1 Department of Obstetrics and Gynecology, Jinhua Maternity and Child Health Care Hospital, 321000 Jinhua, Zhejiang, China
2 Department of Obstetrics and Gynecology, Affiliated Jinhua Hospital, Zhejiang University School of Medicine, 321000 Jinhua, Zhejiang, China
Abstract
Background: The aim of the present study was to evaluate the clinical
efficacy of a vaginal dilator in patients with cervical cancer
after radiotherapy. Methods: A total of 128 patients with cervical
cancer (stages I–III) after radical hysterectomy combined with vaginal
lengthening surgery were evaluated between January 2018 and January 2021. All
eligible patients were told that their radiotherapy treatment resulted in vaginal
stenosis and that vaginal dilators may improve this condition. After the patients
were informed of the potential benefits, complications, and alternatives, those
who agreed to undergo the vaginal dilator manipulation were assigned into Group A
(n = 66), while those who refused were included in Group B (n = 62) and received
routine treatment. Vaginal diameter and length were measured during the follow-up
period and quality of life changes were determined on the basis of the European
Organization for Research and Treatment of Cancer (EORTC) Quality-of-Life
questionnaire cervical cancer module (EORT QLQ-CX24). Results: Three
patients were lost to follow-up over the course of 14 months, while two patients
in Group A refused treatment after radical hysterectomy. A total of 123 cases
with cervical cancer were analyzed in the course of the study. The vaginal
diameter and length were significantly different between the two groups during
the follow-up period. Sexual/vaginal function, sexual worry, sexual activity, and
sexual enjoyment were also significantly different after radiotherapy in groups A
and B (p
Keywords
- cervical cancer
- radiotherapy
- sexual life
- vaginal model
Cervical cancer is one of the most frequent causes of death from all female cancers worldwide. The majority of deaths occur in less developed countries or in individuals who live in low-resource areas, representing a serious threat to women’s health [1]. The treatment methods for cervical cancer include radiotherapy, chemotherapy, and surgery, which are selected based on the stage of the disease. Radical hysterectomy and systematic pelvic lymphadenectomy are preferred for early stages of cervical cancer, while pelvic external beam radiotherapy and/or brachytherapy is the standard treatment for bulky tumors in stages IB3 or greater than IIB [2]. However, the risks associated with radiation therapy, such as urinary or bowel complications, sexual dysfunction, or carcinogenesis, have a significant impact on patients’ quality of life [3]. Vaginal stenosis is one of the most frequently occurring late side effects of pelvic radiotherapy among genital complications. It is defined by a decreased vaginal diameter and/or length, with cited vaginal stenosis rates ranging from 1.25% to 88% [4]. Vaginal stenosis results from decreased collagen deposition and elasticity and increased collagen and fibrous tissue production, especially in patients treated with high radiation doses [5]. Vaginal stenosis may hinder early detection of cancer recurrence [4]. Moreover, many women experience dyspareunia, which impairs sexual function and has a negative impact on patient well-being [6].
Currently, vaginal stenosis prevention and treatment are based on the use of
topical estrogen or a vaginal dilator [7]. Topical estrogen has been demonstrated
to be effective in treating menopausal genitourinary syndrome [8]. However, its
specific use in stenosis after radiotherapy remains unclear [9]. Pitkin
et al. [10] showed that vaginal estrogen-treated patients had few alterations in
the vaginal narrowing and epithelium. Galuppi et al. [11] showed that
vaginal application of
Vaginal dilation is widely recommended by many international guidelines [12, 13]. Stahl et al. [14] reported that extended duration of vaginal dilator use beyond one year could reduce vaginal stenosis after radiotherapy in endometrial carcinoma. Due to heterogeneous treatments, patients with cervical cancer need to remove longer vagina compared to those with endometrial cancer. However, embarrassment and discomfort were the most reported barriers to vaginal stenosis therapy in clinical practice. In the present study, the traditional vaginal dilator was improved to provide patients with more comfort and enhance their treatment compliance. The participants were followed up for 14 months after surgery and a detailed description of the novel vaginal dilator’s clinical effect in patients with cervical cancer after treatment was provided.
A retrospective investigation was carried out within the framework of a single-center study. A total of 128 patients with cervical cancer enrolled into the study between January 2018 and January 2021. The inclusion criteria were as follows: (1) women aged 20–45 years; (2) women with stage I–III cervical cancer (the International Federation of Gynaecology and Obstetrics (FIGO) 2018) who underwent surgery (type B–C radical hysterectomy); and (3) living spouse with normal sexual function. Cases of previous history of pelvic radiation therapy, congenital vaginal stenosis, and patients reporting no sexual activity were excluded. All subjects gave their informed consent for inclusion before they participated in the present study. The study was approved by the Ethics Committee of Jinhua Hospital Zhejiang University School of Medicine (ethics approval No. 2022-258). A total of 128 participants were included in the present study and assigned into two groups (Fig. 1). Group A consisted of 66 cases, and Group B included 62 cases. The patient basic characteristics and related materials are summarized in Table 1.
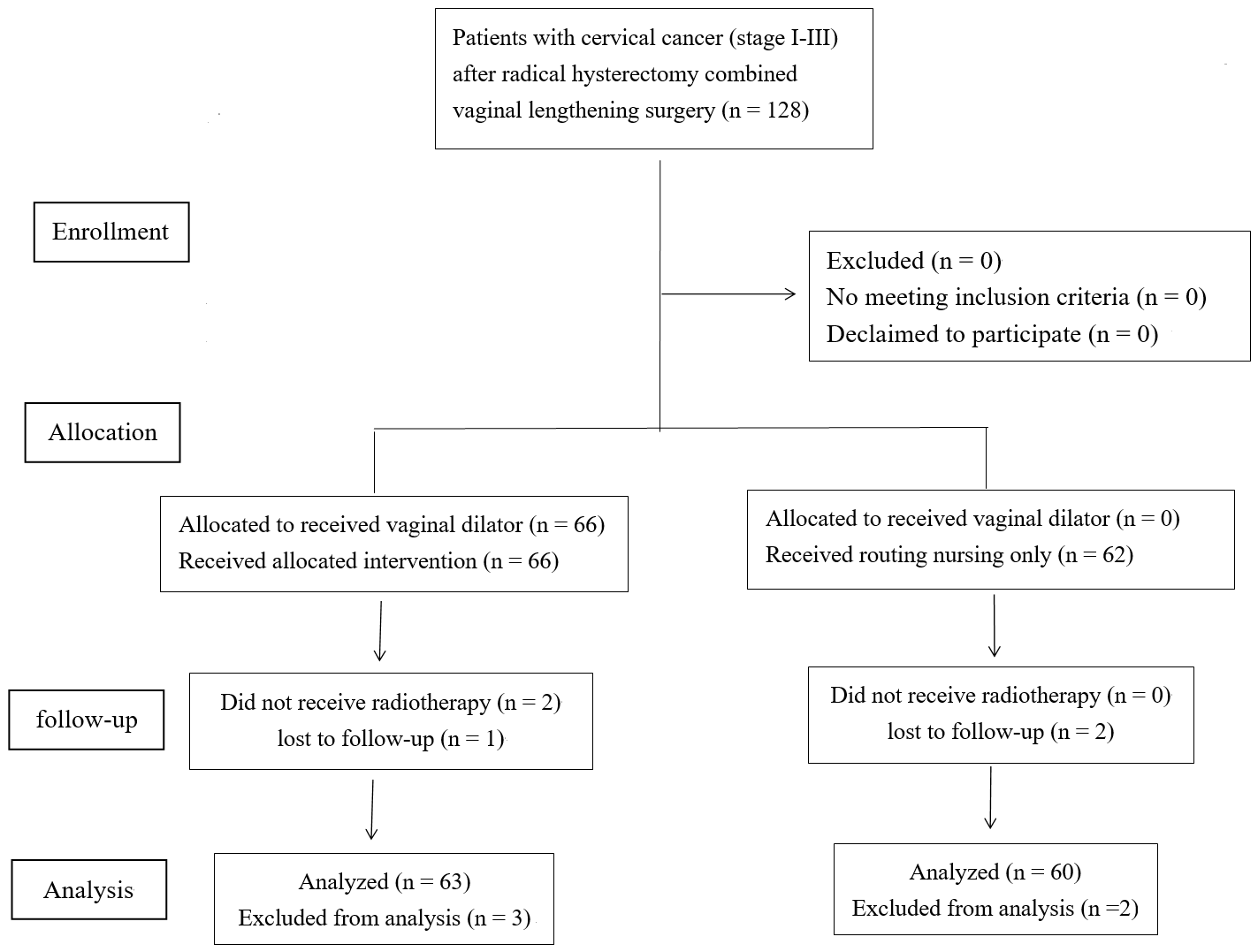 Fig. 1.
Fig. 1.
Consort flow diagram.
| Characteristics | Group A | Group B | p value | |
| Age (years) | 38.81 |
39.20 |
0.709 | |
| No. of previous spontaneous labors | 0–4 (2) | 1–3 (2) | 0.207 | |
| No. of patients (n) | 63 | 60 | ||
| Body mass index (kg/m2) | 21.36 |
20.59 |
0.105 | |
| Histology | 0.845 | |||
| Squamous | 48 | 46 | ||
| Adeno | 13 | 11 | ||
| Adenosquamous | 2 | 3 | ||
| Stage TNM (Tumor node metastasis) | 0.978 | |||
| IA1 LVSI (+) | 3 (4.76%) | 4 (6.67%) | ||
| IA2 | 6 (9.52%) | 7 (11.67%) | ||
| IB1 | 25 (39.68%) | 22 (36.67%) | ||
| IB2 | 16 (25.40%) | 17 (28.33%) | ||
| IIA1 | 10 (15.87%) | 8 (13.33%) | ||
| IIIC1 | 3 (4.76%) | 2 (3.33%) | ||
There were no difference in body mass index, age, histology, stage and
number of spontaneous labors between two groups (p
The dilator was inserted into the patient’s vagina two weeks after surgery. The patients were encouraged to place the vaginal dilator after waking up in the morning and remove it before going to bed at night (treatment window from about 9:00 am to 9:00 pm) and to regularly visit the hospital for examination and care. The three-part vaginal dilator used in the present study consisted of the handheld, connecting, and supporting segments. The connecting and supporting portions were seamlessly put together, and there was a gap between the connecting and handheld portions of the device to prevent the handheld segment from touching the inner side of the vagina during placement, ensuring cleanliness during the procedure. When the supporting and connecting portions were completely inserted into the vagina, the handheld part was rotated to remove it, and the bottom outer side of the connecting part was wrapped with a soft pad to reduce discomfort inside the vagina (Fig. 2).
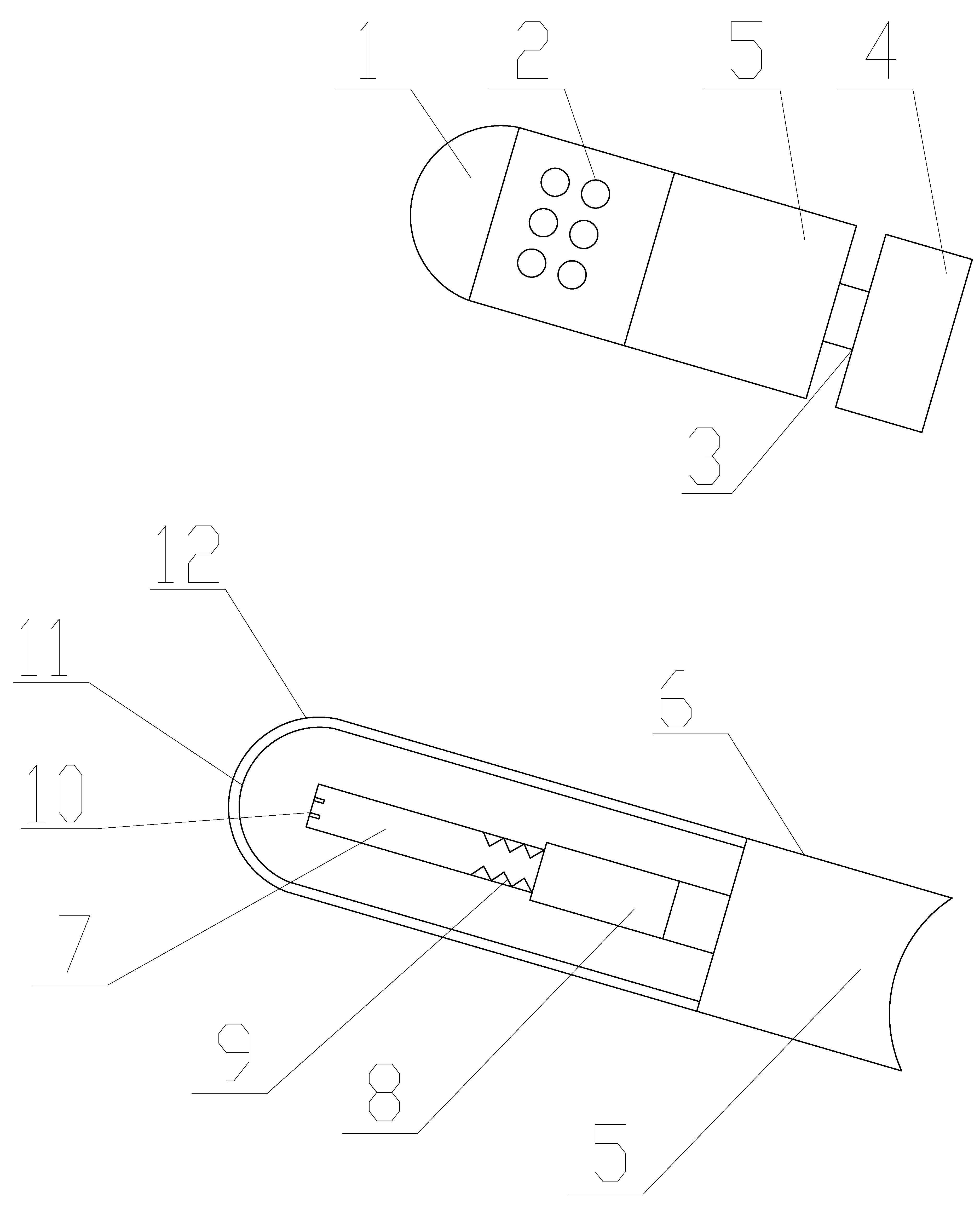 Fig. 2.
Fig. 2.
The structure of the dilator. 1: hollow plastic top of the dilator; 2: several small holes on the dilator; 3: gap between the connecting and handheld parts; 4: handheld part of the dilator; 5: connecting part of the dilator; 6: clamping pad of the dilator; 7: medical liquid supply chamber; 8: medical liquid containment chamber; 9: serrated protrusions in medical liquid chamber; 10: sensor placement slot; 11: medical sponge; 12: sealing film.
Measurements were taken at four time points: at baseline (about one week before radiation therapy) and then three, six, and 12 months after radiation therapy. The patients were followed-up for approximately 14 months.
The assessment of vaginal diameter and length with a special cylindrical dilator
set served as the primary study outcome [15]. The following dilator sizes were
utilized: (1) 69 mm
The EORTC QLQ-CX24 consisting of three multi-item and five single-item scales was used to identify symptoms related to cervical cancer and sexual health in order to evaluate quality of life. The Crobach’s alpha in the present study was 0.802. The standard scoring algorithm recommended by the EORTC was used to linearly transform all scales and item scores to a 0–100 scale. A high score on a functional scale represented a high/healthy level of function, whereas a high score on a symptom scale or item represented a high symptom level or difficulties [16].
All statistical analyses were carried out using the Statistical Package for
Social Science software version 13.0 (Inc., IBM, Chicago, IL, USA). The data were
represented as mean
A total of 128 cases were enrolled in the present study with a follow-up of 14 months. Three patients were lost to follow-up (one patient in Group A and two patients in Group B), and two patients in Group A refused treatment after surgery. A total of 123 women (63 patients in Group A and 60 patients in Group B) were analyzed in the course of the study.
Table 1 shows the participants’ clinical characteristics. There were no
statistical differences (p
Table 2 shows no differences between participants in terms of vaginal diameter
and length after surgery (p
| Characteristics | Group | Before radiotherapy | 3 months | 6 months | 12 months |
| Vaginal diameter (mm) | A | 31.37 |
30.98 |
32.02 |
32.04 |
| B | 32.10 |
29.94 |
30.87 |
30.93 | |
| Vaginal length (cm) | A | 10.78 |
10.40 |
10.96 |
10.89 |
| B | 10.45 |
9.68 |
9.97 |
9.85 | |
| Symptom experience | A | 41.90 |
47.81 |
36.16 |
29.29 |
| B | 45.73 |
54.67 |
40.13 |
33.67 | |
| Body image | A | 38.94 |
42.11 |
35.52 |
29.84 |
| B | 40.37 |
43.55 |
36.05 |
32.33 | |
| Sexual/vaginal function | A | – | 59.24 |
69.46 |
71.56 |
| B | – | 50.73 |
54.28 |
55.35 | |
| Lymphedema | A | 15.02 |
17.75 |
15.29 |
14.81 |
| B | 14.53 |
18.07 |
16.25 |
15.32 | |
| Peripheral neuropathy | A | 29.65 |
31.95 |
34.70 |
30.49 |
| B | 27.12 |
32.37 |
35.25 |
31.55 | |
| Menopausal symptoms | A | 35.24 |
39.06 |
38.95 |
38.46 |
| B | 38.57 |
40.87 |
43.37 |
42.60 | |
| Sexual worry | A | 29.32 |
31.89 |
30.94 |
32.10 |
| B | 35.95 |
37.10 |
38.45 |
39.35 | |
| Sexual activity | A | – | 46.57 |
51.51 |
54.33 |
| B | – | 40.53 |
42.20 |
45.02 | |
| Sexual enjoyment | A | – | 17.44 |
25.10 |
27.41 |
| B | – | 14.82 |
17.37 |
19.33 |
a: p
There was no difference in vaginal diameter and length after surgery (p
Patient performance status was obtained from the EORTC QLQ-CX24.
Radiation therapy used for cervical cancer treatment may have the risk of sexual dysfunction, bowel or urinary complication, or carcinogenesis, which have a significant impact on the quality of life of patients [3]. Currently, there are two main treatments for vaginal stenosis: vaginal dilator or topical estrogen [7]. Vaginal dilation is beneficial due to good contact with the vaginal mucosa and because it is better able to stretch the vaginal mucosa and break down adhesions to maintain vaginal patency [17]. A recent review concluded that topical estrogen was unable to prevent the development of vaginal stenosis and vaginal volume change. Patients who used vaginal dilators had a lower frequency and severity of vaginal stenosis compared to those treated with hormonal therapy [18]. Observational data in the study by Law et al. [19] showed that regular dilator use following radiotherapy was associated with lower rates of self-reported vaginal stenosis.
Vaginal stenosis may not only hinder early detection of cancer recurrence but
also impair sexual function and have a negative impact on the patient’s
well-being [4, 6]. In the present study, women in group B without vaginal dilator
use experienced a significant decrease in vaginal length and diameter compared to
the baseline levels (p
Daily vaginal dilator use improved vaginal length and diameter in the present study, and numerous current international reviews and guidelines recommend its use. However, poor compliance with utilizing vaginal dilator may be present due to psychological distress and lack of consistent or adequate information regarding dilator use [20]. Compared to conventional vaginal dilation, the novel method used in the present study had several advantages. First, there are several small holes on the dilator, which allow vaginal secretions to flow out. Second, the connecting and supporting parts of the dilator are inserted into the vagina during placement and the device can be adjusted by holding the handheld portion that can be disassembled, while the remaining part can be left completely inside the vagina. No other fixing devices are needed to ensure that the entire device is fixed inside the vagina, avoiding bacterial infection caused by the handheld portion entering the vagina. Third, during the process of rotating and disassembling the handheld part of the device, the connection and support segments are fixed more tightly. while tearing part the sealing film ruptures, the medical fluid contains the liquid in the cavity penetrating into the sponge and entering the vagina from the tearing area, improving the vaginal endocrine environment and effectively promoting vaginal recovery. Due to the structural improvements mentioned above, patients do not feel as much pain when inserting the device, improving their comfort level after insertion and reducing the possibility of infection.
Moroney et al. [21] reported that sexual dysfunction was prevalent among cervical cancer patients after radiotherapy. However, Lubotzky et al. [22] showed that only 20% of patients with a partner reported being sexually inactive after radiotherapy due to treatment-related physical or emotional challenges making sexual intercourse uncomfortable or difficult. This proportion decreased to 8% three months after treatment [22]. Since premature sexual activity after surgery may lead to vaginal stump rupture, sexual activity-related factors, such as sexual/vaginal function, sexual activity, and sexual engagement, were all evaluated three months after radiotherapy in the present study. During the follow-up period, daily use of the vaginal dilator was encouraged in Group A patients two weeks after surgery. All of the enrolled patient were suggested to maintain regular sexual intercourse three months after surgery. The results showed that more women felt that sexual activity was enjoyable four months after surgery (approximately one month after radiotherapy) in group A. Vaginal dilator use significantly improved the sexual activity-related factors, such as sexual/vaginal function, sexual worry, and sexual activity in Group A. A possible reason for this observation is that genital-pelvic pain/penetration disorder is very common in cervical cancer patients after surgery, with persistent vaginal penetration difficulty during intercourse [15]. Daily vaginal dilator use was able to regularly dilate the vagina to overcome this condition.
The strengths of the present study included the ability of the novel dilator to decrease vaginal stenosis. Moreover, dilator use is simple and convenient compared to the traditional instrument. The study results showed good patient compliance and effective recovery of patients’ quality of life. However, there were some limitations that should be taken into consideration. First, this was a retrospective study and patient characteristics were heterogeneous. Second, the follow-up period was short. To overcome these drawbacks, a randomized controlled study involving a larger number of cases is needed to evaluate vaginal dilator efficacy with a longer follow-up period.
Using the novel vaginal dilator in patients with cervical cancer after radiotherapy is simple and convenient and can effectively improve vaginal diameter and length and promote the recovery of sexual activity and quality of life.
The datasets generated and/or analyzed during the current study are not publicly available due to further research is needed in the future, but datasets are available from the corresponding author on reasonable request.
MH and LJS designed the research study. MH and LJS performed the research. XYZ, MZ, XLC and PJJ analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All subjects gave their informed consent for inclusion before they participated in the study. The study was approved by the Ethics Committee of Jinhua Hospital Zhejiang University School of Medicine (ethics approval No. 2022-258).
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This study received fees from the research Jinhua Science and Technology Bureau, Zhejiang Province (2022-3-079 and 2021-3-139).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


