1 Department of Obstetrics and Gynecology, and CERICSAL (CEntro di RIcerca Clinico SALentino), “Veris Delli Ponti" Hospital, 73020 Scorrano, Italy
2 R & D Department, Ekuberg Pharma, 73020 Carpignano Salentino, Italy
†These authors contributed equally.
Abstract
Background: One of the most popular surgical procedures is the cesarean
section (CS), and one of the most prevalent side effects of this procedure is
wound and scar issues. The aim of this prospective, short-term,
placebo-controlled study was to evaluate the safety and effectiveness of a
silicone-based medical device (Lecoxen cream, Ekuberg Pharma, Carpignano
Salentino, Italy) in healing wounds and scars derived from cesarean sections in
nulliparous women. Methods: Seventy-four nulliparous women, ranging in
age from 18 to 45, and underwent CSs, were divided in two groups of treatment
(Lecoxen cream or Vaseline). The quality of scar was examined using the Vancouver
scar scale after 28 days, while wound healing was evaluated using the Redness,
Edema, Ecchymosis, Discharge, and Approximation (REEDA) scale on the 14th day
following CS. Results: The treatment with Lecoxen cream determined a
statistically significant improvement (p
Keywords
- cesarean section
- wound healing
- cesarean scar
- Lecoxen cream
- pregnancy
Nowadays, one of the most prevalent surgeries is the cesarean section (CS), and in most countries, the current rate of these procedures has tremendously increased [1, 2]. This type of obstetric surgery is being performed on an ever-increasing number of women, which has sparked worries about possible problems with the mother’s post-operative condition. Consequently, the scar from the CS results from the surgical incisions made in the abdomen and uterine wall to extract the baby from the mother’s womb. The incision can be made either transversely or longitudinally, depending on the type of surgery that is required.
According to clinical data, wound complications are among the most frequent post-CS morbidities, ranging from 2.5% to 34% of percentage [3, 4]. Most of the time, what happens is an aberrant healing of the wound, which not only increases the risk of hypertrophic scar formation but also slows down the wound’s natural healing process [5]. A hypertrophic scar is a significant scar due to the generation of a large amount of fibrotic tissue, which makes the skin thicker at that point. This scar, which can result in pain, itching, contractures, obstructions to mobility, and other functional and cosmetic discomforts, symbolizes an aberrant and heightened healing response to skin lesions [6, 7].
Abnormal healing may also be associated with an infectious process that initiates in the afflicted area of the wound. Numerous studies have shown that the prevalence of wound infection after CS ranges from 3 to 15%, with an average of 6% and 2–42% [8, 9]. Because of the infected wound, the mother’s post-operative clinical status is typically worrying, and this can occasionally have a detrimental effect on both physical and psychological well-being. As a matter of fact, the mother may experience tension, anxiety, a post-operative wound complication, a delay in her recuperation, and overall pain [10].
Depending on the needs of the patient, prevention can be used in a variety of invasive and non-invasive therapeutical ways to keep the clinical picture from worsening. Good wound care is required prior to employing these therapies, since gynecologists advise keeping the area affected by the wound well-protected and out of the sun for the following few months.
Because to its affordability, convenience, non-invasiveness, and ease of administration, topical therapies have grown in popularity. Topical medications have become standard practice in the treatment of CS wounds because they accelerate and promote healing, which reduces the likelihood of abnormal and unsightly scars forming. The patient’s overall health and quality of life are also enhanced by the use of this kind of product. One of the frequently employed methods involves the use of silicone in topical sheets or gels.
In this context, a medical device based on topical treatment, the Lecoxen cream (Ekuberg Pharma, Carpignano Salentino, Italy), is currently used in wound care treatment. This product, thanks to its water-in-silicone emulsion, is able to maintain a correct wet microenvironment at the level of the wounds and encourages the healing process by preventing the formation of keloids.
This short-term, placebo-controlled trial sought to determine the safety and effectiveness of Lecoxen cream in treating the wounds caused by cesarean sections in nulliparous women in order to enhance the healing process and, ultimately, the quality of the scar that results.
The research was conducted from June 2023 to December 2023. The Ethics Committee (ASL Lecce-N° 75839/2023) accepted the study, and each participant provided written consent. The ethical guidelines for medical research involving human subjects (Ethical Guidelines for Medical Research Involving Human Subjects, approved by the 18th World Medical Association, General Assembly in Helsinki, Finland, June 1964, and subsequent updates) were followed throughout the entire study process. All nulliparous women, aged from 18 to 45, submitting to CS surgery in the Department of Gynecology and Obstetrics of the “Veris Delli Ponti” Hospital in Scorrano (Lecce, Italy), were considered to be enrolled. The study protocol has been registered in the ISRCTN registry https://www.isrctn.com/ (ISRCTN51125312). The inclusion criteria were the following: healthy women scheduled for CS, without a diagnosis of autoimmune diseases, skin problems or known hypersensitivity to at least one of the components of the formulation Lecoxen cream or Vaseline. The exclusion criteria were smoking women, patients at risk for premature birth, obesity, hypertension, diabetes and infections, women who use systemic and topical corticosteroids and with presence of previous surgical scarring near the incision site. The general characteristics of enrolled women were shown in Table 1.
| Total | Lecoxen | Vaseline | p-value | |
| N = 74 | N = 37 | N = 37 | ||
| Age | 32.1 |
32.5 |
31.6 |
0.44 (t = –0.76) |
| Weight | 68 (63, 73) | 67 (63, 72) | 70 (63, 73) | 0.58 (z = 0.55) |
| Height | 1.63 |
1.61 |
1.63 |
0.28 (t = 1.08) |
| BMI | 25.6 (24.5, 27.7) | 26 (24.6, 27.6) | 25.6 (24.5, 27.7) | 0.63 (z = –0.48) |
NOTE: BMI, body mass index.
All pregnant women submitted to CS were operated by a Pfannenstiel skin incision, and then the skin was closured by agrafes (Agrafes Appose, Medtronic Italy, Milano, Italy). After surgery, patients were divided into two groups: 37 mothers were allocated to the treatment group with Lecoxen cream, 37 to the placebo group treated with Vaseline (Vaseline F.U., Marco Viti, Vicenza, Italy). Participants were assigned in a 1:1 ratio with sequential assignment, depending on time and date of enrolment. Treatment with Vaseline (pharmaceutical grade) is suggested in normal clinical practice to keep the wound environment under control and avoid excessive transcutaneous water loss, that could result in the formation of aberrant scar.
After the stitches’ removal, normally on the seventh post-operative day, patients were instructed to apply respective treatments from the first day to the twenty-eighth day, with a frequency of once a day, preferably at the morning. Patients were asked not to use any other medicament for wound healing. Patients were instructed to apply daily the product administrating a sufficient dose of product to cover both damaged skin and perilesional skin, after a normal cleaning with isotonic solution and removing eventual residuals of the previous application.
A board-certified gynecologist, all doctors working in the department where the study was performed, supervised the screening, the enrollment of study participants from a panel of candidates, and followed the patients during the study.
The state of wound healing was evaluated at baseline and after 14 days (T1) of the treatment using Redness, Edema, Ecchymosis, Discharge, and Approximation (REEDA) scale [11]. The degree of quality of the resulting scar was finally evaluated after 28 days (T2), using Vancouver Scar Scale (VSS). The VSS consists of four variables including pigmentation, height, flexibility and vascularity [12, 13]. At each timepoint relative questionnaires were filled in and the data were statistically processed. In case of non-expected/adverse reaction occurrence, the medical experimenter evaluated the severity of the reaction (reporting it in the data collecting sheet) and consequently an appropriate therapy was prescribed. Primary outcome included evaluation of wound healing process and quality of resulting scar together with an evaluation of any adverse event.
Data analysis was performed with Stata® software (version 12; StataCorp LLC; College Station, TX, USA).
We perform the Shapiro-Wilk test to verify if the quantitative variables were
normally distributed. We rejected the null hypothesis of normal distribution
except for age and height. Normally distributed quantitative variables were
reported as mean and standard deviation. Non-normally distributed data as weight
and body mass index (BMI) were reported as median (P25, P75) with 25th and 75th
percentiles. Data in Table 2, due to the distribution of the raw data, were
reported as mean and standard deviation that is more intuitive. Comparisons
between the groups for normally distributed variables were performed by using
t-test. Non-normally distributed variables as weight and BMI were
compared by using non-parametric Kruskal-Wallis test while remaining variables
were compared by using non-parametric Wilcoxon signed-rank test. The t-values for
the parametric tests and the z-values for the non-parametric tests were reported.
A p-value lower than 5% (p
| Lecoxen | Vaseline | p-value | ||
| N = 37 | N = 37 | |||
| REEDA T0 | ||||
| Redness | 1 (1, 2) | 1 (1, 2) | 0.89 (z = 0.14) | |
| Edema | 1 (1, 1) | 1 (0, 2) | 0.87 (z = 0.16) | |
| Ecchymosis | 1 (0, 1) | 1 (0, 1) | 0.93 (z = 0.08) | |
| Discharge | 1 (0, 1) | 1 (0, 1) | 0.80 (z = 0.26) | |
| Approximation | 1 (0, 1) | 1 (0, 1) | 0.86 (z = 0.20) | |
| REEDA T14 | ||||
| Redness | 0 (0, 0) | 1 (0, 1) | ||
| Edema | 0 (0, 0) | 1 (0, 1) | ||
| Ecchymosis | 0 (0, 0) | 1 (0, 1) | ||
| Discharge | 0 (0, 0) | 1 (0, 1) | ||
| Approximation | 0 (0, 0) | 1 (0, 1) | ||
| VSS T0 | ||||
| Vascularization | 1 (0, 2) | 1 (0, 2) | 0.88 (z = 0.16) | |
| Pigmentation | 1 (0, 2) | 1 (0, 2) | 0.92 (z = 0.10) | |
| Flexibility | 1 (1, 2) | 1 (1, 2) | 0.76 (z = –0.31) | |
| Height | 1 (0, 1) | 1 (0, 1) | 0.60 (z = 0.53) | |
| VSS T28 | ||||
| Vascularization | 0 (0, 0) | 1 (0, 1) | ||
| Pigmentation | 0 (0, 0) | 0 (0, 1) | ||
| Flexibility | 0 (0, 0) | 1 (1, 1) | ||
| Height | 0 (0, 0) | 1 (0, 1) | ||
NOTE: REEDA, Redness, Edema, Ecchymosis, Discharge, and Approximation; VSS, Vancouver Scar Scale.
On a total of eighty-two pregnant women, seventy-four patients completed the study. The study flowchart is shown in Fig. 1.
 Fig. 1.
Fig. 1.Flowchart of the patient selection. CS (cesarean section), prospective observational clinical trial.
There was no significant difference between the two groups in terms of demographic features such as age, weight, height, BMI and pregnancy characteristics (p ˃ 0.05) (Table 1), and clinical data were compared between baseline and final measurements for each patient, as assessed by medical professionals (Table 2).
The treatment with Lecoxen cream determined a statistically significant
improvement of the wound healing state, in terms of redness, edema, ecchymoses
and discharge. The redness parameter was 1 (1, 2) at baseline (T0) and 0 (0, 0)
at the end of the analysis (T14) (Table 2). The edema parameter was 1 (1, 1) at
baseline and 0 (0, 0) at the end of the analysis (Table 2). The ecchymosis
parameter was 1 (0, 1) at baseline and 0 (0, 0) at the end of the analysis (Table 2). The discharge parameter was 1 (0, 1) at baseline and 0 (0, 0) at the end of
the analysis (Table 2). The approximation parameter was 1 (0, 1) at baseline and
0 (0, 0) at the end of the analysis (Table 2). There was a significative
improvement in the treatment group for each parameter (p
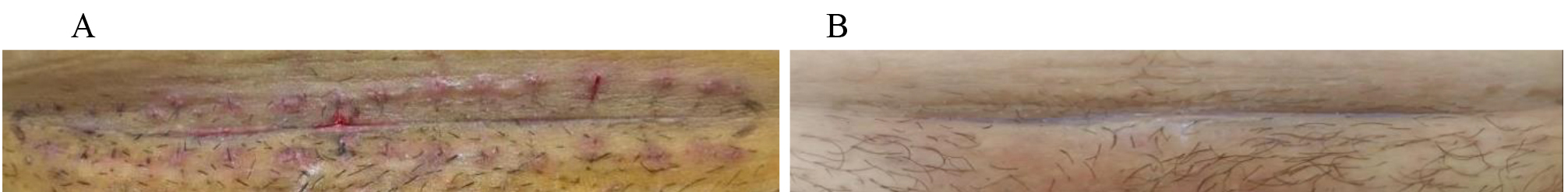 Fig. 2.
Fig. 2.Cesarean section treated with Lecoxen cream (Group 1). At T0, after stitches removal (A) and after 14 days of treatment (B).
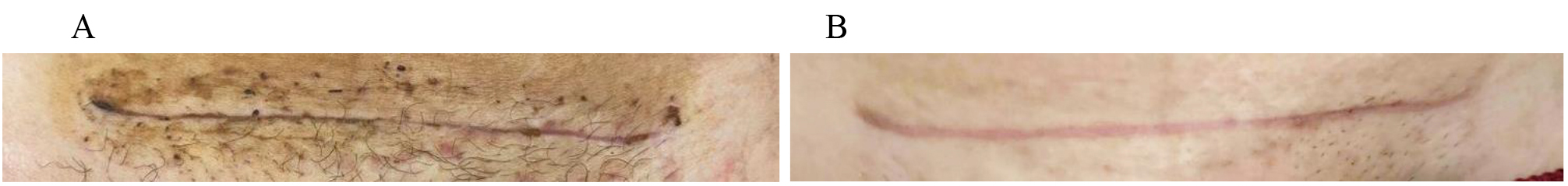 Fig. 3.
Fig. 3.Cesarean section treated with Vaseline (Group 2). At T0, after stitches removal (A) and after 14 days of treatment (B).
Resulting scar appeared absolutely approximated and without sign of redness or oedema. The wound and the perilesional margins appeared clean and the deep was reduced. Redness around the wound area (Fig. 2A) was significantly reduced (Fig. 2B), thus indicating proper tissue restoration in the affected area. Visible swelling (Fig. 2A) resulting from the incision was decreased at every point of the wound (Fig. 2B). The absence of visible ecchymosis and discharge at the site of the incision (Fig. 2B) allowed us to confirm the complete healing of the wound resulting from the caesarean section, also restoring a compliant skin tone and aesthetic appearance in the area. These photos represents an example of the significative improvement observed in Group 1 in comparison with Group 2 for the parameters evaluated through REEDA scale, as showed in Table 2.
Despite wound was completely closed, clear signs of redness and slight oedema was present. Redness, assessed according to the REEDA scale, was visible even after 14 days of treatment (Fig. 3B). Edema, although reduced, was visible at the perilesional margins (Fig. 3B). Discharge was still present in some areas of the wound, and visible ecchymosis was visible in the wound area, as well as in the surrounding area (Fig. 3B). These photos represents an example of the significative improvement observed in Group 1 in comparison with Group 2 for the parameters evaluated through REEDA scale, as showed in Table 2.
Furthermore, the treatment with Lecoxen cream determined a statistically
significant improvement of the assessment of the resulting scar’s quality, in
terms of vascularization, pigmentation, flexibility and height. The
vascularization parameter was 1 (0, 2) at baseline (T0) and 0 (0, 0) at the end
of the analysis (T28) (Table 2). The pigmentation parameter was 1 (0, 2) at
baseline and 0 (0, 0) at the end of the analysis (Table 2). The flexibility
parameter was 1 (1, 2) at baseline and 0 (0, 0) at the end of the analysis (Table 2). The height parameter was 1 (0, 1) at baseline and 0 (0, 0) at the end of the
analysis (Table 2). Even about VSS score there was a significative improvement in
the observed group for each parameter (p
Among the most frequent complications of CSs, which patients generally complain about, are wound healings, which cause physical blemishes and skin discomfort. What occurs in most cases is an abnormal healing of the wound, which in addition to predisposing to the formation of hypertrophic scars, also delays the healing process of the wound itself [5]. Up to now, several studies have been conducted on cesarean wound healer.
However, none of these studies has investigated the effect of this ointment on abdominal surgery wound healing.
A skin scar is the physiological result of damage or injury to the skin. A CS scar results from a longitudinal or transverse incision depending on the type of surgery that is required. Wound healing of the skin is a very complex and dynamic biological activity. In the event of inadequate or insufficient treatment, complications may develop following wound healing, such as the formation of aberrant or keloid scars. This type of complication is associated with the appearance of symptoms such as itching, burning and discomfort in the wound area, which disrupt the patient’s physical and psychological well-being. The quality of life is compromised by the persistence of the wound and the cost of care is manifested both psychologically and in prolonged hospitalization time, as well as in morbidity and even the cost of care [14].
According to the literature, wound healing is a complex dynamic process and patient’s recovery and quality of life could be highly affected by protocols of treatment [15]. Currently, researchers are paying attention to the use of methods that are inexpensive, effective, appropriate and accessible in the health care setting, and which, in the meantime, are acceptable to postpartum women [16]. Due to its affordability, convenience, non-invasiveness and ease of administration, topical therapies, associated with the administration of antibiotics, have grown in popularity. In particular hydrogel wound dressings and creams can improve healing, furnishing a positive effect on the final outcome. These topical medications enable real- time wound monitoring and can be used as a conveyance vehicle for bioactive compounds [17, 18]. Caesarean section rates are progressively rising around the world. Cause of this increase is represented by frequent requests by women for caesarean section in the absence of clear medical indications, such as placenta previa, human immunodeficiency virus (HIV) infection, contracted pelvis and, arguably, breech presentation or previous caesarean section [19].
In this context, Lecoxen cream, a medical device based on substances is suggested for the treatment of skin wounds, including caesarean sections. The mechanism of action of Lecoxen cream is mainly represented by the ability to form a protective and moisturizing film on wound site, favoring the maintenance of a proper wet environment, useful for the promotion of healing as theorized by Winter [20]. Lecoxen is characterized by a water in silicone emulsion that is able to protect the wound site and peri wound margins, and simultaneously provide a long-lasting release of water.
Results of this clinical trial showed a positive effect of Lecoxen cream on wound healing, resulting effective in ameliorating the scar formation. All evaluated wound parameters improved significantly after treatment with Lecoxen cream after 14 days compared to placebo. Furthermore, patients treated with Lecoxen cream reported a significative higher quality of resulting scar tissue after 28 days compared to placebo. No adverse effects were reported from the use of Lecoxen cream in participants.
Results confirmed the properties of Lecoxen cream both in terms of safety and efficacy as topical wound care treatment even in CS.
The present study showed that Lecoxen cream was safe and effective in CS wound healing, and it can be considered as an adjunctive treatment to the standard treatments of cesarean wound. Lecoxen cream proved to be an effective medical intervention for accelerating healing in CS, even with short-term use. Compared to the placebo formulation, Lecoxen cream treatment speeded up wound closure and improved the resultant scar tissue. Both medical professionals and patients concurred that using Lecoxen cream correctly to treat wounds. In particular, the benefits observed in this clinical study could be justified by the formulation characteristics of Lecoxen cream, which as a water-in-silicone emulsion, is able to create a moist environment suitable for the wound by promoting healing.
Absence of notable side effects attests to Lecoxen cream’s high degree of safety and tolerability, and it may inspire future research into the cream’s potential as a useful therapy to promote the biological healing of a wider range of wounds. Further studies are suggested to make Lecoxen cream standardized and to evaluate the effects of its dosage, duration and the repetition of treatments, the stability and potency of the medication on storage and the costs. The inability to accurately assess the status of caesarean wound healing by biopsy was one of the limitations of this study.
Further studies are suggested to make Lecoxen cream standardized and to evaluate the effects of its dosage, duration and the repetition of treatments, the stability and potency of the medication on storage and the costs, including a proper sample size calculation a priori. The inability to accurately assess the status of caesarean wound healing by biopsy was one of the limitations of this study, since most measurements were subjective and based on qualitative assessment of the scar. Furthermore, it should be considered that prevention or emergent deliveries may have a role in improvement of short and long term surgical outcomes.
The data sets generated and/or analyzed during the current study are available after proper request to the authors and to CERICSAL research organization.
Conceptualization—AT and DC; methodology—SG, GP, ML and GD; formal analysis—CS and DC; preparate and writing original draft—GP; writing review and editing—AT, CS and DC; supervision—DC. All authors contributed to editorial changes in the manuscript. All authors have read and agreed to the published version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was approved by the Local Ethics Committee—ASL Lecce-N° 75839/2023. All patients signed an informed consent.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Davide Carati and Camilla Schinzari employed in Ekuberg Pharma, Via Tito Schipa 6, Carpignano Salentino, 73020 Lecce, Italy. They collaborated in the processing of the data (collected by clinicians) and in the statistical evaluation of the data, contributing to structure the manuscript. The analysis, interpretation of data, and writing the manuscript have all been done by the authors and Ekuberg Pharma had no control on them. Their employment did not affect their judgment in interpreting the results.
Andrea Tinelli is serving as one of the Editorial Board members/Guest editors of this journal. We declare that Andrea Tinelli had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Paolo Ivo Cavoretto.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



