1 Division of Maternal-Fetal Medicine, Department of Obstetrics and Gynecology, University of Cincinnati Medical Center, Cincinnati, OH 45219, USA
2 Cincinnati Children's Fetal Care Center, Cincinnati Children's Hospital Medical Center (CCHMC), Cincinnati, OH 45229, USA
3 Department of Obstetrics and Gynecology, University of Cincinnati College of Medicine, Cincinnati, OH 45267, USA
4 Department of Surgery, University of Cincinnati College of Medicine, Cincinnati, OH 45267, USA
5 Division of Pediatric General and Thoracic Surgery, Cincinnati Children's Hospital Medical Center (CCHMC), Cincinnati, OH 45229, USA
Abstract
Background: Significant interplay exists between the amniotic fluid and the developing fetus. However the true nature of the relationship between the amniotic fluid and the fetus, as well as the fetal membranes, is very poorly understood due to the lack of a feasible small animal model for amniotic fluid replacement, drainage, or oligohydramnios. We sought to create and present the feasibility of a rodent model of amniotic fluid exchange. Methods: Pregnant rats underwent midline laparotomy, trans-amniotic suture placement via either a U-stitch or a purse-string. Amniotic fluid was attempted to be drained via needle, and the remainder of the undrained fluid was allowed to drain passively, until the amniotic fluid was then replaced with either Normal Saline, Lactated Ringer’s, or a synthetic amniotic fluid. When oligohydramnios is desired, a catheter could be left transuterine to allow for continued fluid drainage. Results: We report the first successful model of complete amniotic fluid exchange in a rat model, successfully replacing the amniotic fluid with various synthetic fluids. A water-tight closure was achievable in all cases. This model is technically sound and reproducible, ensuring minimal fetal and maternal harm. Post procedure, there were no instances of preterm birth or rupture of membranes. In trials of this model, allowing for a brief period of surgical learning curve, we observed a less than 10% fetal loss rate following the procedure and a 0% preterm delivery rate. Conclusions: This novel model paves the way for more controlled examination of the intrauterine environment. It will allow researchers to examine the interplay between the components of the amniotic fluid and the developing fetus. It also opens potential for amniotic fluid exchange to be considered as a treatment option for various fetal conditions.
Keywords
- amniotic fluid exchange
- amniotic fluid
- animal model for fetal therapy
Initially derived from diffusion of maternal plasma, by mid-gestation the amniotic fluid is almost completely fetal-derived. Historically, amniotic fluid was once thought of a static environment, however, as work in preterm birth, intrauterine inflammation, and various fetal conditions expanded, the role of the amniotic fluid has been highlighted and thusly, the interplay between the amniotic fluid and the developing fetus is an area of burgeoning research interest [1]. The amniotic fluid is believed to play a vital role in fetal development and is correlated with neonatal outcomes [2] and in the response to intrauterine inflammation [3] (Fig. 1). Amniotic fluid contains a significant number of cytokines, growth factors, and proteins, and alterations in the normal amniotic protein profiling have been implicated in conditions such as spontaneous preterm birth as well as abnormal fetal organ development. Particularly, varying disease states in the preterm period can lead to intrauterine inflammation and have varying fetal effects. For example, chorioamnionitis leads to both inflammation of the fetal membranes and inflammation and edema in the fetal lungs. Some of this is triggered by aberrant interleukin production. Meconium in the amniotic fluid can enter the fetal lungs and cause lung inflammation through a different mechanism. Increased reactive oxygen species in the amniotic fluid have been implicated in triggering imbalances in matrix metalloproteinases relative to tissue inhibitors of matrix metalloproteinases, leading to amniotic membrane degradation. There are numerous causes of intrauterine inflammation and thus the content of amniotic fluid with abnormal cytokine and protein content can lead to common pathways of preterm birth [4], rupture of membranes, and fetal inflammation, fetal lung injury, fetal gastrointestinal tract development, and may even impact fetal neurological development [5, 6, 7, 8]. Amniotic fluid contains a significant number of cytokines, growth factors, and proteins, and alterations in the normal amniotic protein profiling (termed “proteomics”) have been implicated in conditions such as spontaneous preterm birth as well as abnormal fetal organ development. For example, it is suspected that interleukin-6 (IL-6) from abnormally high in the amniotic fluid, may cause neuroinflammation, as well as lung injury, however it is not known if the IL-6 is causing the neuroinflammation or lung injury or rather if it is a sequala of chorioamnionitis that led to the IL-6 production in response. On the other hand, a pregnancy without amniotic fluid will develop abnormalities of various organ systems, such as the lungs being hypoplastic, the heart becoming hypertrophied, and the bones becoming abnormally shaped. Furthermore, amniotic fluid provides both mechanical and chemical stimulation to the gastrointestinal tract (i.e., epidermal growth factor promotes development and proliferation of gastrointestinal tract production).
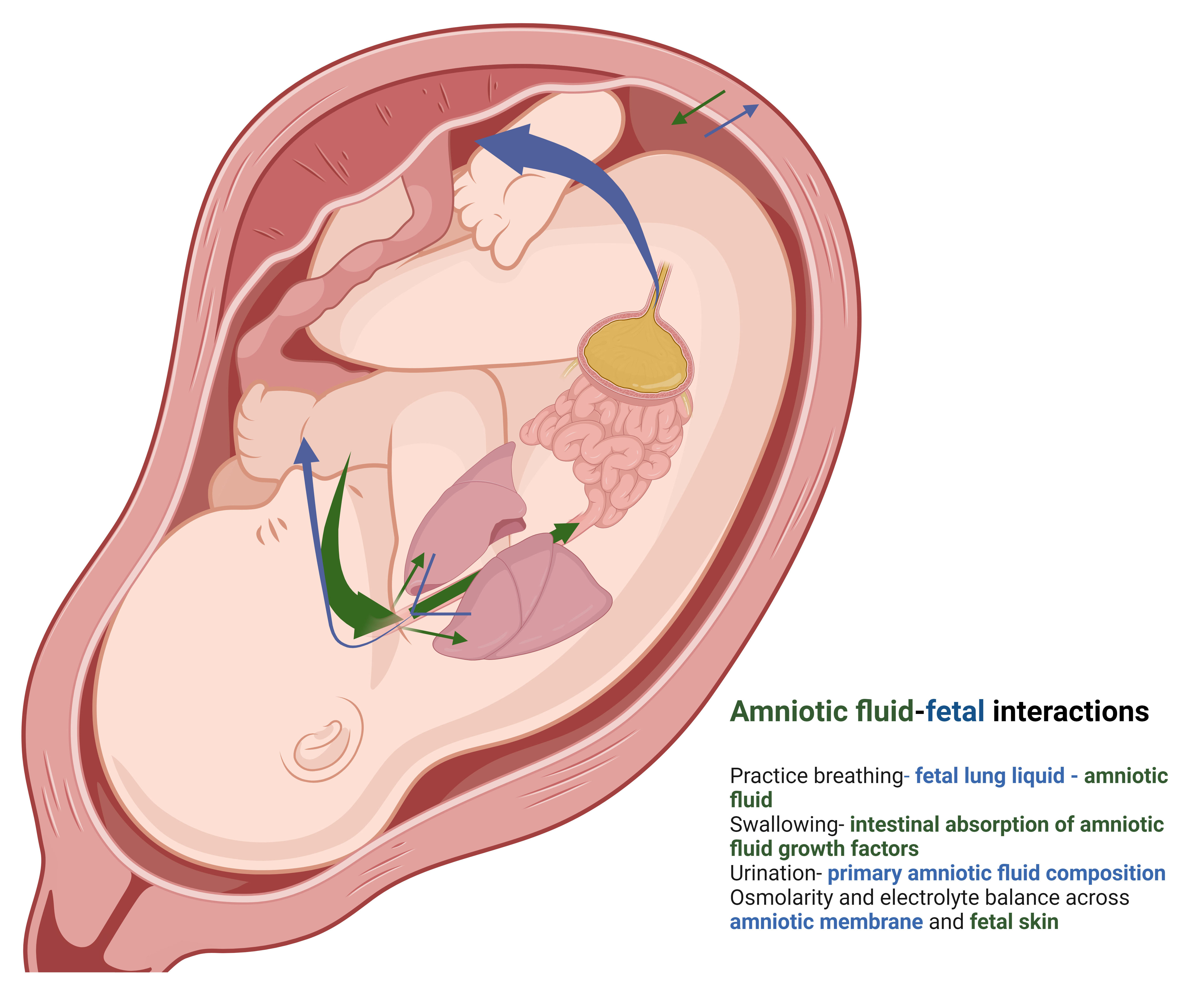 Fig. 1.
Fig. 1.
Amniotic fluid interplay between fetus and mother. The vast
majority (
However, the understanding of interplay between the amniotic fluid and the amniotic membranes and the amniotic fluid and the fetus is very poorly understood. The completely replace the amniotic fluid with synthetic fluids in a successful model of amniotic fluid exchange, can control every element of what a fetus is exposed to in the intrauterine space, without exposing the placenta or the mother to an insulting agent, and will allow for researchers to more accurately study the interplay between abnormalities in amniotic fluid and the fetus, as well abnormalities in the amniotic fluid and the fetal membranes, because amniotic fluid exchanges can be performed and every component of what the fetus is exposed to can be controlled. Despite this, much mystery still remains around the specific factors in the amniotic fluid which lead to various positive (and negative) outcomes in the fetus because there is not a very reliable model of amniotic fluid disorders in small animals. Furthermore, many models which create intrauterine inflammation due so by exposing the exposing the fetus and uterus to significant inflammation [9]. This limits understanding, as specific factors cannot be added or removed individually to know their impact upon the fetus, and a consistent model of oligohydramnios has not been created in the rodent model. Therefore, there is an unmet need for the design of a functional animal model where the amniotic fluid could be safely removed or replaced, or a model of oligohydramnios could be created. Without these tools, it is very challenging for researchers to study amniotic fluid disorders, as well as amniotic fluid interplay with the fetus or fetal membranes. Thusly, we have developed and here-in describe our surgical model of amniotic fluid exchange in a pregnant rat, with the goal that other researchers can use this model for further study of the amniotic fluid.
This was part of an Institutional Animal Care and Use Committee (IACUC) approved study (Cincinnati Children’s Hospital Medical Center IACUC# 2022-0087) examining the impact of various amnioinfusion fluids upon the developing rat fetus and amniotic membrane. Timed pregnant rats were purchased from Charles River and at E17.5, rats underwent midline laparotomy under inhaled and local anesthesia. Inhaled anesthesia was isofluorane, run at 2 L/min. Local anesthesia was 1% plain lidocaine. Mothers also received subcutaneous buprenorphine for postoperative analgesia. After laparotomy, uterine horns were exposed and the fetus, umbilical cord and placenta were located via trans-illumination. The pregnancies are counted and amniotic fluid exchange are carried out on 50% or less of the fetal-placental units in the rats and occur in both horns. In an area of the uterus overlying the fetal flank, away from the fetal organs and any yolk sac blood vessels, a U-stitch was placed trans-uterine, trans-amniotic, with 5-0 or 6-0 Prolene suture. The suture is left loose and a 24-gauge needle is inserted into the amniotic space in the center of the sutured area. Pressure is then gently placed on the uterine sac and the fluid can be completely expressed from the uterus at the needle and suture insertion site. Next, the fluid of interest is injected via the needle and the suture is pulled taught while the needle remains in place. This is followed by tying the suture as the needle is removed (Fig. 2). This technique development was part of a larger project in which we were injecting various isotonic fluids (normal saline, lactated ringer’s, or a synthetic amniotic fluid matched to normal human midtrimester amniotic fluid) to evaluate the impact of various amnioinfusion content upon the fetus and amniotic membranes, however in our experience, given the wide differences in the fluids we injected, any isotonic fluid can be used to replace the amniotic fluid for study, without preterm birth.
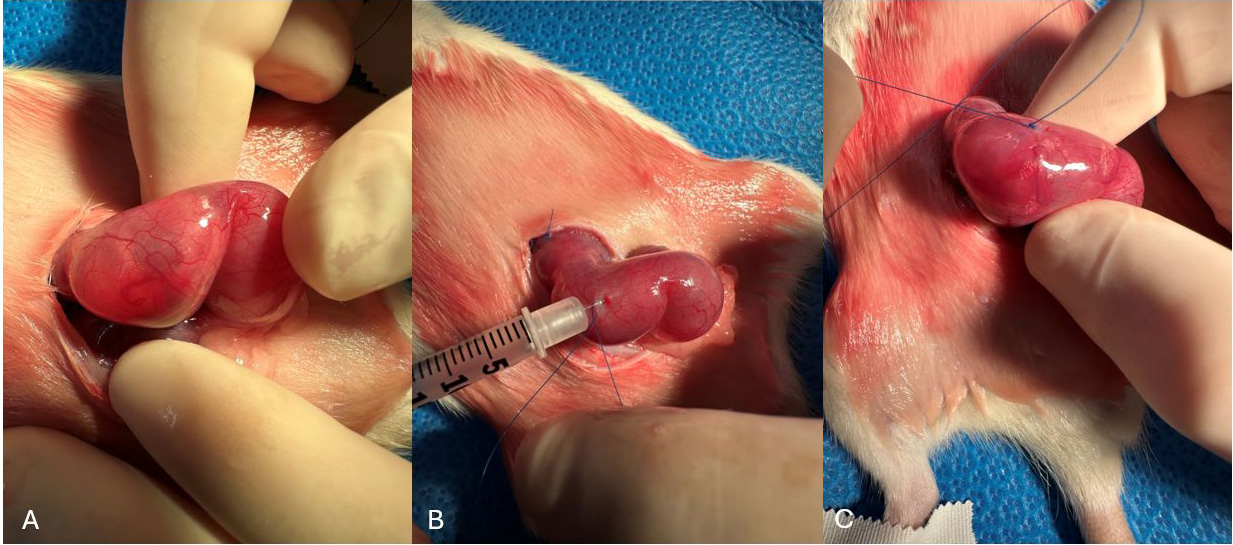 Fig. 2.
Fig. 2.
Method of water-tight amniotic fluid exchage. (A) Rat fetus E17.5. (B) U-stitch and needle placement. (C) Post amniotic fluid exchange. Air bubbles placed intentionally to show airtight closure.
If the amniotic fluid is desired to be removed and stored at the time of amniotic fluid exchange (i.e., amniocentesis is needed), the following is performed: Laparotomy and uterine horn is exposed as described above. Next, a 31-gauge needle with the planned amnioinfusion fluid is inserted into the amniotic space and left in place. In a separate location for the same gestational sac, uterine and transamniotic stitch is performed as above and a 1 mL or less syringe connected to 24 or 22-gauge needle is inserted into the center of the sutured area with care to avoid the fetal organs. While an assistant then holds the suture taut, Fluid is gently removed from the amniotic space until no longer possible via the needle. Next, any remaining amniotic fluid is expressed via the uterus as described above. The suture is then tied while the 24 or 22-gauge needle is removed. The infusion fluid is then injected via the 31-gauge needle and the needle removed. If continued leaking of fluid is planned or required (i.e., to create a model of oligo or anhydramnios, via the 24 or 22-gauge needle, a 24 or 22-gauge catheter can be placed intra-amniotic with passage to the maternal abdomen). The purse string suture tails can be left long and then used to tie around the catheter, keeping it in place and creating a functioning oligo-amnio model as well.
We report here our outcomes of our first 26 pregnant rats that underwent amniotic fluid exchanges. Of those pregnant rats, there were a total of 205 rat fetuses, and 88 underwent amniotic fluid exchanges, and herein we focus on the technical aspects of the procedure as well as the success or failure of amniotic fluid exchange and the risk upon the pregnancy regarding the fetal loss rate, the amniotic fluid.
All rat feto-placental units were collected via repeat maternal laparotomy at E20.5–21.5. Zero rats of the 26 experienced rupture of membranes or preterm birth prior to the said laparotomy. The rodent amnion at E17.5 can accommodate up to 750 µL of fluid without over-distension using this methodology. While the entire gestational sac can be emptied of amniotic fluid via technique 1, given the close proximity of fetal tissue to the needle as the fluid is removed, while fluid can consistently be removed for analysis, it is typically under 200 µL because by that point, fetal parts begin to inhibit the ability to suction fluid. Regarding the fetal demise rate, a total of 205 pregnancies were identified at E17.5 in a total of 26 rodents. Of those 205, 81 underwent complete amniotic fluid exchange without fetal demise in utero, with a total of 8/88 (9.1%) of fetuses experiencing demise in utero (Fig. 3). Maternal rodents tolerated the surgery well, with no rats experiencing preterm delivery. One maternal rodent passed from a presumed respiratory infection and another passed away from an oxygen malfunction to their containment tank. Every single closure was water-tight and air-tight (Supplementary Video). It is presumed that the infused fluid will be replaced by the rodent fetus’s own amniotic fluid within 2 days, however if the researcher wishes to create and oligo-amnio model after exchange, this can be done as described in the methods above, leaving a catheter which is sutured in place.
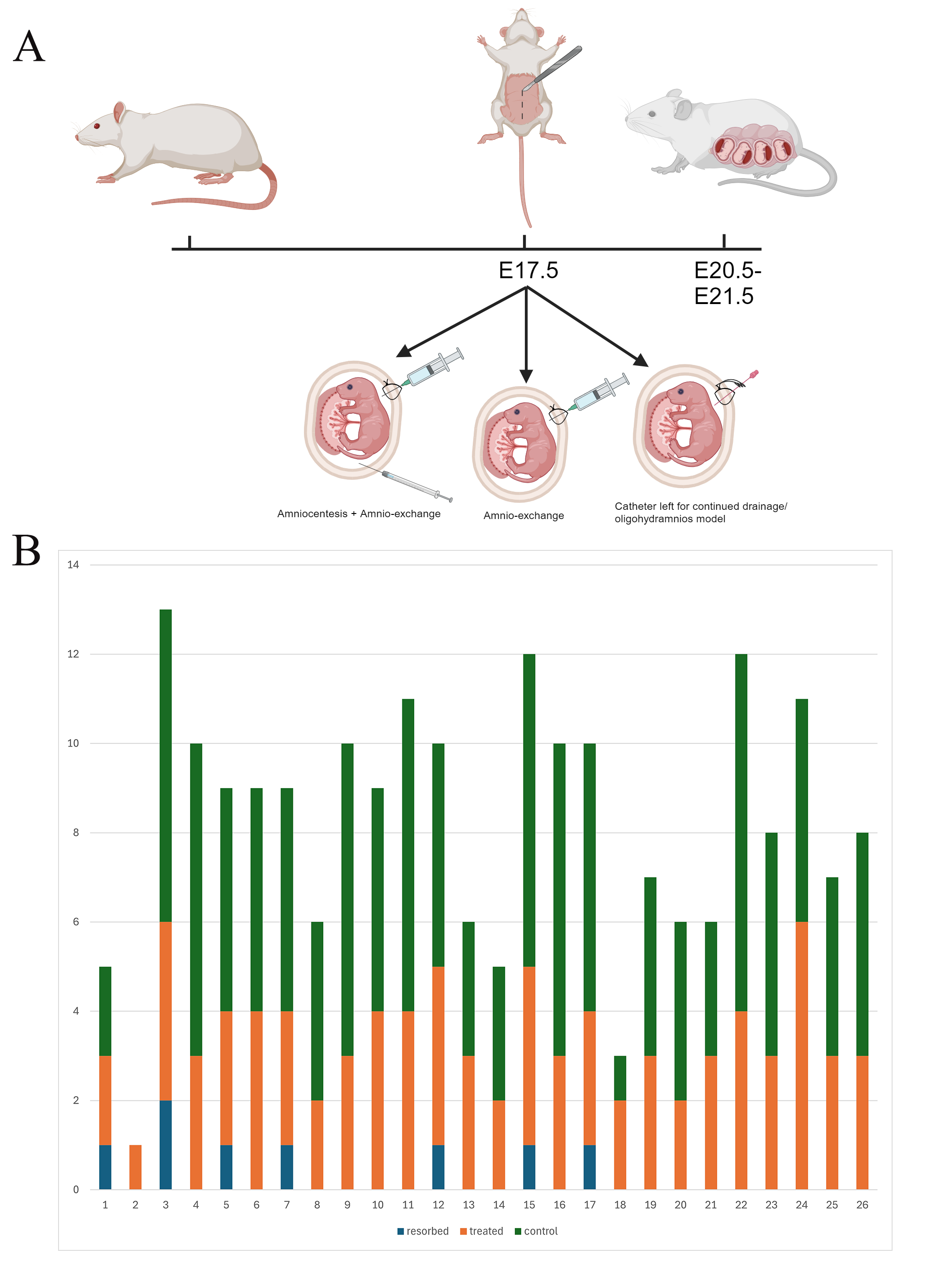 Fig. 3.
Fig. 3.
Timeline of interventions and fetal survival. (A) A design of the rat model with the various options for creation of the surgical model. (B) The number of fetuses overall and the number that underwent surgery. The X-axis depicts the rat number. The Y-axis depicts the total number of rats in the pregnancy, with each color corresponding to the number of rats experiencing either nothing (green), amniotic fluid exchange and survival (orange), or fetal demise in utero (blue).
This is the first rodent model of amniotic fluid exchange. It successfully allows for complete amniotic fluid replacement with the fluid of the researcher’s choice without significant maternal or fetal harm. This opens the door for testing a variety of substances upon the developing fetus in a model that carries several advantages over current models of amniotic fluid disorders. It will allow for mid-gestational amniocentesis and continued pregnancy. Furthermore, as amniotic fluid replacement has served as a fetal therapy in the past as well as been suggested as a fetal therapy in the future [10, 11], this study can serve as a model for studying amniotic fluid exchange as a treatment for amniotic fluid infusions. Further, it can serve as a model for oligo-amnios when a catheter is left in situ for amniotic fluid drainage. Or perhaps more significantly, this model can allow for cytokine specific impact upon the developing amniotic membranes, fetal lungs, or fetal gastrointestinal tract by replacing the amniotic fluid with any fluid of interest.
This study’s findings are significant when viewed against the backdrop of
existing research on both amniotic fluid exchange and the amniotic fluid’s role
in fetal development. Prior studies of amniotic fluid exchange have required
larger animals, speifically rabbits or chickens [12, 13]. Sheep are also a
frequent fetal model due to their very favorable fetal loss rate and low risk of
pregnancy complications. The relative cost for work in sheep and rabbits,
however, compared to murine models, is often more expensive by orders of
magnitude, limiting research in pregnancy related fields. Additionally, the
relative timeline of a pregnancy for a rat is 21–23 days, 31 days in a rabbit,
and 145 days in a sheep. Rabbits also carry a very high rate of fetal loss (as
high as
Luton et al. [11], in their randomized controlled trial of amniotic fluid exchange in humans for gastroschisis, noted that significant inflammatory markers were noted at the time of attempted exchange and that research was needed to understand the optimal treatment strategy to reduce amniotic fluid inflammation as well as the optimal frequency of amniotic fluid exchanges [12]. Furthermore, current studies of intra-amniotic inflammation in a rat or mouse model rely upon the injection of an endotoxin into the amniotic cavity. This is fraught with issues, with a risk of preterm labor and birth, as well as an inability to specifically study the cytokines which trigger preterm labor, fetal lung inflammation, and fetal gastrointestinal tract inflammation. Instead, the entire cascade must be studied at once as an endotoxin is administered. We show that an amniotic fluid exchange can be performed without harm to the developing fetus nor the fetal membranes, and thusly, can perform amniotic fluid exchanges with synthetic fluid that is loaded with high concentrations of a various cytokines which have been implicated in preterm labor, preterm premature rupture of membranes, or fetal lung and gastrointestinal tract injury. By being able to control every aspect of the intra-amniotic inflammatory pathway and expose one cytokine at a time, we can identify which cytokines are responsible for which perinatal complications, thus greatly increasing our understanding of perinatal disease.
Beyond the importance in preterm labor and intra-amniotic inflammation, the successful creation of an amniotic fluid exchange model in rats has far-reaching implications for fetal medicine research. It enables controlled studies on the impact of different substances introduced into the amniotic fluid, facilitating a deeper understanding of intrauterine development and the response to intrauterine treatments. This is critical as many studies of the amniotic fluid in normal and abnormal states reflect vast changes in the proteomic activity of the amniotic fluid [10]. It is entirely unclear how well in utero therapies currently being developed will act in the various states of intrauterine inflammation. There is great promise with intra-amniotc nanoparticle for delivery of targeted therapies, however the impact of the amniotic fluid’s activity in an inflammatory state (or even with the changes that occur through gestation) are unknown [15]. The ability to have a consistent amniotic fluid exchange to stablize the inflammatory state of the intrauterine environment thus has ability to allow for evaluation of various in utero therapies in various intrauterine environments. This could lead to significant advancements in prenatal therapies, particularly for conditions affecting the fetal lung, gastrointestinal tract or preterm labor.
One of the main strengths of this study is its innovative approach, which fills a notable gap in fetal medicine research. The technique’s high success rate, coupled with minimal impact on fetal and maternal health, underscores its potential for broader application. However, there are limitations to consider. The findings also raise questions about the long-term impact of amniotic fluid exchange on fetal development, which requires further investigation. Additionally, the technical skill required for the procedure could limit its widespread adoption in research settings. There are some limitations of this technique however, which should be noted. Firstly, these surgeries were done at E17.5, which is equivalent to the being of the preterm period in most murine models, and would translate to the late second/early third trimester in humans. Thus, this model is for evaluting late changes in a rat fetus model, not early changes secondary to amniotic fluid disorders. Next, these amnio-exchanges were undertaken only once per pregnancy, and thusly, the amniotic fluid produced by the fetus would eventually replace the fluid infused into the amniotic sac.
We present the first successful model of amniotic fluid exchange in rodents, allowing for further investigation of the amniotic fluid in a cheaper model than previously. This will open the door for significant research into the amniotic-fluid fetal interface.
The data supporting the findings of this study are included within the manuscript. Reasonable data inquiry request will be supported by the author, please contact the corresponding author.
BF—study design and performance of the procedure; writing of the manuscript; SM—performance of the procedure; JLP—study design and manuscript editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by Cincinnati Children’s Hospital Medical Center IACUC# 2022-0087. All animal research was conducted in accordance with the Institutional Animal Care and Use Committee and handling of the animals, including surgery and harvest were in accordance with humane treatment of animals.
The authors respect and appreciate the comments from the peer reviewers to improve the quality of this work. The authors thank the North American Fetal Therapy Network for their support of this work and their continued work in improving fetal and perinatal outcomes.
This research was supported by the North American Fetal Therapy Network Bill Polzin Seed Grant (SPR300815).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



