1 Department of Surgery, Yongkang Hospital of Traditional Chinese Medicine, 321300 Jinhua, Zhejiang, China
2 The First Clinical College, Zhejiang Chinese Medical University, 310053 Hangzhou, Zhejiang, China
3 Department of Medical Oncology, The First Affiliated Hospital of Zhejiang Chinese Medical University, 310060 Hangzhou, Zhejiang, China
4 Department of Breast Surgery, The First Affiliated Hospital of Zhejiang Chinese Medical University, 310060 Hangzhou, Zhejiang, China
Abstract
To evaluate the efficacy of immunochemotherapy in advanced triple-negative breast cancer (aTNBC) or metastatic triple-negative breast cancer (mTNBC) by assessing overall survival (OS) and progression-free survival (PFS).
Randomized controlled trials (RCTs) of immunochemotherapy in aTNBC or mTNBC were identified through a systematic literature search from different databases. The primary endpoint included OS and PFS. Grade 3/4 adverse events were included in the toxicity analysis, with 95% confidence intervals (CIs) retrieved into the meta-analysis for hazard ratios (HRs).
A total of 7 publications with 3287 patients with aTNBC or mTNBC were enrolled. In the programmed death ligand 1 (PD-L1)-positive aTNBC or mTNBC population, immunochemotherapy was associated with significantly improved PFS than chemotherapy alone ([hazard ratio] HR = 0.84; 95% CI: 0.78–0.91; p < 0.0001). In the intention-to-treat population, immunotherapy effectively prolonged PFS in aTNBC or mTNBC patients (HR = 0.91; 95% CI = 0.88–0.94; p < 0.00001), and OS benefits were limited to combined positive score (CPS) ≥10/20 subgroups. Although immunochemotherapy was found to have some efficacy on PD-L1-positive patients, the improvement in OS was not statistically significant in either population (HR = 0.93; 95% CI = 0.82–1.05; p = 0.24; HR = 0.96; 95% CI = 0.92–1.01; p = 0.09). Regarding adverse events, immunochemotherapy was not associated with a significantly different risk compared to placebo or chemotherapy alone (HR = 0.91; 95% CI = 0.43–1.92; p = 0.73).
PD-L1 inhibitors prolong PFS in PD-L1-positive patients, with a greater effect observed in those with higher CPS.
The study has been registered on https://www.crd.york.ac.uk/prospero/ (registration number: CRD420251067972; registration link: https://www.crd.york.ac.uk/PROSPERO/view/CRD420251067972).
Keywords
- immunochemotherapy
- programmed death ligand 1
- survival
- triple-negative breast cancer
As of 2020, breast cancer was the most frequently diagnosed cancer worldwide, representing 30% of all cancers in women [1]. Since 2014, the incidence rate has been rising at an average annual rate of 0.5% [2]. Triple-negative breast cancer (TNBC) is defined by the lack of expression of human epidermal growth factor receptor 2 (HER2), progesterone receptor (PR), and estrogen receptor (ER), accounting for approximately 24% of all breast cancer cases. Compared to other breast cancer subtypes, TNBC is more common in younger women and is usually accompanied by a breast cancer (BRCA) gene mutation. It is characterized by poor differentiation, high invasiveness, early recurrence, and greater susceptibility to recurrence and metastasis [3]. The five-year survival rate is approximately 85% when diagnosed at an early stage, but drops to 12.2% if detected at an advanced stage [4]. TNBC is unresponsive to endocrine or molecular targeted therapies due to a particular molecular profile [5]. Traditional endocrine therapies (e.g., tamoxifen or aromatase inhibitors) target the ER/PR signaling pathways, whereas HER2-targeted drugs (e.g., trastuzumab) are ineffective in treating TNBC [6, 7]. Most currently available targeted therapies for breast cancer focus on HER2 or hormone receptor pathways, but TNBC does not benefit from these treatments [8, 9]. Chemotherapy is frequently used in clinical settings to extend patient survival, with combined treatment outperforming single-agent chemotherapy [10]. However, combination chemotherapy is associated with increased toxicity, highlighting the need for novel therapeutic options for TNBC.
Currently, immunotherapy mainly focuses on immune checkpoint inhibitors (ICIs), such as programmed death receptor 1 (PD-1) and programmed death ligand 1 (PD-L1) inhibitors. PD-1 is an immunosuppressive receptor that, when targeting its ligand PD-L1, leads to immunization against cancers [11]. Immunotherapeutic agents have been demonstrated to modulate this apoptotic process, especially when combined with chemotherapy, in patients with PD-L1 expression [12]. The effect of durvalumab on advanced breast cancer is still under investigation. However, the US Food and Drug Administration (FDA) approved atezolizumab and pembrolizumab for PD-L1-positive, locally metastatic TNBC (mTNBC) in 2019 and 2020, respectively [13, 14]. For patients with mTNBC, immunotherapy holds significant therapeutic value by prolonging survival. PD-1 inhibitors are currently recognized to have a therapeutic impact on the survival of TNBC patients. As such, we conducted a meta-analysis of immunotherapy combined with chemotherapy, comparing therapeutic effects based on overall survival (OS) and progression-free survival (PFS) in intention-to-treat (ITT) and PD-L1-positive populations, selected from randomized controlled trials (RCTs).
This study was previously registered with PROSPERO (CRD420251067972) and followed PRISMA guidelines [15].
The PubMed, Embase, Medline, and Cochrane Library databases were searched for relevant studies published between 1 January 2010, and 1 July 2023. In PubMed, the following search strategy was employed: (“triple-negative breast cancer” [MeSH Term]) AND (“immunotherapy” OR “PD-1 inhibitor” OR “PD-L1 inhibitor” OR “anti-PD-1” OR “anti-PDL-1” OR “atezolizumab” OR “durvalumab” OR “pembrolizumab” OR “immune checkpoint inhibitor”). In Embase, the term(s): “PD-1 inhibitor” OR “PD-L1 inhibitor” OR “anti-PD-1” OR “anti-PDL-1” OR “atezolizumab” OR “durvalumab” OR “pembrolizumab” OR “immune checkpoint inhibitors”; Year(s): 2010–2023; Title, abstract or author-specified keywords: immunotherapy; Title: “triple-negative breast cancer”, were employed. In Medline, the following search method was employed: (TS = (triple-negative breast cancer)) AND (TS = (immune checkpoint inhibitors)) AND (TS = (atezolizumab)) OR (TS = (pembrolizumab)) OR (TS = (durvalumab)), Year(s): 2010–2023. The Cochrane Library employed: (“triple-negative breast cancer” in Record Title) AND (“immunotherapy” in All Text OR “PD-L1 inhibitor” in All Text OR “PD-1 inhibitor” in All Text - in Trials).
The inclusion criteria for the literature were as follows: (1) availability of full text and raw data from publicly accessible RCTs; (2) patients diagnosed with mTNBC or advance TNBC (aTNBC); (3) immunotherapy, such as PD-1 or PD-L1 inhibitors, was employed as the primary intervention, and the observation group’s interventions included chemotherapy alone or in combination with a placebo; and (4) for the analysis of OS and PFS feasibility studies, hazard ratios (HRs) and 95% confidence intervals (CIs) were utilized. The exclusion criteria were: (1) the literature’s content is irrelevant to TNBC; (2) treatment in the included literature is inconsistent with immunotherapy; (3) literature without full text or raw data; and (4) non-RCT literature, and republished articles or reviews.
The quality of the selected studies was assessed using the Newcastle-Ottawa scale. The results were obtained through the selection of case and control groups, comparability, and the selected articles were rated. Scores ranged from 0 to 9 points, with studies scoring 7 points or more were considered high-quality studies. Since no more than 10 articles were included, a formal bias analysis was not conducted [16].
Data were collected from the included studies by two unbiased reviewers (ML and
YZ). Disagreements were resolved through consensus between the reviewers. The
primary data extracted included the study name, study phase, number of TNBC
patients enrolled, duration of follow-up, pharmacological intervention, PD-L1
status, and adverse events graded as grade three or higher. For OS and PFS, HRs
and 95% CI were calculated, with PD-L1 positivity defined as a combined positive
score (CPS) of
For the ITT population receiving PD-1/PD-L1 inhibitors in addition to chemotherapy, as well as the PD-L1-positive population, OS and PFS were the primary endpoints. The secondary endpoint was the frequency of adverse events graded as grade three or higher. CPS was used to perform subgroup analysis.
The Cochrane Collaboration RevMan 5.4 (The Cochrane Collaboration, The Nordic
Cochrane Centre, Copenhagen, Denmark) was employed. Odds ratios and HRs were used
for the analysis of primary endpoints. The
A total of 3007 papers was obtained after searching PubMed, Embase, Medline, and The Cochrane Library. After initial data screening, 62 studies were initially selected for inclusion. After reviewing the full texts, 9 papers were excluded based on the non-RCT inclusion and exclusion criteria, 16 papers had irrelevant outcome indicators, 4 papers were post-hoc analyses or early reports, full-text data for 9 papers were single arm studies, and 17 papers were neoadjuvant chemotherapy. 7 papers were finally included [18, 19, 20, 21, 22, 23, 24]. The data collection process is depicted in Fig. 1.
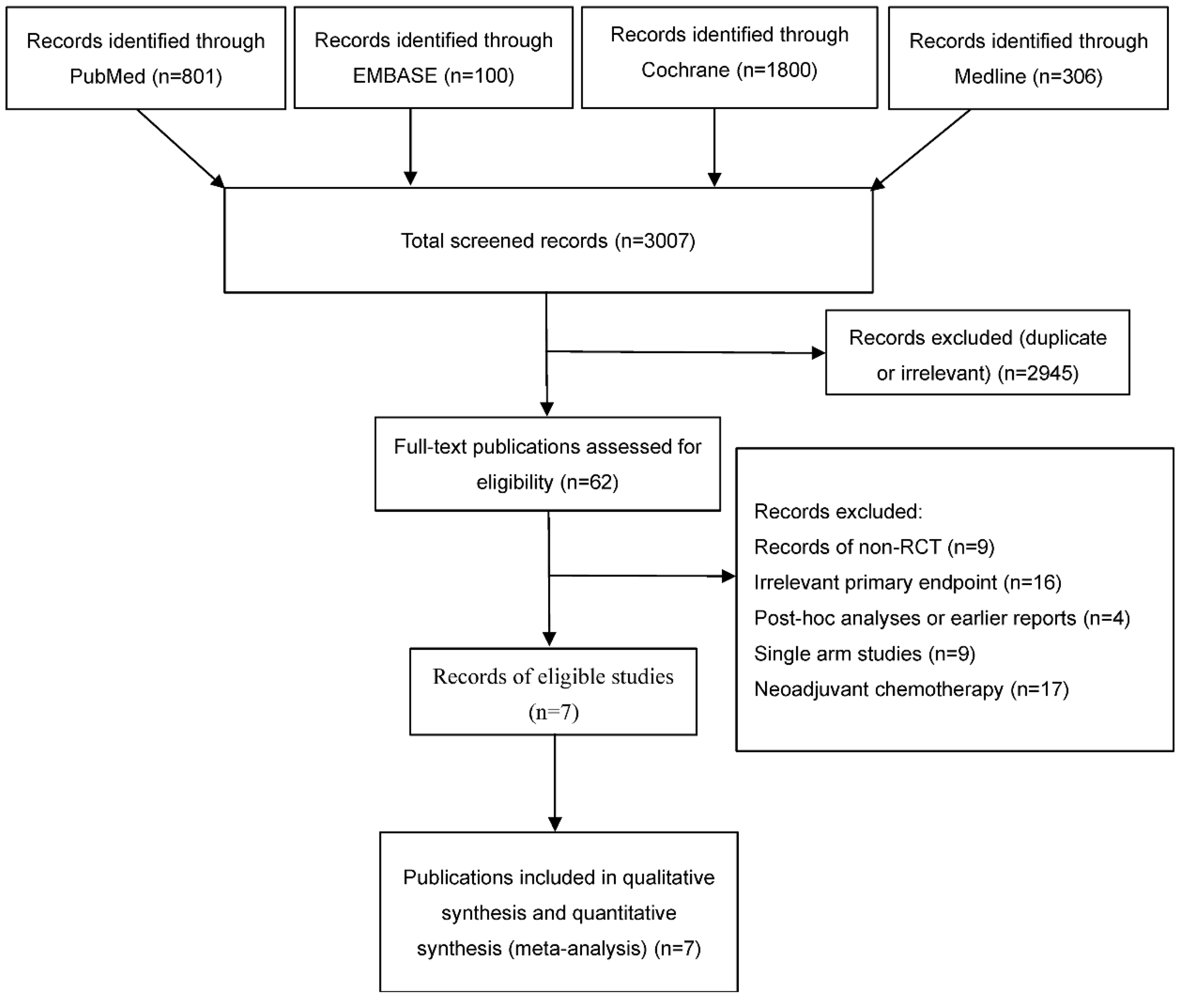 Fig. 1.
Fig. 1.
Flow chart of literature screening. RCT, randomized controlled trial.
All these studies were either retrospective or prospective cohort studies published from 2020 to 2022. A total of 3287 TNBC patients were included across studies, with 1932 patients in the trial group receiving immunotherapy combined with chemotherapy, and 1355 patients in the control group receiving chemotherapy alone, with or without placebo. The general data of the included studies are summarized in Table 1 (Ref. [18, 19, 20, 21, 22, 23, 24]). Table 2 (Ref. [18, 19, 20, 21, 22, 23, 24]) shows the primary endpoints of the included studies. The quality of the included articles was assessed according to the Newcastle-Ottawa scale: 2 articles were classified as medium quality (4–6 points) and 5 articles were classified as high quality (7–9 points).
| Trial | Trial alias | Phase | Study arm (n) | Control arm (n) | Median follow-up (month) | PD-L1-positive (n) | Grade |
| Ref. [18] | IMpassion130 | III | Atezolizumab plus Nab-Paclitaxel (451) | Nab-Paclitaxel plus placebo (451) | 13.0/12.5 | 185.0/184.0 | 230.0/190.0 |
| Ref. [19] | IMpassion131 | III | Atezolizumab plus paclitaxel (432) | Placebo plus paclitaxel (217) | 9.0/8.6 | 191.0/101.0 | 49.0/11.0 |
| Ref. [20] | SAFIR02-BREAST IMMUNO | II | Durvalumab plus chemotherapy (131) | Maintenance chemotherapy (68) | 19.7 | 18.0/14.0 | / |
| Ref. [21] | KEYNOTE-119 | III | Pembrolizumab plus chemotherapy (312) | Chemotherapy (310) | 31.4/31.5 | 352.0 | 60.0/199.0 |
| Ref. [22] | IMpassion130 | III | Atezolizumab plus Nab-Paclitaxel (451) | Nab-Paclitaxel plus placebo (451) | 21.0/18.7 | 185.0/184.0 | 239.0/186.0 |
| Ref. [23] | KEYNOTE-355 | III | Pembrolizumab plus chemotherapy (566) | Placebo plus chemotherapy (281) | 25.9/26.3 | 645.0/314.0 | 383.0/181.0 |
| Ref. [24] | ALICE | IIb | Atezolizumab plus chemotherapy (40) | Placebo plus chemotherapy (28) | 32.2 | 36.0/23.0 | 25.0/12.0 |
AEs, adverse events; PD-L1, programmed cell death ligand 1. “/” means not reported
| Trial (I + C/C (m)) | ITT OS | ITT PFS | PD-L1-positive OS | PD-L1-positive PFS | CPS |
CPS |
CPS |
| Ref. [18] | 21.0/18.7 | 7.2/5.5 | 25.0/18.0 | 7.5/5.3 | / | / | / |
| Ref. [19] | 19.2/22.8 | 5.9/5.6 | 22.1/28.3 | 7.2/6.4 | / | / | / |
| Ref. [20] | 21.2/14.0 | / | 27.3/12.1 | / | / | / | / |
| Ref. [21] | 9.9/10.8 | / | / | / | OS 10.7/10.2 | OS 12.7/11.6 | OS 14.9/12.5 |
| Ref. [22] | 21.0/18.7 | / | 25.4/17.9 | / | / | / | / |
| Ref. [23] | 17.2/15.5 | 7.5/5.6 | / | / | OS 17.6/16.0 | OS 23.0/16.1 | / |
| PFS 7.6/5.6 | PFS 9.7/5.6 | ||||||
| Ref. [24] | Not mentioned | 4.3/3.5 | / | 4.7/1.8 | / | / | / |
CPS, combined positive score; I+C/C, immunochemotherapy + chemotherapy/chemotherapy; ITT, intention-to-treat; OS, overall survival; PFS, progression-free survival. “/” means not reported.
Six trials comparing the OS of immunotherapy between chemotherapy and
chemotherapy plus placebo were included. Overall, there was significant
heterogeneity between the two groups regarding ITT patients ([hazard ratio] HR,
0.96; 95% CI, 0.94–0.98; p
For further analysis, we performed subgroup analysis of OS (Fig. 2). Subgroup
analysis by chemotherapy regimen showed that a combined HR of 0.96 (95% CI:
0.92–1.01, p = 0.09, I2 = 37%, p for
heterogeneity = 0.20) for the paclitaxel group, and 0.96 (95% CI: 0.90–1.03,
p = 0.25, I2 = 51%, p for heterogeneity = 0.13)
for the non-paclitaxel group. Subgroup analysis by sample size showed a combined
HR of 0.98 (95% CI: 0.94–1.03, p = 0.46, I2 = 17%,
p for heterogeneity = 0.30) for studies with sample size
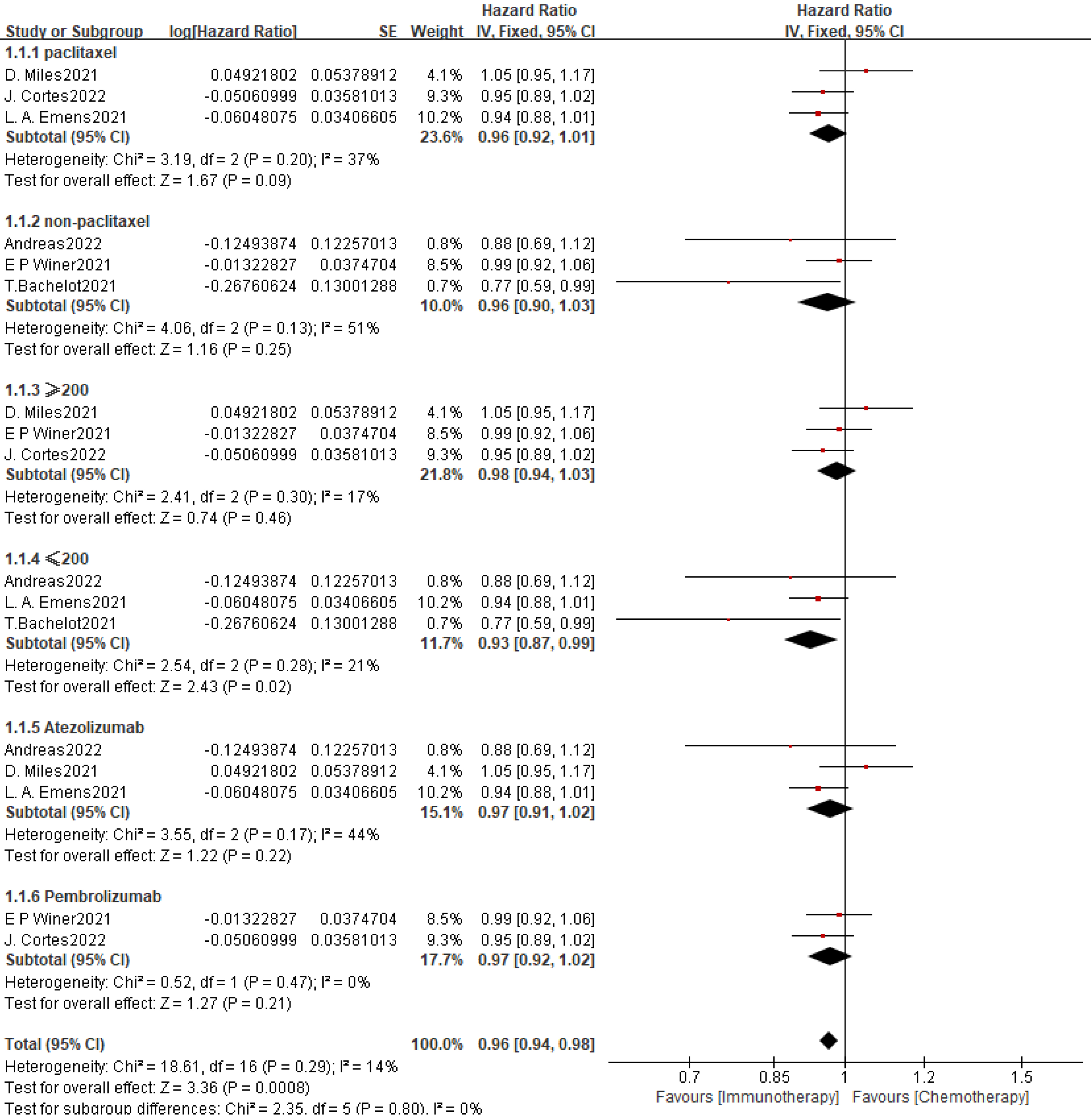 Fig. 2.
Fig. 2.
Summary and subgroup analysis of effects of PD-L1 inhibitors in
combination with chemotherapy on OS in the ITT population of mTNBC patients.
Subgroup analyses were performed according to chemotherapy regimen (paclitaxel
vs. non-paclitaxel), sample size (
In the PD-L1-positive subgroup, the pooled HR for OS was 0.93 (95% CI: 0.82–1.05, p = 0.24), indicating no statistically significant survival advantage of immunotherapy over chemotherapy. However, there was significant heterogeneity among the included studies (I2 = 70%, pheterogeneity = 0.02), and a random-effect model was applied.
For further analysis, we did subgroup analysis of OS (Fig. 3). A total of three
subgroups were included to compare the OS of mTNBC patients with CPS
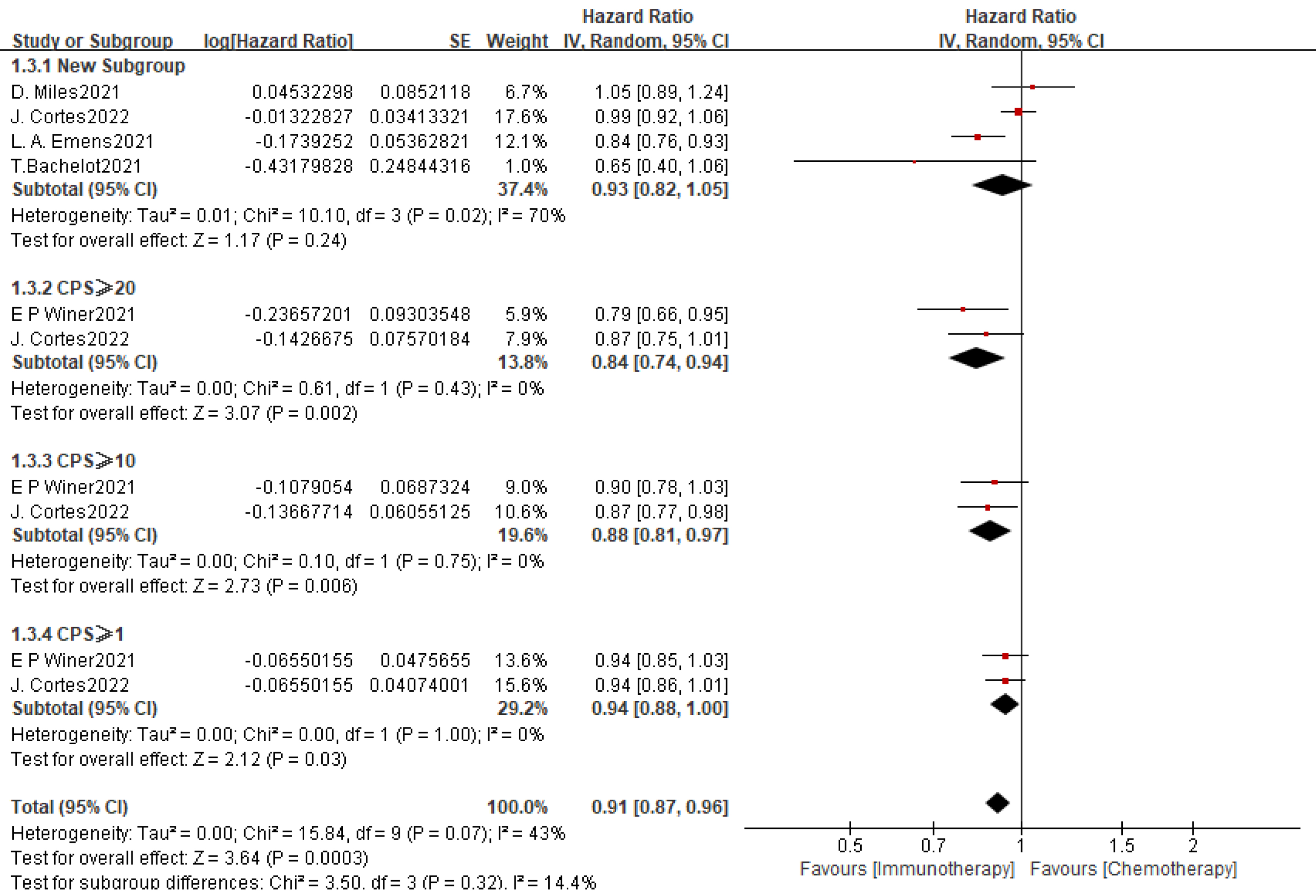 Fig. 3.
Fig. 3.
Summary and subgroup analysis of effects of PD-L1-inhibitors in
combination with chemotherapy on OS in the PD-L1-positive population of mTNBC
patients. This figure presents OS outcomes in PD-L1-positive subgroups,
including an overall pooled analysis of 4 studies (HR = 0.93, 95% CI:
0.82–1.05, p = 0.24; I2 = 70%, p = 0.02), and
stratified analyses by PD-L1 CPS (CPS
| PD-L1 Threshold | PFS HR (95% CI) | p | OS HR (95% CI) | p |
| CPS |
0.89 (0.82–0.97) | 0.94 (0.86–1.02) | 0.14 | |
| CPS |
0.72 (0.64–0.82) | 0.85 (0.74–0.98) | 0.03 | |
| CPS |
0.78 (0.60–1.02) | 0.07 | 0.88 (0.68–1.15) | 0.35 |
CI, confidence interval; HR, hazard ratio.
Four studies compared PFS with and without immunotherapy in the mTNBC treatment.
Patients with mTNBC receiving chemotherapy combined with PD-1 inhibitors had
longer PFS (HR, 0.91; 95% CI, 0.88–0.94; p
| Factor | Number of studies | Number of patients | HR (95% CI) | Overall effect p-value | Heterogeneity | ||
| I2 (%) | p | ||||||
| ITT OS | |||||||
| Overall | 6 | 3287 | 0.96 (0.94, 0.98) | 14% | 0.29000 | ||
| Chemotherapy | |||||||
| Paclitaxel | 3 | 2400 | 0.97 (0.91, 1.02) | 0.26000 | 37% | 0.20000 | |
| Non-paclitaxel | 3 | 887 | 0.91 (0.78, 1.05) | 0.21000 | 51% | 0.13000 | |
| Sample size | |||||||
| 3 | 2120 | 0.98 (0.94, 1.04) | 0.54000 | 17% | 0.30000 | ||
| 3 | 1167 | 0.91 (0.82, 1.00) | 0.05000 | 21% | 0.28000 | ||
| ICIs | |||||||
| Atezolizumab | 3 | 1612 | 0.97 (0.89, 1.06) | 0.53000 | 44% | 0.17000 | |
| Pembrolizumab | 2 | 1469 | 0.97 (0.92, 1.02) | 0.21000 | 0% | 0.47000 | |
| PD-L1+ OS | |||||||
| Overall | 4 | 1540 | 0.93 (0.82, 1.05) | 0.24000 | 70% | 0.02000 | |
| CPS | |||||||
| CPS |
2 | 1041 | 0.94 (0.88, 1.00) | 0.03000 | 0% | 1.00000 | |
| CPS |
2 | 517 | 0.88 (0.81, 0.97) | 0% | 0.75000 | ||
| CPS |
2 | 313 | 0.84 (0.74, 0.94) | 0% | 0.43000 | ||
| ITT PFS | |||||||
| Overall | 4 | 2459 | 0.91 (0.88, 0.94) | 0% | 0.58000 | ||
| PD-L1+ PFS | |||||||
| Overall | 3 | 729 | 0.84 (0.78, 0.91) | 0% | 0.72000 | ||
| AEs | |||||||
| Overall | 6 | 3287 | 0.91 (0.43, 1.92) | 0.73000 | 96% | ||
| ICIs | |||||||
| Atezolizumab | 4 | 1612 | 1.38 (1.18, 1.62) | 0% | 0.78000 | ||
| Pembrolizumab | 2 | 1469 | 0.37 (0.04, 3.67) | 0.39000 | 99% | ||
PD-L1+, protein programmed cell death ligand 1positive expression.
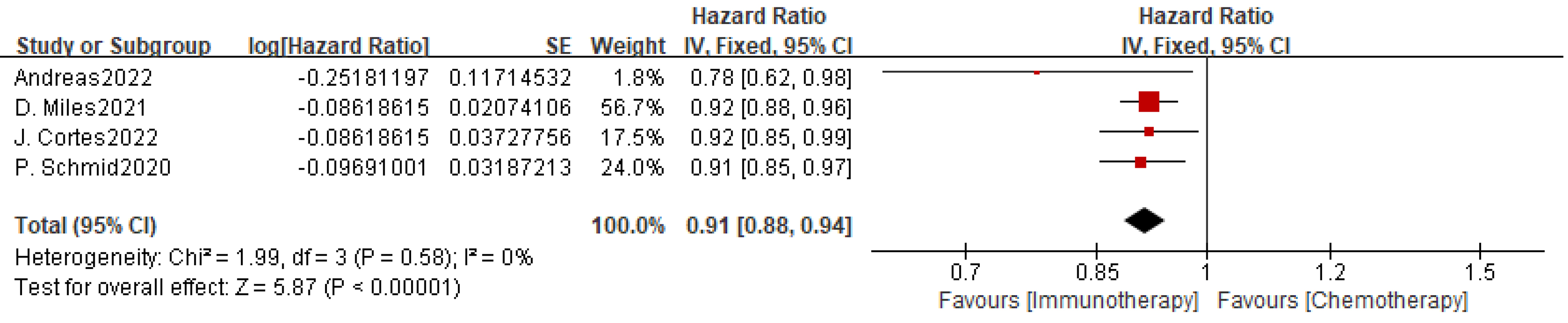 Fig. 4.
Fig. 4.
Effect of PD-L1 inhibitors combined with chemotherapy on PFS in the ITT population of patients with mTNBC.
There were only three studies that analyzed PFS with or without applying
PD-1/PD-L1 inhibitors to chemotherapy in the PD-L1-positive mTNBC population. The
outcomes demonstrated significantly increased PFS when PD-1 inhibitors were
combined with chemotherapy (HR, 0.84; 95% CI, 0.78–0.91; p
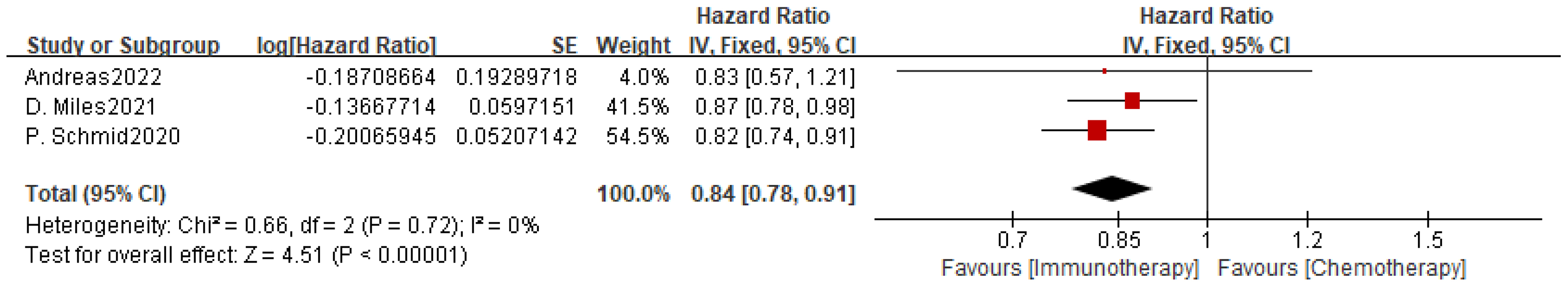 Fig. 5.
Fig. 5.
Effect of PD-L1 inhibitors in combination with chemotherapy on PFS in PD-L1-positive patients with mTNBC.
PD-L1 inhibitors mainly included durvalumab, atezolizumab, and pembrolizumab. A
total of five studies were included (Table 5), demonstrating that grade 3–4
adverse reactions to atezolizumab primarily involved neutropenia, peripheral
neuropathy, decreased neutrophil count, fatigue, and occasional hyper- or
hypothyroidism. The paclitaxel plus placebo group had fewer side effects than the
atezolizumab plus paclitaxel group (HR, 1.38; 95% CI, 1.18–1.62; p
| Trial (AEs (ICI + C/C) (%)) | Common adverse events | Immune-mediated adverse events | ||
| Grade |
Grade 3, 4, or 5 | Grade |
Grade 3, 4, or 5 | |
| IMpassion130 | Alopecia (57.2%/57.4%) | Neutropenia (8.0%/8.0%) | Rash (35.9%/26.0%) | Hepatitis (1.5%/0.2%) |
| Fatigue (47.0%/45.1%) | Peripheral neuropathy (6.0%/3.0%) | Hypothyroidism (18.3%/4.4%) | Rash (1.1%/0.5%) | |
| Nausea (46.7%/38.4%) | Decreased neutrophil count (5.0%/4.0%), fatigue (4.0%/3.0%) | Hyperthyroidism (4.8%/1.2%), pneumonitis (3.9%/0.2%) | Colitis (0.4%/0.2%) | |
| IMpassion131 | Alopecia, anaemia, peripheral neuropathy, diarrhoea, fatigue, nausea (25.0%/18.0%) | Diarrhea, vomiting, decreased appetite, hypothyroidism, hyperthyroidism (53.0%/46.0%) | Rash (30.0%/33.0%), hypothyroidism (6.0%/14.0%), hepatitis (0.9%/2.0%) | Diabetes mellitus (0.9%/0.9%) |
| Colitis (0.9%/0.2%), rash (0.9%/0.9%) | ||||
| SAFIR02-BREAST IMMUNO | Not mentioned | Not mentioned | Not mentioned | Not mentioned |
| KEYNOTE-119 | Fatigue (11.0%/13.0%) | Increased aspartate aminotransferase (3.0%/1.0%) | Hypothyroidism (7.0%/1.0%) | Pneumonitis (1.0%/0) |
| Nausea (10.0%/21.0%) | Asthenia (1.0%/ |
Hyperthyroidism (4.0%/0) | Severe skin reaction (1.0%/ | |
| Hypothyroidism (7.0%/0) | Pneumonitis (1.0%/0) | |||
| KEYNOTE-355 | Anemia (49.1%/45.9%), neutropenia (41.1%/38.1%) | Neutropenia (29.7%/29.9%), neutrophil count decrease (17.4%/20.3%), anemia (16.5%/14.6%) | Hypothyroidism (15.8%/3.2%), hyperthyroidism (4.3%/1.1%), pneumonitis (2.5%/0) | Severe skin reactions (1.8%/0), pneumonitis (1.1%/0) |
| Nausea (39.3%/41.3%) | ||||
| ALICE | Rash (65.0%/39.0%) | Decreased lymphocyte count (15.0%/18.0%) | Hypothyroidism (12.0%/7.0%) | Pneumonitis (5.0%/0) |
| Nausea (57.0%/54.0%) | Rash (18.0%/0) | Pneumonitis (10.0%/4.0%) | Rash (5.0%/0) | |
| Palmar-plantar erythrodysaesthesia syndrome (52.0%/11.0%) | Palmar-plantar erythrodysesthesia syndrome (8.0%/4.0%) | Rash (8.0%/4.0%) | Pancreatitis (2.0%/0) | |
| Pyrexia (2.0%/0) | ||||
ICI, immune checkpoint inhibitors.
| Drug type | AEs | No. of patients (%) | HR (95% CI) | p |
| Atezolizumab | All-grade | 70.20% | 1.25 (1.10–1.42) | |
| Atezolizumab | Grade 3/4 | 28.50% | 1.38 (1.18–1.62) | |
| Pembrolizumab | All-grade | 64.80% | 0.95 (0.78–1.15) | 0.60 |
| Pembrolizumab | Grade 3/4 | 22.10% | 0.37 (0.04–3.67) | 0.39 |
This meta-analysis systematically evaluated the efficacy and safety of ICIs combined with chemotherapy in aTNBC or mTNBC. TNBC is a highly invasive and genetically unstable cancer characterized by negative expression of both hormone receptors and HER2, which is prone to germline BRCA1 mutations [25]. Since the antitumor activity of ICIs was demonstrated by the KEYNOTE-012 in 2015 [26], the efficacy of ICIs combined with chemotherapy in aTNBC has been increasingly explored. However, RCTs articles and comprehensive analyses on this topic remain limited.
This meta-analysis included seven RCTs, comprising a total of 3219 patients with
aTNBC or mTNBC. We demonstrated that combining ICIs, such as atezolizumab,
pembrolizumab, and durvalumab, with chemotherapy significantly improved PFS in
both the ITT and PD-L1-positive populations. Importantly, the analysis
highlighted that pembrolizumab combined with chemotherapy delivered a
significantly greater PFS benefit, particularly in patients with higher CPS
scores (CPS
Previous studies, such as IMpassion130, demonstrated clinical benefits of atezolizumab combined with nab-paclitaxel in PD-L1-positive patients [23, 27]. The concurrent studies validated it through SP263, 22C3, or CPS analyses [28]. In 2019, Adams et al. [29] evaluated the efficacy of atezolizumab combined with nab-paclitaxel in 33 women with stage IV or locally recurrent TNBC who had received prior chemotherapy, finding a median PFS and OS of 5.5 and 14.7 months, respectively. PD-L1-positive patients had OS and PFS of 21.9 and 6.9 months, respectively, compared to 11.4 and 5.1 months in PD-L1–negative patients. The combination of atezolizumab and ipatasertib with capecitabine extended PFS to 8.2 months in mTNBC patients naive to platinum-based chemotherapy and ICIs, which was twice as long as the 3.9 months observed with chemotherapy alone [30].
Voorwerk et al. [31] demonstrated that atezolizumab in combination with platinum agents also exhibited promising antitumor activity in patients with TNBC. Among the six individuals, four were TNBC patients who had increased expression of cluster of differentiation 8 positive T cells (CD8+ T ) cells and immune checkpoint markers. Furthermore, Røssevold et al. [24] conducted a pioneering study using atezolizumab combined with pegylated liposomal doxorubicin (PLD) for mTNBC treatment. The results indicated that this combination prolonged OS and PFS in mTNBC patients, with lower cardiac toxicity compared to anthracycline-based drugs [32]. In contradiction to the above findings, the IMpassion131 study reported that atezolizumab in combination with paclitaxel did not significantly benefit OS or PFS [19], which may be attributed to paclitaxel’s sensitizing effects and the concomitant use of dexamethasone [33].
In addition to atezolizumab, pembrolizumab for mTNBC treatment has been
gradually studied in recent years. In 2021, Winer et al. [21] conducted
a study that categorized tumor status into CPS
In 2018, Santa-Maria et al. [44] carried out a pilot study using durvalumab and demonstrated a high benefit for TNBC. To further explore the efficacy of durvalumab, Al Sayed et al. [45] combined it with paclitaxel, resulting in increased CD8+ T-cell infiltration, enhanced immune checkpoint expression and reduced T-cell exhaustion, as well as fewer adverse effects on mTNBC patients. In 2021, Ghebeh et al. [46] conducted a 2-year follow-up after combining every-5-week paclitaxel with every-2-week durvalumab, giving PFS and OS of 5 months and 20.7 months, respectively. They attributed the results to increased peripheral blood eosinophil counts [47]. This study further confirmed the efficacy of durvalumab on treating aTNBC.
Considering the high heterogeneity (I2 = 96%) observed in the
pooled analysis of adverse events, and recognizing the distinct toxicity profiles
among different ICIs, we further stratified the analysis by drug type and by
severity of adverse events (grade 3/4 versus all grades). When analyzed
separately, atezolizumab combined with chemotherapy demonstrated a higher
incidence of grade 3/4 adverse events (HR, 1.38; 95% CI, 1.18–1.62; p
However, this meta-analysis has some limitations. First, the included studies
employed different chemotherapy regimens, and comprehensive studies evaluating
various combinations of ICIs and chemotherapy in mTNBC remain limited,
introducing potential bias. Additionally, the number of studies involving
durvalumab and PLD is limited, which may cause bias. Second, although
pembrolizumab has shown OS benefits in patients with CPS
Immunotherapy using PD-L1 inhibitors prolong PFS in the PD-L1-positive population, with a more pronounced effect observed at higher CPS levels. Understanding and utilizing CPS thresholds allow clinicians to personalize treatment strategies, ensuring that patients most likely to benefit from immunotherapy are appropriately identified. It also helps prevent unnecessary exposure to potential side effects in patients less likely to respond. Incorporating CPS assessment into clinical decision-making potentially enhances the precision of TNBC management.
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
HC, XL and ML designed the research study. The table was conducted by HC, while the graphic figures were created by LS; the data from studies were compiled by XL. YZ and LS acquired, analyzed, and interpreted the data. HC and ML contributed to preparing the draft and editorial revisions. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
The study was supported by the Industry-University-Research Innovation Fund for Chinese Universities (No. 2023HT083).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG37642.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





