1 Department of Obstetrics and Gynecology, Second Affiliated Hospital of Soochow University, 215123 Suzhou, Jiangsu, China
2 Department of Obstetrics and Gynecology, The Affiliated Suzhou Hospital of Nanjing Medical University, 215123 Suzhou, Jiangsu, China
3 Department of Forensic Medicine, School of Basic Medicine, Suzhou Medical College of Soochow University, 215123 Suzhou, Jiangsu, China
4 College of Life Sciences, Mudanjiang Medical University, 157011 Mudanjiang, Heilongjiang, China
5 MOE Key Laboratory of Geriatric Diseases and Immunology, School of Basic Medicine, Soochow University, 215123 Suzhou, Jiangsu, China
†These authors contributed equally.
Abstract
Severe endometrial injury constitutes a significant risk to female fertility, often leading to the development of intrauterine adhesions. Pyroptosis, a form of programmed cell death associated with inflammation, is initiated by the cleavage of gasdermin family proteins by caspase, which has been implicated in endometrial injury. In recent years, hydrogen sulfide (H2S), a gaseous signaling molecule, exerts significant regulatory effects on pyroptosis in diverse pathological processes. The aim of this study is to elucidate the role of H2S in facilitating functional recovery of the endometrium following injury in a murine model.
A prospective laboratory based randomized control trial was performed to evaluate the protective role of H2S in endometrial injury. Ethanol-induced endometrial injury mouse models were established and H2S donor sodium hydrosulfide (NaHS) was randomly administered to the injured mice. Western blot analysis was conducted to assess changes in the expression of endogenous H2S metabolism and pyroptosis-related markers and histological analysis (Hematoxylin & Eosin and Masson staining) was employed to examine alterations in endometrial morphology. Finally, a fertility test was performed to evaluate the restoration of the uterine function.
Following endometrial injury, the expression of key endogenous H2S enzymes-cystathionine β-synthase (CBS), cystathionine γ-lyase (CSE), and 3-mercaptopyruvate sulfurtransferase (MST)-is significantly reduced, while the levels of pyroptosis-related proteins are elevated (p < 0.05). However, treatment with H2S resulted in an increase in the expression of endogenous H2S enzymes and a decrease in pyroptosis-associated proteins compared to the Model group (p < 0.05). Moreover, endometrial morphology and embryo count showed the most pronounced improvement in the H2S treatment group (p < 0.05).
This study confirms the therapeutic efficacy of H2S in facilitating the recovery of injured endometrium and revealed its potential therapeutic mechanism, offering a promising therapeutic avenue for patients with severe endometrial injury.
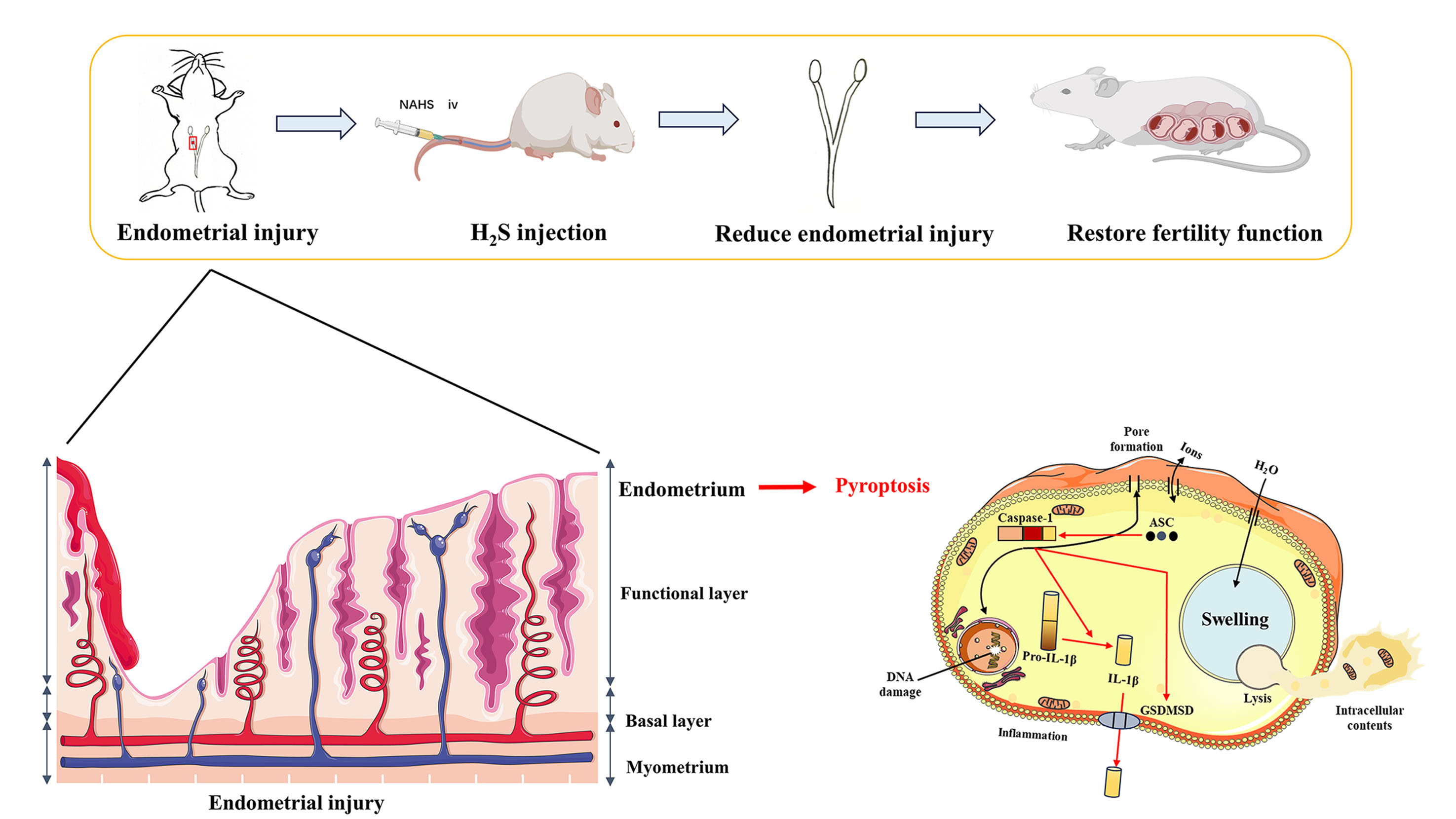
Graphical Abstract
Keywords
- endometrial injury
- hydrogen sulfide
- intrauterine adhesions
- pyroptosis
With the rise in uterine surgeries, endometrial injury has become a common cause of intrauterine adhesions (IUA), resulting from impaired repair of the endometrial basal layer. During this process, damage to the basal lamina is usually irreversible and accompanied by fibrosis, which led to irregular bleeding, hypomenorrhea, secondary dysmenorrhea and amenorrhea, reduced fertility and premature birth [1]. Transcervical resection of uterine adhesions is the standard clinical treatment for IUA, but it is often associated with poor reproductive outcomes post-surgery [2]. Both intravenous and intrauterine administration of stem cells have proven effective in treating endometrial dysfunction, as evidenced by preclinical and clinical studies. The efficacy of cell transplantation for uterine repair, particularly in the context of IUA, has been shown to have variable results, with only a small percentage of transplanted cells successfully engrafting in the uterus [3]. Consequently, there is a pressing need for innovative therapeutic strategies to expedite endometrial repair and enhance its receptivity.
Hydrogen sulfide (H2S), a gaseous signaling molecule, is involved in multiple pathophysiological processes, including inflammation, oxidative stress, abnormal angiogenesis and cell death [4].
Emerging evidence suggests that H2S may offer therapeutic benefits in the management of various female reproductive disorders, including endometriosis, intrauterine adhesions, and preeclampsia [5, 6, 7]. Xia et al. [7] reported that H2S has demonstrated the potential to mitigate adhesion formation after uterine horn post-operative model. However, the detailed molecular healing mechanism of H2S in endometrial injury has not been reported to date.
Pyroptosis, a type of inflammatory cell death, can play an important role in
cell and tissue damage, which is involved in the pathogenesis of multiple
endometrial diseases [8]. Pyroptosis, a highly inflammatory form of cell death,
is triggered by the cleavage of gasdermin proteins, resulting in the formation of
pores in the cell membrane. This process leads to the release of pro-inflammatory
cytokines, such as interleukin-1
Endometrial cells employ various adaptive strategies, such as the inhibition of pyroptosis, to ensure their survival and function within the demanding uterine environment. While significant progress has been made in understanding the role of pyroptosis in endometriosis and endometrial cancer, the precise mechanisms that regulate this process are still not fully elucidated in endometrial injury. Our group recently reported on the regulatory effects of H2S on pyroptosis in brain injury, suggesting that there is a mutual relationship between hydrogen sulfide and pyroptosis [11, 12, 13]. However, to date, the mechanisms regulating the interplay between hydrogen sulfide and pyroptosis in the endometrial injury are still poorly understood. Building on these studies, we hypothesize that pyroptosis and endogenous H2S metabolism are involved in the molecular mechanism of endometrial injury. Thus, we sought to investigate the potential therapeutic benefits of exogenous H2S in a mouse model of endometrial injury and to elucidate the underlying molecular protective mechanisms.
Adult female ICR mice (specific pathogen free [SPF] grade), aged 6–8 weeks and weighing 20–25 g, were obtained from the Laboratory Animal Center at Soochow University. The mice were housed under controlled conditions of 50% humidity, 25 °C, and a 12-hour light/dark cycle with ad libitum access to food and water. Mice were randomly assigned to experimental groups using a computer-generated random number table. All animal procedures were carried out in compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Prior to experimentation, the protocols were reviewed and approved by the Soochow University Animal Use and Care Committee (reference number: SYXK (Su) 2021-0065).
A prospective laboratory based randomized control trial was performed to evaluate the protective role of H2S in endometrial injury. Mice were coded with computer-generated random numbers prior to the experiment, and randomly divided into a sham operation group (Sham group), endometrial injury group (Model group), and sodium hydrosulfide (NaHS) treatment after injury group (Model+NaHS group). It is important to ensure that reducing the number of animals used is balanced against any additional suffering that might be caused by their repeated use according to the welfare of animals. Therefore, we choose n = 6/group.
Mice in the control group underwent a sham laparotomy and were fed a standard laboratory diet. Uterine tissue samples were harvested concurrently with the experimental groups. 95% ethanol (1590100500, Merck KGaA, Shanghai, China) was used to establish the mouse endometrial injury model, and a visual representation of the established endometrial injury model is provided in Fig. 1 [14, 15]. Mice were anesthetized using an isoflurane gas anesthesia machine (VetFlo-1205SP, Kent Scientific Corporation, Torrington, CT, USA) first with 2–3% isoflurane and then with 1.5–2% isoflurane to maintain the anesthetic effect, and allow the mice to breathe spontaneously during the procedure. After the mouse were anesthetized, the mouse abdominal mouse hair was removed with a shaver, and the mouse abdomen was sterilized with 75% alcohol, and then the animals were immobilized, and then, in the middle of the rectus abdominis muscle along the lower abdomen, the abdominal wall was carefully incised in layers, and the sterile gauze pads were placed around the incision, to expose the pink Y-ring. Gauze was placed around the incision to expose the pink Y-shaped uterus. The uterine horns were relocated out of the abdominal cavity with sterile ophthalmic forceps, after which the right uterus of the mouse was gently clamped at the pro-ovarian end and the pro-cervical end with sterile forceps to form a closed uterine cavity. A 1 mL insulin needle aspirating 95% ethanol was inserted from the proximal cervical opening, and 95% ethanol was slowly injected until the uterus filled up and turned white (about 20 µL) and kept for 1 min to induce endometrial injury in mouse, and then the uterine cavity and the abdominal cavity were rinsed with physiological saline in each layer three times to remove the residual ethanol. Meanwhile, the left uterus was kept intact.
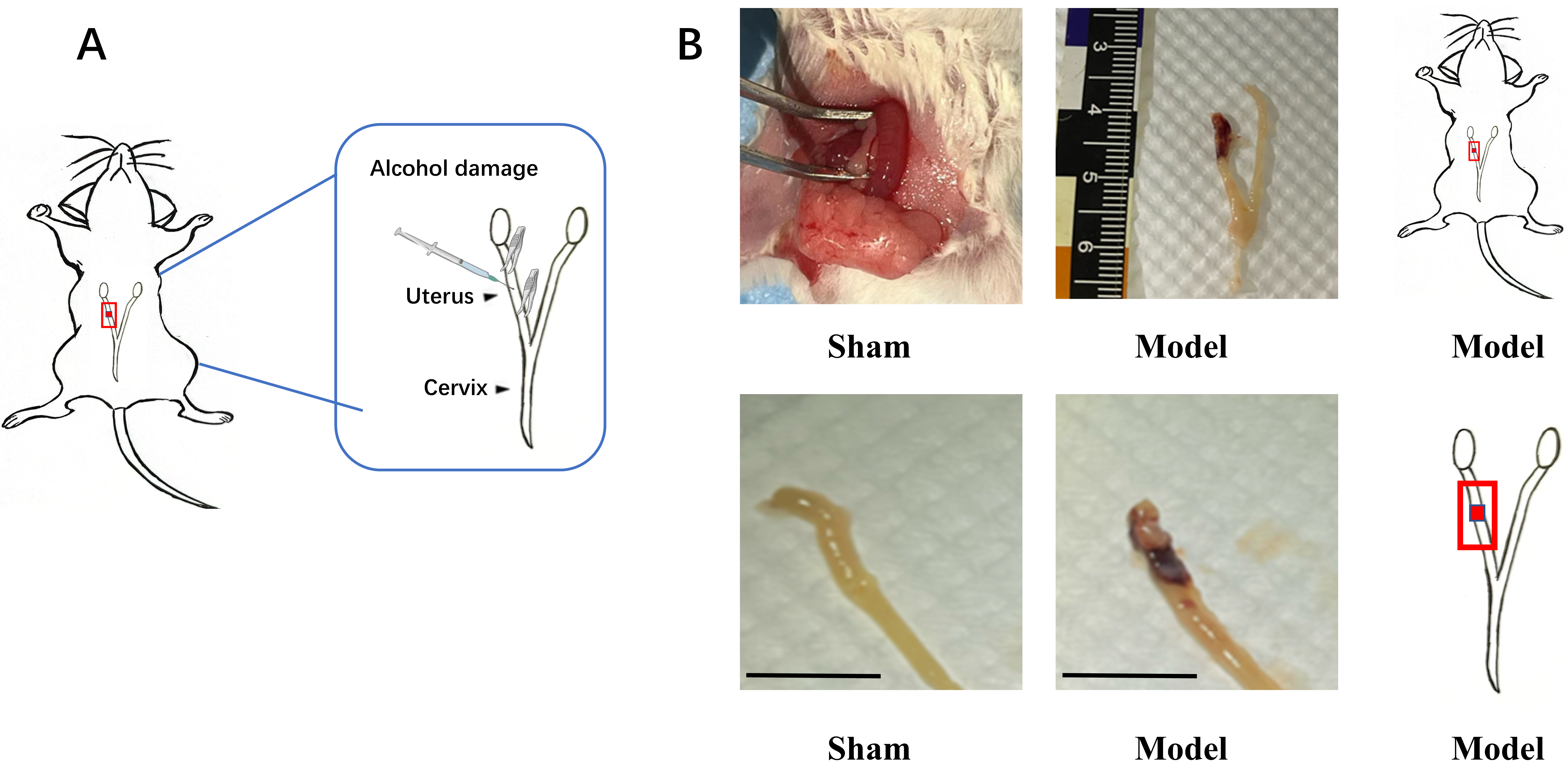 Fig. 1.
Fig. 1.
Schematic representation of the mouse endometrial injury model and the morphology of uterus after injury. (A) Schematic illustration of the experimental procedure for inducing endometrial injury in mice by intrauterine injection of 95% ethanol for a duration of 20 seconds. (B) The morphology of the uterus was observed under visible light and schematic of the endometrial injury site. Scale bar = 1 cm. The schematic illustration of mice model is created by PowerPoint (21.0, Microsoft Inc., Washington, USA) using pictures drawn by our team.
After establishing the mouse endometrial injury model, the model group (Model group) was given 200 µL of phosphate-buffered saline (PBS) (C0221A, Beyotime, Shanghai, China) tail vein injection immediately after suturing the abdominal cavity. The dosage of NaHS (161527, Sigma-Aldrich, Shanghai, China) is based on previous literature reports [7, 13, 16]. For the NaHS treatment group (Model+NaHS group), mice were immediately administered 200 µL of 1.2 mg/kg NaHS via tail vein injection post-surgery. The sham operation group was given 200 µL of PBS via tail vein injection. After completion of the surgery, the mice were placed on a heating pad at 37 °C. After the mice were awakened, they were returned to the cage. Mice in all three groups were euthanized after one estrous cycle (5 d), and the right uterus was removed and placed in a refrigerator at –80 °C for subsequent processing. Female mouse used for fertility tests were modeled at the same time and were allowed to recover for 15 d for subsequent experiments. After these experiments, the mice were euthanized using carbon dioxide inhalation. The mice were placed in a suitable euthanasia chamber and exposed to a slow flow of 100% CO2 gas that displaces the chamber’s air volume in 30–70 seconds. Once the mice became unresponsive and ceased breathing, death was confirmed by palpating for a heartbeat and checking for a corneal reflex. The deceased animals were then disposed of according to institutional guidelines.
Fresh uterine tissues were homogenized on ice in radio immunoprecipitation assay (RIPA) buffer (P0013B, Beyotime, Shanghai, China) supplemented with
phenylmethylsulfonyl fluoride (PMSF, ST506, Beyotime, Shanghai, China) using an
ultrasonic histocytometer (JY99-IIDN, Scientz Biotechnology, Ningbo, Zhejiang, China) to
obtain a tissue lysate. The tissue homogenate was centrifuged at 14,000 rpm for
25 minutes at 4 °C, and the supernatant was collected. The protein
concentration in different samples was detected by Nanodrop-2000 ultra-micro
spectrophotometer (Thermo Fisher, Shanghai, China), and a mixture of RIPA and
PMSF was used for leveling to adjust the total concentration of the samples to 20
µg/µL. 5
To quantify H2S levels in uterine tissue, we employed a previously
described method using Micro Sulfide Ion Electrode (LIS-146AGSCM, Lazar Research
Laboratories Inc., Los Angeles, CA, USA) [13]. Before data acquisition, the probe
was calibrated using a standard curve generated with NaHS solutions ranging from
10-2 to 10-6 M, prepared in an antioxidant buffer containing 250 g sodium
salicylate (S3007, Merck, Shanghai, China), 65 g ascorbic acid (PHR1008, Merck, Shanghai, China), and 85 g sodium hydroxide (NaOH) (655104, Merck, Shanghai, China) per liter of distilled water.
Subsequently, the probe was carefully inserted into 2 mm below the tissue aqueous
solution and the value of the probe was monitored and recorded for each
experimental group. The H2S level (%) = [(value at Sham group – value at
Model group or Model+NaHS group)/value at Sham group]
The dehydrated uteri were oriented and embedded in optimal cutting temperature compound (OCT). Subsequently, the uteri were sectioned longitudinally at a thickness of 10 µm. The uterine morphology was observed with a microscope (DS-Ri2, Nikon Instruments Inc, Shanghai, China). The thickness of endometrial and the number of endometrial glands in each uterine section was calculated using Image J software (1.54, National Institutes of Health, Bethesda, MD, USA) with hematoxylin-eosin (HE) staining (C0105M, Beyotime, Shanghai, China). Masson’s trichrome staining was used to assess endometrial fibrosis. The staining procedure was carried out according to the manufacturer’s instructions for the Masson staining kit (G1346; Solarbio, Beijing, China). Gland number, endometrial thickness, and fibrotic area were quantified in at least 10 random images per slide using ImageJ software. These photographs were processed using Photograph software (22.0, Adobe Inc., San Jose, CA, USA).
Fertility was assessed in female mouse after three estrous cycles, approximately 15 d after modeling, according to the grouping in Method 2.2. Female mice (n = 6) in each group were housed in a 2:1 ratio with sexually mature male on the day of estrus. Female mice were observed to develop vaginal plugs marked for 0.5 d. These mice were then isolated and euthanized at 10 d after the development of vaginal plugs, and data on the embryo count were recorded.
The data obtained in the study were statistically analyzed using GraphPad Prism
(9.0, Dotmatics Inc., Boston, MA, USA). All data were presented as mean
An in vivo model of endometrial damage was established. Briefly, 95% ethanol was injected into the uterine horn to induce endometrial injury, with the severity of injury depending on the duration of ethanol exposure. The uteri of sham-operated mice exhibited normal histological features. In contrast, the uteri of the model group displayed significant pathological changes, including tissue degeneration, reduced tissue elasticity, fluid accumulation, and bleeding (Fig. 1).
H2S has been implicated in the regulation and therapeutic applications in
the treatment of female reproductive disorders. Cystathionine
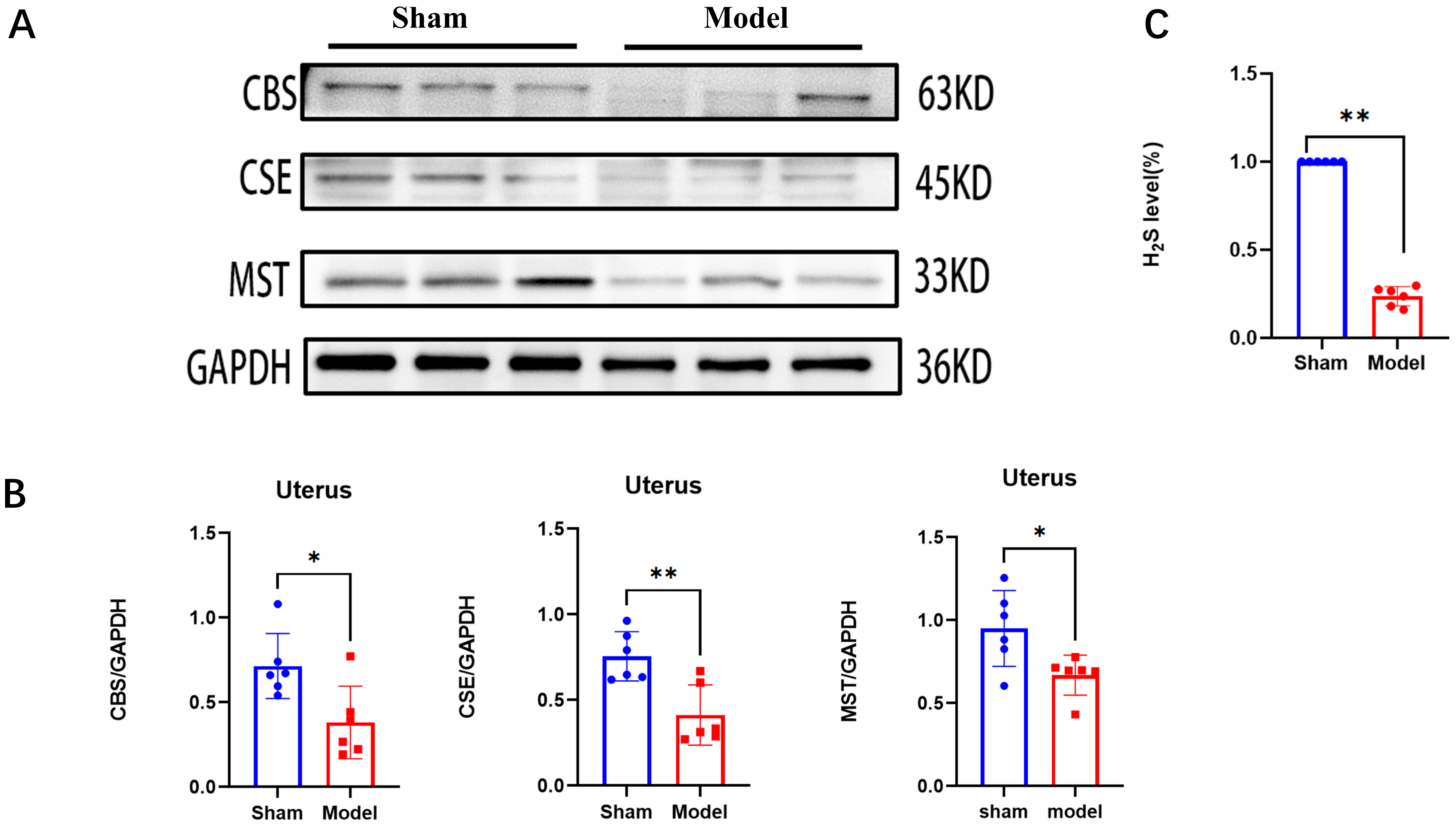 Fig. 2.
Fig. 2.
The changes of the level of endogenous hydrogen sulfide
(H2S) and H2S-synthesizing enzymes after endometrial injury. (A)
Representative Western blot images illustrating the protein expression levels of
cystathionine
Pyroptosis, a proinflammatory cell death pathway, plays a crucial role in the
onset and progression of endometrial injury. To validate the occurrence of
pyroptosis, we detected gasdermin D (GSDMD), Caspase-1, IL-1
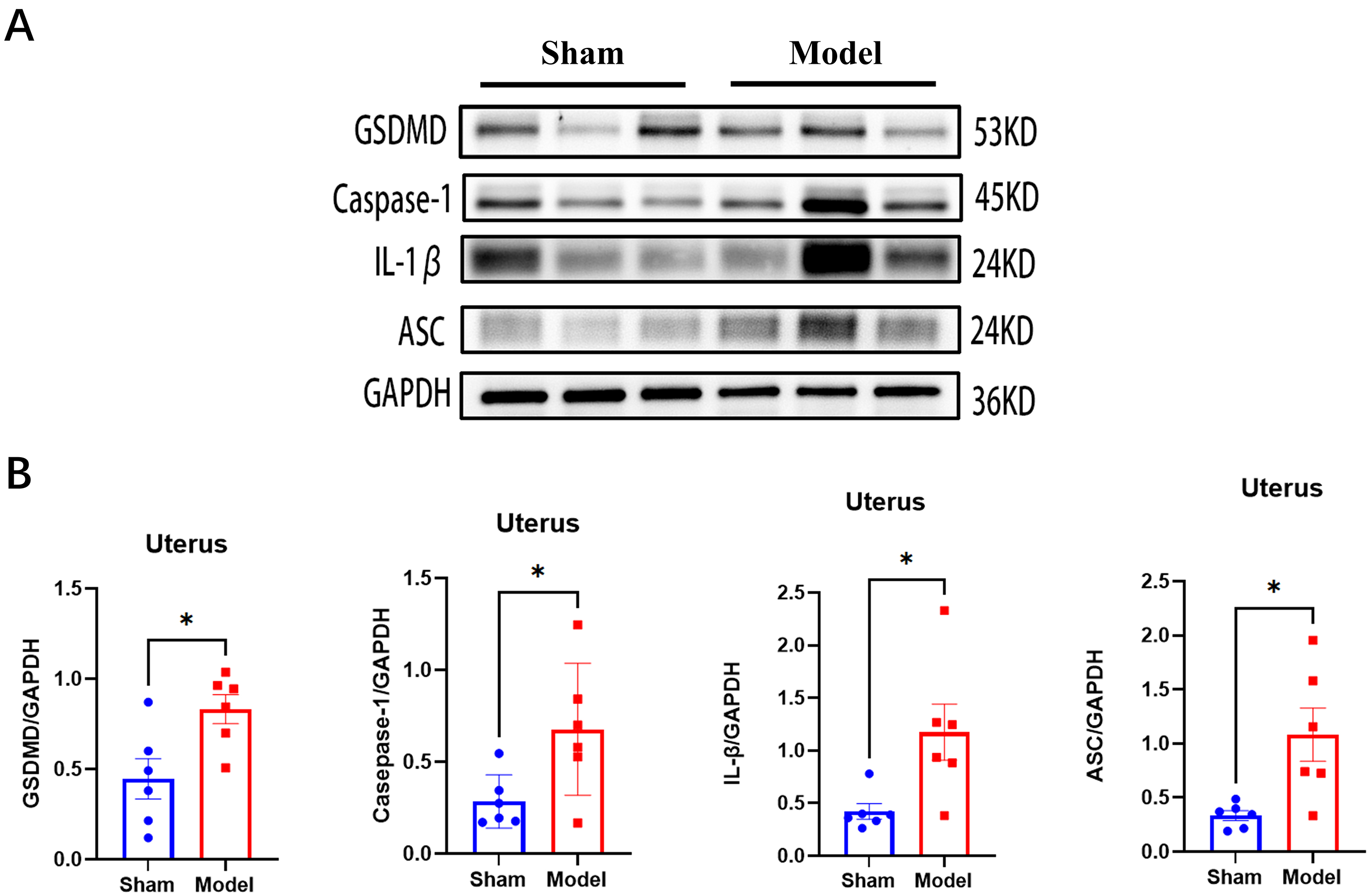 Fig. 3.
Fig. 3.
The changes of pyroptosis-related proteins after endometrial
injury. (A) Representative Western blot images illustrating the protein
expression levels of GSDMD, Caspase-1, IL-1
To investigate the effect of H2S on pyroptosis triggered by endometrial injury,
we analyzed the expression of pyroptosis-related proteins following the injury.
Firstly, we observed that exogenous H2S treatment reversed the decreased
expression of H2S production-related proteins (CBS, CSE and MST) and the
level of endogenous levels (p
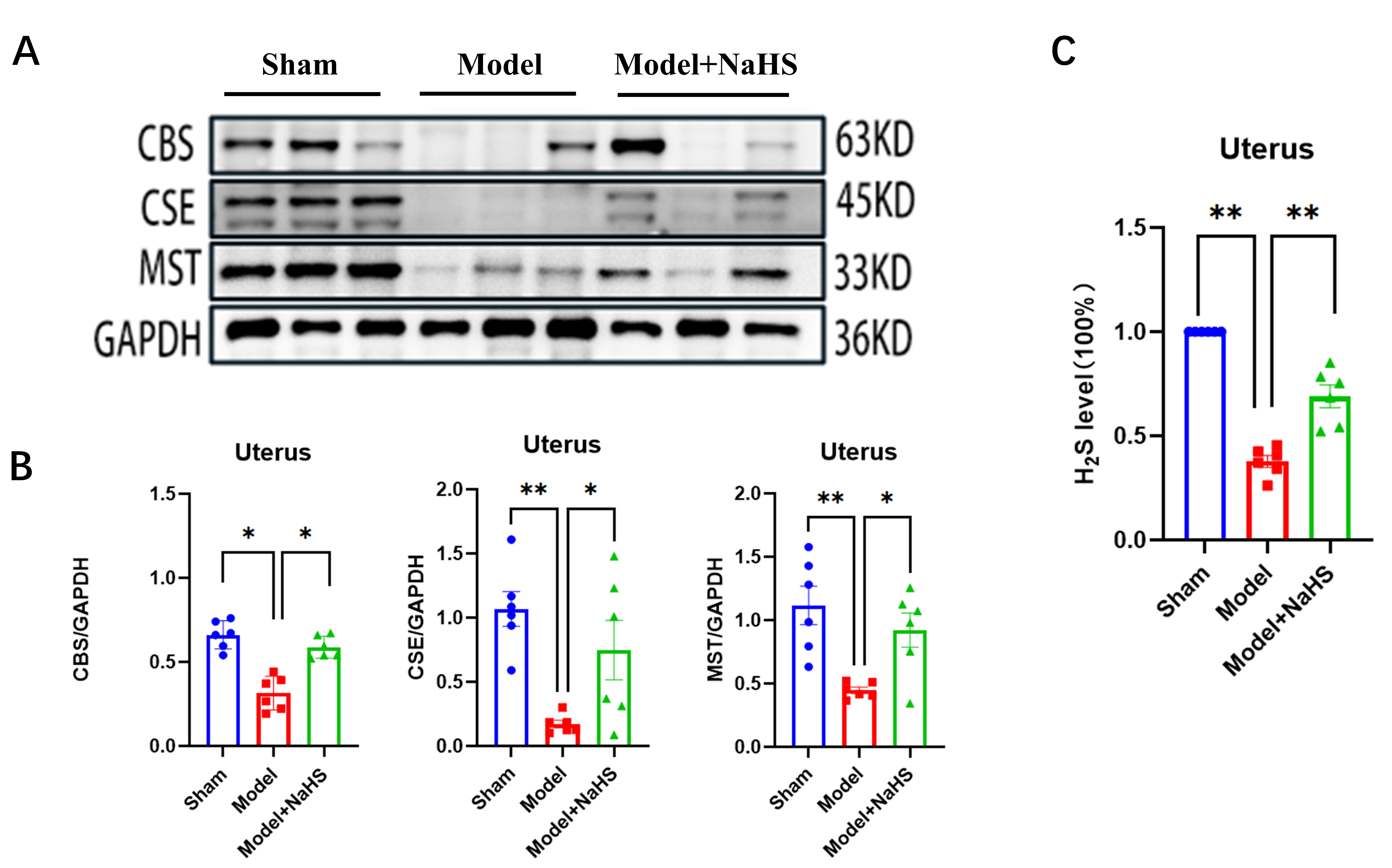 Fig. 4.
Fig. 4.
Treatment with NaHS ameliorated decrease in the expression of
endogenous H2S enzyme and H2S levels induced by EI. (A) Representative
western blot analysis illustrating the altered expression of CBS, CSE, and MST
proteins in the injured uterus of the Sham group, Model group, and Model+NaHS
group, respectively. (B) The levels of CBS, CSE, and MST in the injured uterine
tissue from different groups were quantified using ImageJ software. (C) The level
of H2S was detected by micro sulfide ion electrode. GAPDH was used as a
loading control for western blotting. Bars represent the mean
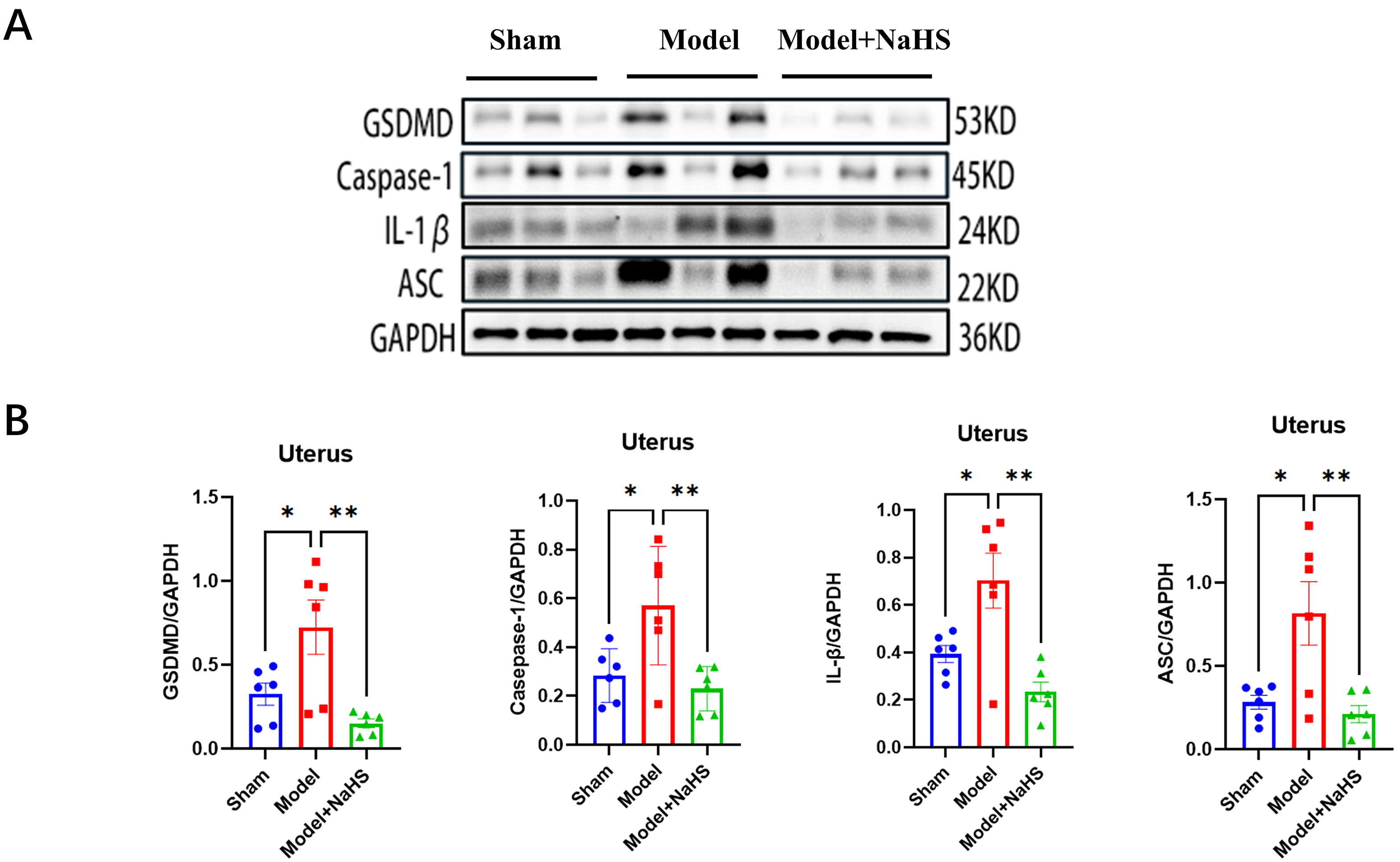 Fig. 5.
Fig. 5.
Exogenous H2S inhibits the increase for pyroptosis. (A)
Representative western blot analysis illustrating the altered expression of
GSDMD, Caspase-1, ASC, and IL-1
HE staining revealed disrupted uterine cavity morphology, thinner endometrium,
and decreased glandular density. Compared with the model group, H2S groups
exhibited increased endometrial thickness (p
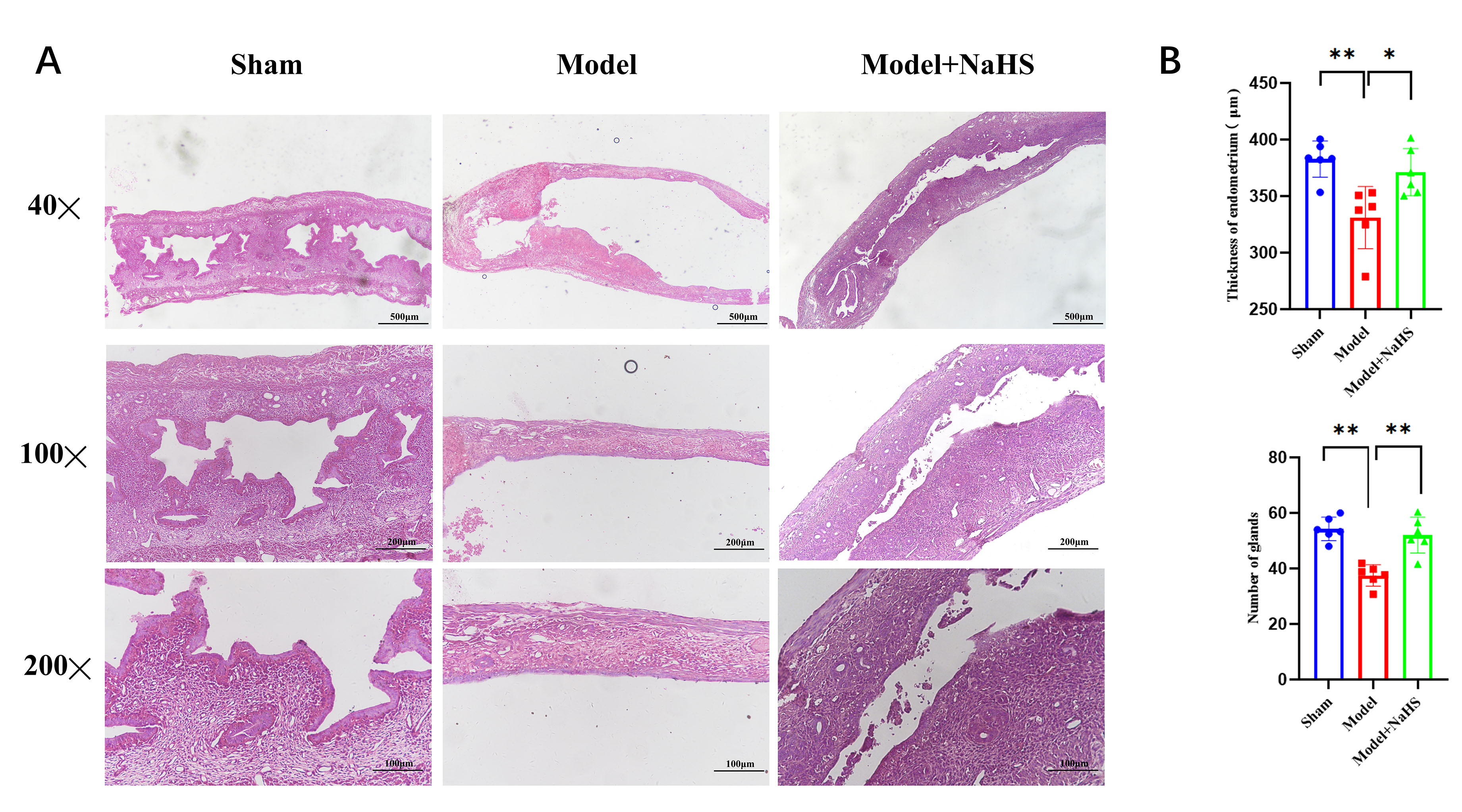 Fig. 6.
Fig. 6.
The endometrial morphology was evaluated by HE
staining after H2S treatment. (A) Hematoxylin-eosin (HE)
staining of mice uterine tissue. (B) Histogram depicting the distribution of
endometrial thickness and gland number across different experimental groups. Bars
represent the mean
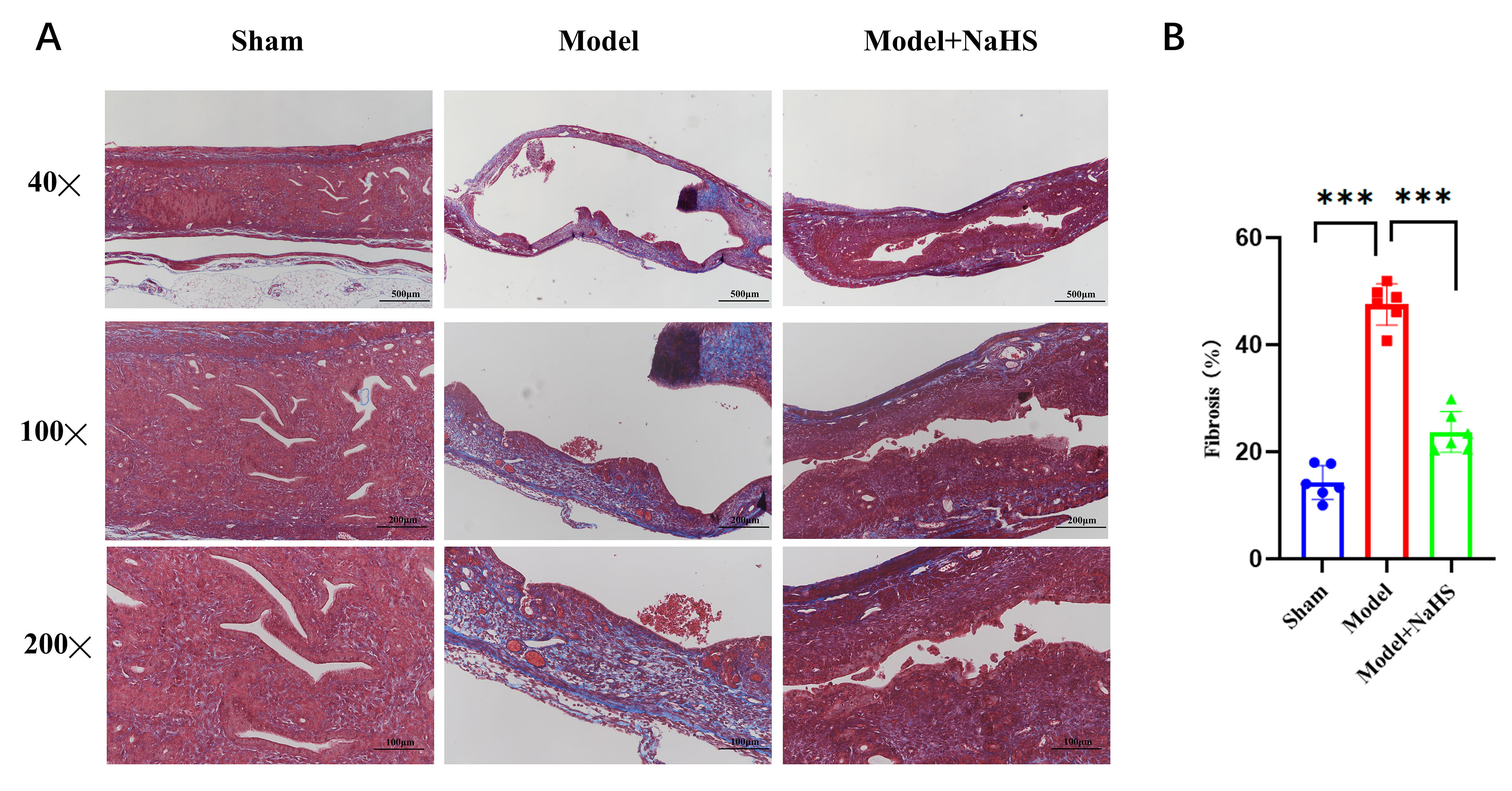 Fig. 7.
Fig. 7.
The endometrial fibrosis was evaluated by Masson staining after
H2S treatment. Masson’s trichrome staining was used to assess endometrial
fibrosis. (A) Masson stain showed fibrotic tissue (stained pale blue) in the
endometrium. (B) Statistical analysis was conducted on the percentage of
endometrial fibrosis area in each group. Bars represent the mean
Treatment with exogenous H2S can significantly improve uterus damage in
female mice after injury. To further assess the impact of exogenous H2S on
endometrial function and recovery, fertility tests were performed. To evaluate
the functionality of the damaged uterus, female mice with unilateral uterine
damage were mated with sexually mature males during estrus following the H2S
treatment period. On the eighth day after the vaginal plug appeared, the
distribution of embryos within the uterine horn was analyzed to evaluate the
effect of exogenous H₂S on fertility. Compared to the Model group, H2S
treatment had more embryos in the Model+NaHS group (p
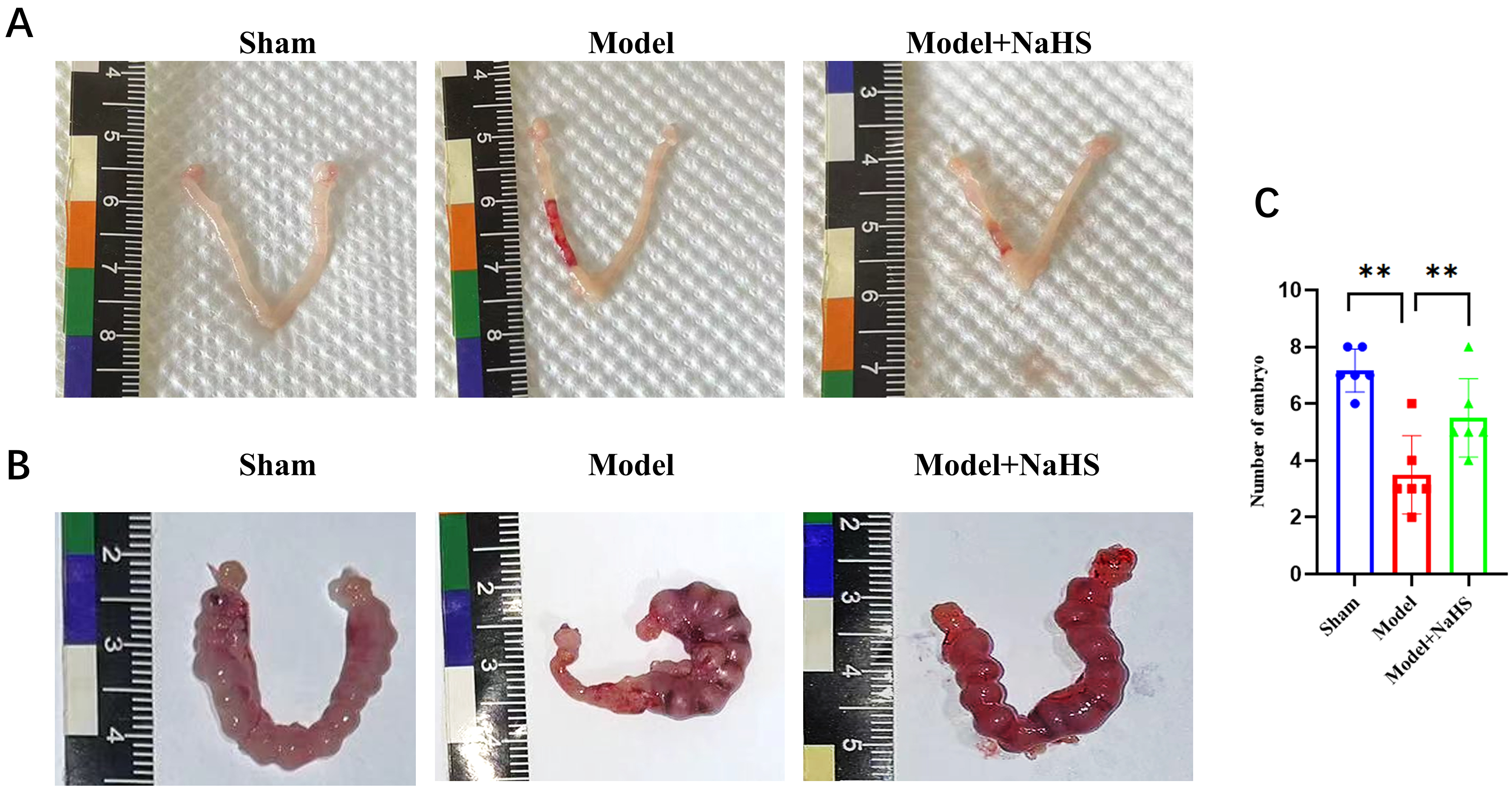 Fig. 8.
Fig. 8.
H2S treatment improve the mice fertility. Two weeks
following the endometrial injury model, female mice were mated with males to
record and analyze the number of embryos in each pregnancy. (A) The protective
role of H2S in uterine after injury in each group. (B) The protective role
of H2S in the mice fertility. (C) Bars represent the mean
In an attempt to understand the role of hydrogen sulfide metabolism and
pyroptosis in the endometrial injury, a murine model of endometrial injury was
established by the intrauterine administration of 95% ethanol.
Our study provides novel evidence for the downregulation of
endogenous H2S-synthesizing enzymes, such as
cystathionine-
A good and receptive endometrium provides an essential internal environment for
embryo implantation and is the prerequisite and guarantee for the achievement of
pregnancy. Endometrial injury plays a crucial role in causing
infertility and also leads to a lot of gynecological diseases, such as
intrauterine adhesions (IUA) and thin endometrium, which can
directly lead to amenorrhea, infertility, abortion or other serious symptoms
[19]. Approximately 90% of patients with endometrial injury develop IUA, a
pathological condition characterized by the formation of endometrial fibrosis.
Persistent inflammation was evident in the endometrium of IUA patients, and the
degree of endometrial fibrosis was positively correlated with the severity of
inflammation [20]. The endometrium is composed of two layers: the functional
layer, which undergoes cyclical changes and is shed during menstruation and
postpartum, and the basal layer, which remains intact and serves as a
regenerative source for the functional layer. Recently, pyroptosis was reported
to play an important role in the endometrium-related disease, such as
endometriosis [21]. Pyroptosis is initiated by inflammasomes, which active
caspases-1, cleave gasdermin D (GSDMD) and
then leading to the release of inflammatory cytokines [22]. GSDMD, caspase-1, and
IL-1
The treatment of severe endometrial injury induced intrauterine adhesion aims to
promote endometrial repair, prevent adhesion recurrence and restore the
fertility. Current treatments include surgical treatment (hysteroscopic
adhesiolysis or transcervical resection of adhesion (TCRA)), pharmacotherapy, and
stem cell therapy [1]. However, surgical treatment is limited to physically
separating the adhesions and may inadvertently cause further damage to the
remaining endometrial tissue [26]. Infection-induced inflammation results in the
damage of endometrial stem cells, compromising their regenerative capacity and
hindering the repair of the uterine cavity [27]. Therefore, pharmacologic
treatment is the priority in treatment of severe endometrial injury. Hydrogen
sulfide (H2S), as an endogenous gasotransmitter, is widely distributed in
many tissues and organs of the human body and contributes to the modulation of a
myriad of biological signaling pathway. Endogenous H2S is
predominantly synthesized by three key enzymes: CBS, CSE, and MST. Studies have
shown endogenous H2S generation systems have been found in female
reproductive system and dysregulation of the endogenous H2S
is involved in various pathophysiological processes in reproductive disease [4].
We have investigated the expression of endogenous H2S synthase and found
that CBS, CSE and MST significantly decreased in the uterus surrounding the
injury compared with the sham groups. A similar trend was observed in the
endogenous H2S levels and the expression of H2S-synthesizing enzymes in
the uterine tissue surrounding the injury. These results suggest that endogenous
H2S is involved in regulating the uterine microenvironment. To determine the
effect of maintaining hydrogen sulfide homeostasis on endometrial damage, we
observed the effects on H2S metabolism and cell pyroptosis after injury by
administering exogenous hydrogen sulfide. Our results showed that exogenous
H2S restored the hydrogen sulfide homeostasis after endometrial damage,
suggesting that exogenous H2S has a protective role in endometrial injury.
Our group previously shown that H2S significantly
inhibited traumatic brain injury-induced neuronal pyroptosis, suggesting that
there is a mutual relationship between hydrogen sulfide and pyroptosis [13]. In
this study, we have confirmed that pyroptosis is significantly upregulated after
endometrial injury. To further verify whether exogenous hydrogen sulfide
regulates the occurrence of pyroptosis after endometrial injury, we examined the
expression of GSDMD, Caspase-1, IL-1
Persistent chronic inflammation can lead to fibrosis, making endometrial recovery challenging [14, 28]. Disruption of the endometrial repair process can result in abnormal fibrosis, leading to uterine cavity occlusion and subsequent infertility. The endometrium plays a vital role in determining uterine receptivity, and a thin endometrium may compromise successful embryo implantation [29]. To further evaluate the effect of exogenous H2S on the endometrium function after injury, HE staining and Masson’s trichrome staining were used to quantify the thickness of endometrium, the number of glands, and the area of fibrosis. We found that exogenous H2S showed an increase in endometrial thickness and glands number. Compared to the control group, the fibrosis area was significantly increased after injury in the model group, while the fibrosis area in the H2S groups showed the greatest reduction in endometrial fibrosis area by Masson’s trichrome staining. These results suggested that H2S may be a key factor in the induction of tissue-specific repair processes. Our results also demonstrate that exogenous H2S can improve fertility efficiency and yield the good effect in injured endometrium in mice, suggesting that treatment with H₂S can enhance fertility and promote the recovery of uterine function after endometrial injuries. While H2S has shown promise as a potential treatment for infertility, it is not currently used in clinical practice. More research is needed to determine the optimal dose and delivery method of H2S for fertility treatment. Additionally, the safety of H2S for long-term use needs to be further investigated. Caspase-1 can also initiate apoptosis in cells that lack GSDMD [30]. Based on current results, it is difficult to rule out the role of apoptosis in endometrial injury. The relationship between different death pathways in endometrial injury should be investigated in the future research.
In conclusion, our study demonstrated that endogenous H2S metabolism and pyroptosis is involved in the pathological process of endometrial injury. This dual action of reducing inflammation and fibrosis while modulating pyroptosis pathways highlights the therapeutic potential of exogenous H₂S in enhancing fertility and treating uterine disorders.
All data points generated or analyzed during this study are included in this article, and no further underlying data is necessary to reproduce the results.
HZ, LYT, MYZ and HYS designed the research study. HYS, MYX, MCX, JX, JN, LZ and XHY performed the research and analyzed the data. All authors are credited with the editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The experiments performed in this study have been carried out according to the rules in the Guide for the Care and Use of Laboratory Animals adopted by the National Institutes of Health Animal Use and Care and Animal Research. The present study was approved by the Ethics Committee of Soochow University (SUDA20240911A04). The mice used in the experiment were obtained from the Experimental Animal Laboratory of Soochow University (SYXK (Su) 2021-0065). Informed consent was N/A.
We would like to express our gratitude to all those who helped us while writing this manuscript. Thanks to all the peer reviewers for their opinions and suggestions. The graphic abstract is created by PowerPoint using pictures from Servier Medical Art, by Servier (http://smart.servier.com). The pictures in the Fig. 1 are drawn by Ziqi Zhang.
The Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD); Jiangsu 333 High Level Talent Training Project (2022); the Jiangsu Maternal and Child Health Research Key Project (F202013); Gusu District Health Talent Training Project (2024145); Project of MOE Key Laboratory of Geriatric Diseases and Immunology (KJS2505).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG26554.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








