1 Department of Pharmacy, Jiangsu Province Hospital of Chinese Medicine, 210029 Nanjing, Jiangsu, China
2 Department of Pharmacy, Women's Hospital of Nanjing Medical University, Nanjing Women and Children's Healthcare Hospital, 210004 Nanjing, Jiangsu, China
Abstract
Background: We conducted a comprehensive systematic review and meta-analysis to evaluate the diverse impacts of combining aspirin with prednisone or prednisolone on patients undergoing in vitro fertilization (IVF) or experiencing recurrent implantation failure (RIF). Our analysis encompassed parameters such as clinical pregnancy rate, implantation rate, live birth rate, miscarriage rate, and fertilization rate. Our primary objective was to resolve the debate regarding the comparative efficacy of prednisone versus prednisolone when administered alongside aspirin in women undergoing assisted reproduction. Methods: A variety of electronic databases were searched between 1984 and December 2023, including PubMed, Web of Science, Embase, the China National Knowledge Infrastructure (CNKI), the China Biology Medicine Disc (CBM), and the CQVIP Database. We employed the Newcastle-Ottawa Scale and the Cochrane Risk of Bias Tool for the quality evaluation. We used Stata 12.0 and Revman 5.4 to pool the data. Results: In this meta-analysis, 10 trials, including 2902 individuals, were considered. Aspirin plus prednisone or prednisolone might improve clinical pregnancy rates (relative risk (RR) = 1.13; 95% confidence interval (95% CI) = 1.03–1.23) and implantation rates (RR = 1.27; 95% CI = 1.01–1.60) compared with the placebo or no treatment group. Conclusions: Our findings suggest that aspirin plus prednisone or prednisolone may improve clinical pregnancy rates and implantation rates in patients with IVF or RIF, and in the subgroup of ANA-positive patients, it may also improve implantation rates. Further design of larger randomized controlled trials is required to determine whether aspirin in combination with prednisone or prednisolone may improve assisted reproductive outcomes in patients undergoing IVF or RIF, considering the limits of study enrollment.
Keywords
- aspirin
- prednisone
- prednisolone
- in vitro fertilization
- repeated implantation failure
With the rise of infertile couples and the openness of China’s two-child policy, assisted reproductive technology is expected to play an increasingly significant role. Although in vitro fertilization (IVF) techniques have achieved notable success, with fertilization rates reaching as high as 80%, the implantation rate per embryo transfer is still outside the ideal range [1, 2, 3].
Reproductive failure could have autoimmune disorders as one of its possible causes. Clinical, subclinical, or preclinical autoimmune diseases may result from abnormal non-specific autoantibody levels, such as those against thyroid peroxidase antibodies and thyroglobulin antibodies (TPOAb and TgAb), anti-nuclear antibodies (ANA), anti-double chain (ds) DNA (AdsDNA), anti-cardiolipin antibodies (ACA), and anti-phospholipid antibodies (APA).
Controversial studies have emerged, such as that by Wang et al. [4] and Kang et al. [5], which suggested that aspirin might exert potential positive effects by enhancing uterine blood perfusion. In a study by Wada et al. [6], aspirin administration led to elevated pregnancy rates and enhanced outcomes in IVF cycles involving cryopreservation among patients exhibiting impaired uterine perfusion (high beat index). Additionally, favorable outcomes were observed in autoantibody-positive IVF patients with the concurrent administration of aspirin and prednisolone [7]. However, immunosuppression with prednisolone did not affect the pregnancy rate in in vitro fertilization and embryo transfer (IVF-ET) patients, according to Lee et al. [8] investigation. According to Laskin et al. [9], administering prednisone and aspirin to individuals with autoantibodies and recurrent fetal loss poses an increased risk of preterm birth, hypertension, and diabetes, rather than enhancing the probability of a live birth.
We conducted a systematic review and meta-analysis to address the present issue of the impact of aspirin with prednisone or prednisolone on in vitro fertilization/recurrent implantation failure (IVF/RIF) outcomes since the previous studies results were conflicting.
This study was previously registered with PROSPERO (CRD42024536645) and followed PRISMA guidelines. We followed the Cochrane handbook guidelines in doing all the steps [10]. All analyses were based on previously published studies; thus, no ethical approval or patient consent were required. We conducted a comprehensive search across multiple databases, including PubMed, Web of Science, Embase, the China National Knowledge Infrastructure (CNKI), the China Biology Medicine disc (CBM), and the CQVIP database, aiming to identify relevant studies published between January 1984 and December 2023. Our search strategy employed MeSH subject terms such as “Fertilization in Vitro”, “Aspirin”, and “Infertility”.
The following were the inclusion criteria: (a) women having IVF or infertility included in the research population; (b) intervention, use of aspirin plus prednisone or prednisolon; (c) comparator, no treatment or placebo; (d) the types of studies were randomized controlled trials or cohort studies. All subjects received a consistent administration of aspirin alongside prednisone or prednisolone until clinical pregnancy was confirmed through fetal heartbeat detection or gestational sac observation. Furthermore, non-original studies, non-human studies, and publications in languages other than English and Chinese were excluded from our analysis.
Using our keyword search, we looked over the abstracts and titles of the studies that we found. By examining the titles and abstracts in accordance with our inclusion and exclusion criteria, irrelevant papers were eliminated. Independently, the findings were evaluated by researchers XT and YT. Additionally, we performed a manual search of the references cited in the included papers to identify potentially relevant literature.
Two investigators, YT. and XT., independently conducted a formal quality assessment of the observational studies using the Newcastle-Ottawa Scale (NOS) [11]. Each of the three categories received a score: ascertainment of exposure or result (three points), comparability of the study groups (two points), and selection (four points). To evaluate the risk of bias in individual studies for randomized controlled trials (RCTs), the Cochrane risk of bias assessment method was employed [10].
We employed both the Mantel-Haenszel random effects model and the fixed model in
our meta-analyses to summarize the data. The outcomes were conveyed through the
computation of the relative risk (RR) and the corresponding 95% confidence
intervals (95% CIs). Indications of significant heterogeneity in the Cochran’s Q
and I2 tests were noted with I2
After careful investigation, 411 pertinent articles during the period of January 1984 to December 2023 were found. Three hundred and ninety articles (334 in English and 56 in Chinese) remained after redundant material was eliminated. After a preliminary screening of titles and abstracts, 324 publications were excluded. Among these, 254 were unrelated to the research subject, 54 were case reports or reviews, and 16 were single-group studies. Following this initial screening, 66 articles remained. However, upon a comprehensive examination of all articles, 56 were deemed unsuitable. Consequently, this meta-analysis ultimately incorporated 10 studies comprising 2902 individuals (Fig. 1).
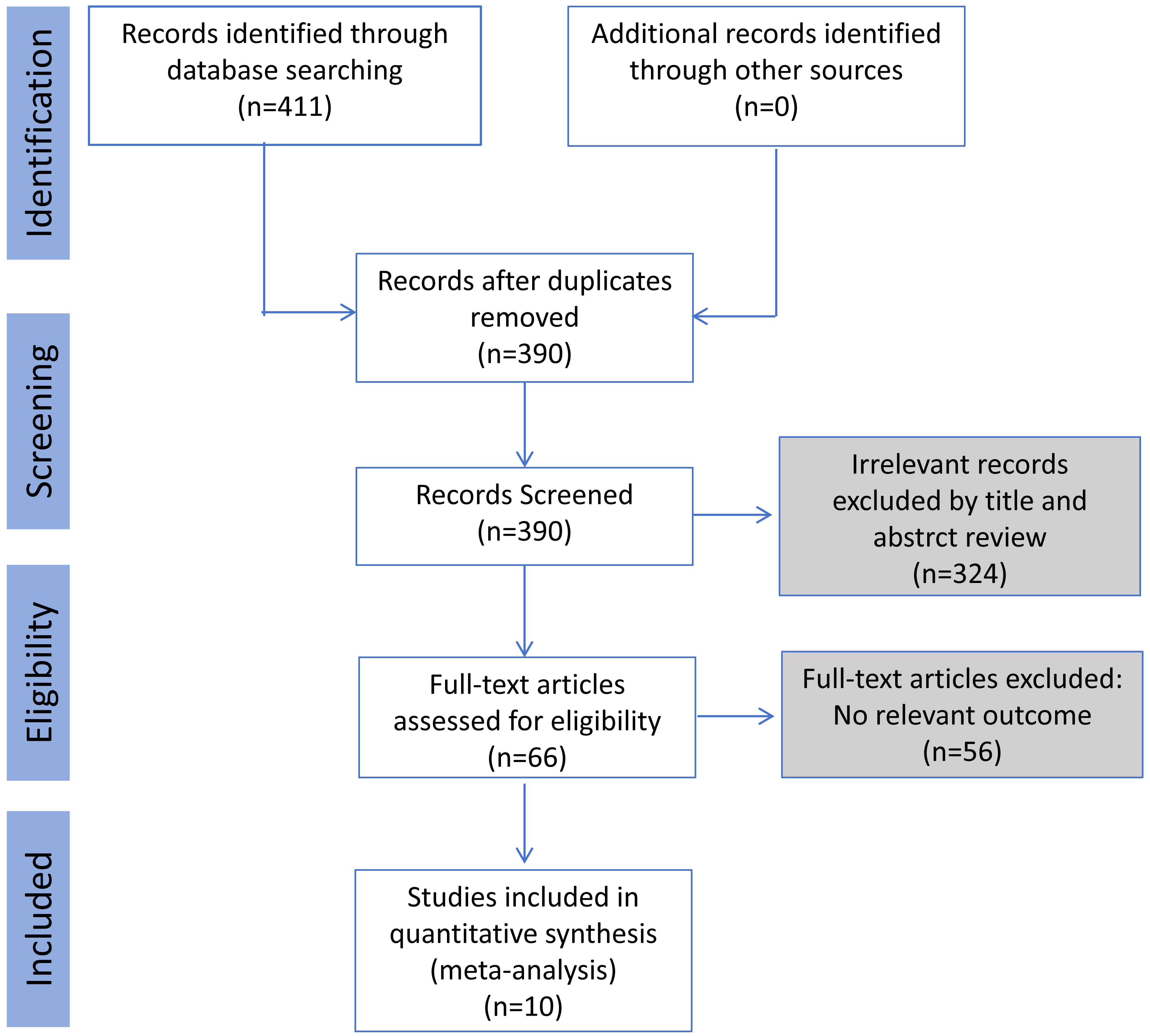 Fig. 1.
Fig. 1.
The PRISMA flow diagram of study searching processes.
An overview of the features of the included studies is shown in Table 1 (Ref. [7, 9, 12, 13, 14, 15, 16, 17, 18, 19]). Between 1997 and 2022, research was conducted with 5 of these studies conducted in China, while one study was conducted in each of the other countries (Canada, Italy, Israel, Turkey, Japan). Seven RCTs [9, 12, 13, 14, 15, 16, 17] and 3 cohort studies [10, 18, 19] comprised the remaining 10 studies (Table 1).
| Study | Year | Country | Study design | Participants | Autoantibody | Drug dose |
| Laskin el al. [9] | 1997 | Canada | RCT | 270 | ANA/ACA/AdsDNA | Aspirin (100 mg/d) + prednisone (40–60 mg/d) |
| Hasegawa et al. [7] | 1998 | Japan | Cohort | 307 | ANA/APA | Aspirin (81 mg/d) + prednisolone (10 mg/d) |
| Geva et al. [12] | 2000 | Israel | RCT | 52 | ACA/ANA/AdsDNA | Aspirin (100 mg/d) + prednisone (10 mg/d) |
| Duvan et al. [13] | 2006 | Turkey | RCT | 200 | Negative | Aspirin (100 mg/d) + prednisolone (10 mg/d) |
| Revelli et al. [14] | 2008 | Italy | RCT | 395 | Negative | Aspirin (100 mg/d) + prednisolone (10–30 mg/d) |
| Zhang et al. [15] | 2013 | China | RCT | 52 | ACA | Aspirin (50–100 mg/d) + prednisone (5–10 mg/d) |
| Fan et al. [16] | 2016 | China | RCT | 133 | ANA | Aspirin (100 mg/d) + prednisone (10 mg/d) |
| Pan et al. [17] | 2020 | China | RCT | 319 | Negative | Aspirin (50 mg/d) + prednisone (2.5 mg/d) |
| Zhao et al. [18] | 2021 | China | Cohort | 828 | ANA | Aspirin (75–100 mg/d) + prednisone (5 mg/d) |
| Zhou et al. [19] | 2022 | China | Cohort | 346 | TPOAb/TgAb | Aspirin (100 mg/d) + prednisone (10 mg/d) |
Notes: RCT, randomized controlled trial; ACA, anti-cardiolipin antibodies; ANA, antin-uclear antibodies; AdsDNA, anti-double chain (ds) DNA; APA, anti-phospholipid antibodies; TPOAb/TgAb, thyroid peroxidase antibodies/thyroglobulin antibodies.
Among the 10 studies included in this article, 7 were randomized controlled trials [9, 12, 13, 14, 15, 16, 17], and 3 were retrospective cohort studies [7, 18, 19]. The included RCTs were evaluated using the Review Manager 5.4 (The Cochrane Collaboration, The Nordic Cochrane Centre, Copenhagen, Denmark), while the cohorts were assessed using the Newcastle-Ottawa Scale. The summary of the quality assessment is presented in Fig. 2 and Table 2 (Ref. [7, 18, 19]).
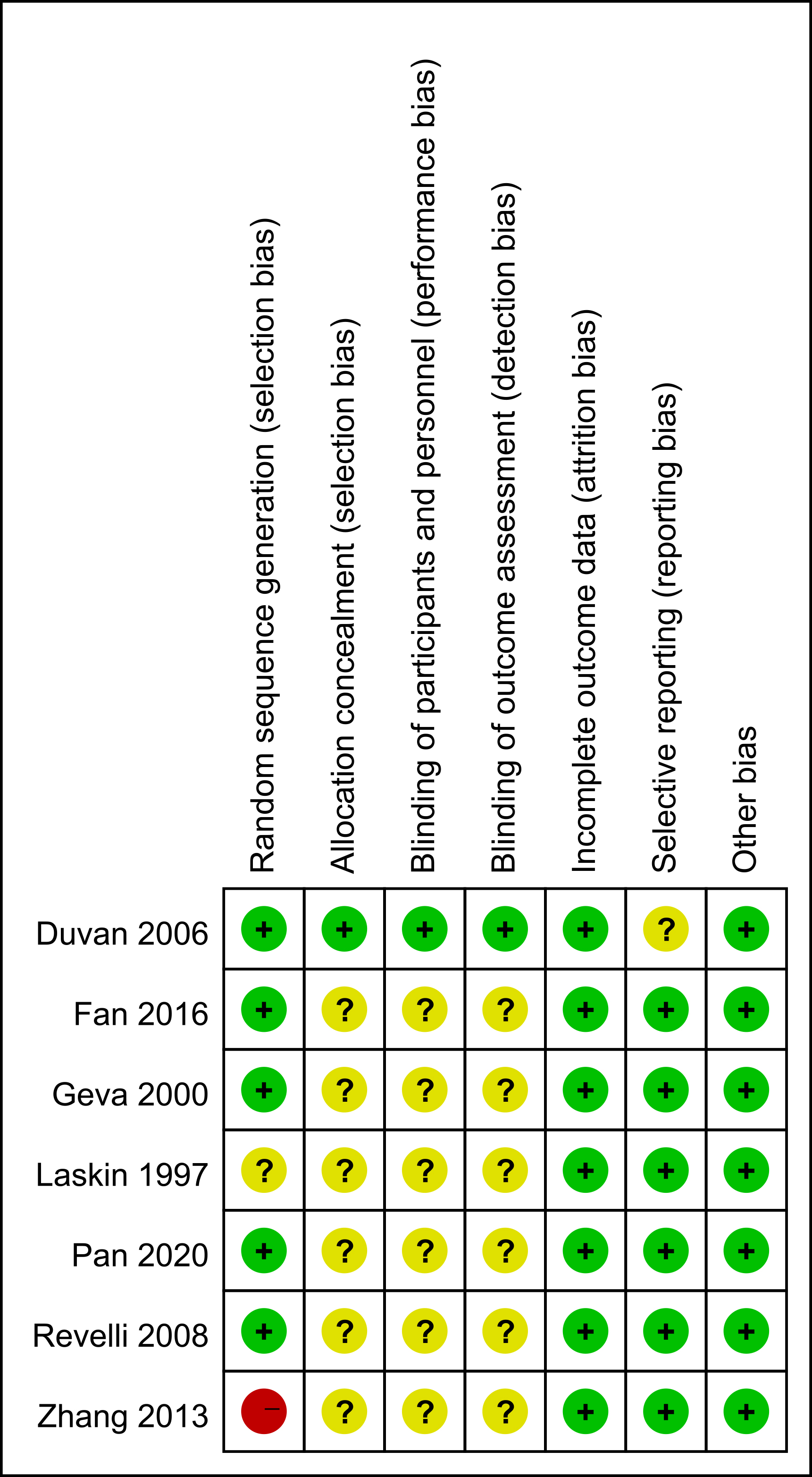 Fig. 2.
Fig. 2.
Quality evaluation chart of the included studies.
Eight studies involving 2325 participants [12, 13, 14, 15, 16, 17, 18, 19] reported the clinical pregnancy rate as an endpoint. The trials showed no significant heterogeneity, as indicated by the heterogeneity test results (I2 = 40%; p = 0.091). The pooled analysis using the fixed-effects model suggested that the use of aspirin in combination with prednisone or prednisolone might enhance the clinical pregnancy rate compared to the control group (RR = 1.13; 95% CI = 1.03–1.23; Fig. 3).
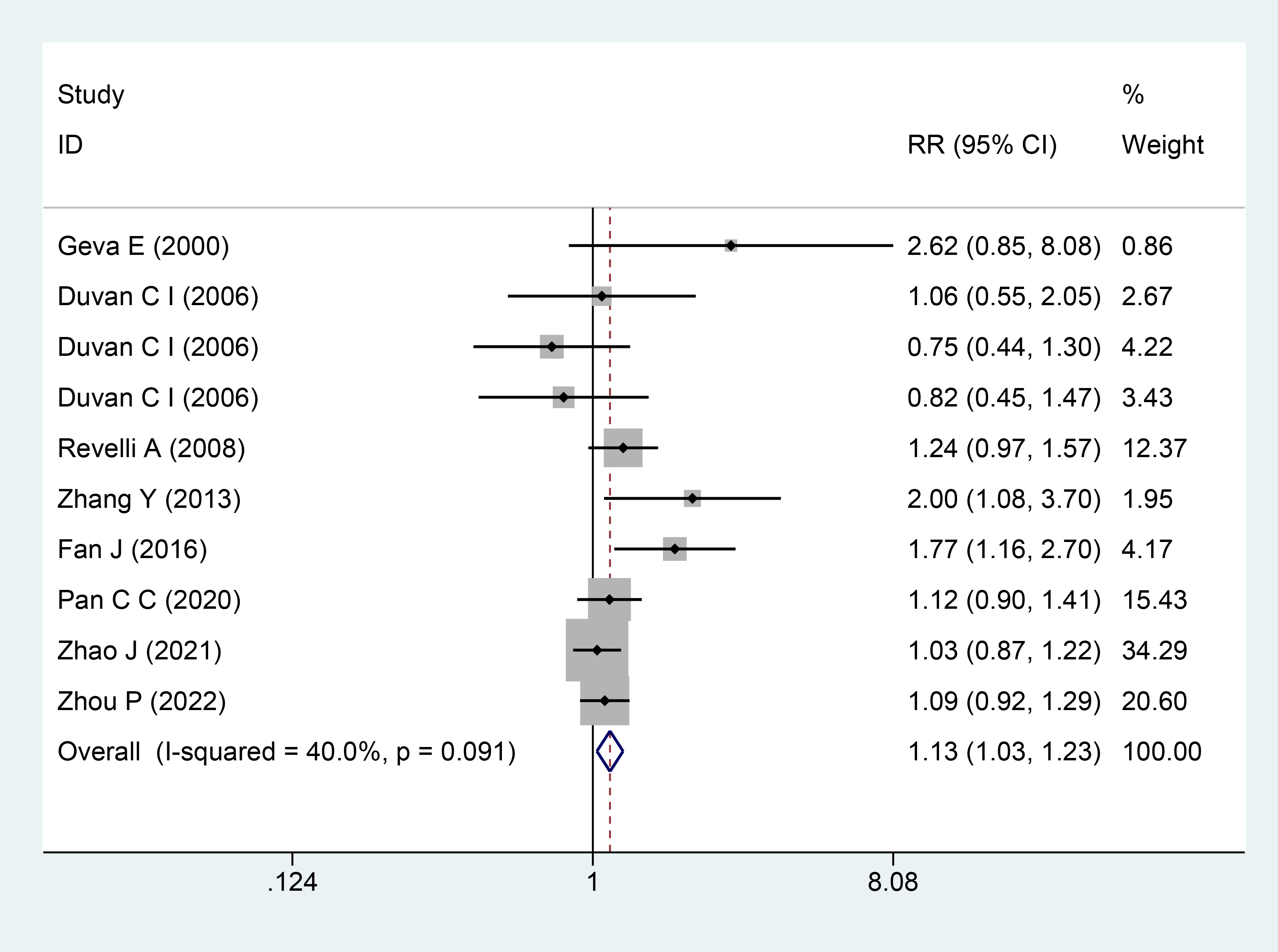 Fig. 3.
Fig. 3.
The Forest plots for clinical pregnancy rate. RR, relative risk; 95% CI, 95% confidence interval.
Seven studies, comprising 2261 participants [7, 13, 14, 15, 16, 18, 19], reported the implantation rate as an outcome. Significant heterogeneity among the studies was observed, as indicated by the heterogeneity test results (I2 = 68.6%; p = 0.001). The pooled analysis using the random-effects model suggested that the use of aspirin in combination with prednisone or prednisolone might enhance the implantation rate compared to the control group (RR = 1.27; 95% CI = 1.01–1.60; Fig. 4).
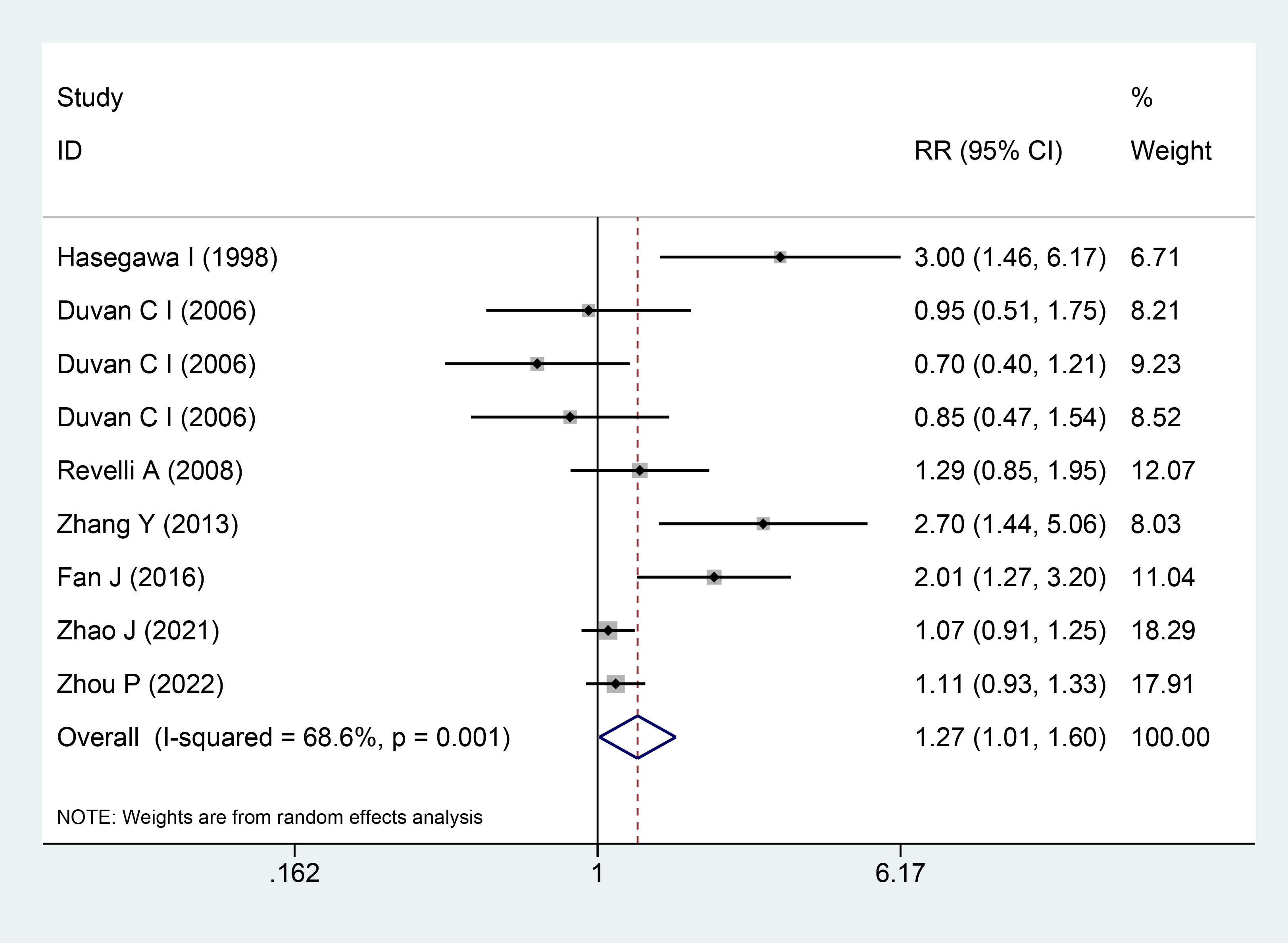 Fig. 4.
Fig. 4.
The Forest plots for implantation rate.
The autoantibody subgroup analysis findings indicated that the ANA subgroup exhibited similarities with the overall meta-analysis. In comparison to the control group, the implantation rate of the ANA subgroup may be higher when aspirin is combined with prednisone or prednisolone, as suggested by the pooled analysis (RR = 2.26; 95% CI = 1.53–3.34; Fig. 5). Notably, no significant heterogeneity was identified in this subgroup analysis (I2 = 0%; p = 0.364). However, there was no discernible difference between the aspirin plus prednisone and control groups in the autoantibody-negative subgroup (RR = 1.04; 95% CI = 0.91–1.19; Fig. 5), and no significant heterogeneity was observed in this subgroup analysis (I2 = 0%; p = 0.450).
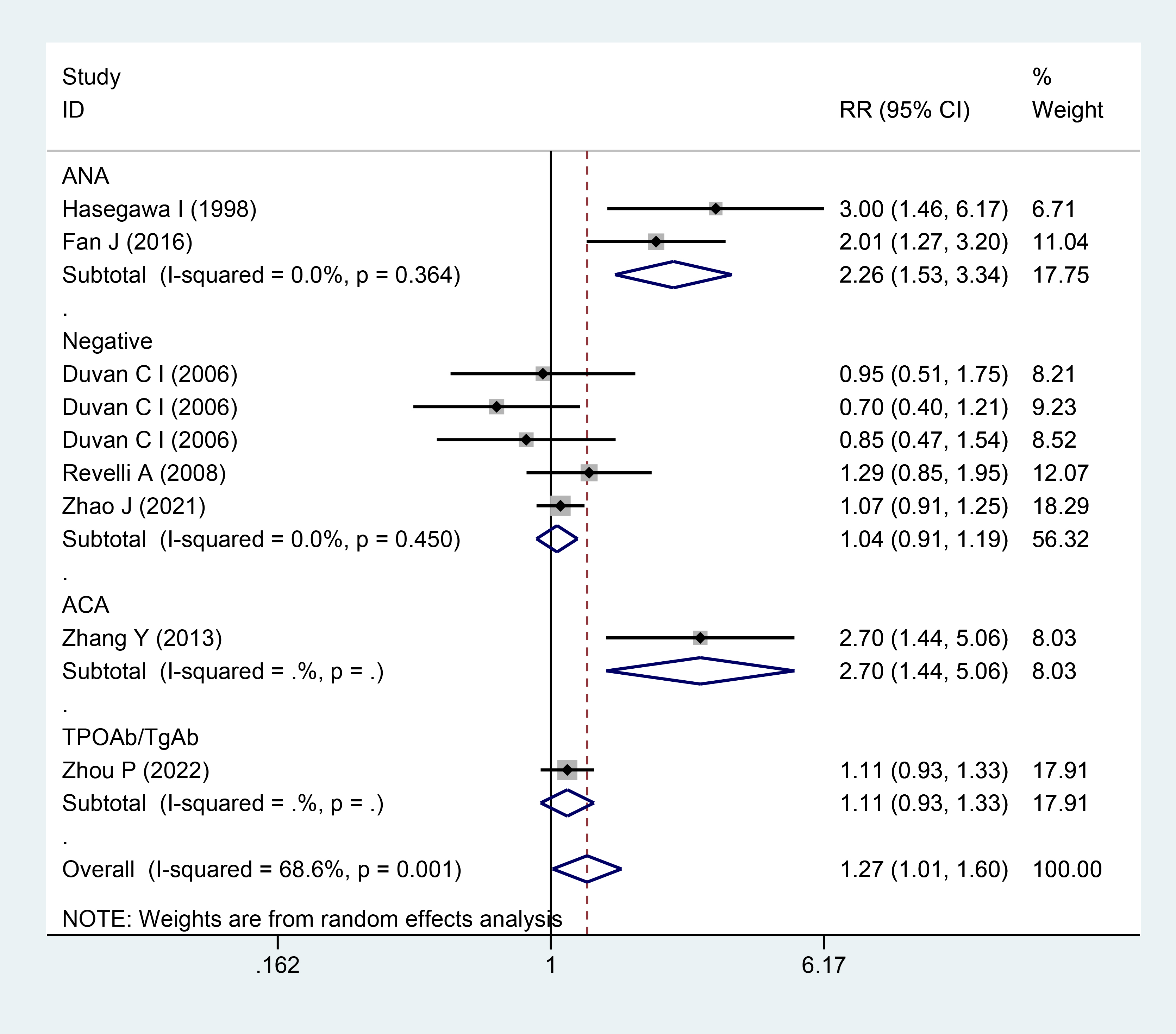 Fig. 5.
Fig. 5.
Forest plots for subgroup analysis for implantation rate.
Seven studies involving 2343 participants [9, 14, 15, 16, 17, 18, 19] reported the miscarriage rate as a result. Significant heterogeneity among the studies was detected, as evidenced by the heterogeneity test findings (I2 = 89.2%; p = 0.000). The pooled analysis using the random-effects model indicated that there was no significant difference in the miscarriage rate between the aspirin plus prednisone or prednisolone and control groups (RR = 0.84; 95% CI = 0.41–1.73; Fig. 6).
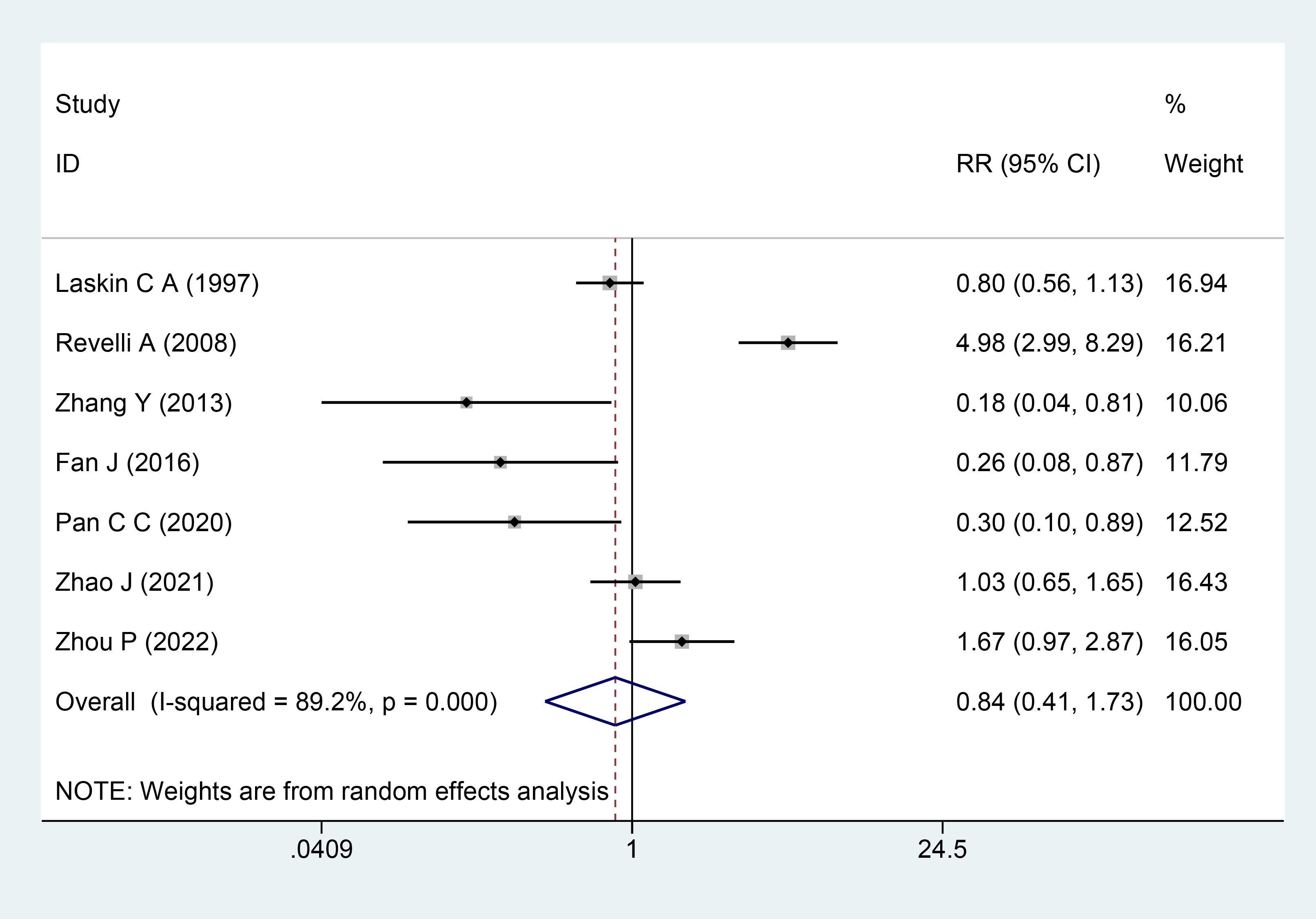 Fig. 6.
Fig. 6.
The Forest plots for miscarriage rate.
Three studies involving 1021 participants [7, 14, 16] reported the fertilization rate as an outcome. Significant heterogeneity among the studies was identified, as indicated by the heterogeneity test findings (I2 = 73.4%; p = 0.023). The results using the random-effects model indicated that there was no significant difference in fertilization rate between the aspirin plus prednisone or prednisolone and control groups (RR = 1.09; 95% CI = 0.93–1.27; Fig. 7).
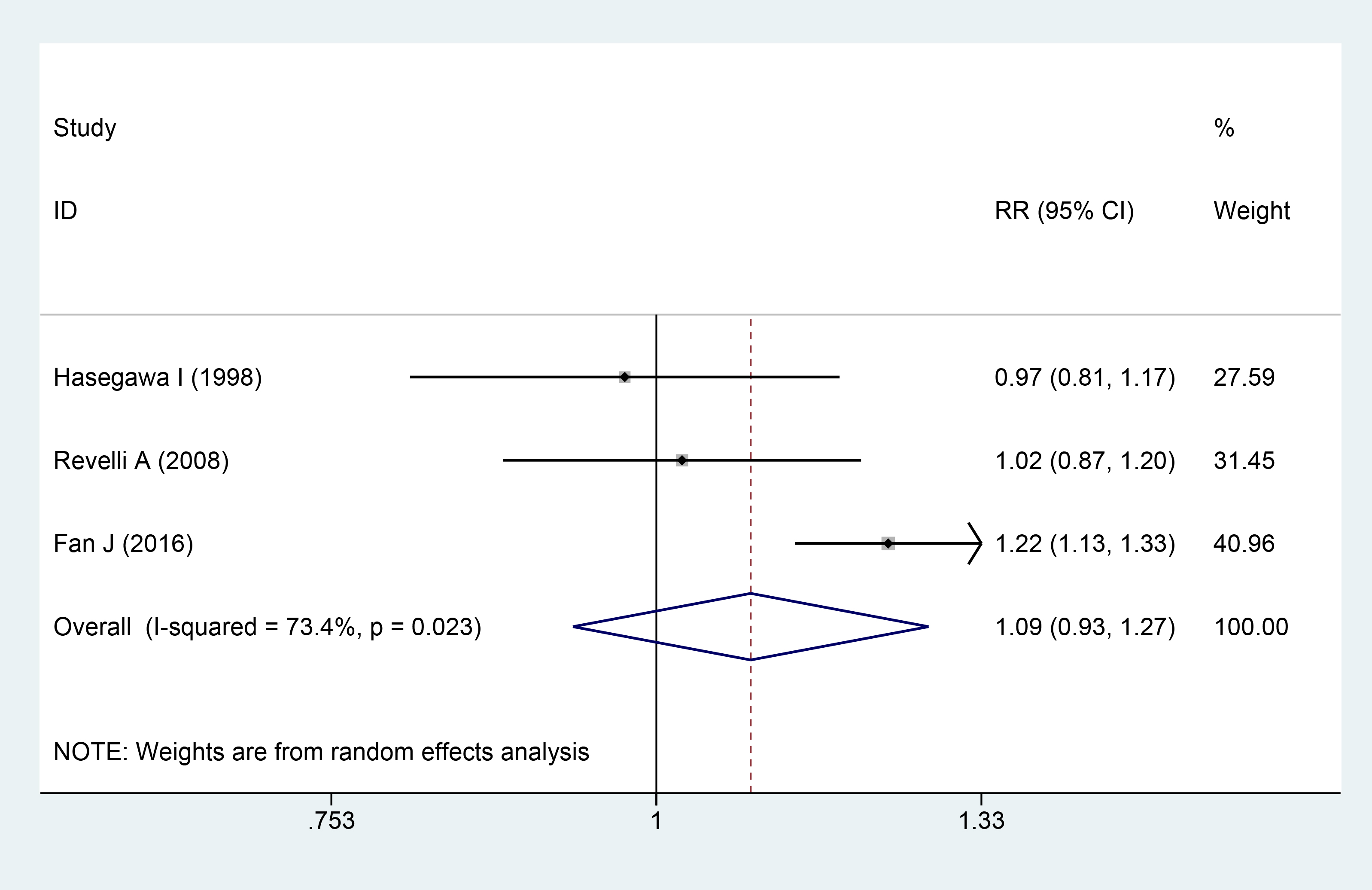 Fig. 7.
Fig. 7.
The Forest plots for fertilization rate.
Two studies involving 616 participants [9, 19] reported the live birth rate as a result. There was no discernible heterogeneity among the trials, as indicated by the heterogeneity test results (I2 = 35.3%; p = 0.214). The results using the fixed-effects model demonstrated that there was no significant difference in the live birth rate between the aspirin plus prednisone or prednisolone and control groups (RR = 1.03; 95% CI = 0.88–1.21; Fig. 8).
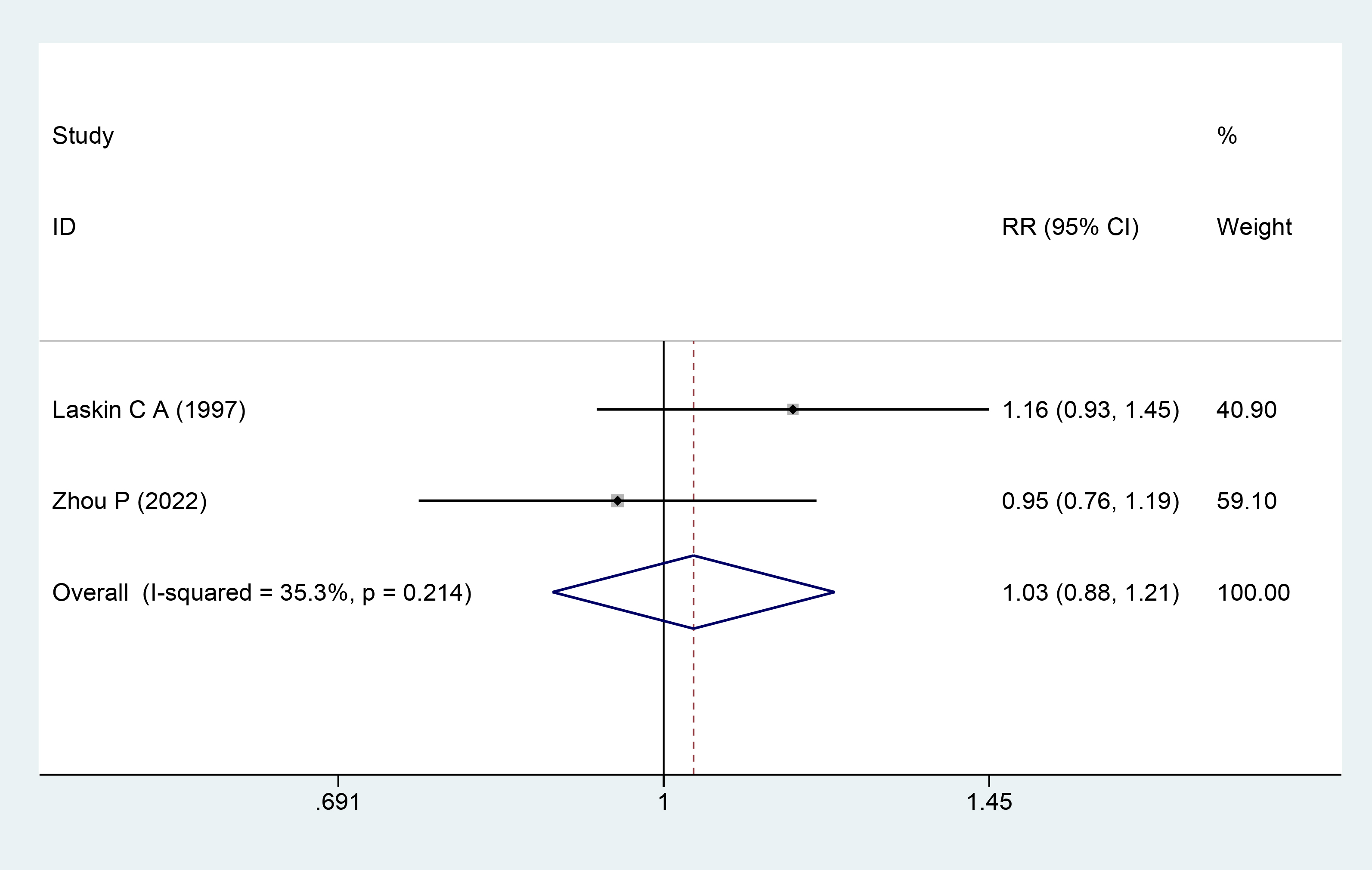 Fig. 8.
Fig. 8.
The Forest plots for live birth rate.
A relative symmetry may be seen in the funnel plot (Fig. 9). Overall, neither the Egger’s (z = 1.59, p = 0.15) nor the Begg’s (z = 1.25, p = 0.210) tests indicated a possible risk of publishing bias.
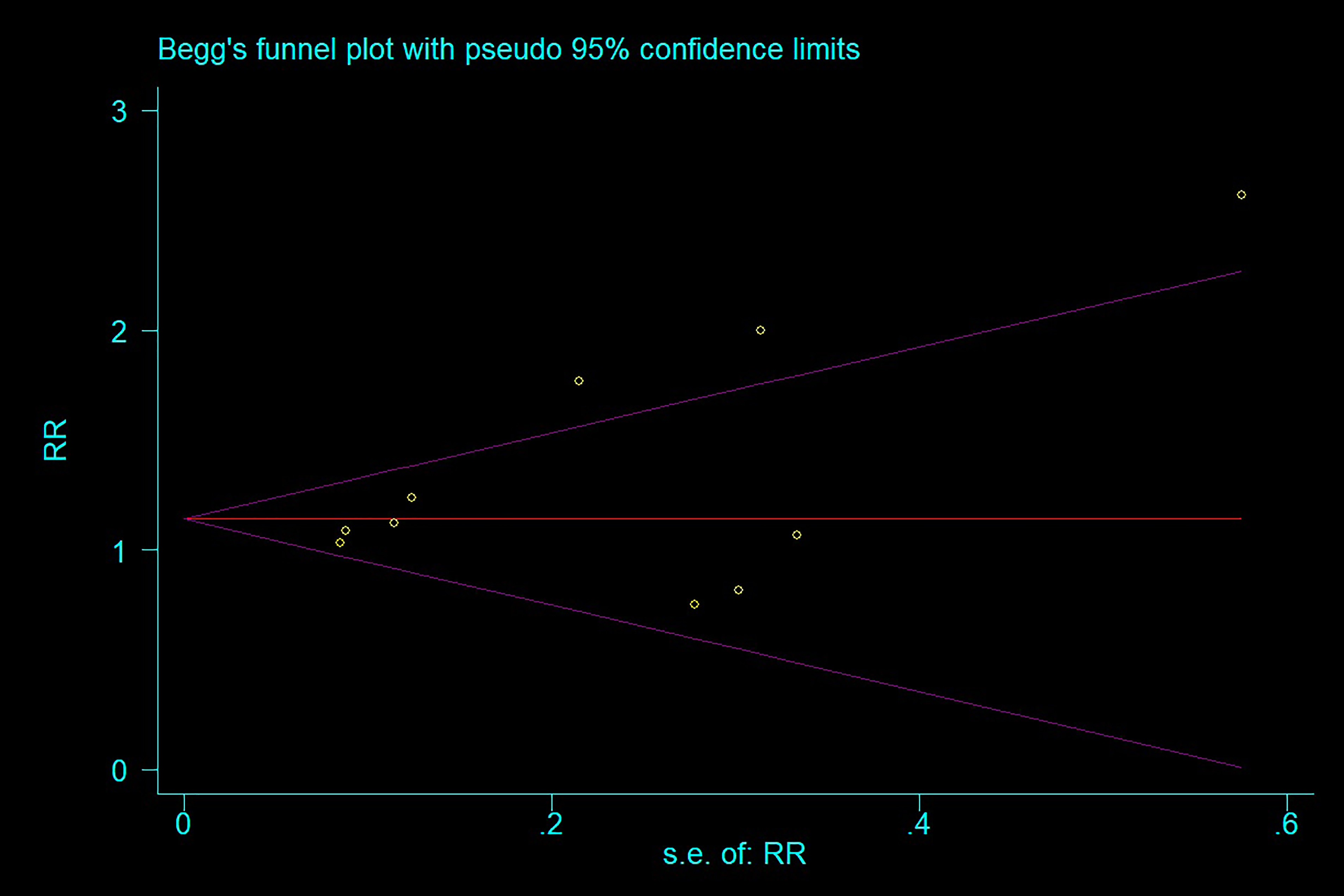 Fig. 9.
Fig. 9.
Begg’s funnel plot.
The success rate of infertility treatment continues to pose challenges. This study primarily examined the impact of aspirin combined with prednisone or prednisolone on IVF pregnancy outcomes. The findings of our systematic review and meta-analysis suggested the absence of potential publication bias in any of the included studies. Moreover, aspirin in combination with prednisone or prednisolone alone may augment the clinical pregnancy and implantation rate in IVF/RIF patients, particularly in those who are ANA positive.
Aspirin functions as a cyclooxygenase inhibitor. A pivotal animal study conducted in rats in 1991 marked the first demonstration that aspirin has the capability to enhance uterine blood flow, suggesting its potential as a drug to elevate the rate of embryo implantation [20]. Kutteh et al. [21] observed that aspirin increased endometrial receptivity, resulting in an increase in the implantation rate. In contrast, Testart and Gauthier [22] showed that low-dose aspirin did not increase endometrial thickness or increase the likelihood of pregnancy.
Autoimmune diseases may also be a cause of reproductive failure. Autoantibody seropositive individuals may have early pregnancy loss due to immunopathologic processes such as vasoconstriction and uteroplacental thrombosis, which are brought on by the antibodies interaction with phospholipids in endothelium membranes and platelets. Prior research demonstrated the connection between ANA and unfavorable reproductive outcomes, such as endometriosis, infertility, and recurrent spontaneous abortion [23, 24, 25, 26, 27]. According to a recent study, ANA may affect the development of embryos and the quality of oocytes, which might lower the likelihood of pregnancy and implantation. However, Lockwood et al. [28] discovered that in individuals showing subclinical autoimmune diseases, no treatment was necessary to attain normal results. Chantilis et al. [29] have indicated that anti-phospholipid antibodies (APL), anti-thyroid antibodies (ATA), and ANA do not appear to exert a significant influence on pregnancy outcomes among individuals undergoing oocyte donation.
As an adjuvant treatment for infertility, aspirin, corticosteroids, or a combination of the two have been suggested by a number of researchers and have gained some traction despite the lack of clear facts to support their use. Because of this, we performed a systematic review and meta-analysis and found a promising prospect that aspirin plus prednisone or prednisolone may prove to be an effective IVF or RIF adjuvant. However, our study is not without its limitations. First, we have to acknowledge the inherent nuance between prospective and retrospective studies. Nevertheless, this study’s lack of publication bias ensures that the main findings remain unaltered. Our conclusions may be applied to a wider range of countries because the papers we included came from different continents.
In conclusion, the combination of prednisone or prednisolone with aspirin demonstrated efficacy in increasing the rates of clinical pregnancy and implantation among IVF/RIF patients. However, to determine whether there exists a disparity in effectiveness between aspirin plus prednisone or prednisolone and a placebo or no treatment group, further RCTs are warranted.
All data generated or analyzed during this study are included in this published article or in the data repositories listed in References.
XT lead the team, formulated the search strategy and collected the data, solved any conflict, conducted the analysis, and writing the manuscript. YT participated in screening, data extraction, quality assessment, and writing the manuscript. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









