1 Department of Obstetrics and Gynecology, West China Second University Hospital of Sichuan University, 610041 Chengdu, Sichuan, China
2 West China School of Medicine, Sichuan University, 610041 Chengdu, Sichuan, China
3 Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, 610065 Chengdu, Sichuan, China
4 NHC Key Laboratory of Chronobiology (Sichuan University), West China School of Basic Medical Sciences & Forensic Medicine, West China Second Hospital, Sichuan University, 610065 Chengdu, Sichuan, China
Abstract
Background:
Immune checkpoint inhibitor
(ICI) therapies have shown promising potential in cervical cancer immunotherapy.
However, its therapeutic efficacy remains to be further evaluated. Our goal was
to evaluate the efficacy and safety of ICI therapies in cervical cancer through a
meta-analysis of relevant studies. Methods: Databases, including PubMed,
Embase, Web of Science, and Cochrane Central Register of Controlled Trials, were
systematically searched for studies on the efficacy and safety of ICI therapy in
cervical cancer to 19 January 2024. Outcomes including objective response rate
(ORR), progression-free survival (PFS),
overall survival (OS), the Europe
Organization for Research and Treatment of Cancer (EORTC) Quality of Life
Questionnaire-Core 30 (QLQ-C30) scores and adverse events (AEs)
were extracted for analysis. Results: Seven studies involving a total of
2514 patients were enrolled in this meta-analysis. ICI therapies significantly
extended OS [hazard ratio (HR) = 0.68, 95% confidence interval
(95% CI) (0.60, 0.77); p
Keywords
- immune checkpoint inhibitors
- cervical cancer
- efficacy
- safety
- meta-analysis
Globally, cervical cancer ranks as one of the leading causes of cancer among women. According to the 2020 global cancer data released by the International Agency for Research on Cancer of the World Health Organization, there are 600,000 new cases and approximately 340,000 deaths due to cervical cancer worldwide. Cervical cancer ranks fourth in incidence and mortality among all female cancers and is the most prevalent gynecologic cancer [1]. Alarmingly, about two-thirds of cervical cancer patients are diagnosed at a locally advanced stage, with more than half of these individuals deemed clinically unsuitable for surgical intervention. This highlights the importance of prevention and early screening. Early human papillomavirus (HPV) vaccination can significantly reduce cervical cancer prevalence, and screening methods such as cervical cytology (Pap detection) and co-testing with HPV and cytology can detect cervical lesions early [2]. However, improving treatment strategies for these patients remains a critical issue.
The concurrent administration of radiotherapy and chemotherapy constitutes the standard treatment regimen [3]. However, despite advancements, the survival rate of cervical cancer has not significantly improved over the past five decades [4, 5]. While early-stage cervical cancer treatment with synchronous radiotherapy and chemotherapy shows promise, with a five-year overall survival (OS) rate of 65%, the prognosis for patients with advanced, recurrent, or metastatic cervical cancer remains bleak [6]. In these cases, the ability to control lesion metastasis falls short, and the five-year OS plummets to approximately 15% [7]. Despite undergoing standard treatment, the overall outlook for patients with locally advanced cervical cancer continues to be dismal [8, 9]. This underscores an urgent need for the development of new therapies to significantly enhance the prognosis for these patients.
In recent years, the field of oncology has seen remarkable advances with the advent of immunotherapy, particularly through the use of immune checkpoint inhibitors (ICIs). These innovative treatments have demonstrated significant efficacy across a broad spectrum of malignant tumors, introducing a novel paradigm in cancer therapy. Immune checkpoints serve as regulatory molecules within the human immune system, functioning as safeguards that temper the immune response by preventing the excessive activation of T cells, which could otherwise cause inflammatory damage. However, cancer cells can exploit this mechanism by over expressing these checkpoint molecules, effectively dampening the immune response and evading detection and destruction by the immune system, thereby facilitating tumor growth [10]. The most extensively researched and clinically applied ICIs target cytotoxic T lymphocyte associate protein-4 (CTLA-4), programmed death 1 (PD-1) and programmed death-ligand 1 (PD-L1). By inhibiting these immune checkpoints activity, ICI therapy lifts the “immune brakes” within the tumor microenvironment, reinvigorating T cells to recognize and attack tumor cells [11]. This approach has been particularly effective in cancers characterized by high mutation rates and dense T-cell infiltration, including melanoma and non-small cell lung cancer [12, 13].
Recently, the potential of ICIs for cervical cancer treatment has garnered attention, with numerous clinical trials highlighting their promising therapeutic impacts [14, 15, 16, 17, 18, 19]. These trials have explored various strategies, including ICI monotherapy, the combination of multiple ICIs, and the integration of ICIs with conventional chemotherapy, pointing toward novel avenues for cervical cancer treatment. However, the efficacy and safety of these treatment regimens are still unclear. Therefore, we conducted a meta-analysis of clinical trials focusing on cervical cancer to assess the effectiveness and safety of ICIs, aiming to provide a robust, evidence-based foundation for their clinical application.
All data included in this study are publicly available in the PubMed, EMBASE, Web of Science, and the Cochrane Central Register of Controlled Trials. The search was performed to 19 January 2024. The key search terms included “Immune Checkpoint inhibitors”, “PD-L1 inhibitors”, “PD-1 inhibitors”, “CTLA-4 Inhibitors”, “Uterine Cervical Neoplasms”, “Cancer of the Uterine Cervix”, “Clinical trials”, and “Randomized Controlled Trials”. The detailed search strategy is displayed in Supplementary File 1. The language was confined to English. This systematic review and meta-analysis was registered in PROSPERO (no. CRD42024508954).
Two researchers independently assessed candidate studies by screening titles, abstracts, and full texts. Any disagreements between the two researchers were resolved by a third researcher to reach a consensus.
Articles meeting the following criteria were included in the meta-analysis: (1) population: patients histologically diagnosed with cervical cancer (all types of histology); (2) intervention: immune checkpoint inhibitors, either alone or in combination with other therapy; (3) comparison: any chemotherapy, targeted therapy, surgery or placebo; (4) outcome: at least one of the following outcomes reported being OS, objective response rate (ORR), progression-free survival (PFS), duration of response (DOR), adverse events (AEs) and quality of life (QOL); (5) study design: randomized controlled trial (RCT).
The exclusive criteria were as follows: (1) case reports, reviews, meta-analyses, conference reports, abstracts without full articles, and commentaries/editorials; (2) studies with the number of patients being less than 10; (3) publications with fewer outcomes from the same study (except when reporting different outcomes); (4) cost-effective analyses.
Two researchers independently reviewed and extracted data from the final included articles using a standardized extraction form. When discrepancy could not be resolved, a third researcher was consulted. Data extracted included study characteristics (first author name, country, year of publication, study design, patient source, masking, study type, randomization method, study phase), demographic information (sample size, median age, pathologic type, stage), intervention model (prior therapeutic regimen, control therapeutic regimen, ICIs), reported endpoints (ORR, PFS, OS, DOR), the Europe Organization for Research and Treatment of Cancer (EORTC) QOL Questionnaire-Core 30 (QLQ-C30) scores (the summary score), and the incidence of grade 3 or worse AEs. The quality of the randomized controlled trials was evaluated using the Cochrane Risk of Bias Tool for RCTs.
All statistical analyses were conducted using R (version
4.3.0, R Foundation for Statistical Computing, Vienna, Austria) with the packages
“meta”, “dmetar”, and “tidyverse”. A p-value
From the systematic database search, we retrieved 1231 articles. Based on the inclusion criteria, 1224 articles were excluded, leaving 7 articles [20, 21, 22, 23, 24, 25, 26] eligible for the meta-analysis (Fig. 1). A total of 2514 patients were evaluated from the included studies. The characteristics of all included studies are presented in Table 1 (Ref. [20, 21, 22, 23, 24, 25, 26]).
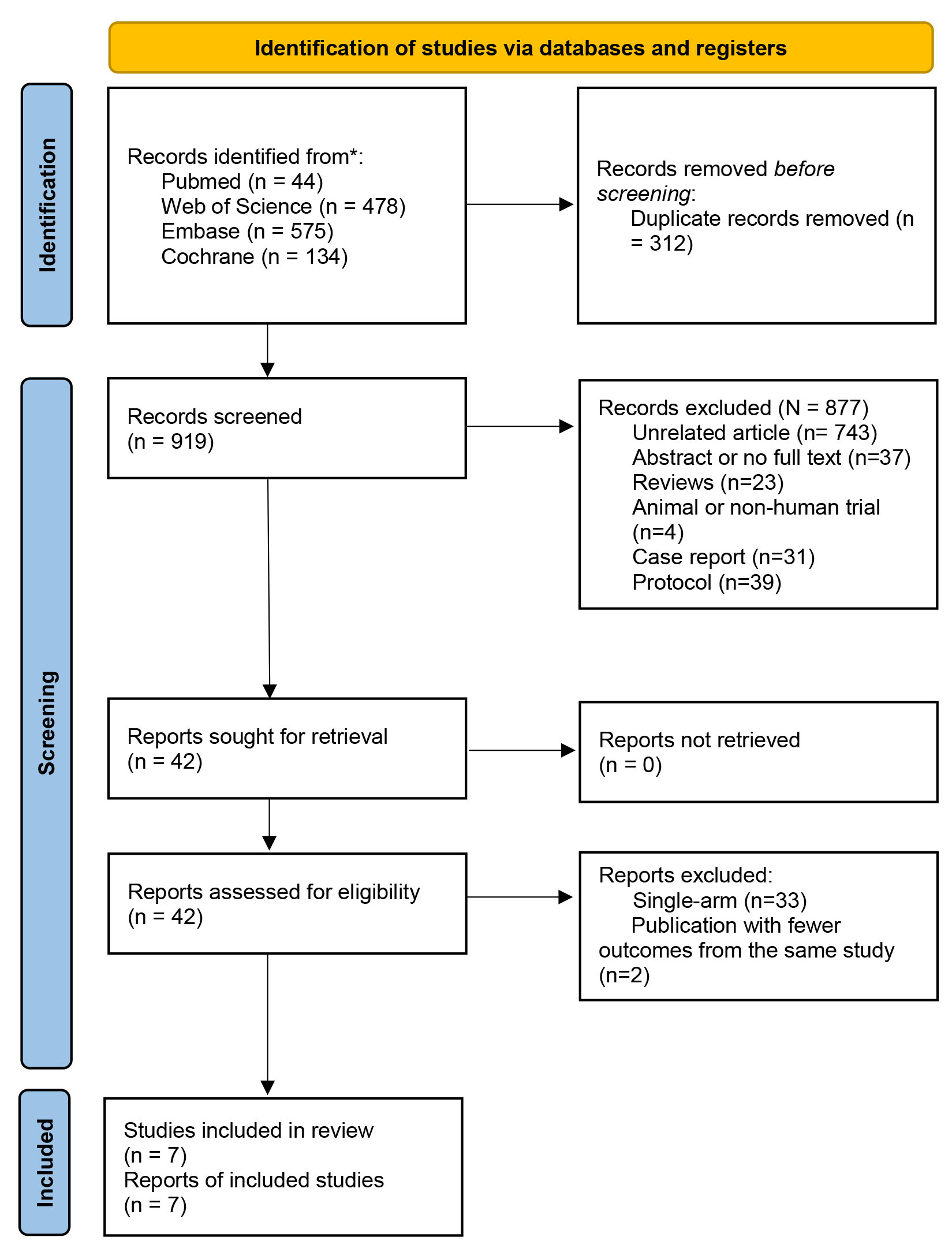 Fig. 1.
Fig. 1.
Flow diagram of studies identified in the meta-analysis.
| Author | Year | Study design | Sample size | Mean age (year) | Median follow-up months | Therapy | ICIs | Endpoints | Grade 3 or above AEs (study group vs. control group) | Other | |||
| Study group | Control group | Study group | Control group | Study group | Control group | ||||||||
| Chen et al. [20] | 2019 | Phase 3 trial, randomized | 23 | 30 | 52 (20–65) | 51 (31–70) | Study group: 46 (16–60); Control group: 42 (2–60) | Nimotuzumab i.v.gtt 200 mg QW + chemotherapy and radiotherapy | Chemotherapy (paclitaxel and cisplatin) and radiotherapy | Anti-EGFR agent | OS, PFS, AEs | Leukocyte reduction (39.1% vs. 70%) (p = 0.025), hemoglobin reduction (17.4% vs. 46.7%) (p = 0.026), thrombocytopenia (4.3% vs. 26.7%) (p = 0.076). | |
| Colombo et al. [21] | 2021 | Double-blind, phase 3 trial, randomized, multicenter, placebo-controlled | 308 | 309 | 51 (25–82) | 50 (22–79) | 22 (15.1–29.4) | Pembrolizumab 200 mg Q3W + chemotherapy |
Chemotherapy (paclitaxel and cisplatin or carboplatin) |
Anti-PD-1 | OS, PFS, DOR, CR, AEs + EQ-5D-5L VAS score | Any event (81.8% vs. 75.1%), the most common: anemia (30.3% vs. 26.9%), neutropenia (12.4% vs. 9.7%). | Control group: CPS |
| Nishio et al. [22] | 2022 | Phase 3 trial, double-blind, randomized, placebo-controlled | 35 | 22 | 54 (26–82) | 50 (33–78) | 23.2 (16.4–27.8) | Pembrolizumab 200 mg Q3W + chemotherapy |
Chemotherapy (paclitaxel and cisplatin or carboplatin) |
Anti-PD-1 | OS, PFS, ORR, DOR, AEs | The most frequently: decreased neutrophil count (60% vs. 59%), anemia (40% vs. 27%), decreased white blood cell count (34% vs. 32%). Serious AEs (43% vs. 46%). | Control group: CPS |
| Tewari et al. [23] | 2022 | Phase 3 trial, open-label, randomised | 304 | 304 | 51 (22–81) | 50 (24–87) | 18.2 (6.0–38.2) | Cemiplimab i.v. 350 mg Q3W | Chemotherapy (Anti-folate therapy: pemetrexed, topoisomerase inhibitors, nucleoside analogue, vinca alkaloid) | Anti-PD-1 | OS, PFS, AEs, DOR, ORR + QLQ-C30 | Any event (45.0% vs. 53.4%), the most common: anemia (12.0% vs. 26.9%), urinary tract infection (5.0% vs. 2.8%), neutropenia (1.0% vs. 9.0%). Serious AEs (23% vs. 22.1%). | |
| Monk et al. [24] | 2023 | Randomised, double-blind, multicenter, placebo-controlled, phase 3 trial | 308 | 309 | 51 (25–82) | 50 (22–79) | 22.0 (IQR: 19.1–24.4) | Pembrolizumab 200 mg Q3W + chemotherapy |
Chemotherapy (paclitaxel and cisplatin or carboplatin) |
Anti-PD-1 | QLQ-C30 | No report | |
| Monk et al. [25] | 2023 | Randomised, double-blind, multicenter, placebo-controlled, phase 3 trial | 385 | 384 | 50 (41–57) | 48 (40–57) | Study group: 18.5 (IQR: 13.2–21.5). Control group: 18.4 (IQR: 13.2–23.7). | Durvalumab i.v. 1500 mg Q4W + chemotherapy and radiotherapy | Chemotherapy (cisplatin or carboplatin) and radiotherapy | PD-L1 inhibitors | OS, PFS, ORR, DOR, AEs + QLQ-C30, QLQ-CX24, TAP score | Grade 3 or 4 AEs (52% vs. 51%), the most frequent: anaemia (20% vs. 15%), decreased white blood cells (10% vs. 13%). | PD-L1 expression was assessed according to the tumour area positivity score. |
| Serious AEs (28% vs. 23%). | |||||||||||||
| Oaknin et al. [26] | 2023 | Randomised, open-label, phase 3 trial, multicenter | 206 | 204 | 51.0 (43.0–60.0) | 52.5 (43.5–61.0) | 32.9 (95% CI: 31.2–34.6) | Atezolizumab 1200 mg Q3W + chemotherapy |
Chemotherapy (paclitaxel and cisplatin or carboplatin) |
PD-L1 inhibitors | OS, PFS, AEs, ORR, DOR | Any event (79% vs. 75%), the most common: hypertension (18% vs. 16%), asthenia (12% vs. 9%), haematological events. | |
| Adverse events were fatal (3% vs. 3%) (1% in the study group were treatment-related). | |||||||||||||
AEs, adverse events; CI, confidence interval; CPS, combined positive score; CR, complete response; DOR, duration of response; EGFR, epidermal growth factor receptor; EQ-5D-5L, 5-level EQ-5D version; ICIs, immune checkpoint inhibitors; IQR, interquartile range; i.v., intravenous injection; i.v.gtt, intravenously guttae; ORR, objective response rate; OS, overall survival; PD-1, programmed death 1; PD-L1, programmed death-ligand 1; PFS, progression-free survival; QLQ-C30, the Europe Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire-Core 30; QLQ-CX24, EORTC Quality-of-Life questionnaire cervical cancer module; QW, quaque week; Q3W, 3 weeks using a; Q4W, 4 weeks using a; TAP, tumor area positivity; VAS, visual analogue scale.
The included studies in our analysis were all RCTs and phase-III trials, most of which were multicenter trials published within the last five years (2019–2023). Our study included four double-blind trials [21, 22, 24, 25] and two open-label trials [23, 26].
Most studies used anti-PD-1 agents [Cemiplimab (n = 1) [23] and Pembrolizumab (n
= 3) [21, 22, 24]] and PD-L1 inhibitors [Durvalumab (n = 1) [25]
and Atezolizumab (n = 1) [26]]. Additionally, Nimotuzumab, an
anti-epidermal growth factor receptor (EGFR) agent, was used in
one trial [20]. The most common therapeutic strategy involved ICIs combined with
chemotherapy (
The Cochrane Collaboration’s tool for evaluating risk of bias was utilized to assess the quality of each study included (Fig. 2) [20, 21, 22, 23, 24, 25, 26]. All studies implemented blind outcome assessment and avoided selective results reporting. The outcome data of all studies were complete. The method of random sequence generation in one study [20] and the allocation concealment in four studies [20, 21, 22, 23] remain unclear. Two studies [23, 26] lacked blinding for both participants and researchers, while blinding status in one study [20] was unclear. Uncertainty persisted regarding the presence of additional sources of bias in one study [20].
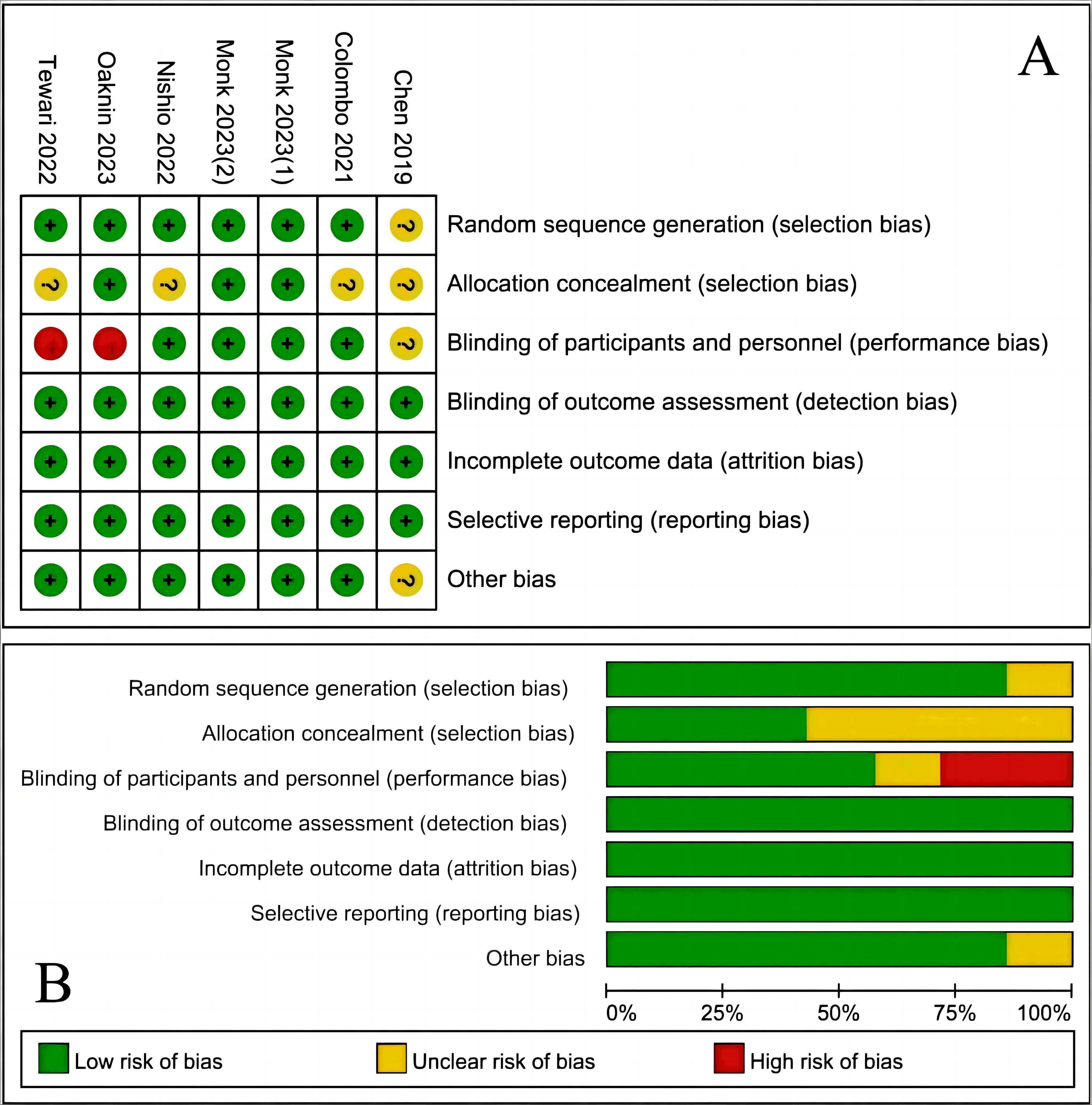 Fig. 2.
Fig. 2.
Assessment of risk of bias. (A) Summary of risk of bias for each trial assessed by Cochrane Collaboration’ tool; plus sign was for a judgment of “yes or low risk of bias”, minus sign was for a judgment of “no or high risk of bias”, and question mark was for a judgment of “unclear, or uncertain risk of bias”, which indicates there was insufficient information to permit a judgment of yes or no. (B) Risk of bias graph about each risk of bias item presented as percentages across all included studies.
Six studies reported on OS [20, 21, 22, 23, 25, 26]. The OS of patients
in the study group significantly exceeded that of the control group
[hazard ratio (HR) = 0.68, 95%
confidence interval (95% CI) (0.60, 0.77); p
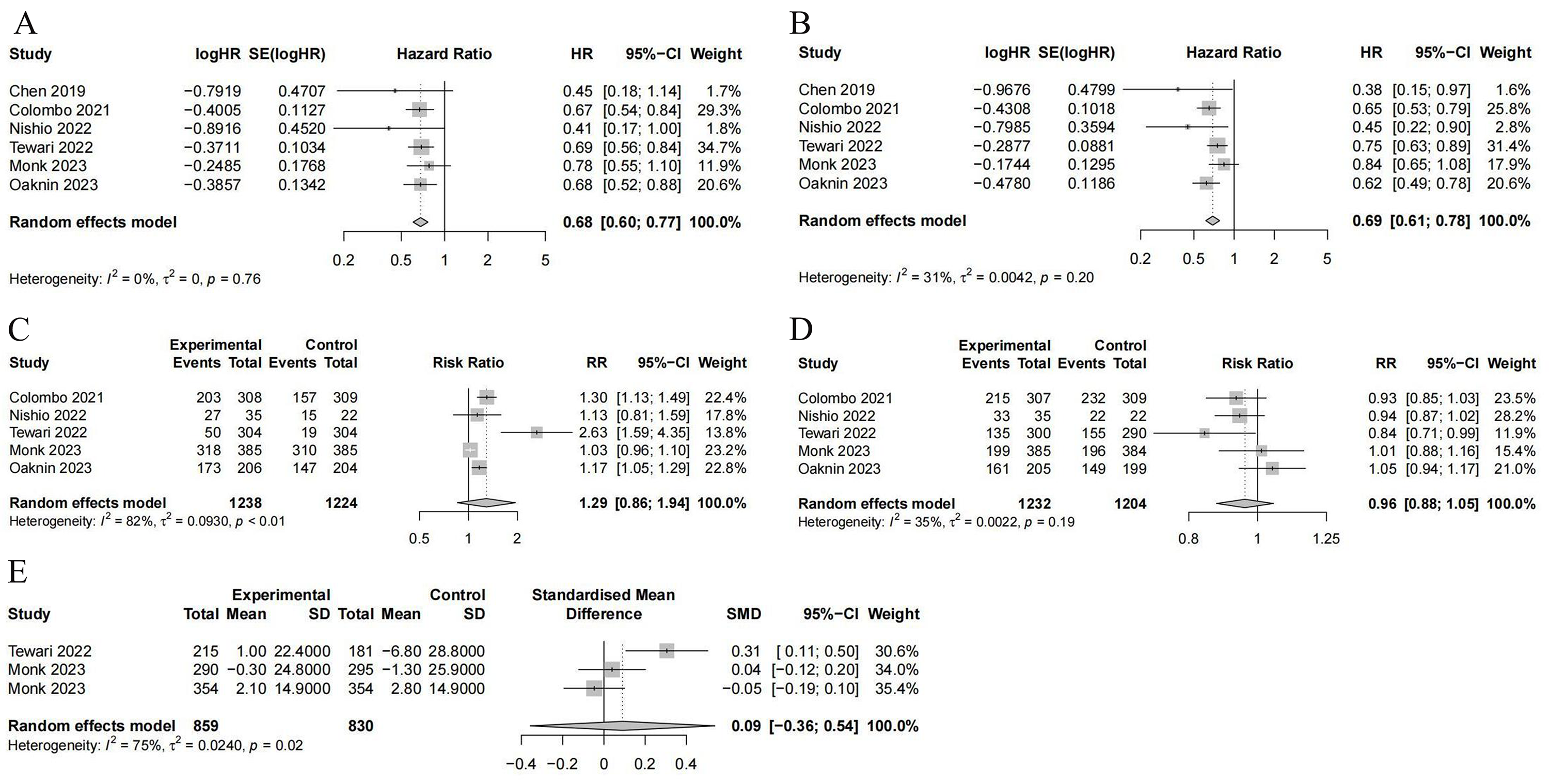 Fig. 3.
Fig. 3.
The forest plots. (A) Overall survival. (B) Progression-free survival. (C) Objective response rate. (D) Grade 3 or above adverse events. (E) The Europe Organization for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30 score. CI, confidence interval; HR, hazard ratio; RR, risk ratio; SD, standard deviation; SE, standard error; SMD, standardized mean difference.
We found that six studies reported on PFS [20, 21, 22, 23, 25, 26]. The findings revealed
that the PFS of immunotherapy was significantly higher than that of the
conventional therapy [HR = 0.69, 95% CI (0.61, 0.78);
p
Five studies reported on ORR [21, 22, 23, 25, 26]. There was no evident difference in
the outcomes between ICI therapy and conventional therapy [HR =
1.29, 95% CI (0.86, 1.94); p = 0.16]. However, significant
heterogeneity was noted among these studies (p
There were five studies reporting on grade 3 or worse AEs [21, 22, 23, 25, 26]. No significant difference was found in the outcomes between ICI therapy and conventional therapy [HR = 0.96, 95% CI (0.88, 1.05); p = 0.28]. These studies did not exhibit significant heterogeneity (p = 0.19; I2 = 35%; Fig. 3D).
Three studies utilizing the EORTC QLQ-C30 to assess QOL were identified [23, 24, 25]. There was no significant difference in the within-group overall least-squares mean change from baseline quality-of-life score between the two groups [standardized mean difference (SMD) = 0.09, 95% CI (–0.36, 0.54); p = 0.48]. However, significant heterogeneity was noted among these studies (p = 0.02; I2 = 75%; Fig. 3E).
Sensitivity analysis was conducted by systematically excluding one study at a time to evaluate its impact on the pooled results (Supplementary Fig. 1) [20, 21, 22, 23, 24, 25, 26]. As indicated by the results of the analysis, none of the pooled results with 95% CIs were remarkably affected by any individual study. This demonstrated that the overall results of the meta-analysis remained relatively stable.
Cervical cancer presents a substantial challenge to global public health, primarily due to its frequent late-stage diagnosis, leading to substantial rates of morbidity and mortality. Consequently, immunotherapy has emerged as a promising novel approach to treatment.
This study aimed to evaluate the efficacy and safety of ICIs
in managing cervical cancer. Using predefined inclusion and exclusion criteria,
we conducted an analysis of seven RCT [20, 21, 22, 23, 24, 25, 26]. The findings from these trials
suggested that immunotherapy yields notable clinical benefits. Compared to
conventional treatments, therapies involving ICIs have significantly prolonged OS
and PFS in cervical cancer patients. However, our analysis did not reveal
statistically significant differences in ORR, quality of life (QLQ-C30 scores),
or the incidence of grade 3 or higher AEs. This means that the safety of ICI
therapy appears to be comparable to conventional therapy. Subsequent sensitivity
analyses confirmed the reliability of these findings. Our
research results showed that the OS [HR = 0.68, 95% CI (0.60, 0.77); p
Previous studies have indicated that combination therapy involving ICIs may yield superior efficacy compared to ICI monotherapy. Some substances released following radiotherapy-induced tumor cell death can induce tumor-specific immune response to continue killing tumor cells, and ICIs can improve the therapeutic effect of radiotherapy by reversing the immunosuppression of tumor microenvironment [30, 31]. The synergistic effect has been validated in clinical studies of stage III non-small cell lung cancer [32]; however, research on cervical cancer remains relatively scarce. Currently, an ongoing phase II clinical study (NCT03614949) has drawn attention because of its novel approach involving atezolizumab combined with radiotherapy for cervical cancer [33]. Chemotherapy can release tumor antigens and alleviate immune suppression induced by tumor cells. The combination of chemotherapy and ICIs has been shown to significantly augment central and effector memory T cell populations, thereby fostering persistence [34].
In contrast, monotherapy is prone to triggering additional immune evasion mechanisms that contribute to primary or adaptive drug resistance [35]. However, this study did not demonstrate the superior efficacy of combination therapy compared to monotherapy with ICIs. However, given the limited number and characteristics of the included studies, it is premature to definitively conclude the absence of such superiority. Further research is warranted to thoroughly investigate this promising therapeutic strategy.
In addition, it is noteworthy that the majority of drugs in
our included studies were PD-1 and PD-L1 inhibitors.
This could be partly attributed to our
deliberate focus on “PD-L1 inhibitors” as a key search term during our
literature review. This reflects the current research trend towards PD-1/PD-L1
inhibitors, which, alongside CTLA-4 inhibitors, constitute the forefront of
immune checkpoint inhibition in cancer treatment. Notably, the approval of the
PD-1 inhibitor Pembrolizumab by the US Food and Drug
Administration (FDA) in 2018 as a second-line treatment for PD-L1 positive
cervical cancer (combined positive score
Indeed, there are other PD-1 and PD-L1 inhibitors undergoing clinical trials, such as the PD-1/CTLA-4 inhibitor Cadonilimab, the PD-1 inhibitor Zimberelimab and Nivolumab [36]. At the same time, recent years have witnessed the discovery of more immune checkpoints, such as lymphocyte activation gene-3 (LAG-3), t cell immunoglobulin and mucin domain-containing protein 3 (TIM-3) and cluster of differentiation 47 (CD47) [37, 38, 39, 40]. Inhibitors targeting these proteins are presently undergoing preclinical or clinical investigation, and hold promise for potential utilization in cervical cancer treatment in the future. These aspects merit further examination and discussion in subsequent investigations.
Regarding the results of our data analysis, we observed significant heterogeneity in ORR and QLQ-C30 scores. Given the limited number of included studies, subgroup analysis was not feasible. The increased heterogeneity might stem from the diversity of drugs and treatment strategies in the evaluated trials. It is noteworthy that two particular studies [23, 25], which significantly contributed to the increase in heterogeneity, were not blinded to participants and researchers. The impact of this lack of blinding cannot be overlooked. Other factors, such as baseline characteristics and histologic classification, may also have contributed to the observed heterogeneity.
In addition, it is worth noting that not all included studies reported the expression of PD-L1 in tumor cells, which can greatly affect the effectiveness of the corresponding drugs. Some studies observed significant anti-tumor activity only in PD-L1 positive patients [41]. High PD-L1 expression in tumor cells suggests a greater possibility of tumor immune escape via PD-1/PD-L1 signaling pathway, increasing the potential efficacy of PD-1/PD-L1 inhibitors. Consequently, the differences in treatment efficacy between studies may be partly due to varying levels of PD-L1 expression in the included populations. This hypothesis requires further investigation.
There were several limitations in this meta-analysis. First, the studies reviewed focused on a narrow range of drug types, limiting our ability to fully assess the efficacy and safety of ICI therapy for cervical cancer treatment. Second, the number of included studies is relatively small. The lack of comprehensive histologic data on cervical cancer restricted our analysis concerning the efficacy of ICIs across different histologic types. For a more thorough evaluation of the efficacy and safety of ICIs in the treatment of cervical cancer and to tailor treatments more precisely, future research should include a broader spectrum of ICIs beyond PD-1/PD-L1 inhibitors. Further investigations into the efficacy of ICI therapy across different histologic types of cervical cancer are also necessary. To refine treatment approaches, more studies comparing the efficacy and safety of combination therapy versus monotherapy are also essential. Finally, further analyses of PD-1/PD-L1 inhibitors should consider the expression of PD-L1 in tumor cells, which will help identify patients who are more suitable for ICI therapy.
In summary, patients receiving immunotherapy reported a delayed onset of symptom deterioration and an improvement in overall survival rate, although the incidence of severe AEs appears to be comparable to those observed with traditional treatments. These findings suggest that immunotherapy may offer a more favorable option for cancer patients. Despite these promising results, more research and randomized clinical trials are needed to validate these initial findings.
All data points generated or analyzed during this study are included in this article and there are no further underlying data necessary to reproduce the results.
CL designed the research study. XZ and YZhan extracted and analyzed the data, and drafted the manuscript. YZhou designed the chart. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was supported by the Natural Science Foundation of Sichuan province (2024NSFSC1673).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



