1 Department of Obstetrics and Gynecology, Faculty of Medicine, Sivas Cumhuriyet University, 58050 Sivas, Turkey
2 Department of Medical Genetics, Faculty of Medicine, Aksaray University, 68100 Aksaray, Turkey
Abstract
Background: Preeclampsia, affecting 2–8% of pregnancies, is
characterized by new-onset hypertension, proteinuria, and edema, stemming from
placental dysfunction and subsequent inflammatory cascade activation. This study
aimed to test the predictive ability of hematological indices in predicting
preeclampsia. Therefore, predicting the diagnosis of preeclampsia earlier with
higher accuracy could potentially preserve the lives of both the mother and the
fetus, while also reducing the rates of complications. Methods: This
retrospective study included two groups: patients with preeclampsia and a matched
control group without additional medical conditions, both having undergone
childbirth in the same gestational week. The primary difference between the
groups was the preeclampsia diagnosis. Hematological parameters and inflammatory
indices were compared for analysis. Results: Statistical significance
was observed between derived neutrophil-lymphocyte ratio (dNLR), systemic
immune-inflammation index (SIRI) and pan-immune inflammation value (PIV)
between groups. The dNLR and PIV showed predictive relevance for preeclampsia (p
Keywords
- preeclampsia
- inflammation
- hematological parameters
- inflammatory indices
- derived neutrophil lymphocyte ratio
- pan-immune inflammation value
- systemic inflammatory index
Preeclampsia (PE) is a complex pregnancy-related disorder, occurring in the latter half of gestation and impacting 2–8% of pregnancies, with an elusive etiology [1]. Typically emerging post the 20th week of gestation, PE affects both maternal and fetal health, escalating the risks of maternal and neonatal mortality and morbidity. Hallmark features of PE include hypertension, edema, and proteinuria [2]. Oxidative stress (OS), denoting an imbalance between reactive oxygen species generation and antioxidant defense mechanisms, is implicated in various pathological processes, including PE [3]. Severe cases of PE can precipitate systemic complications such as hemolysis, thrombocytopenia, and hepatic dysfunction, stemming from organ malfunctions in the maternal body [4, 5, 6, 7, 8, 9].
PE can be conceptualized as excessive activation during fetal acceptance’s abnormal immune tolerance process, culminating in a multisystem inflammatory cascade, adversely impacting all organs. This inflammatory milieu associated with PE arises from aberrant maternal immune responses, potentially inciting widespread inflammation across various organ systems [10]. It is hypothesized that this inflammatory cascade contributes to the pathogenesis and progression of PE, exacerbating complications for both the mother and the fetus. Recent research has increasingly focused on systemic inflammatory indices, encompassing various disorders, including cataracts [11], chronic spontaneous urticaria [12], asthma [13], and PE [14]. Indices derived from systemic immune-inflammatory responses, such as derived neutrophil-lymphocyte ratio (dNLR), pan-immune inflammation value (PIV), systemic inflammation index (SII), and systemic inflammation response index (SIRI), computed from basic parameters, have garnered attention due to their simplicity and accessibility. However, the precise role of systemic inflammatory markers in clinically assessing, differentially diagnosing, and prognostically evaluating PE remains uncertain.
PE arises from placental dysfunction and closely correlates with placental stress, inciting an inflammatory milieu. Timely identification and intervention in PE are paramount for preserving maternal and fetal well-being. Nonetheless, dependable and sensitive markers for predicting and diagnosing PE remain scarce. In recent years, some studies have suggested the potential of inflammatory indices in predicting PE [14, 15].
The primary objective of this investigation is to enhance comprehension of the intricate interplay between PE and inflammatory processes, alongside evaluating the predictive capacity of hematological parameters in routine antenatal assessments for anticipating PE development among pregnant individuals. This manuscript holds significance due to the scarcity of comparative studies evaluating various hematological parameters and utilizing contemporary indices. By identifying these markers, the study seeks to provide a reliable, non-invasive method for anticipating the onset of PE, thereby facilitating earlier interventions and potentially reducing the adverse outcomes associated with this condition.
This retrospective case-control study enrolled 116 pregnant women who delivered at the Obstetrics and Gynecology clinic of Sivas Cumhuriyet University between May 2022 and May 2023. Among them, the study group comprised 58 pregnant women diagnosed with preeclampsia, while the control group consisted of 58 normotensive pregnant women matched for gestational age. The diagnoses were made according to the American College of Obstetrics & Gynecology (ACOG) Pregnancy Hypertension and Preeclampsia Statement published in June 2020 [16]. To mitigate the potential impact of inflammatory measurements on gestational age, pregnant women delivering at equivalent gestational ages were selected for both groups. Routine blood tests were conducted, and hematological indices were derived from the admission tests conducted upon hospitalization for delivery.
Preeclampsia Group: participants were included if they had a diagnosis of preeclampsia, were aged between 18 and 45 years, and had a gestational age between 26 and 41 weeks based on the last menstrual period.
Normal Pregnancy Group: participants were included if they had no history of preeclampsia or severe obstetric complications, were matched with preeclampsia patients in the same age range (18–45 years), and delivered at the same gestational age.
Both Groups: participants with serious health problems such as chronic blood diseases or diabetes, and those with active infectious diseases, were excluded from the study.
Preeclampsia Group: participants with serious hematological problems such as advanced anemia, and those with additional systemic diseases such as diabetes or systemic lupus erythematosus were excluded.
Normal Pregnancy Group: cases where any obstetric complications developed, and those with excessive blood loss or bleeding disorders, were excluded.
The blood samples routinely collected from pregnant women upon their admission to the delivery room for the calculation of hematological parameters. Using the venous blood sampling method, hematological parameters (neutrophil, lymphocyte, platelet, monocyte, and eosinophil count) were measured with an automated analyzer.
The calculated hematological indices were as follows: the NLR (neutrophil lymphocyte ratio) is calculated by dividing the neutrophil count by the lymphocyte count; PLR (platelet lymphocyte ratio): calculated by dividing the platelet count by the lymphocyte count; dNLR (derived neutrophil-to-lymphocyte ratio): calculated by subtracting the neutrophil count from the leukocyte count and then dividing the result by the neutrophil count; SII (systemic inflammatory index): calculated by multiplying the neutrophil count by the platelet count and then dividing the result by the lymphocyte count; SIRI (systemic inflammatory response index): calculated by multiplying the neutrophil count by the monocyte count and then dividing the result by the lymphocyte count; PIV (pan-immune inflammation value): calculated by multiplying the neutrophil, platelet, and monocyte counts and then dividing the result by the lymphocyte count; CoI (composite index): calculated by summing the dNLR, the SIRI, and the PIV.
These measurements were evaluated as potential markers for predicting preeclampsia.
All data analysis was conducted using SPSS version 23.0 (IBM Corp, Armonk, NY,
USA). Descriptive statistics were presented as medians and interquartile range
values (IQR). Normal distribution was assessed using the Kolmogorov-Smirnov test.
Non-normally distributed variables were compared using the Mann-Whitney U test
with statistical significance set at
Initially, demographic variables including age, parity (number of pregnancies), gravidity (number of births), and gestational age were assessed. Pregnant individuals in both cohorts delivering at equivalent gestational ages were compared. The results indicate no statistically significant disparity between the preeclampsia and control groups regarding age (p = 0.424), parity (p = 0.312), and gravidity (p = 0.788) (Table 1).
| Parameters | Patient group (n = 58) | Healthy controls (n = 58) | p |
| Median (IQR) | Median (IQR) | ||
| Age (years) | 29.0 (25.75–36.25) | 29.0 (25.00–34.00) | 0.424 |
| Gravida | 2.0 (1–5) | 2.0 (1–4) | 0.312 |
| Parity | 2.0 (1–4) | 2.0 (1–3) | 0.788 |
| Birth weight (grams) | 2585.00 (1798.25–3036.25) | 2845.00 (2180.00–3265.00) | 0.271 |
| Gestational age (weeks) | 36 (33–38) | 36 (33–38) | 1.000 |
Mann-Whitney U test. IQR (interquartil range) = Q1–Q3.
When hematological parameters were assessed, there were no significant differences between the two groups in terms of hemoglobin, neutrophil, leukocyte, lymphocyte, monocyte, and platelet counts (p = 0.659, 0.174, 0.213, 0.531, 0.103, 0.407, respectively) (Table 2).
| Parameters | Patient group (n = 58) | Healthy controls (n = 58) | p |
| Median (IQR) | Median (IQR) | ||
| Hemoglobin (g/dL) | 12.10 (10.65–12.98) | 11.80 (11.08–12.73) | 0.659 |
| Neutrophil (10 |
7.66 (6.00–10.42) | 8.84 (6.75–11.20) | 0.174 |
| Leukocyte (10 |
10.74 (9.11–13.13) | 11.84(9.09–15.02) | 0.213 |
| Lymphocyte (10 |
2.00 (1.37–2.28) | 1.83 (1.36–2.30) | 0.531 |
| Monocyte (10 |
0.63 (0.45–0.81) | 0.70 (0.54–0.87) | 0.103 |
| Platelet (10 |
221.00 (186.75–283.50) | 232.00 (201.50–278.00) | 0.407 |
Mann-Whitney U test. IQR (interquartil range) = Q1–Q3.
Inflammatory indices were evaluated as well (Table 3). The dNLR showed a significantly decreased level in the preeclampsia group compared to the control group (p = 0.034). Conversely, the SIRI and PIV demonstrated significantly decreased levels in the preeclampsia group (p = 0.034, p = 0.023) (Table 3).
| Hematologic indexes | Patient group (n = 58) | Healthy controls (n = 58) | p |
| Median (IQR) | Median (IQR) | ||
| dNLR | 1.36 (1.29–1.48) | 1.43 (1.34–1.56) | 0.034* |
| SII | 957.90 (664.09–1628.30) | 1018.30 (674.23–1355.55) | 0.183 |
| SIRI | 2.47 (1.70–3.63) | 3.02 (2.33–5.36) | 0.034* |
| PIV | 578.11 (376.36–812.57) | 760.38 (455.98–1406.15) | 0.023* |
| PLR | 126.66 (91.30–177.22) | 123.56 (103.45–189.18) | 0.608 |
| NLR | 3.92 (3.02–6.38) | 4.10 (3.25–8.53) | 0.193 |
Mann-Whitney U test. *p
NLR, neutrophil lymphocyte ratio; PLR, platelet lymphocyte ratio; dNLR, derived
neutrophil-to-lymphocyte ratio (neutrophil count divided by the result of
leukocyte count minus neutrophil count); SII, systemic inflammatory index
(neutrophil
The area under the curve (AUC) for dNLR was 0.374, with p
| Hematologic indexes | AUC | Cut off | Sensitivity | Specificity | p |
| dNLR | 0.374 | 1.41 | 0.386 | 0.414 | 0.020* |
| SIRI | 0.392 | 2.66 | 0.404 | 0.397 | 0.046* |
| PIV | 0.384 | 666.81 | 0.439 | 0.414 | 0.032* |
ROC, receiver operating characteristic; AUC, area under curve; dNLR, derived
neutrophil-to-lymphocyte ratio (neutrophil count divided by the result of
leukocyte count minus neutrophil count); SIRI, systemic inflammatory response
index (neutrophil
The findings of this study enrich our comprehension of the intricate interplay between PE and inflammation. In response to the limitations observed with individual inflammatory markers where AUC were below 0.5, indicating minimal diagnostic value, a novel composite index was devised. This composite index is derived from the sum of the dNLR, the SIRI, and the PIV. The creation of this composite index, tested for the first time in this study, represents an innovative approach aimed at enhancing predictive accuracy by integrating multiple inflammatory metrics into a single diagnostic tool.
The AUC for CoI was 0.62, with p
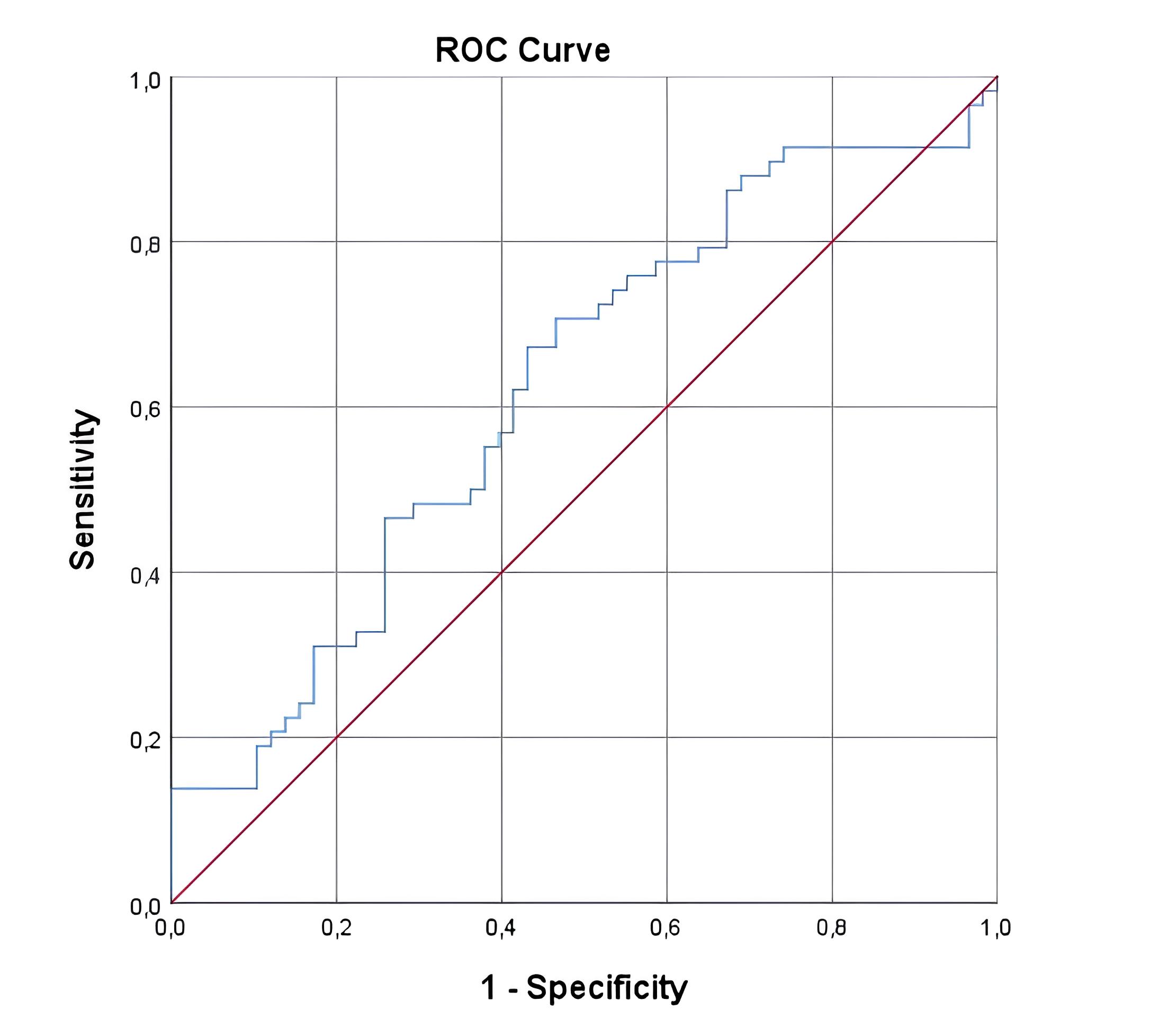 Fig. 1.
Fig. 1.Receiver operating characteristic (ROC) analysis of composite index (CoI) in preeclampsia.
PE poses substantial risks to both maternal and fetal health, underscoring the importance of early diagnosis for optimal disease management and improved pregnancy outcomes. The International Federation of Gynecology and Obstetrics (FIGO) has developed an online risk calculator leveraging serum placental growth factor, uterine artery pulsatility index, and mean arterial pressure to predict PE [17]. Recent advancements suggest that specific biomarkers such as soluble fms-like tyrosine kinase 1 and placental growth factor may serve as predictors of PE [18, 19]. Incorporating blood cell indices for PE prediction offers cost-effective, expedited, and readily accessible alternatives. Inflammatory parameters like PIV, SII, SIRI, and dNLR, commonly employed in predicting various diseases including cancer prognosis, acute coronary syndrome, first-trimester pregnancy losses, and hemolysis, elevated liver enzymes, low platelet (HELLP) syndrome, present promising avenues for PE prediction [20, 21, 22, 23].
However, literature presents conflicting evidence regarding the predictive value of NLR and PLR for PE. While some studies assert significant differences in NLR among PE patients compared to others [24], conflicting publications report non-significance [15, 25, 26]. Similar inconsistencies exist for PLR, with some study demonstrating significance [25] while other refuse it [26]. In our study, while no significant differences were observed in NLR, PLR, and SII indices, dNLR, SIRI, and PIV exhibited significant differences in the preeclamptic group.
In a study by Onat et al. [14], evaluating NLR, PLR, and lymphocyte to monocyte ratio (LMR) in PE patients, no significant differences were found compared to the control group. Similarly, Toptaş et al. [27] observed higher NLR levels in severe PE patients compared to mild cases, though not statistically significant. Consistent with these findings, our study did not reveal statistically significant differences in NLR and PLR levels between patient and control groups. Contrarily, Akdulum et al. [28] reported significantly elevated SII levels in PE patients, suggesting its potential utility in PE prediction. However, unlike their study, our investigation did not find statistically significant differences in SII levels.
Overall, our study underscores the association between PE and heightened inflammation. While non-significantly, PE patients exhibited higher hemoglobin levels and lower neutrophil counts. Notably, hematological parameters, particularly dNLR, SIRI, and PIV, demonstrated significant predictive value for PE. Nonetheless, these findings necessitate further refinement, particularly concerning their integration into routine pregnancy monitoring for early PE diagnosis and treatment initiation. However, it is crucial to acknowledge certain limitations, including the retrospective study design and limited sample size, warranting the need for prospective studies with larger cohorts. Moreover, future research should elucidate the clinical applicability of our findings and the role of inflammatory indices in PE diagnosis and management. While this study provides a promising trajectory for predicting PE, further specific research and clinical validation are imperative.
Based on the current literature, the role of eosinophils in preeclampsia (PE) has been explored in several studies, albeit with varied findings. Recent studies have investigated the role of eosinophils, traditionally associated with allergic reactions, in predicting preeclampsia due to their involvement in immune regulation and inflammation [29, 30]. The reviewed studies illustrate the complexity of immune changes in preeclampsia and the potential of comprehensive leukocyte profiling in its prediction and management. Çintesun et al. (2020) [31] noted no significant difference in eosinophil counts between PE patients and controls, although basophil counts were elevated in the PE group, suggesting a complex interplay of different immune cells in preeclampsia. Gelaw et al. (2022) [29] focused on the clinical significance of eosinophil counts in pregnancy, particularly in preeclampsia, finding a potential for these counts to serve as markers for the disease. Herzog et al. (2016) [32] highlighted that while eosinophil counts did not significantly differ between early-onset and late-onset preeclampsia, the overall leukocyte profiles were indicative of underlying inflammatory processes.
The discussion of the CoI within this study is pivotal, as it represents a significant evolution in the approach to predicting preeclampsia. While individual markers such as dNLR, SIRI, and PIV have shown statistical significance, their clinical application has been limited by suboptimal sensitivity and specificity. The CoI, by integrating these indices, seeks to mitigate these limitations and offer a more reliable diagnostic tool. Preliminary results suggest that the CoI improves the predictive accuracy, underscoring its potential to transform clinical practices. However, further validation through longitudinal studies is essential to confirm its efficacy and to refine its application, ensuring it can be effectively integrated into routine clinical workflows. This integration could potentially lead to more timely and accurate interventions, ultimately enhancing patient outcomes in prenatal care.
This study employs a retrospective design, utilizing historical data not initially gathered with the intention of exploring the specific hypotheses presented. This limits the ability to control for all confounding variables that may impact the results. Although the matched cohort methodology strengthens the comparative analysis, the overall sample size of 116 subjects may limit the study’s statistical power, diminishing the capacity to detect smaller yet clinically relevant differences between the preeclampsia and control groups. Data collection from a single medical center may not reflect the wider population, potentially limiting the generalizability of the findings to different geographical locations or demographic groups. The absence of longitudinal follow-up restricts the ability to assess long-term outcomes and monitor changes in inflammatory markers over time. Moreover, the control group consisted solely of normotensive pregnant women; including subjects with different hypertensive disorders might have allowed for a broader comparison across the hypertensive spectrum. And one of the limitations of this study is the use of blood samples collected in the delivery room to test the predictive ability of hematological indicators. Future research could support our findings by conducting similar analyses with blood samples taken at earlier stages of pregnancy.
In summary, this study underscores the significance of elucidating the interplay between inflammation and hematological parameters through the utilization of inflammatory indices. Hematological parameters, notably the dNLR, SIRI, and PIV, hold promise in predicting preeclampsia, thus facilitating early diagnosis and intervention. The introduction of the CoI, which amalgamates these indices, marks a significant advancement. This newly developed CoI has demonstrated enhanced diagnostic performance in preliminary assessments, suggesting a superior predictive capability over the individual indices. However, refining the formulations of these indices, including the CoI, to enhance their clinical utility and conducting comprehensive prospective studies to validate their effectiveness are imperative. This integrated approach may offer a more robust framework for managing preeclampsia, emphasizing the importance of a multifaceted diagnostic strategy in clinical settings.
The original contributions presented in this study are detailed within the article itself, if applicable. For further inquiries or access to the data, please contact the corresponding author.
SOG and HE designed the research study. SOG performed the research, HE analyzed the data. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The research underwent review and approval by the Ethics Committee of Sivas Cumhuriyet University under protocol number 2023-09/53. Written informed consent was obtained from all participants prior to their involvement in the study.
We would like to acknowledge the support of the Obstetrics Clinic at Sivas Cumhuriyet University for providing access to essential equipment and facilities necessary for data collection and analysis.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

