1 Department of Gynecology and Obstetrics, Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, West China Second Hospital, Sichuan University, 610066 Chengdu, Sichuan, China
2 Department of Gynecology and Obstetrics, The Fourth People's Hospital, 643000 Zigong, Sichuan, China
†These authors contributed equally.
Abstract
Background: Since the global outbreak of coronavirus disease 2019
(COVID-19), its rapid spread has had a dramatic impact on the physical/mental
health of people of all ages, including lactating women. We chose to study
whether COVID-19 affected breastfeeding of in China. Since there has been no
relevant research on this topic in China our team planned to study this topic.
Methods: The study population was drawn from the postpartum clinic of
the Fourth People’s Hospital of Zigong City, Sichuan province, China, from
07/12/2022 to 01/04/2023. This group comprised lactating women who manually
expressed their milk due to various reasons, rather than directly nursing their
infants. The population was classified into two groups: the infected group and
uninfected group, based on their current COVID-19 status. COVID-19 infection was
defined as a positive result in either the COVID-19 nucleic acid test (reverse
transcription-polymerase chain reaction/polymerase chain reaction (RT-PCR/PCR))
or the COVID-19 antigen test. We retrospectively assessed their breast milk
production and infant growth and development over a subsequent 4-month period.
Results: The study encompassed a total of 79 lactating women, comprising
45 in the infected group and 34 in the uninfected group. There was a
statistically significant difference in the rate of breast milk production
between the COVID-19 infected and uninfected groups (
Keywords
- COVID-19
- breastmilk production
- influence factor
Breast milk is the preferred nutrition for infants and young children (0–6 months old). It is rich in nutrients and its natural immune antibodies play a very important role in defending against diseases and reducing the prevalence of a variety of diseases [1, 2, 3, 4, 5, 6]. In reality, there are many reasons why breastfeeding may not be successful [7, 8].
Coronavirus disease 2019 (COVID-19), which occurred globally, spread in a number of ways [9, 10, 11]. Symptoms included fever, cough, dyspnea, sore throat, nasal congestion, musculoskeletal pain, chills, headaches, nausea and vomiting and other gastrointestinal symptoms [12, 13, 14, 15, 16, 17, 18]. Its Omicron strain has strong transmissibility and is susceptible to all populations, including lactating women [17].
Although studies have shown that if the health of the mother and infant permits, breastfeeding is recommended for COVID-19 infected mothers, with appropriate protective measures being maintained [18, 19]. Several studies have confirmed the safety of breastfeeding in women infected with the COVID-19 [20, 21]. However, the author found in clinical work that many nursing women infected with the COVID-19 will have reduced milk production, insufficient breastfeeding, while we lack of relevant clinical research reports. The objective of the study is to investigate the impact of COVlD-19 on breast milk secretion and breastfeeding continuation among infected individuals who have not ceased breastfeeding post-infection, as well as its effects on infant health, through retrospective analysis. The aim is to provide valuable insights for clinical management.
The study population was drawn from the postpartum clinic of the Fourth People’s Hospital in Zigong City, Sichuan Province, China. This group comprised lactating women who expressed their milk rather than directly nursing their infants due to various reasons. We conducted a retrospective analysis of their subsequent 4 months of breastfeeding. This study received ethical approval from the Institutional Review Board of the Fourth People’s Hospital in Zigong City, Sichuan Province, China (approval number: (003), 2022). The need for obtaining informed consent was waived due to the retrospective nature of the study.
We studied lactating women who had regular follow-up visits at the
postpartum clinic of the Fourth People’s Hospital in Zigong City, Sichuan
Province, from 07/12/2022 to 01/04/2023. Inclusion criteria for the study
population were as follows: ① Women who had regular follow-up visits at
the postpartum clinic of the Fourth People’s Hospital in Zigong City, Sichuan
Province; ② Inability to breastfeed directly due to various reasons,
necessitating milk expression, with a breast milk yield that had reached a
balanced supply and did not require artificial feeding; ③ Age
Clinical data were collected using self-designed structured network questionnaires, encompassing relevant demographic and clinical characteristics of the participants. Baseline information including age, height, weight, body mass index (BMI), gravidity, parity, labor time (preterm/full-term, delivery mode (vaginal birth/cesarean birth/cesarean birth after vaginal birth), educational level (no undergraduate/undergraduate and above), COVID-19 vaccination history, milking collection method (machine/others), sleep quality, breast pain conditions (using Visual Analogue Scoring (VAS) [22], results were divided into painless (0), mild (1–3), moderate (4–6), and severe (7–10)), breast swelling conditions (no swelling-normal breasts, mild-does not affect maternal rest, moderate-interferes with maternal rest, severe-unable to rest)). For the infected group, we collected the sleep quality [23], breast pain conditions, and breast swelling conditions a month before (1) and after infection (2); for the uninfected group, we collected the information a month before (1) and after enrollment (2). During the follow-up questionnaire, we collected both COVID-19-related data and breastfeeding-related data. The volume of breast milk was documented based on the subjective judgment of the lactating woman. The milk volume 1 week before infection or enrollment was designated as 100%, and subsequent weekly production was recorded and compared to this baseline (due to poor compliance, consecutive recording was not maintained over 2–3 months) (See Supplementary Material for data).
Data were processed and analyzed using SPSS statistical analysis software
(version 27.0, IBM Corp., Armonk, NY, USA). Continuous data were assessed using
the independent t-test or Mann-Whitney rank sum test, while categorical
data were analyzed using the
Our study comprised 79 lactating women, with 45 in the infected group and 34 in the uninfected group. The infected group exhibited statistically significant differences in age (younger), educational level (higher), and sleep quality (better) compared to the non-infected group. However, there were no statistically significant differences between the two groups in terms of mothers’ height, weight, BMI, gravidity and parity, delivery mode, delivery time, and COVID-19 vaccine history (Table 1).
| Infected group (N = 45) | Uninfected group (N = 34) | p | ||
| Age-years ( |
27.60 |
30.74 |
0.020* | |
| Height-cm ( |
158.51 |
158.68 |
0.676 | |
| Weight-kg ( |
71.64 |
73.15 |
0.384 | |
| BMI-kg/m |
28.576 |
29.071 |
0.519 | |
| Gravida number | 0.255 | |||
| 1 (No, %) | 23 (63.9) | 13 (36.1) | ||
| 22 (51.2) | 21 (48.8) | |||
| Para number | 0.662 | |||
| 1 (No, %) | 35 (58.3) | 25 (41.7) | ||
| 2 (No, %) | 10 (52.6) | 9 (47.4) | ||
| Labor time | 0.529 | |||
| Full term (No, %) | 37 (58.7) | 26 (41.3) | ||
| Preterm (No, %) | 8 (50.0) | 8 (50.0) | ||
| Delivery mode | 0.189 | |||
| Vaginal birth (No, %) | 25 (50.0) | 25 (50.0) | ||
| Cesarean birth (No, %) | 15 (83.3) | 8 (34.8) | ||
| Cesarean birth after vaginal birth (No, %) | 5 (83.3) | 1 (16.7) | ||
| Educational level | 0.042* | |||
| Under undergraduate (No, %) | 14 (77.8) | 4 (22.2) | ||
| Undergraduate and above (No, %) | 31 (50.8) | 30 (49.2) | ||
| COVID-19 vaccination injection history | 0.634 | |||
| Yes (No, %) | 40 (58.0) | 29 (42.0) | ||
| No (No, %) | 5 (50.0) | 5 (50.0) | ||
| Milking collection method | 0.371 | |||
| Breast pump (No, %) | 26 (53.1) | 23 (46.9) | ||
| Others (No, %) | 19 (63.3) | 11 (36.7) | ||
| Sleep quality (1) | 0.017* | |||
| Best (No, %) | 3 (100.0) | 0 (0) | ||
| Better (No, %) | 20 (51.3) | 19 (38.7) | ||
| Worse (No, %) | 20 (71.4) | 8 (28.6) | ||
| Worst (No, %) | 2 (22.2) | 7 (77.8) | ||
| Breast pain conditions (1) | 0.742 | |||
| Painless (0) (No, %) | 1 (50.0) | 1 (50.0) | ||
| Mild (1–3) (No, %) | 27 (54.0) | 23 (46.0) | ||
| Moderate (4–6) (No, %) | 17 (63.0) | 10 (37.0) | ||
| Breast swelling conditions (1) | 0.402 | |||
| Normal (No, %) | 2 (40.0) | 3 (60.0) | ||
| Mild (No, %) | 29 (63.0) | 17 (37.0) | ||
| Moderate (No, %) | 14 (50.0) | 14 (50.0) | ||
| Sleep quality (2) | ||||
| Best (No, %) | 0 (0) | 0 (0) | ||
| Better (No, %) | 6 (27.3) | 16 (72.7) | ||
| Worse (No, %) | 36 (76.6) | 11 (23.4) | ||
| Worst (No, %) | 3 (30.0) | 7 (70.0) | ||
| Breast pain conditions (2) | 0.449 | |||
| Painless (0) (No, %) | 5 (62.5) | 3 (37.5) | ||
| Mild (1–3) (No, %) | 39 (58.2) | 28 (41.8) | ||
| Moderate (4–6) (No, %) | 1 (25.0) | 3 (75.0) | ||
| Breast swelling conditions (2) | 0.206 | |||
| Normal (No, %) | 5 (50.0) | 5 (50.0) | ||
| Mild (No, %) | 39 (60.9) | 25 (39.1) | ||
| Moderate (No, %) | 1 (20.0) | 4 (80.0) | ||
* the two sets of data have statistically significant difference. SD, standard deviation; BMI, body mass index; COVID-19, coronavirus disease 2019.
In our study, 45 women experienced a decrease in breast milk production during
the follow-up period: 31 cases in the COVID-19 infected group and 14 cases in the
non-infected group. The rate of breast milk production decrease exhibited a
statistically significant difference between the COVID-19 infected and uninfected
groups (
In our research, the average time of COVID-19 infection occurred during the fourteenth week after delivery. The majority of infections were of the mild type. The decline in breast milk production commenced on the fifth day following COVID-19 infection and reached its nadir during the second week post-infection (Fig. 1). Among lactating women experiencing reduced breast milk production 67.7% opted to increase their rest to facilitate breast milk recovery. However, by the end of our retrospective analysis, only 15.2% of women had seen their breast milk production return to normal levels.
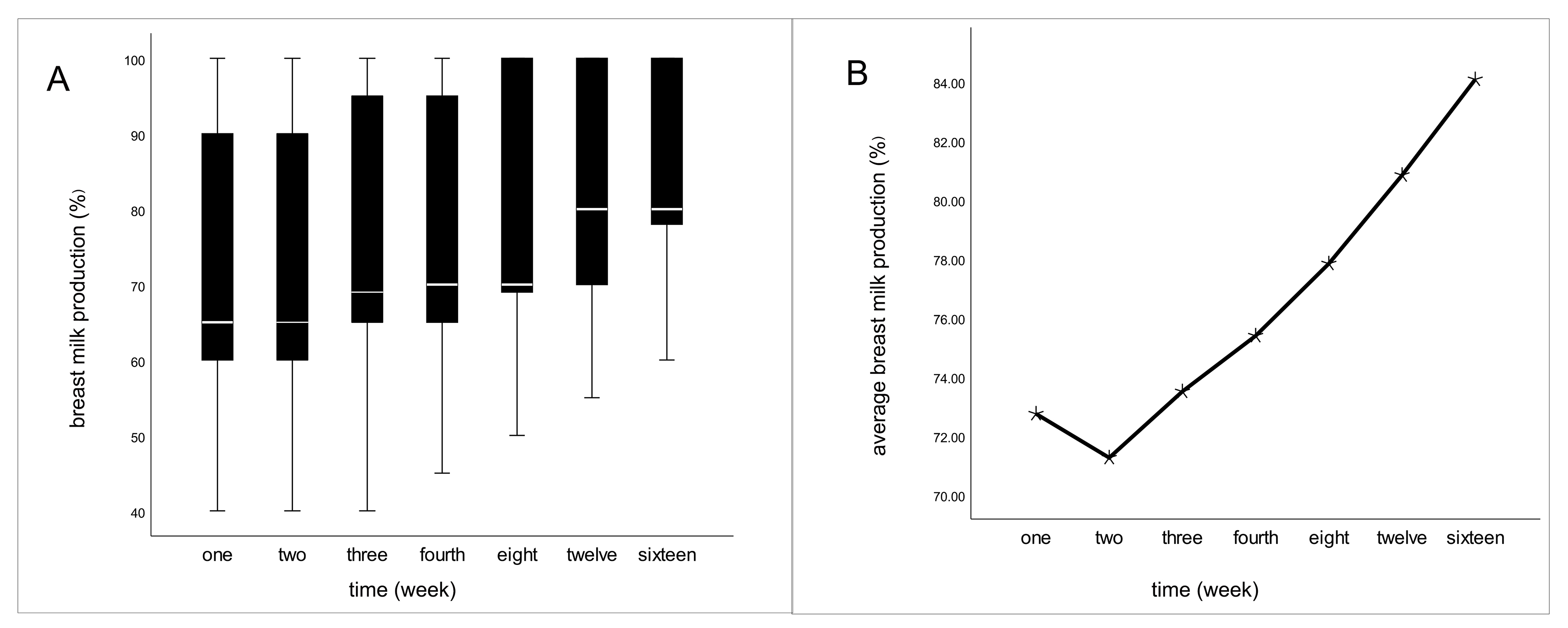 Fig. 1.
Fig. 1.Fluctuations in milk production after infection of COVID-19 in
the infected group. (A) The boxes represent the interquartile range with breast
milk production up to 16 weeks after infection. Whiskers present the data range
(Q1/Q3
In our study, 37.8% of infected women opted to discontinue breastfeeding and switch to artificial feeding, while 62.2% of infected women chose to continue breastfeeding. Among those who continued breastfeeding, 25% experienced insufficient breastfeeding during the period of decreased milk production (insufficient means that artificial feeding milk is more than half) (Table 2). During the follow-up period, only 1 case involved a child with sleep disturbances, while the others did not exhibit abnormal symptoms such as irritability, drowsiness, abdominal pain, or diarrhea.
| Number | Percentage (%) | ||
| Before infection | |||
| Enough* | 40 | 88.9 | |
| General | 5 | 11.1 | |
| Insufficient | 0 | 0 | |
| After infection | |||
| Enough | 5 | 17.9 | |
| General | 16 | 57.1 | |
| Insufficient | 7 | 25.0 | |
* general means needs increase of 1–2 artificial feedings; insufficient means that artificial feeding is more than half of the feedings.
In our study, the proportion of people with sufficient milk decreased from 88.9% to 17.9% after infected, and the proportion of people with insufficient milk increased from 0 to 25%. We speculate that infection with the novel coronavirus may affect the production of breast milk. The average age, height, and weight of children in the COVID-19 infected group who experienced continued breastfeeding did not exhibit a statistically significant difference when compared to the COVID-19 uninfected group at the baseline level. Furthermore, during the 4-month follow-up period, there remained no statistically significant difference in height between the two groups. Concerning infant weight, the infected group was heavier than the uninfected group in the third and fourth months, and this difference was statistically significant (Table 3 and Fig. 2). This study concluded that COVID-19-infected women’s breast milk did not have a significant negative impact on short-term growth and development in their children.
| Infected group (N = 28) | Uninfected group (N = 34) | p | ||
| Age-months ( |
3.89 |
4.94 |
0.305 | |
| Weight-kg ( |
||||
| Begin | 11.46 |
10.07 |
0.108 | |
| First month | 12.21 |
10.90 |
0.117 | |
| Second month | 13.00 |
11.70 |
0.108 | |
| Third month | 14.25 |
12.60 |
0.036* | |
| Fourth month | 15.29 |
13.79 |
0.045* | |
| Height-cm ( |
||||
| Begin | 60.64 |
60.85 |
0.943 | |
| First month | 62.00 |
62.17 |
0.977 | |
| Second month | 63.39 |
63.32 |
0.915 | |
| Third month | 64.93 |
64.63 |
0.755 | |
| Fourth month | 66.57 |
66.20 |
0.686 | |
* the two sets of data have statistically significant differences.
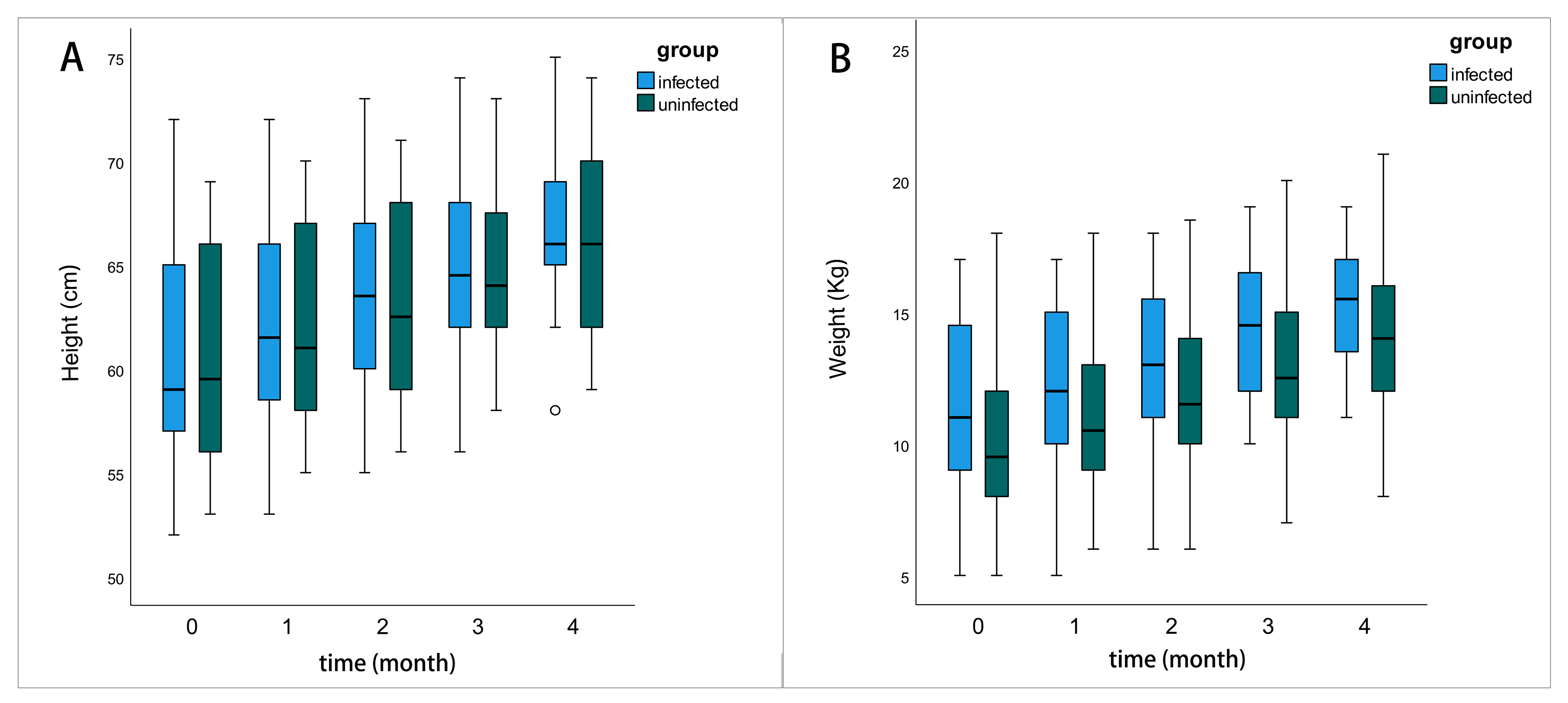 Fig. 2.
Fig. 2.The comparison of the growth and development status of the
infants. The boxes represent the interquartile range with height/weight up to 4
months after follow-up. Whiskers present the data range (Q1/Q3
Our research revealed an elevated risk of reduced breast milk production in the COVID-19 infected group compared to the non-infected group (OR 3.163, 95% CI: 1.248–8.017). Even after adjusting for confounding factors, this increased risk persisted (OR 3.339, 95% CI: 1.134–9.833). Among women who continued breastfeeding without interruption, we observed that their breast milk had no adverse impact on short-term growth and development in their children.
Within the first 24 hours postpartum, breast milk production increases rapidly, and it stabilizes at approximately 750–800 mL/day after one month [24]. In our research, 91.1% of women entered into the relatively stable stage. The breast milk secreted by the mammary epithelial cell (MEC), upon the effect of oxytocin (OT), the milk expelled out of the alveoli into the ducts, and simultaneously under the function of milk ejection reflex mediated by OT, ultimately leads to milk ejection [25]. Our research findings indicated that the risk of reduced breast milk production increased following COVID-19 infection. In our study, all women used a breast pump to express milk, and after infection, there were no significant differences in breast symptoms between the infected and non-infected groups. This supports our finding that the reduction in milk production is primarily due to decreased breast milk secretion rather than abnormal breast emptying. Several factors influence breast milk production, including genetics, diet, hormones, and the environment [26]. One possible reason is that the majority of COVID-19 infected women experienced increased body temperature. In our study, 77.8% of women in the infected group had fever (defined as a body temperature exceeding 37.3 °C). This fever can accelerate metabolism and increase the body’s water consumption, which may partly contribute to the reduced breast milk production. Another significant factor is mental health, which can influence the secretion of OT, a hormone crucial for breast milk ejection. Additionally, psychologic distress can lead to elevated cortisol levels and decreased insulin levels in the serum, both of which are associated with reduced milk production [27, 28]. During the COVID-19 pandemic, women’s mental health is significantly affected, with a noticeable increase in symptoms of depression, anxiety, and stress. This increase is particularly pronounced among infected women, who exhibit a higher incidence of depression and anxiety [29, 30, 31, 32]. A meta-analysis showed that the ORs of COVID-19 pandemic for postpartum depression and anxiety symptoms were 1.54 (95 % CI: 1.00–2.36) and 2.56 (95% CI: 1.62–4.04) [33]. Hormones levels, except for those indirectly influenced by the mental factors, were also found to be directly influenced by the COVID-19 infection. One crucial hormone is insulin. Research has reported that mothers with insulin resistance are at an elevated risk of insufficient milk production and delayed lactogenesis [34, 35]. Reports have indicated that COVID-19 can induce insulin resistance in patients. Furthermore, the angiotensin-converting enzyme 2 (ACE2), which serves as the receptor for the severe acute respiratory syndrome (SARS) coronavirus, has been found to be expressed in the endocrine part of the pancreas. This suggests that the virus may enter islets using ACE2 and potentially harm them [36]. The reduction in insulin levels, is associated with decreased milk production. However, a meta-analysis demonstrated that COVID-19 infection has no impact on prolactin (PRL) levels [37]. The influence of COVID-19 infection for other hormones needs further exploration.
Breast milk is rich in energy and nutrients, offering children appropriate
amounts of protein, fat, lactose, vitamins, iron, minerals, enzymes, and water.
These nutrients are easily digested and absorbed, meeting all the nutritional
needs of infants under 6 months of age. In light of this, during the COVID-19
pandemic, international and national health organizations, including the World
Health Organization (WHO) and the American Academy of Pediatrics, advocated for
mothers to continue breastfeeding, even if they are infected [21]. In contrast,
the Chinese consensus differs, as they recommend breastfeeding only when both the
PCR test for mothers and breast milk is negative [38]. Regarding the safety of
breastfeeding among COVID-19 infected women, there is no explicit evidence
demonstrating that the virus can be transmitted through breast milk [18]. Several
case reports have identified the presence of viral nucleic acid in breast milk
[39, 40]. However, none of these cases have cultured the virus. Therefore, it
remains uncertain whether breast milk contains active infectious viruses and
whether the virus originates from the milk rather than the infected environment
or droplets from infected mothers. According to recommendations from
international organizations, when a mother is infected with COVID-19, it is
considered safe for an uninfected caregiver to handle feeding. Alternatively,
mothers should wear masks and practice breast and hand hygiene. Additionally,
breast pumps should be thoroughly cleaned following pumping sessions [19].
Several studies have found that the existence of antibodies against SARS-CoV-2 in
breastmilk after being infected or receiving a vaccine, allowing the infant to
directly acquire the antibodies against SARS-CoV-2 and confer effective passive
immunity [41]. In our research, all the mothers use the pump breastmilk to feed.
Following COVID-19 infection, 68.8% of mothers experienced a decrease in breast
milk production, reaching its lowest point during the second week after
infection. This timeframe is later than the average duration of the worst symptom
days among infected individuals (4.9
The limitations of our study include: first, it is a retrospective study, and retrospective bias is inevitable, potentially impacting result accuracy; second, regarding psychological factors, we intended to collect data on depression and anxiety using standardized scales. However, due to poor compliance among women, we were unable to collect this information.
Previous studies have primarily focused on the safety of breast milk from COVID-19 infected women, mainly discussing whether these mothers should interrupt breastfeeding. Few studies have explored changes in milk volume following infection. Our retrospective study demonstrated that COVID-19 infection can result in decreased breast milk production (OR 3.339, 95% CI: 1.134–9.833). Among infected mothers, breast milk volume reaches its lowest point during the second week after infection and gradually increased thereafter. Additionally, breast volume does not fully recover to pre-infection levels by the end of our follow-up period. In the short term, breast milk from infected mothers does not have a negative impact on the height and weight gain of infants. However, long-term follow-up is still necessary.
The datasets used and analyzed during the current study are available from the corresponding author (XQ) on reasonable request.
XH, JY and XQ designed the research study. XH, JY performed the research. XQ provided help and advice on the experiments. JY analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The need for obtaining informed consent was waived due to the retrospective nature of the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Zigong Fourth People’s Hospital (approval number: (003), 2022).
Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


