1 Department of Obstetrics, The International Peace Maternity and Child Health Hospital, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
2 Shanghai Key Laboratory of Embryo Original Disease, 200030 Shanghai, China
3 Department of Radiology, Shanghai Jiaotong University Affiliated Sixth People’s Hospital, 200233 Shanghai, China
4 Department of Radiology, The International Peace Maternity and Child Health Hospital, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
Abstract
Placenta previa (PP) combined with placenta accreta spectrum (PAS) is a serious obstetric condition that increases the risk of maternal and fetal complications. Early diagnosis is therefore crucial for improving outcomes. The aim of this study was to evaluate the accuracy of magnetic resonance imaging (MRI) combined with three serological markers (alpha-fetoprotein [AFP], creatine kinase [CK], and soluble fms-like tyrosine kinase-1 [sFlt-1]) for the diagnosis of PP with PAS.
This retrospective study included 243 patients with suspected PP and PAS treated at two hospitals between August 2018 and August 2023. Patients were divided into two groups based on surgical and pathological findings: PP+PAS (+) and PP+PAS (–). The serum levels of AFP, CK, and sFlt-1 were measured, and optimal diagnostic thresholds were determined using receiver operating characteristic (ROC) curve analysis. The diagnostic performance of MRI, serological markers, and a combination of the two was compared with that of surgical pathology.
Serum AFP, CK, and sFlt-1 levels were significantly higher in the PP+PAS (+) group compared to the PP+PAS (–) group. The optimal thresholds of AFP, CK, and sFlt-1 for the diagnosis of PP+PAS (+) were 58.49 U/mL, 168.25 U/mL, and 2.28 ng/mL, respectively. A combined diagnostic approach using MRI and serological markers showed good accuracy, with a Kappa coefficient of 0.850, indicating good agreement with surgical pathology.
Combining MRI with serological markers (AFP, CK, and sFlt-1) provides enhanced diagnostic accuracy for detecting PP with PAS as compared to either method alone. This approach can facilitate early diagnosis, improve clinical decision-making, and reduce the risks associated with surgical intervention.
Keywords
- placenta previa
- placenta accreta spectrum
- magnetic resonance imaging
- serological indicators
- diagnostic value
Placenta previa (PP) occurs when the placenta attaches to the lower segment of the uterus and fully or partially covers the cervical os [1, 2]. This condition often leads to vaginal bleeding in the second and third trimesters of pregnancy. Placenta accreta spectrum (PAS) represents a range of disorders whereby the placental villi abnormally invade the myometrium. Severe forms of PAS, such as placenta percreta, extend through the uterine wall and even into adjacent organs [3]. These conditions pose significant risks of massive hemorrhage during delivery, frequently necessitating hysterectomy to control the bleeding [4]. When PP is accompanied by PAS, the risks to maternal and fetal health are considerably heightened, highlighting the need for early and accurate diagnosis. Magnetic resonance imaging (MRI) is a non-invasive diagnostic tool that provides detailed images of the placenta and its relationship to surrounding tissues, making it useful for identifying PAS [5]. However, the sensitivity of MRI for the detection of less severe forms of placental invasion is limited [6]. This has prompted research into additional diagnostic methods, such as the use of serological biomarkers [7]. Several biomarkers have shown potential in the diagnosis of PAS due to their association with placental dysfunction. Alpha-fetoprotein (AFP) is a glycoprotein produced by the fetal liver and yolk sac, and elevated maternal serum levels of AFP have been associated with placental abnormalities, including PAS. Creatine kinase (CK), an enzyme involved in energy metabolism, is released into the maternal circulation when the placental tissue invades the myometrium, leading to increased levels in cases of PAS. Soluble fms-like tyrosine kinase-1 (sFlt-1), an anti-angiogenic factor, is also associated with placental pathologies, including preeclampsia and PAS. Elevated levels of sFlt-1 reflect endothelial dysfunction and abnormal placentation. Hence, the rationale for selecting AFP, CK and sFlt-1 as diagnostic markers for PAS lies in their biological roles in placental development and invasion [8]. These biomarkers provide insight into the extent of placental tissue invasion and vascular disruption, which are key features of PAS [9, 10]. Although MRI provides valuable anatomical detail, the combination of MRI with these biomarkers may improve diagnostic accuracy, especially in complex cases where imaging alone is inconclusive [11, 12]. The aim of this study was therefore to evaluate the combined diagnostic value of MRI and three serological biomarkers (AFP, CK, and sFlt-1) for the detection of PP with PAS. By integrating these diagnostic modalities, we sought to improve the early detection of high-risk pregnancies, thereby helping clinical decision-making and reducing the risks of maternal and neonatal complications.
This study retrospectively analyzed clinical data from 243 patients suspected of having PP in combination with PAS. The patients were treated between August 2018 and August 2023 at the International Peace Maternity and Child Health Hospital, and the Sixth People’s Hospital, both affiliated with the Shanghai Jiao Tong University School of Medicine. All patients underwent both MRI and serum biomarker testing (AFP, CK, sFlt-1) prior to surgical treatment.
Inclusion criteria for the study were: (1) singleton pregnancy; (2) complete
clinical data; and (3) availability of MRI and biomarker test results. Patients
were excluded if they had primary psychiatric disorders, significant organ
dysfunction, pregnancy complications, malignancies, poor MRI image quality,
immune disorders, or abnormal fetal development. A total of 177 cases were
confirmed as PP+PAS (+) based on surgical pathology, while 66 cases were
identified as PP+PAS (–) and served as the control group. The general data showed
no significant differences (p
| Group | Age (years) | Pre-pregnancy body mass index (BMI) (kg/m2) | Primigravid | Previous cesarean delivery | Placenta position | Centralized placenta previa | |
| Anterior wall | Posterior wall | ||||||
| PP+PAS (+) (n = 177) | 28.64 |
23.28 |
75 (42.37%) | 107 (60.45%) | 79 (44.63%) | 98 (55.37%) | 107 (60.45%) |
| PP+PAS (–) (n = 66) | 28.89 |
23.35 |
35 (53.03%) | 35 (53.03%) | 35 (53.03%) | 31 (46.97%) | 43 (65.15%) |
| t/ |
0.533 | 0.203 | 2.204 | 1.090 | 1.361 | 0.449 | |
| p-value | 0.594 | 0.839 | 0.138 | 0.296 | 0.203 | 0.839 | |
PP, placenta previa; PAS, placenta accreta spectrum.
Serum samples were collected from all patients before surgery. Enzyme-linked immunosorbent assay (ELISA, EK1175S, Liankebio, Hangzhou, Zhejiang, China) was used to measure AFP, CK, and sFlt-1 levels with the Multiskan FC Analyzer (1410101, Thermo Fisher Technology (China) Co., Ltd., Shanghai, China). MRI scans were performed with a PHILIPS 3.0T nuclear magnetometer (Philips Achieva, Philips Healthcare, Best, The Netherlands) to obtain axial, sagittal, and coronal T2-weighted sequences. Image quality was independently assessed by two radiologists, and any discrepancies were resolved through consultation with a third radiologist.
Data were analyzed using SPSS 23.0 (IBM Corp., Armonk, NY, USA). Categorical
variables were expressed as frequencies and percentages. Comparisons between
groups for categorical variables were performed using the Chi-square test or
Fisher’s exact test, where appropriate. The diagnostic efficacy of AFP, CK, and
sFlt-1 was evaluated using receiver operating characteristic (ROC) curves, with
optimal diagnostic thresholds determined using the Youden index. Agreement
between MRI findings, serological markers, and surgical pathology was assessed
using Kappa statistics, where a Kappa value
The PP+PAS (+) group had higher AFP, CK and sFlt-1 levels than the PP+PAS (–) group (Table 2).
| Group (n) | AFP (U/mL) | CK (U/mL) | sFlt-1 (ng/mL) |
| PP+PAS (+) (177) | 62.92 |
168.26 |
2.51 |
| PP+ PAS (–) (66) | 38.75 |
131.31 |
1.56 |
| t value | 10.125 | 7.734 | 9.830 |
| p-value |
AFP, alpha-fetoprotein; CK, creatine kinase; sFlt-1, soluble fms-like tyrosine kinase-1.
ROC curve analysis showed that the area under the curve (AUC) values of serum AFP, CK, and sFlt-1 for the diagnosis of PP+PAS (+) were 0.775, 0.759, and 0.793, with sensitivities of
51.4%, 49.7%, and 50.8%, specificities of 100.0%, 90.9%, and 100.0%, and
optimal thresholds of 58.49 U/mL, 168.25 U/mL, and 2.28 ng/mL, respectively (Fig. 1). Therefore, cases with a serum AFP level
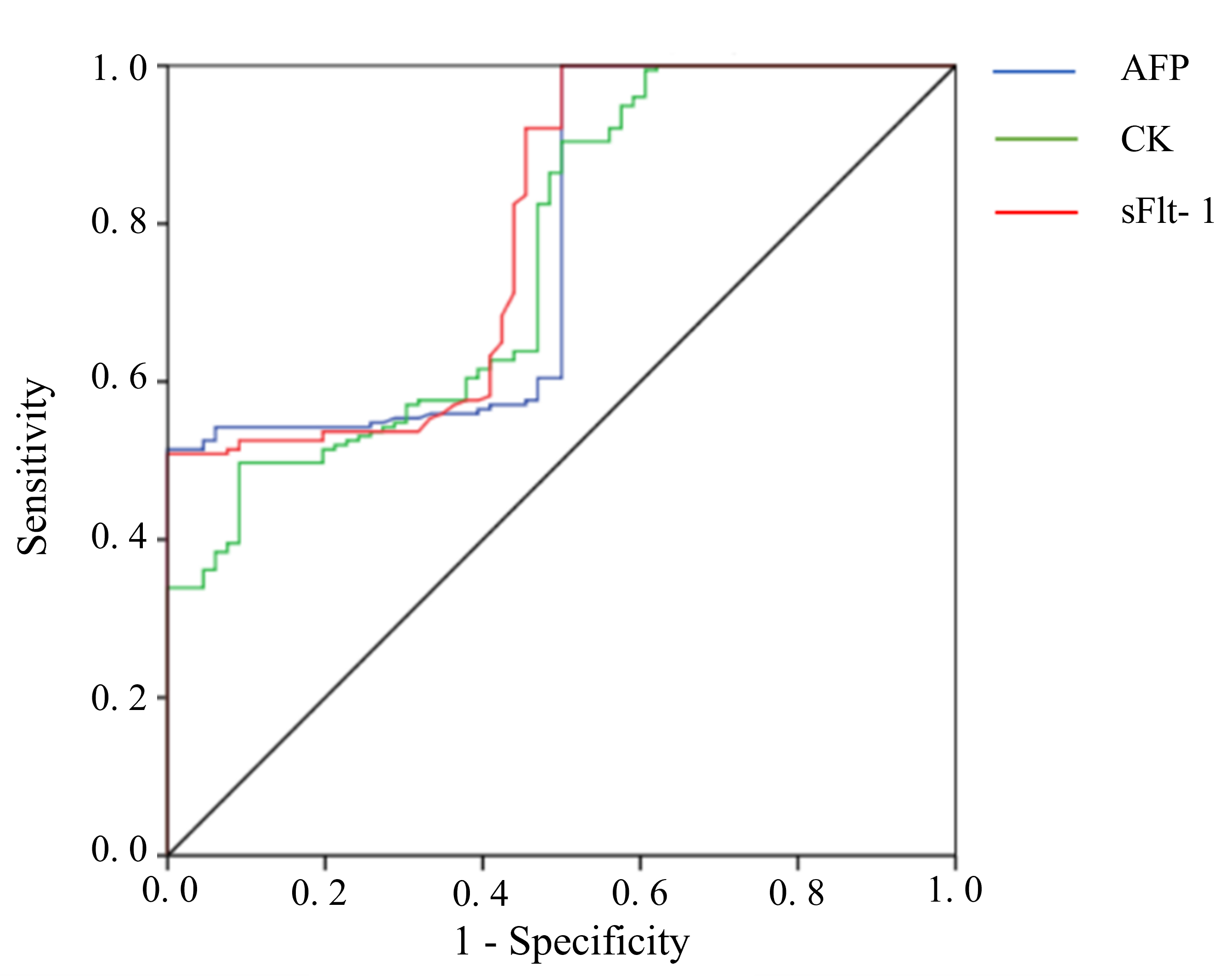 Fig. 1.
Fig. 1.
Receiver operating characteristic (ROC) curve of serologic indicators for PP+PAS (+).
Using surgical pathology as the gold standard, the accuracy of MRI for the diagnosis of PP+PAS (+) was 88.89% (216/243), the sensitivity was 90.40% (160/177), and the specificity was 84.85% (56/66). The accuracy of the combination of three serologic indicators was 88.48% (215/243), the sensitivity was 87.57% (155/177), and the specificity was 90.91% (60/66). The accuracy of the combination of the four features (MRI and three serological indicators) for the diagnosis of PP+PAS (+) was 94.24% (229/243), the sensitivity was 97.74% (173/177), and the specificity was 84.85% (56/66). Compared with surgical pathology examination, the Kappa value of MRI for the diagnosis of PP+PAS (+) was 0.728, thus showing fair agreement between the two. The Kappa value of the combined serologic indicators for the diagnosis of PP+PAS (+) was 0.729, again showing fair agreement with surgical pathology examination. However, the four combined indicators (MRI and three serological indicators) showed a Kappa value of 0.850 for the diagnosis of PP+PAS (+), thus providing the best agreement (Tables 3,4).
| Examination method | Surgical pathology results | All | ||
| Positive | Negative | |||
| MRI examination | + | 160 | 10 | 170 |
| – | 17 | 56 | 73 | |
| Combined serological indicators | + | 155 | 6 | 161 |
| – | 22 | 60 | 82 | |
| Quadrilateral system | + | 173 | 10 | 183 |
| – | 4 | 56 | 60 | |
| All | 177 | 66 | 243 | |
MRI, magnetic resonance imaging.
| Measurement of rank | Value | SD | T | p-value | |
| MRI examination | Kappa | 0.728 | 0.049 | 11.38 | |
| Combined serological indicators | Kappa | 0.729 | 0.047 | 11.508 | |
| Quadrilateral system | Kappa | 0.850 | 0.039 | 13.279 | |
SD, standard deviation. “T” in the table means one sample t-test.
Table 5 shows the levels of the three serological indicators in different PAS
groups. The levels were highest in the percreta group, followed by the increta
group, and lowest in the creta group (p
| Placenta group | n | AFP (U/mL) | CK (U/mL) | sFlt-1 (ng/mL) |
| Creta | 50 | 45.63 |
158.82 |
1.93 |
| Increta | 65 | 71.60 |
180.18 |
2.95 |
| Percreta | 62 | 94.12 |
218.61 |
3.80 |
| F-value | - | 142.63 | 32.93 | 56.34 |
| p-value | - |
Note: a p
Table 6 shows the comparison of MRI imaging features between patients with
different types of PAS. Significant differences in the following indicators were
found between the three types of PAS: bladder protrusion, convexity of the
uterine silhouette, and T2-weighted imaging (T2WI) placental ripple low signal shadow. A consistent
trend was observed with the serological indicators, with the percreta group
| Placenta group | n | Uneven intraplacental signaling | Increased and thickened vascularization in the placenta | Bladder protrusion | Poor demarcation of placenta and uterus | Convexity of the uterine silhouette | T2WI intraplacental striated low signal shadow |
| Creta | 50 | 50 (100.00%) | 50 (100.00%) | 28 (56.00%) | 50 (100.00%) | 31 (62.00%) | 30 (60.00%) |
| Increta | 65 | 65 (100.00%) | 65 (100.00%) | 48 (73.85%) a | 65 (100.00%) | 52 (80.00%) a | 51 (78.46%) a |
| Percreta | 62 | 62 (100.00%) | 62 (100.00%) | 57 (91.94%) a,b | 62 (100.00%) | 58 (93.55%) a,b | 58 (93.55%) a,b |
| - | - | - | 19.227 | - | 17.010 | 18.477 | |
| p-value | - | - | - | - |
Note: a p
PP occurs when the placenta is located in the lower uterine segment and partially or completely covers the cervical os [2]. It is the most common cause of vaginal bleeding in late pregnancy [3]. PP is also one of the most serious complications in the third trimester. PAS refers to a range of conditions where placental tissue invades the uterine myometrium to varying degrees. Clinically, PAS can result in severe postpartum hemorrhage, shock, and in many cases also hysterectomy, with an increased risk of postpartum infection [1, 6]. In severe cases, PAS can be life-threatening. The incidence of PP combined with PAS (PP+PAS) is estimated to range between 1% and 5%. Given the high mortality and morbidity associated with these conditions, early and accurate diagnosis is crucial for improving maternal and neonatal outcomes [4, 13].
Surgical pathology is considered to be the gold standard for diagnosing PP+PAS. However, in the present study the combination of MRI and serological markers demonstrated a Kappa value of 0.729 for the diagnosis of PP+PAS, indicating moderate agreement with surgical pathology. This highlights the need for more accurate diagnostic methods to improve detection. MRI is a non-invasive imaging technique that provides detailed visualization of the placenta’s location and morphology, as well as its relationship with the uterine wall [14, 15]. This technique can detect abnormal placental contact and infiltration into the myometrium, which is essential for diagnosing PAS [16]. When combined with serum biomarkers such as AFP, CK, and sFlt-1, MRI provides valuable diagnostic clues, aiding in the assessment of PAS before surgery [11, 17, 18]. This approach can potentially reduce surgical trauma and associated risks [19, 20, 21]. The present study showed a Kappa value of 0.850 for the diagnosis of PP+PAS when using the combination of MRI and serological markers. This result indicates a high degree of consistency with surgical pathology.
The high-resolution capabilities of MRI allow clear visualization of the extent and type of PAS, including the depth of placental invasion. Different forms of PAS exhibit distinct MRI features. For example, the primary distinction between partial and complete placental implantation lies in the size of the affected area, while adhesive and penetrating placentas involve different depths of myometrial invasion, each of which affects the MRI imaging characteristics [22]. In cases of placenta creta, placental villi adhere to the uterine muscle without penetrating the uterine wall, and typically show no significant impact on the bladder or uterine contour. In placenta increta, localized uterine protrusion may occur, but without bladder involvement. The most severe form of PAS, placenta percreta, can lead to uterine perforation and massive bleeding, often necessitating surgical intervention. In such cases, MRI can detect bladder protrusion and uterine contour abnormalities [23, 24].
In the current study, significant differences between the different PAS groups were observed in terms of bladder protrusion, uterine contour abnormalities, and T2-weighted imaging (T2WI) low-signal shadows. These differences were positively correlated with the depth of placental invasion, with the placenta percreta group showing the most pronounced features, followed by the placenta increta and placenta creta groups. Our results further highlight the importance of MRI in accurately diagnosing the type and severity of PAS. T2WI is particularly useful in identifying low-signal shadows within the placenta. These may be caused by factors such as fibrous tissue proliferation, vascular proliferation, or inflammatory reactions. Such imaging characteristics are likely to vary according to the type of PAS, although further research is required to elucidate the underlying mechanisms (Fig. 2). This study is the first to explore the value of combining MRI with serological markers for the diagnosis of PP with PAS. Our findings demonstrate that a combined approach can increase the diagnostic accuracy, thereby improving clinical decision-making in the management of pregnancies complicated by PP+PAS.
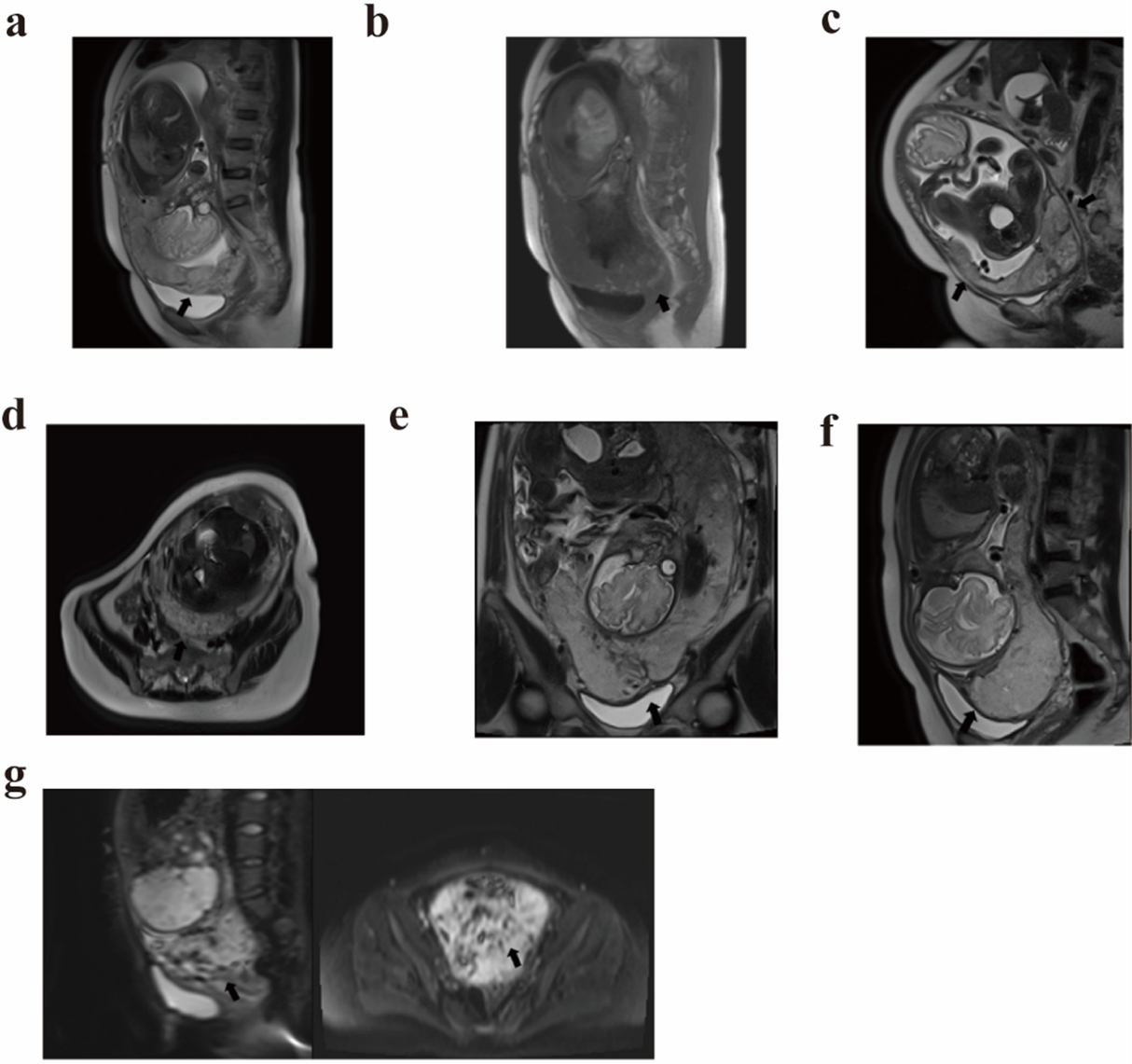 Fig. 2.
Fig. 2.
Representative MRI images of placenta previa with placenta accreta spectrum. (a) Sagittal T2W showing the placenta located in the anterior wall of the uterus, completely covering the internal orifice and reaching the posterior wall, with interruption of the continuity of the myometrium in the anterior wall, and tight adhesion of the placenta to the uterus. (b) Sagittal T1W showing abundant and awry blood vessels in the lower uterine segment. (c) Sagittal T2W showing the placenta located in the lower wall of the uterus, completely covering the internal os of the cervix. The continuity of the myometrium of the anterior and right posterior walls of the uterus was interrupted. (d) Transverse T2W showing a thin myometrium and interruption of continuity in the right posterior wall of the uterus. The placenta was tightly adhered to the uterus. (e) Coronal T2W showing that the signal in the lower part of the placenta was not homogeneous. The lower part of the muscle layer of the anterior wall of the uterus was poorly demarcated from the lower part of the uterine wall, and protruded downward to the bladder without penetrating it. (f) Sagittal T2W showing the placenta was poorly demarcated from the lower muscular layer of the anterior wall of the uterus, and protruding downward into the bladder without penetrating it. (g) Sagittal and transverse diffusion-weighted imaging (DWI) sequences showing abnormal and disorganized vascularization of the placenta and placental interuterine wall. The placenta was implanted in the myometrium above the endocervical opening, and in the anterior and posterior walls of the lower uterine segment and the lateral wall.
The combination of MRI and serological biomarkers (AFP, CK, sFlt-1) offers a reliable and non-invasive method for diagnosing PP with PAS. This approach enhances early detection and improves clinical outcomes by facilitating timely interventions. Future studies should explore the potential of integrating additional biomarkers and advanced imaging techniques to further refine the diagnostic accuracy of complex obstetric conditions such as PAS.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
YW, ZH, and BX designed the research study. YW and YH performed the research. BX provided help and advice on the MRI imaging. XX, ZH analyzed the data. XX performed the statistical analysis and interpretation of the results. All authors contributed to the manuscript revision, and all approved the final version. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki. The protocol was approved by the Ethics Committees of the International Peace Maternity and Child Health Hospital and the Sixth People’s Hospital, both affiliated with Shanghai Jiaotong University School of Medicine ((GKLW)2020-03 and 2023YFF0722204).
We would like to express our gratitude to all those who helped us during the writing of this manuscript. We also thank the peer reviewers for their valuable comments and suggestions.
This project was supported by the Shanghai Municipal Health Commission research project (Grant No.: 20194Y0050).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


