1 Clinical Medical College, Guizhou Medical University, 550004 Guiyang, Guizhou, China
2 Department of Gynecological, The Affiliated Hospital of Guizhou Medical University, 550001 Guiyang, Guizhou, China
†These authors contributed equally.
Abstract
Perimenopausal syndrome (PMS) significantly impacts the quality of life in women during the perimenopausal phase. Despite being the primary treatment, hormone replacement therapy has notable limitations. Consequently, dietary interventions, particularly low-fat diets, have garnered international attention as an alternative.
The prospective Randomized Controlled Trial (RCT), approved by the Ethics Committee of the Affiliated Hospital of Guizhou Medical University, enrolled 62 perimenopausal women. Participants were divided into two groups: a low-fat diet group and a menopausal hormone therapy (MHT) group, with interventions lasting eight weeks. Both groups underwent assessments for perimenopausal symptoms, lipid profile, and oxidative stress markers, including superoxide dismutase (SOD), malondialdehyde (MDA), and reactive oxygen species (ROS), before and after the interventions. Additionally, DNA methylation levels of the PDE4C and NPAS2 genes were measured in 10 women from the low-fat diet group before and after the intervention.
Both the low-fat diet and hormone supplementation improved specific perimenopausal symptoms, reduced specific lipid levels, and had a positive effect on oxidative stress markers. The low-fat diet proved more effective than hormone replacement in improving the serum lipid profile and also resulted in reductions in methylation at specific sites of the PDE4C and NPAS2 genes.
A low-fat diet may alleviate perimenopausal symptoms, lower lipid levels, and alter gene methylation sites.
The study has been registered on https://www.chictr.org.cn/ (registration number: ChiCTR2400084007).
Keywords
- dietary interventions
- DNA methylation
- low-fat diet
- NPAS2
- oxidative stress markers
- PDE4C
- perimenopausal syndrome
Perimenopausal syndrome (PMS) encompasses a range of physical and psychological symptoms in women, triggered by estrogen fluctuations around menopause [1, 2]. The primary etiological factors include hormonal imbalances and cytokine disorders [3, 4, 5]. While hormone replacement therapy remains the predominant treatment, its effectiveness is limited by contraindications and potential adverse effects [6]. Extended use increases the risk of hormone-dependent malignancies, particularly endometrial and breast cancers [7, 8].
Estrogens play a pivotal role in cardiovascular protection by enhancing lipoprotein structure, reducing low-density lipoprotein (LDL) cholesterol levels, increasing high-density lipoprotein (HDL) cholesterol levels, and combatting atherosclerosis [9]. The notable decline in estrogen during perimenopause triggers various pathways, with oxidative stress emerging as a prominent pathway. Consequently, perimenopausal women often exhibit increased oxidative and inflammatory response [10, 11]. This predisposes them to a variety of ailments, including perimenopausal conditions such as cardiovascular and metabolic diseases [12, 13]. Additionally, DNA methylation, an epigenetic modification influenced by external factors, is gaining prominence in aging studies [14, 15, 16]. Changes in DNA methylation patterns in leukocytes and serum DNA may serve as indicators of menopausal age and correlate with the progression of menopausal [17, 18].
During perimenopause and menopause, hormonal changes significantly decrease women’s basal metabolic rate [19, 20], which may potentially lead to a weight gain of approximately 2 to 2.5 kilograms (kg) over a period of three years [20]. The Mediterranean diet, known for its anti-inflammatory and antioxidant properties [21], has been extensively proven to improve the health of perimenopausal women [22, 23, 24]. A recent study demonstrated that high adherence to the Mediterranean diet is associated with healthier body composition, characterized by lower body mass index (BMI), reduced fat mass, and decreased visceral adipose tissue. This is particularly beneficial during perimenopause, a period when women are prone to weight gain and changes in body composition [22]. However, many women find it challenging to maintain long-term adherence to this diet. Moderate adherence to the Mediterranean diet does not significantly reduce cardiometabolic risks, whereas high adherence provides notable cardiovascular protection [25]. Some components of the Mediterranean diet, such as high-quality olive oil, seafood, and nuts, may be expensive in some regions, posing affordability challenges for families with lower economic status [26, 27]. On the other hand, low-fat diets have been shown to effectively reduce oxidative stress [28]. Conversely, high-fat diets have been reported to exacerbate oxidative stress, thereby contributing to multiple diseases [28, 29, 30]. A low-fat diet is a simple, cost-effective, and non-invasive remedy for perimenopausal symptoms. This research aims to investigate the effectiveness of a low-fat diet in alleviating perimenopausal symptoms by modulating oxidative stress, DNA methylation, and associated pathways.
We conducted a prospective Randomized Controlled Trial (RCT) involving 82 women from the Menopausal Health Clinic at the Affiliated Hospital of Guizhou Medical University. The study protocol has been registered at https://www.chictr.org.cn/ (ChiCTR2400084007). The Ethics Committee of the Affiliated Hospital of Guizhou Medical University approved this study (ethics approval number: GZWJ2022-149). All participants provided signed informed consent.
For our study, specific inclusion and exclusion criteria were defined for each group. For the low-fat diet group, the inclusion criteria were as follows: (1) no prior use of hormone supplementation; (2) informed consent obtained from both the patient and their family; and (3) permanent residency in Guiyang City. The exclusion criteria for this group include: (1) presence of hepatic, pulmonary, or renal insufficiency; and (2) severe internal medical conditions. For the menopausal hormone therapy (MHT) group, the inclusion criteria were: (1) absence of organic diseases and mental disorders; and (2) no contraindications to hormone replacement therapy and willingness to undergo treatment. The exclusion criteria were: (1) use of hormone replacement, traditional Chinese medicine, or other treatments in the past three months for alleviating perimenopausal symptoms; (2) presence of serious gynecological diseases such as hypertension, diabetes, dyslipidemia, and malignant tumors; and (3) history of breast or ovarian tumors, or past ovarian resection.
We used the Modified Kupperman Index (MKI) to assess the intensity and frequency
of perimenopausal symptoms. This tool, originally developed by Kupperman
et al. [31] in the 1950s and widely adopted in our region in a modified version,
consists of 13 items, each with a different weighting. It yields a total score
ranging from 0 to 63, where higher scores indicate more severe symptoms. Scores
are interpreted as follows:
Demographic and lifestyle data of participants from both groups were collected prior to the intervention. This data included age, marital status, ethnicity, height, weight, BMI, education level, monthly income, employment status, family relationships, smoking habits, alcohol consumption, and dietary habits. This information was organized in tabular form and recorded before the intervention began.
Fasting blood samples were collected and sent to the laboratory department of the Affiliated Hospital of Guizhou Medical University for immediate testing to assess lipid levels, including triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), apolipoprotein A1 (ApoA1), and apolipoprotein B (ApoB). Oxidative stress marker assays were performed according to the specified kit (Vazyme-innovation in enzyme technology, Nanjing, Jiangsu, China) procedures.
Given their critical roles in biological aging and the regulation of the biological clock, we selected the methylation status of PDE4C and NPAS2 for detection. The methylation status of PDE4C is associated with biological age, indicating it as a potential biomarker for aging and age-related diseases [32]. This association is crucial for understanding and potentially intervening in age-related physiological changes and diseases. NPAS2, a circadian clock gene, plays a key role in regulating the body’s internal circadian rhythm [33]. This regulation is vital for maintaining various biological rhythms, including sleep-wake cycles, hormone release, and metabolic processes. To accurately measure these DNA methylation levels, we used specialized assay kits (Vazyme-innovation in enzyme technology, Nanjing, Jiangsu, China).
The experimental data were statistically analyzed using R 4.1.3 software (R
Foundation for Statistical Computing, Vienna, Austria). Data conforming to a
normal distribution were described by mean
After excluding 18 participants who met the exclusion criteria, the remaining participants were randomly assigned to groups in this prospective RCT. 32 individuals were initially allocated to both the low-fat diet and MHT groups. However, 2 participants from the diet group withdrew, resulting in 30 participants by the end of the study. Table 1 shows that the pre-intervention baseline characteristics were comparable between the two groups in terms of age, marital status, race, height, weight, BMI, education, monthly income, work status, familial relationships, smoking habits, alcohol consumption, and dietary patterns. These findings suggest that the groups were well-matched for the concurrent control and non-inferiority study design.
| Intervention method | p-value | |||
| Low-fat diet (N = 30) | MHT (N = 32) | |||
| Age (years) | 50 (48.75, 52) | 50 (49, 51) | 0.875 | |
| Weight (kg) | 58.50 (54.00, 62.25) | 58.00 (53.00, 62.75) | 0.724 | |
| Height (cm) | 159.63 |
158.94 |
0.465 | |
| BMI (kg/m2) | 23.04 |
23.04 |
0.997 | |
| Married, n (%) | 0.205 | |||
| Yes | 24 (80.00%) | 21 (65.63%) | ||
| No | 6 (20.00%) | 11 (34.38%) | ||
| Race, n (%) | 0.891 | |||
| Han | 23 (76.67%) | 25 (78.13%) | ||
| Others | 7 (23.33%) | 7 (21.88%) | ||
| Education, n (%) | 0.562 | |||
| Upper secondary or less | 4 (13.33%) | 6 (18.75%) | ||
| Tertiary | 26 (86.67%) | 26 (81.25%) | ||
| Smoking status, n (%) | 0.928 | |||
| Yes | 3 (10.00%) | 4 (12.50%) | ||
| No | 27 (90.00%) | 28 (87.50%) | ||
| Alcohol abuse, n (%) | 0.659 | |||
| Yes | 9 (30.00%) | 8 (25.00%) | ||
| No | 21 (70.00%) | 24 (75.00%) | ||
| Employment, n (%) | 0.878 | |||
| Yes | 23 (76.67%) | 24 (75.00%) | ||
| No | 7 (23.33%) | 8 (25.00%) | ||
| Income (RMB/MS), n (%) | 0.990 | |||
| 3 (10.00%) | 3 (9.38%) | |||
| 3000–5000 | 10 (33.33%) | 12 (37.50%) | ||
| 5000–10,000 | 13 (43.33%) | 13 (40.63%) | ||
| 4 (13.33%) | 4 (12.50%) | |||
Note: data that conformed to the normal distribution were expressed as mean
The mean score on the MKI scale for the dietary intervention group was 25.47
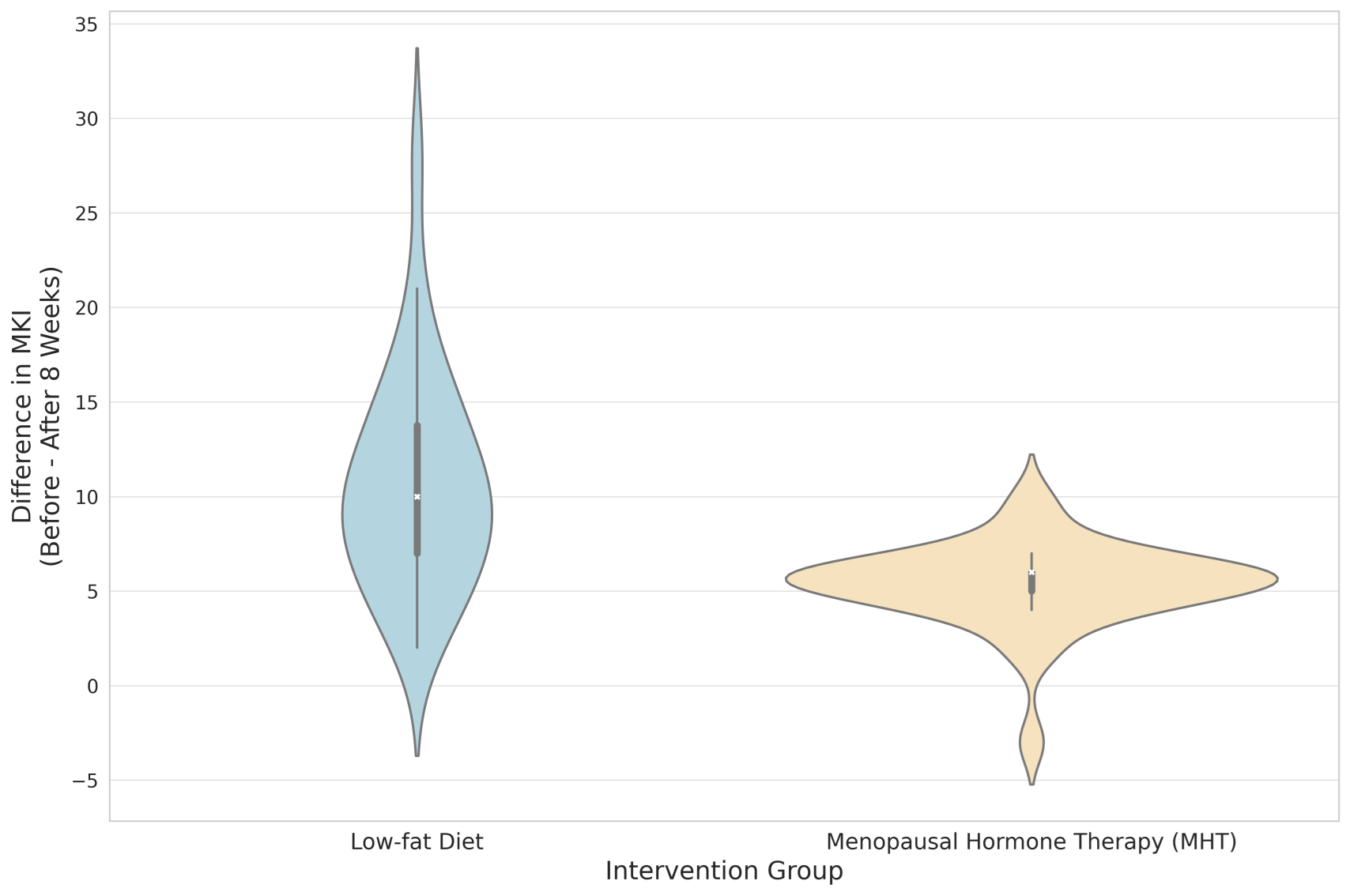 Fig. 1.
Fig. 1.
Difference in MKI before and after 8 weeks of intervention. The violin plots represent the difference in MKI scores (pre-intervention minus post-8-week intervention) for the two groups: low-fat diet and MHT. The width of the plot indicates the density of the data points. MKI, Modified Kupperman Index; MHT, menopausal hormone therapy.
| Low-fat diet | p-value* | MHT | p-value* | p-value** | |||
| Pre-test | Post-test | Pre-test | Post-test | ||||
| MKI | 25.47 |
14.93 |
25.19 |
19.84 |
0.003 | ||
| HDL-C | 1.48 |
1.65 |
0.086 | 1.45 |
1.40 |
0.422 | 0.008 |
| LDL-C | 2.49 |
1.91 |
2.69 |
2.52 |
0.018 | ||
| TG | 1.93 |
1.04 |
2.48 |
1.88 |
|||
| TC | 5.89 |
4.15 |
5.81 |
5.31 |
0.006 | ||
| ApoA1 | 1.55 |
1.78 |
1.48 |
1.47 |
0.834 | ||
| ApoB | 0.94 |
0.73 |
0.93 |
0.83 |
0.035 | 0.010 | |
| Lp(a) | 126.89 |
211.52 |
165.42 |
159.47 |
0.001 | ||
| ROS | 4960.38 |
4419.66 |
0.027 | 4934.30 |
4466.51 |
0.415 | |
| MDA | 3.85 (5.38, 2.73) | 2.10 (3.42, 1.41) | 3.62 (5.31, 3.17) | 2.20 (3.48, 1.42) | 0.239 | ||
| SOD | 10.81 |
14.06 |
10.42 |
15.40 |
|||
Note: *: paired t-test was used to calculate the within-group pre- and post-intervention differences. **: independent t-test was used to calculate the differences between groups post-intervention. The data for the variable MDA did not follow a normal distribution, so we used median and IQR for description and the Wilcoxon rank-sum test for analysis. Units for HDL-C, LDL-C, TG, and TC are in mmol/L; units for ApoA1 and ApoB are in g/L. MHT, menopausal hormone therapy; MKI, modified Kupperman index; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TG, triglycerides; TC, total cholesterol; ApoA1, apolipoprotein A1; ApoB, apolipoprotein B; Lp(a), lipoprotein(a); ROS, reactive oxygen species; MDA, malondialdehyde; SOD, superoxide dismutase.
| Low-fat diet | MHT | |||
| Cohen’s d with 95% CI | Corrected Cohen’s d | Cohen’s d with 95% CI | Corrected Cohen’s d | |
| MKI | –1.51 (–1.95, –1.08) | –1.49 | –0.77 (–0.89, –0.64) | –0.76 |
| HDL-C | 0.47 (0.07, 0.86) | 0.46 | –0.21 (–0.56, 0.14) | –0.21 |
| LDL-C | –0.93 (–1.24, –0.62) | –0.92 | –0.60 (–0.85, –0.36) | –0.60 |
| TG | –1.67 (–2.36, –0.98) | –1.65 | –1.16 (–1.46, –0.86) | –1.14 |
| TC | –1.57 (–2.12, –1.03) | –1.55 | –0.70 (–0.95, –0.45) | –0.69 |
| ApoA1 | 1.01 (0.72, 1.30) | 1.00 | –0.07 (–0.34, 0.21) | –0.07 |
| ApoB | –0.97 (–1.37, –0.56) | –0.96 | –0.50 (–0.78, –0.22) | –0.49 |
| Lp(a) | 1.07 (0.55, 1.59) | 1.06 | –0.07 (–0.56, 0.42) | –0.07 |
| ROS | –0.42 (–0.50, –0.35) | –0.42 | –0.40 (–0.50, –0.29) | –0.39 |
| MDA | –1.04 (–1.25, –0.83) | –1.03 | –0.99 (–1.28, –0.70) | –0.98 |
| SOD | 1.51 (1.14, 1.88) | 1.49 | 3.39 (2.42, 4.36) | 3.35 |
Note: this table presents the Cohen’s d values along with their 95% confidence interval (95% CI) for various biomarkers in the low-fat diet and MHT groups. Additionally, the corrected Cohen’s d values are provided. Units for HDL-C, LDL-C, TG, and TC are in mmol/L; units for ApoA1 and ApoB are in g/L. MHT, menopausal hormone therapy; MKI, modified Kupperman index; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TG, triglycerides; TC, total cholesterol; ApoA1, apolipoprotein A1; ApoB, apolipoprotein B; Lp(a), lipoprotein(a); ROS, reactive oxygen species; MDA, malondialdehyde; SOD, superoxide dismutase.
Lipid metabolism levels in the low-fat diet group and the MHT group were
compared before and after treatment by assessing serum levels of TG, TC, HDL-C,
LDL-C, ApoA1, and ApoB. Both interventions showed significant improvements in
serum TG, TC, LDL-C, and ApoB (p
A comparative analysis of aging-related biomarkers, including superoxide dismutase (SOD) activity, malondialdehyde (MDA)
content, and reactive oxygen species (ROS) content, was conducted for the low-fat diet group and MHT group
before and after treatment (Fig. 2 and Table 2). The data, as presented in the
Fig. 2 and Table 2, indicate that both the low-fat diet nor the MHT group
significantly reduced ROS content. However, both groups exhibited significant
improvements in SOD activity and a reduction in MDA content (p
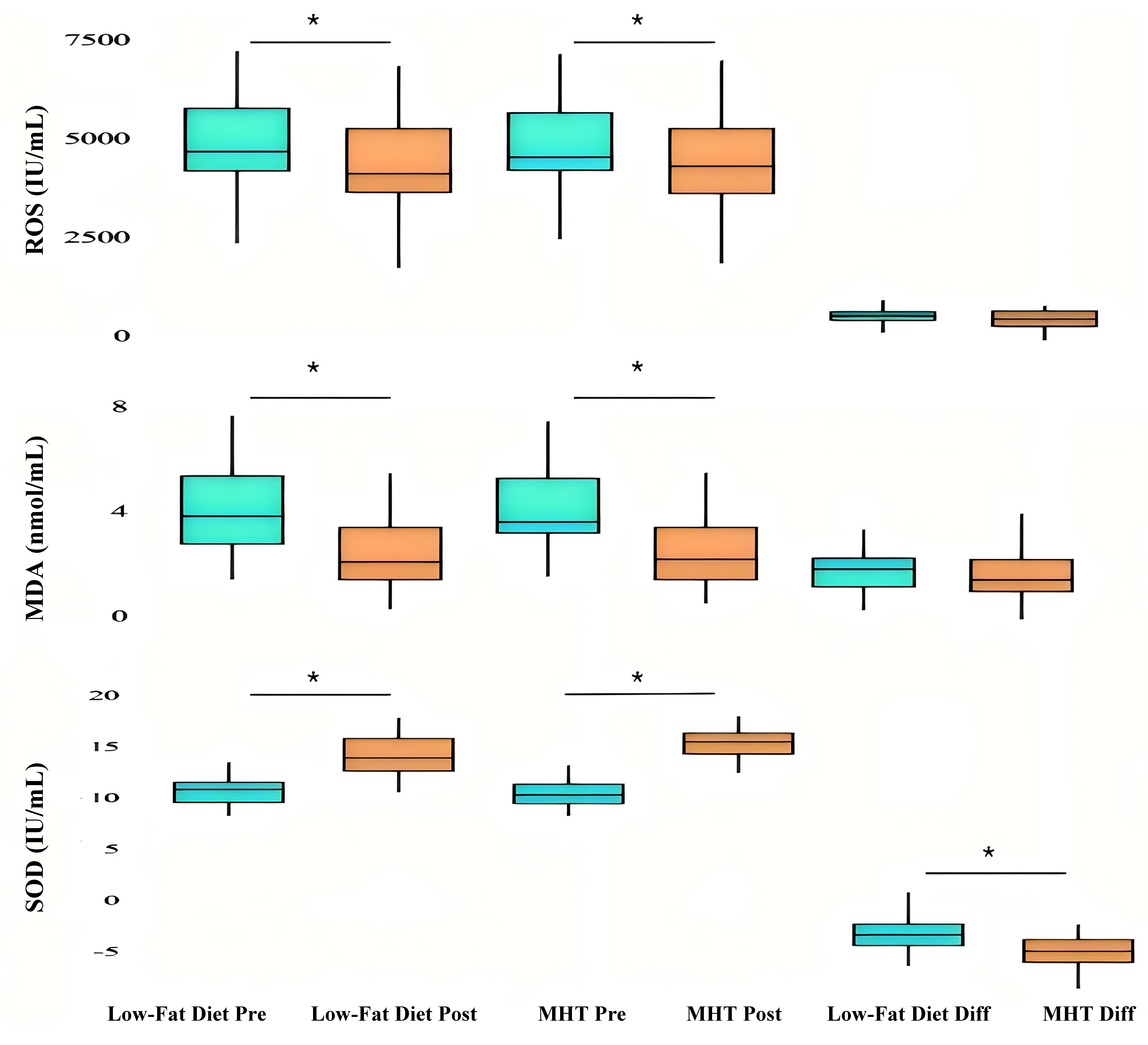 Fig. 2.
Fig. 2.
Comparative analysis of ROS, MDA, and SOD levels before and after intervention. Note: *Represents a significant difference. The boxes show the interquartile range of the data, while the upper and lower “whiskers” of the boxes indicate the range of variation in the data, excluding potential outliers. Low-fat diet Diff, MHT Diff refers to the difference between pre- and post-intervention. ROS (IU/mL): reactive oxygen species level. MDA (nmol/mL): malondialdehyde level, a marker of oxidative stress. SOD (IU/mL): superoxide dismutase activity, an important antioxidant defense in nearly all cells exposed to oxygen. MHT, menopausal hormone therapy.
In a preliminary study, the methylation levels of the PDE4C and
NPAS2 genes were analyzed in 10 perimenopausal women from the dietary
intervention group. While there were no significant overall changes in
methylation levels, site-specific analyses were performed. For the PDE4C
gene, sites 159, 226, 234, 98, 112, 193, and 195 showed significant methylation
differences both pre-intervention and at 8 weeks post-intervention (p
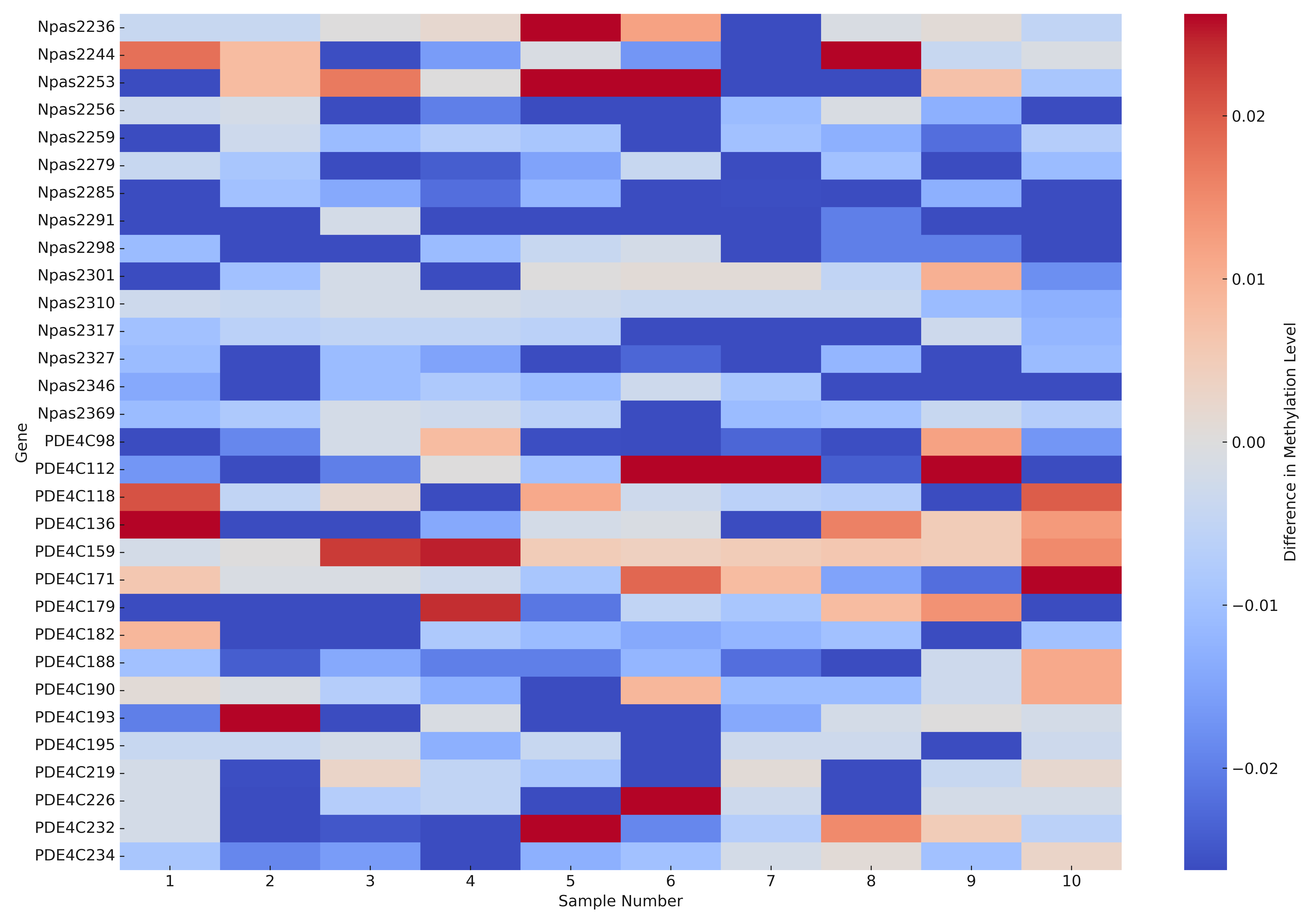 Fig. 3.
Fig. 3.
Differential methylation levels in NPAS2 and PDE4C gene sites pre- and post-intervention for low-fat diet group. This heatmap visualizes the differences in methylation levels at specific gene sites associated with NPAS2 and PDE4C for the low-dat diet group. The color intensity represents the magnitude of the difference in methylation levels between pre- and post-intervention, where warmer colors indicate increased methylation post-intervention and cooler colors indicate decreased methylation post-intervention.
Perimenopause represents a significant physiological transition as women progress from their reproductive years to old age [34]. In China, a substantial proportion of perimenopausal women, ranging from 60% to 80%, experience perimenopausal syndrome. Abnormal body fat distribution among this population, combined with excessive synthesis and secretion of active substances such as adipocytokines and ROS, often leads to complications such as obesity and depression, thereby prolonging the duration of these conditions [35]. While MHT is widely acknowledged as the most effective treatment for mucopolysaccharidosis (MPS), its use during the perimenopausal period has been proven to ameliorate associated symptoms [36, 37]. Significantly, hormone replacement therapy has been demonstrated to reduce the risk of colorectal cancer in healthy postmenopausal women. However, meta-analyses indicate that prolonged use of hormone medications increases the risk of hormone-dependent cancers, including endometrial and breast cancer [38]. Given these risks, alternative approaches are crucial for perimenopausal women who have contraindications to hormone therapy. Dietary intervention emerges as a viable, cost-effective, and convenient alternative that is comparable in efficacy to hormone supplementation for alleviating perimenopausal symptoms.
Our study findings reveal that before dietary intervention and hormone supplementation treatment, most participants exhibited severe and moderate scores on the MKI scale. However, following an 8-week intervention, scores predominantly shifted to moderate and mild categories, indicating a significant improvement in perimenopausal symptoms. The diet used in this research, which includes soy products, provides supplementary estrogens that helped mitigate perimenopausal symptoms. The onset of symptoms in perimenopausal women, such as hot flashes, sweating, palpitations, and insomnia, can be attributed to fluctuating estrogen levels and progesterone deficiencies. Hormone supplementation addresses these imbalances, thereby enhancing the overall well-being of perimenopausal women. This research evaluates the efficacy of both dietary interventions and hormone supplementation treatments, concluding that each approach effectively alleviates the severity of perimenopausal symptoms.
After menopause, the diminished protective metabolic influence of estrogen contributes to an increase in metabolic syndrome (MetS) occurrences among perimenopausal women. According to a meta-analysis by Hallajzadeh et al. [39], nearly 40% of postmenopausal women globally exhibit MetS symptoms, marking them as a high-risk group. Our study findings indicate that both dietary interventions and hormone supplementation therapies are effective in reducing serum TG, TC, LDL-C, and ApoB levels. Notably, dietary intervention surpassed hormone supplementation in lowering serum TG, TC, LDL-C, and ApoB levels, and in elevating HDL-C and ApoA1 levels. This aligns with a study that reported significant reductions in serum TG, TC, LDL-C, and ApoB levels after hormone supplementation therapy, alongside notable increases in HDL-C and ApoA1 [40]. These findings are consistent with our study results. Recognizing its effectiveness, dietary therapy is widely considered a primary defense against the onset and progression of MetS [41]. However, a contrasting study suggests that hormone supplementation leads to a more pronounced reduction in serum TG and LDL-C concentrations, alongside a marked increase in HDL-C, compared to dietary interventions [42]. This disparity might arise from variations in the recipes of dietary intervention or differences in the duration of interventions. Future endeavors should therefore emphasize refining the content of recipes and prolonging the duration of interventions to achieve optimal outcomes.
The escalation of oxidative stress, coupled with defects in the antioxidant system, is intricately linked to the onset and progression of various diseases. Therefore, enhancing the body’s antioxidant capability and mitigating oxidative damage are crucial for decelerating the aging process. Estrogen boosts the activity of mesangial cells by upregulating the expression of vascular endothelial growth factor, while also influencing antioxidant synthesis [43]. Our research findings indicate that both dietary intervention and hormone supplementation therapies are effective in increasing SOD activity and reducing MDA levels. Notably, hormone supplementation therapy exhibited superior efficacy in enhancing SOD activity compared to dietary intervention. A study conducted by Yan et al. [44] investigated the effects of Chuanxiong extract on D-galactose-induced aging mice, demonstrating increase SOD activity and reduced MDA content in mouse tissues, thereby highlighting the significance of oxidative stress modulation.
The presented research validates that dietary interventions can mitigate oxidative stress markers and contribute to delaying aging, consistent with the outcomes of this investigation. We observed that hormone supplementation therapy exhibited superior efficacy in improving SOD activity compared to dietary intervention. To date, no studies have discussed the comparative impacts of dietary intervention and hormone supplementation therapy on SOD activity. Considering the relatively brief intervention duration in this study and the gradual effectiveness of dietary interventions, future investigations should consider longer intervention periods. This would allow a more comprehensive evaluation of the influence of both dietary and hormonal treatments on oxidative stress markers, specifically those associated with the aging process. Such insights might help elucidate the observed superiority of hormone supplementation therapy over dietary intervention in enhancing SOD activity.
A decrease in the prevalence of associated diseases within the population suggests a link between DNA methylation and longevity [45]. Recently, DNA methylation age (DNAmAge), determined using the human DNA methylation map, has emerged as a precise measure for assessing an individual’s biological age [46]. Epigenetic age can ascertain and predict the impact of various intrinsic and extrinsic factors on human aging processes. Considering the reversible nature of epigenetic changes and their significance, targeting aging or age-associated diseases through epigenetic regulation has emerged as a novel therapeutic avenue. Neuronal PAS domain protein 2 (NPAS2) [47], a member of the basic helix-loop-helix PAS domain transcription factors, is predominantly expressed in mammals. NPAS2 is recognized as the most significant clock gene identified globally and primarily functions as a transcription factor found in the mammalian forebrain [48]. Its diminished expression has been linked to neurodegenerative diseases [49]. Furthermore, Phosphodiesterase 4C (PDE4C), a crucial member of the Phosphodiesterase 4 (PDE4) family, is ubiquitously distributed among various inflammatory and immune cells. It acts on intracellular targets to mitigate inflammation [50]. Notably, research has unveiled a direct correlation between PDE4C methylation levels and age [51].
In this study, the methylation status of the PDE4C and NPAS2 genes was assessed in 10 perimenopausal women undergoing dietary intervention. After 8 weeks, no significant alterations were observed in the overall DNA methylation levels of these genes. However, upon detailed examination of specific methylation sites, it was revealed that certain sites exhibited a significant reduction in methylation, while others remained largely unchanged. Given that methylation levels can serve as an indirect marker of aging, these findings suggest that dietary interventions may influence DNA methylation at specific sites within the PDE4C and NPAS2 genes, potentially contributing to the deceleration of aging. Previous studies have shown varying DNA methylation levels in human adipose tissue in response to different dietary patterns [52]. Additionally, international research indicates that dietary and lifestyle interventions can modulate DNAmAge [53]. Nonetheless, this study presents several limitations regarding DNA methylation assessment: it primarily focuses on populations with a higher BMI in international studies, whereas Chinese populations generally exhibit lower BMIs; there are genetic variations between international and Chinese populations; and the study addresses only a subset of the vast number of methylation sites available for investigation.
Both the low-fat diet and MHT were effective in improving certain perimenopausal symptoms, reducing specific lipid levels, and positively influencing oxidative stress markers. The low-fat diet was more effective than MHT in improving the serum lipid profile and also resulted in decreased methylation at specific sites of the PDE4C and NPAS2 genes. This study supports a low-fat diet as a key component of lifestyle management for perimenopausal women, especially for those with contraindications to MHT. Further investigation is required to assess the impact of body composition and other variables.
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy concerns related to human participant data.
XZ was central to patient recruitment, the completion of questionnaires, and the collection of specimens. XZ also conducted all laboratory tests and authored the majority of the manuscript. XL and KL handled the statistical analysis and contributed to part of the manuscript. MX and LZ provided technical advice, supervised the statistical analysis process, and made critical revisions to the manuscript. LY orchestrated and directed the project, meticulously reviewing the manuscript before giving his final approval. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study adhered to the principles of the Declaration of Helsinki. The Ethics Committee of The Affiliated Hospital of Guizhou Medical University granted approval for this study (ethics approval number: GZWJ2022-149). Informed consent was obtained from all participants prior to the study.
The authors thank all the participants of this work.
This work was supported by the Health Commission of Guizhou Province (gzwkj2022-149), and the Guizhou Provincial Science and Technology Department (qkhjc-ZK [2023] yb351).
The authors declare no conflict of interest.
AI and AI-assisted Technologies in the Writing Process: During the preparation of this work the authors used ChatGpt-3.5 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



