1 Department of Obstetrics and Gynecology, West China Second University Hospital, 610041 Chengdu, Sichuan, China
2 Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, 610041 Chengdu, Sichuan, China
3 West China School of Medicine, West China Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
Abstract
Objective: This review aims to examine the role of telemedicine in facilitating access to medication-induced abortion services, in the context of legal restrictions, geographical barriers, and the impact of the coronavirus disease 2019 (COVID-19) pandemic on healthcare delivery. Mechanism: This study conducted a narrative literature review focusing on the application of telemedicine in abortion care. It included analyzing various study types and evidence from peer-reviewed articles to evaluate the effectiveness, safety, and patient reception of telemedicine in providing medication-induced abortion. Findings in Brief: Medication-induced abortion via telemedicine is safe, effective, and well received by patients, offering a viable alternative to traditional in-person services. Telemedicine enables remote eligibility assessment, medication prescription, and follow-up care, significantly benefiting isolated or underserved populations. Conclusions: Telemedicine has emerged as a critical innovation in abortion care, enhancing access, safety, and equity in abortion services. It addresses significant barriers such as travel expenses and missed work, providing a patient-centered approach to abortion care. Future directions should focus on optimizing telemedicine protocols and expanding access to underserved populations, ensuring comprehensive abortion care through telemedicine.
Keywords
- telemedicine
- abortion care
- medication-induced abortion
- COVID-19 pandemic
Abortion techniques have been practiced since antiquity. In 2020, approximately 800 women worldwide die from preventable causes related to pregnancy and childbirth [1]. Therefore, prompt access to abortion services is imperative for the preservation of women’s health, lives and well-being. Medication-induced abortion has emerged as a viable alternative approach for pregnancy termination following the availability of prostaglandins in the 1970s and further advanced with the development of progesterone antagonists in the 1980s [2]. These pharmaceuticals are the most extensively studied and have been shown to be safe and efficacious for clinician-supervised termination of pregnancies, and are highly recommended by the World Health Organization (WHO) [3, 4, 5, 6, 7, 8, 9, 10].
Regardless of the legal framework governing abortion, women may seek alternative avenues for terminating pregnancies, including self-managed abortions, when they face barriers to or are reluctant to utilize institutionally provided abortion services [11]. Thus, telemedicine, emerged as an easily accessible approach to a safe abortion, is vital to women’s reproductive health. Telemedicine refers to the use of electronic communications and software to provide clinical services to patients without an in-person visit. This technology supports a range of healthcare activities, including diagnosis, treatment, consultation, medication management, and monitoring, in order to enable remote patient care. The coronavirus disease 2019 (COVID-19) pandemic was a catalyst to shift the delivery of healthcare toward telemedicine to ensure ongoing access to abortion services [12]. For obstetrics and gynecology, telemedicine has become integral to modern healthcare delivery, offering a critical solution particularly in areas with limited access to medical facilities [13].
Although there are sufficient high-quality studies confirming the effectiveness and safety of telemedicine abortion [14, 15, 16], many pregnant patients are still skeptical about this technique, possibly because telemedicine abortion has not been widely known to the public. In addition, with various telemedicine abortion provision models having been developed, there has been a notable absence of comprehensive synthesis or comparative reviews of these models. This narrative aims to discuss the safety and efficacy of medication-induced abortion through telemedicine from the service user’s perspective, to address that gap by demonstrating merits and pitfalls of each mode, and to expand telemedicine to abortion care and health management for service users. Although self-managed abortion uses much of the same evidence base, we will focus instead on telemedicine abortion delivered through formalized health care systems.
Our review meticulously identified and selected relevant literature using a method that is both thorough and aligned with scientific principles, ensuring the transparency and reproducibility of findings. We conducted an exhaustive literature search across a variety of databases to encompass a wide range of studies pertinent to medication-induced abortion and care through telemedicine. Based on the inclusion and exclusion criteria and assessment, this review encompasses a total of 133 studies.
Our search strategy was designed to explicitly explore the intersection of telemedicine and abortion care. We conducted a comprehensive literature search using a combination of keywords to ensure the focus on both telemedicine and abortion. The keywords included “Abortion”, “Abortion procedure”, “Abortion care”, “Induced abortion”, “Abortion technique”, “Medical abortion”, “Medication-induced abortion”, “Online”, “Telemedicine”, “Telehealth”, “Online abortion”, and “Teleconsultation”. These terms were combined using Boolean operators to capture studies that specifically addressed the provision of abortion services via telemedicine.
Databases searched included PubMed, Embase, Cochrane Library, and Web of Science, covering publications from January 2000 to January 2024. The search was limited to articles published in English and present in peer-reviewed journals to ensure the reliability and academic rigor of the sources. Further articles were identified through the reference lists of the initially selected manuscripts, ensuring a thorough exploration of the topic.
The inclusion criteria were: (1) participants were pregnant persons seeking medication-induced abortion; (2) participants received medication-induced abortion provided through telemedicine; (3) peer-reviewed empirical studies, including randomized controlled trials, cohort studies, case-control studies, and qualitative studies that provide data on patient experiences and outcomes; (4) studies conducted in the United States of America.
Exclusion criteria: (1) participants with issues other than pregnancy; (2) articles that did not directly relate to the use of telemedicine for abortion services, for example, those focusing broadly on telemedicine for other medical services and those focusing on in-person abortion procedures without aspects of telemedicine; (3) commentaries, letters to the editor, news articles, and conference abstracts that did not provide empirical evidence or detailed analysis; (4) studies focused on regions where telemedicine or abortion is not legally practiced, unless these provide comparative insights that can be generalized or adapted to the context under study; (5) articles not published in English or outside the specified time range of January 2000 to January 2024; (6) animal studies.
Medication-induced abortion is a procedure that uses prescription pills to end a pregnancy in the early stages, and the most common regimen involves taking two pills—mifepristone and misoprostol [17]. Telemedicine represents a paradigm of healthcare provision in which there is physical separation between healthcare providers and patients or between providers and specialists. Interactions between these parties can occur synchronously, facilitated through real-time communication technologies such as telephone or video connections, or asynchronously via the store-and-forward method. This method involves the submission of inquiries to which responses are later furnished, potentially through media such as email, text, or voice/audio messages [18]. Within this framework, the provision of medication-induced abortion via telemedicine encompasses a comprehensive suite of services for such abortions. These services may include, but are not limited to, assessing patient eligibility for medication-induced abortion, offering related counseling, providing detailed instructions and active support for the procedure, and conducting follow-up care—all executed through telemedical platforms.
An obstetrician with expertise in literature critical appraisal was responsible for reviewing the identified articles. This expert selected articles for inclusion based on consensus and relevance to medical practice in obstetrics, ensuring that the review focused on high-quality, relevant evidence pertaining to the use of telemedicine in abortion care. In assessing the quality of the included studies, the following criteria were used: (1) Methodological Rigor: the study must use clear and appropriate methods for data collection and analysis; (2) Relevance: the study must focus on telemedicine applications specific to abortion care, including patient outcomes and service delivery; (3) Credibility of Sources: sources must be published in reputable scientific journals; (4) Consensus Process: selection of articles was based on a consensus among the researchers. This involved discussions within our research team to agree on the inclusion of studies, ensuring that each study met our relevance and methodological rigor criteria. Discrepancies were resolved through discussion.
Based on the inclusion and exclusion criteria and evaluation, this review encompasses a total of 133 studies, and the flow chart of study selection is shown in the Fig. 1.
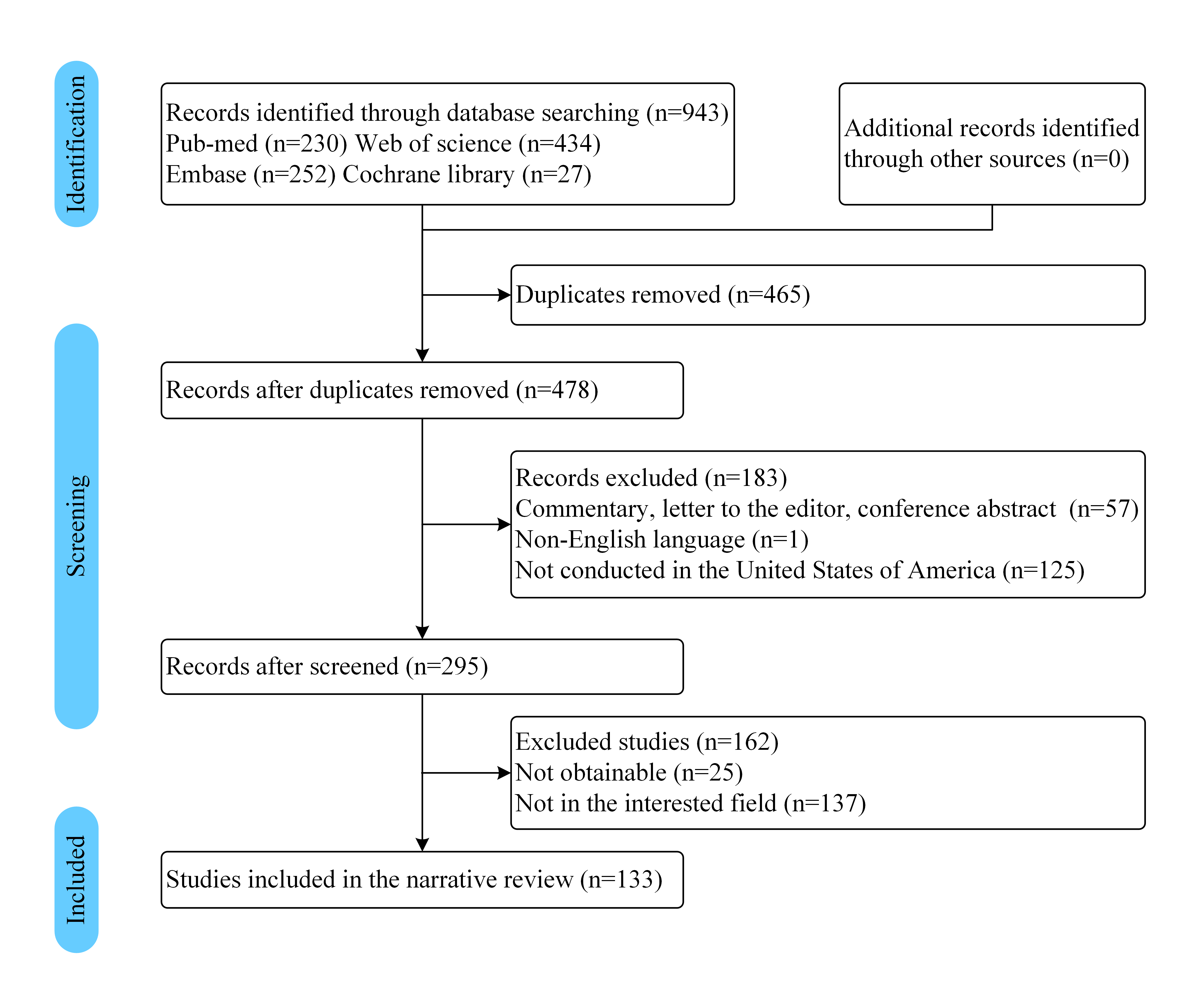 Fig. 1.
Fig. 1.Flow chart showing the study selection process.
The WHO advocates for telemedicine as a viable alternative to traditional in-person services for medication-induced abortion care [19]. Medication-induced abortion through telemedicine encompasses eligibility assessment for the procedure, guidance, medication prescriptions and management, and subsequent care, which allows for the secure and efficacious administration of medication-induced abortion and access to isolated or underserved populations. Moreover, in some circumstances, medication-induced abortion through telemedicine may be preferred, primarily because of self-determination, discretion, and secrecy [20].
Guaranteeing the safety of medication-induced abortion via telemedicine is a fundamental prerequisite for its effectiveness. A comprehensive meta-analysis revealed that telemedicine-mediated medication abortion is highly regarded by both patients and practitioners, exhibiting comparable success and safety metrics to in-person services, albeit with a noted increase in the necessity for surgical interventions (1%–19%) [21]. Moreover, a systematic review that included four studies on medication abortion underscored that the utilization of telehealth not only enhances access to early abortion services but also guarantees clinical outcomes comparable to those achieved through conventional care [22].
However, apprehensions concerning the potential adverse effects and overall safety of medication for abortions continue to pose challenges to widespread adoption. A detailed retrospective cohort study comparing 8765 telemedicine-facilitated abortions to 10,405 in-person medication-facilitated abortions indicated a low incidence of adverse events, confirming the noninferiority of telemedicine in preventing significant adverse outcomes [23]. Furthermore, the rate of mortality associated with medication-induced abortions is approximately 0.65 deaths per 100,000 procedures, rendering medication-induced abortion more than 13 times safer than childbirth in the United States [24, 25].
Clarifying the optimal medication-induced abortion regimen is critical to
obtaining a secure abortion. An update of a review demonstrated that the
combination of mifepristone and misoprostol has been proven to be more efficient
than utilizing these medications individually [26]. When the combination of
mifepristone and misoprostol is administered under the careful supervision of
clinical professionals, the occurrence of significant complications such as
hospitalization, surgical intervention, or the need for blood transfusion is
infrequent, affecting only a small portion of patients, specifically 0.3% [24].
The combination of mifepristone along with misoprostol or misoprostol alone
facilitates abortions, as indicated by the typical medication protocols listed in
Table 1 [17], which is also supported by the WHO [27, 28, 29, 30]. For medication-induced
abortion at
| Gestation period | Medication protocol |
| Up to 9 weeks (63 days) | Mifepristone, 200 mg administered orally, followed by misoprostol, 800 µg administered vaginally, buccally or sublingually 24–48 hours later. |
| Misoprostol 800 µg, vaginal or sublingual, every 3–12 hours for 3 doses. | |
| 9–12 weeks (63–84 days) | Mifepristone 200 mg orally, followed by misoprostol 800 µg buccally, vaginally, or sublingually after 36–48 hours, then misoprostol 400 µg every 3 hours for up to 5 doses. |
| Misoprostol 800 µg vaginally or sublingually, every 3–12 hours, 3 doses. | |
| Mifepristone 200 mg orally, followed in 36–48 hours by misoprostol 800 µg vaginally, then misoprostol 400 µg buccally, vaginally or sublingually every 3 hours for up to 5 doses. | |
| Mifepristone 200 mg orally, followed in 36–48 hours by misoprostol 400 µg orally, then misoprostol 400 µg buccally, vaginally or sublingually every 3 hours for up to 5 doses. | |
| Misoprostol, 400 µg administered vaginally or sublingually every 3 hours up to 5 doses. |
Discussion around the use of mifepristone in the context of telemedicine-enabled abortion services often focuses on its regulatory history and clinical application. It is critical to specify that the approval and use of mifepristone discussed here pertains to its role in telemedicine practices in specific country or region, e.g., the United States, where regulatory adjustments have had significant impacts on the accessibility of medication-induced abortion. This context is essential to understanding the barriers and facilitators in the adoption of telemedicine for abortion care, reflecting recent changes that allow providers to prescribe and send mifepristone through the mail following a telemedicine consultation.
The focus of this review on the efficiencies of clinic/providers in implementing telemedicine is aimed at highlighting how telehealth can streamline operations and reduce resource burdens. This emphasis is chosen to illustrate the potential for telemedicine to enhance healthcare delivery systems, especially in abortion services, where provider shortages and clinic accessibility are ongoing challenges. However, this does not preclude the importance of service user perspectives, which are also considered throughout the paper to provide a balanced view of the implications of telemedicine in abortion care.
After its initial approval in 2000, mifepristone mandated the completion of all procedural steps, barring the termination itself, within a clinical setting, constrained under the US Food and Drug Administration (FDA) [26]. Subsequent relaxations in regulatory frameworks governing the medical abortion protocol have facilitated a more patient-centric approach, enabling the utilization of mifepristone and misoprostol outside the clinic with remote follow-up assessments, related to the COVID-19 pandemic in 2020 [31]. This transformation underscores the pivotal role of telemedicine in diversifying abortion care models, notably characterized by spatial separation between the healthcare provider issuing the prescriptions and the patient [32].
This evolution has given rise to two primary telemedicine abortion models: direct-to-clinic and direct-to-consumer approaches [33]. The direct-to-clinic model permits patients to access care within local facilities while engaging in telecommunication with off-site physicians, thereby eliminating the necessity for onsite medical personnel [34]. This model is particularly beneficial in rural or underserved areas, where it addresses the disparity in provider-to-patient ratios [35]. Data from a prospective cohort study carried out by Planned Parenthood of the Heartland indicated that there were no notable differences in the completion rates of abortions, adverse events, or patient satisfaction when comparing traditional in-clinic services to the direct-to-clinic telemedicine approach [14]. Furthermore, a retrospective cohort study corroborated that telemedicine-facilitated medical abortion did not result in a greater incidence of adverse events than in-person medical abortion [16, 25, 36]. Despite the direct-to-clinic model’s efficacy in mitigating provider shortages and enhancing scheduling flexibility, it does not substantially alleviate the associated burdens of transportation, time, and financial costs for patients [32].
Conversely, the direct-to-consumer model presents a paradigm shift toward cost efficiency and operational streamlining for clinics [37]. This model facilitates direct interactions between women and healthcare providers via video calls, with abortifacient medications being dispatched through mail or prescription, bypassing the necessity for physical clinic visits [32]. The inherent flexibility of this model, unbound by geographical constraints for either party or specific locales for screening procedures, holds particular promise in areas where the availability of abortion services is constrained [38]. Its utility is further amplified in contexts where screening and laboratory testing capabilities are available, albeit disconnected from traditional abortion service locations [21]. This direct-to-consumer approach not only broadens the accessibility and convenience of abortion care but also represents a significant stride toward mitigating logistical barriers, thereby enhancing the overall efficacy of medication-induced abortion provision in underserved areas [39].
Further endeavors to streamline the medication abortion process are underway, including the potential elimination of certain preliminary procedures such as pretreatment ultrasound or Rh (Rhesus) factor blood test. The American College of Obstetricians and Gynecologists (ACOG) supports the practice of estimating gestational age through remote assessment for patients who have confirmed their pregnancy via a home urine test, who report regular menstrual cycles, who are certain about their last menstrual period date, and who exhibit no risk factors associated with ectopic pregnancy, without the necessity for ultrasound confirmation [40]. A recent study examining the safety and appropriateness of omitting pretreatment ultrasounds or pelvic examinations before medication-induced abortion revealed a low rate of serious adverse events (1%), with a significant majority (90%) of women expressing satisfaction with the elimination of these preliminary procedures [41, 42, 43, 44, 45]. Moreover, the Society of Family Planning revised its guidelines in 2022 regarding Rh testing and the administration of Rh immunoglobulin for patients under 12 weeks of gestation seeking medication abortions. This revision was informed by research indicating the unlikelihood of fetal red blood cells to induce an alloimmunization response before the 12-week gestation threshold, underscoring a shift toward a more streamlined and patient-centric approach in the provision of medication-induced abortion services [46]. Conclusions of the traditional model, direct-to-clinic model, and direct-to-consumer model are listed in the Table 2.
| Aspect | Traditional model | Direct-to-clinic model | Direct-to-consumer model |
| Site | Local clinic | Local clinic & digital platform | Digital platform (home-based) |
| Screening | In-person: lab tests, ultrasound | Remote: lab tests, video-assisted ultrasound | At-home: self-administered tests and assessments |
| Counseling | In-person agreement | Videoconference with physician | Videoconference with physician |
| Medication | Mifepristone at clinic, misoprostol at home | Dispensed on-site | Mailed to home with instructions |
| Follow-up | In-clinic confirmation, 1–2 weeks later | Remote follow-up using ultrasound or |
Remote follow-up with home pregnancy test |
Hemoglobin, blood type and the quantitative serum human chorionic gonadotropin
concentration are measured via laboratory tests.
Building upon the detailed exploration of service user perspectives previously introduced, this discussion section seeks to further analyze how these insights integrate with broader themes of accessibility and efficiency in telemedicine for abortion care. We also considered the implications of these user experiences for future policy development and research directions in telemedicine applications.
Telemedicine has emerged as a pivotal innovation in abortion care, offering medication abortion services that are both safe and effective while also being highly valued by patients and healthcare providers alike [15, 47, 48]. This approach is especially beneficial for those residing far from abortion facilities, as it addresses significant barriers, including childcare, travel expenses, and missed work, thereby enhancing accessibility and equity in abortion care [14, 15, 47, 49]. Convenience is often cited as a primary reason for selecting telemedicine, with patients appreciating the ability to undergo the process within the privacy and comfort of their own homes [50]. Moreover, telemedicine mitigates the financial and logistical strains imposed by legal restrictions such as mandatory delays and informed consent, which can exacerbate psychological distress and health risks by delaying access to abortion services [48, 51, 52, 53].
Although not universally suitable, particularly for individuals preferring in-person consultations or lacking the technological means for videoconferencing, telemedicine nonetheless represents a critical adjunct to traditional care models. It promises improvement over time, contrasting with the immutable challenges of distance and financial constraints inherent to in-person visits [54]. The COVID-19 pandemic has underscored telemedicine’s role in maintaining access to abortion services amid heightened demand and operational restrictions, paralleling its adoption across other healthcare domains for preprocedural consultations [55]. As telemedicine continues to evolve, it holds the potential to fulfill unmet needs for safe abortion access, especially in resource-constrained environments lacking comprehensive medical facilities, thereby ensuring a broader reach to those facing substantial barriers to clinic-based care [56, 57, 58].
This review has explored the pivotal role of telemedicine in enhancing access to abortion services, particularly focusing on its operational efficiencies and impact on service delivery. While telemedicine presents a promising solution to circumvent geographical barriers and improve provider workflows, our review has also highlighted the complexities introduced by varying legal landscapes. Although the specific interactions between telemedicine and legal restrictions were not extensively covered in the body of the paper, the implications are significant and merit further detailed exploration in future research.
We conclude that telemedicine in abortion care not only offers potential benefits in terms of accessibility and efficiency but also requires careful consideration of legal, ethical, and social dimensions. To optimize the integration of telemedicine into abortion services, policymakers and healthcare providers must navigate these challenges thoughtfully, ensuring that telehealth initiatives align with legal standards and fully support patient needs. Future studies should focus on the legal implications of telemedicine to provide clearer guidance for practice and policy, ensuring that telemedicine not only supports but enhances the provision of safe and accessible abortion care.
Both the authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by AW and KZ. The first draft of the manuscript was written by AW and KZ, and both the authors commented on previous versions of the manuscript. Both the authors have read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

