRP has clinical value, especially since the 95% CI ranged up to 23%.
We observed that early miscarriage rates in both the control and PRP groups were relatively high in this study, at 16.1% (5/31) and 26.5% (9/34), respectively. A previous study has shown that conditions such as chronic endometritis, adenomyosis, and endometriosis can lead to RIF and may increase the risk of early miscarriage [26]. A recent endometrial transcriptomic study revealed a large overlap in abnormally expressed gene profiles between women with RIF and those with recurrent early pregnancy loss [27]. In order to identify possible factors that influence the high rate of early miscarriage in our study, we first analyzed whether the three endometrial preparation protocols could potentially impact treatment outcomes. The use of the three pretreatment methods (GnRHa downregulation HRT, HRT, COS) between the two groups was 54.24%, 30.51%, and 15.25%, respectively, for the PRP group, and 50.63%, 36.71%, and 12.66%, respectively for the control group. There was no significant difference in the proportion of pretreatment methods used between the two groups. However, a study has shown that the natural cycle endometrial preparation protocol may be more effective at reducing the miscarriage rate in RIF patients compared with the HRT protocol [28]. This could be one of the factors contributing to the relatively high miscarriage rate. Second, we analyzed the potential impact of embryo quality. Since preimplantation genetic screening (PGS) was not performed on the embryos in this study, we cannot completely rule out the possibility of aneuploid embryos, which could be another contributing factor to the relatively high miscarriage rate. We next analyzed the data for the 14 miscarriage cycles. The overall PRP and control groups showed no significant differences in high-quality embryos (57% vs. 65%, respectively) and average number of transferred embryos (1.63 vs. 1.52). However, amongst the 14 miscarriage cycles, the PRP group had fewer high-quality embryos (54.55% vs. 87.5%) and average number of transferred embryos (1.22 vs. 1.60) compared to the control group. This result suggests that embryo quality factors may have been an important cause of early miscarriage in the current study. A recent study also found that the number of high-quality embryos is an independent prognostic factor for early miscarriage [29]. Finally, the potential risks of PRP cannot be ignored completely, with larger studies needed to properly evaluate the safety profile of PRP treatment.
Univariate logistic regression analysis revealed that PRP had some impact on clinical pregnancy outcomes. In multivariate analysis, the OR for PRP was very unstable, but the adjusted OR for PRP in the final model again showed a significant impact. A possible reason for this is that after variable screening and adjustment, some confounding factors were excluded, thus allowing a more accurate reflection of the effect of PRP on clinical pregnancy. Variables such as the duration of infertility and AMH also showed near-significant results in the final model, suggesting they could have some impact on clinical pregnancy outcomes when all relevant factors are comprehensively considered.
After further subgrouping of patients, the biochemical pregnancy rate of subgroup 1 (patients with a history of one failed embryo transfer) was found to be significantly higher than that of subgroup 2, with a history of two failed transfers (68.97% vs. 47.14%, respectively, p = 0.048). This result indicates that PRP can improve the clinical outcomes of ART even in women with a history of only one failed embryo transfer. Based on the results before subgrouping in the current study, and together with the use of PRP in RIF patients in previous studies, it appears that PRP not only benefits standard RIF patients, but may also have significant benefit for atypical RIF patients. These findings support early PRP intervention in patients with a history of only one failed embryo transfer in future clinical practice.
Despite not reaching statistical significance, the substantial improvements seen
in subgroup 2 with PRP compared to controls (clinical pregnancy rate: 60.00% vs.
33.33%; embryo implantation rate: 53.33% vs. 25.83%) represent clinically
meaningful differences. In order to clarify whether the non-statistical
significance was due to the small sample size of subgroup 2, we conducted a
post-hoc power analysis of these key indicators. For the biochemical pregnancy
rate, with Cohen’s h = 0.48 and
Several limitations to this study should be acknowledged. Firstly, as a retrospective study, it relied on existing data records. Due to the incompleteness of data, some indicators that may have affected the conclusions (e.g., endometrial blood flow, PRP-related growth factor levels, and differences in chromosomal status) could not be included in the statistical analysis. In subsequent studies, we will incorporate more complete data derived from a larger sample size in order to increase the reliability of conclusions. Secondly, there may have been some uncontrollable factors, such as individual variations in ovarian stimulation protocols and endometrial preparation protocols. When designing subsequent studies, researchers should strive to ensure these factors remain consistent, thereby reducing their potential impact on the results and conclusions. Thirdly, long-term indicators such as obstetric outcomes or neonatal outcomes were not included in the study. This limitation prevented us from tracking the longer-term effects and safety of PRP treatment.
In summary, intrauterine infusion with autologous PRP during FET cycles in atypical RIF patients may help to increase the biochemical pregnancy rate, embryo implantation rate, and clinical pregnancy rate.
The data analyzed during the current study are not publicly available due to the confidentiality factors of patient information, but are available from the corresponding author on reasonable request.
YL designed this study, JS conducted the study, JL collected data for the study, and FY and LL analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and was approved by the Ethics Committee of Zhongda Hospital Affiliated with Southeast University (2024ZDSYLL486-P01). Informed patient consent was waived, as the study was retrospective in nature and analyzed patient data anonymously.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

