1 Department of Gynecology, The Fifth Affiliated Hospital of Sun Yat-sen University, 519000 Zhuhai, Guangdong, China
Abstract
Background: To comprehensively investigate the diagnostic effect of intraoperative indocyanine green (ICG) imaging for endometriosis (EMs). Methods: We systematically retrieved relevant literature from a series of databases (Embase, Web of Science, PubMed, Clinical Trials, Cochrane Library, Chinese National Knowledge Infrastructure (CNKI), and WanFang Database) up to January 2024. We also performed manual searches using the reference lists from selected articles, along with reports from relevant meetings and Google Scholar. Data were stored and analyzed by Review Manager version 5.3 and Stata/MP version 14.0. Results: Systematic searches of the literature identified six articles comparing the diagnostic effect of ICG imaging to white light (WL) imaging for EMs during laparoscopic surgery. Two of these studies found that ICG imaging improved the diagnostic rate of EMs while the remaining four studies reported that the diagnostic value of ICG imaging was minimal. Cumulative analysis determined that the sensitivity was 0.88 (95% confidence interval [95% CI]: 0.81–0.93) for WL and 0.64 (95% CI: 0.36–0.84) for ICG. The specificity was 0.85 (95% CI: 0.49–0.97) for WL and 0.88 (95% CI: 0.66–0.97) for ICG. The positive likelihood ratio (LR+) was 5.8 (95% CI: 1.4–24.5) for WL and 5.4 (95% CI: 1.2–24.1) for ICG. The negative likelihood ratio (LR–) was 0.14 (95% CI: 0.09–0.20) for WL and 0.41 (95% CI: 0.18–0.94) for ICG, while the diagnostic odds ratio (DOR) was 42 (95% CI: 10–182) for WL and 13 (95% CI: 1–124) for ICG. Conclusions: Although we only analyzed a limited number of publications, our analysis demonstrated that ICG may be helpful for the visualization of occult EMs, although the diagnostic effect of ICG is not superior to WL. Our findings need to be confirmed by additional studies involving larger sample sizes from multiple centers.
Keywords
- endometriosis
- indocyanine green imaging
- white light imaging
- diagnostic effect
- meta-analysis
Endometriosis (EMs) is typically characterized by the adhesion, growth, implantation, infiltration, and erosion of viable ectopic endometrial tissue outside of the uterus. This condition can cause chronic pain, subfertility, nodules, or masses due to repeated bleeding and inflammatory reactions. EMs is a common disease and is affects almost 10% of women of childbearing age. Globally, approximately 196 million women suffer from this disease [1, 2, 3]. The diagnosis of EMs is challenging and often delayed due to the lack of sensitive non-invasive diagnostic methods. Laparoscopic exploration with histological confirmation is considered to represent the current gold standard for the diagnosis of EMs. However, the diagnostic ability of this methodology is highly dependent on the experience of the surgeon and the specific type of EMs as small, hidden or non-pigmented endometriotic lesions (known as occult EMs) are very challenging to recognize [4]. Laparoscopic radical excision of lesions is currently considered as the standard treatment for EMs that do not respond to pharmacological treatment or in cases experiencing infertility. However, simple laparoscopic excision is associated with a high recurrence rate and a high risk of further surgical episodes; these issues may be explained by the by inadequate visualization of occult EMs, at least in part [5]. Therefore, there is an urgent need to improve the intra-operative diagnosis of EMs so that surgeons can recognize occult EMs. The development of new diagnostic methods may facilitate the radical reduction of lesion, improve the efficacy of treatment, and reduce the risk of recurrence.
EMs is a hyper-vascular disease; consequently, the visualization of hyper-vascular lesions may facilitate the detection of EMs [6, 7]. Indocyanine green (ICG) is a dye that can bind to plasma proteins and be distributed by the blood vessels to serve as an efficient tool for vascular imaging. New or immature vascular vessels are highly permeable and can therefore permit the diffusion and retention of ICG in patients with EMs. These characteristics could provide an efficient tool to identify EMs intraoperatively following an intravenous injection of ICG [8, 9]. In 2014, Levey [10] reported the first application of intraoperative ICG imaging technology to diagnose a case of peritoneal EMs during robot-assisted laparoscopy. Subsequently, Raimondo et al. [11, 12] reported that ICG imaging was feasible for evaluating anastomotic perfusion following the discoid or segmental resection of rectosigmoid EMs and for evaluating ureteral perfusion and guiding surgical decision-making in cases with ureteral EMs. Bruneel et al. [13] also reported that ICG imaging facilitated the precise resection of EMs in the bladder wall. Several prospective studies have evaluated the diagnostic effect of ICG imaging for EMs; however, the findings arising from these studies were inconsistent. There is still no consensus as to whether the application of ICG imaging can improve the diagnostic rate for EMs [14, 15, 16, 17, 18].
Herein, we present a systematic review and meta-analysis that summarizes the existing literature that compared diagnostic ability between white light (WL) imaging and ICG imaging for EMs. Our intention was to provide specific recommendations for the clinical application of ICG imaging for patients with EMs.
A normative protocol was generated in accordance with the standards required for meta-analysis. This meta-analysis was also registered and approved by the International Prospective Register of Systematic Reviews (ID: CRD42024512567) [19] and was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [20]. First, we identified relevant literature by systematically searching multiple electronic databases up to January 2024 without any geographic limitation, including Embase, Web of Science, PubMed, Clinical Trials, Cochrane Library, Chinese National Knowledge Infrastructure (CNKI), and the WanFang Database. We attempted to identify all research publications that compared the diagnostic effects of laparoscopic exploration between WL and ICG imaging for EMs. We also performed manual searches of Biosis Previews [21], the reference lists of the identified literature, OpenGrey [22], Google Scholar, OpenDOAR [23], reports of scientific meetings, and the Bielefeld Academic Search Engine [24] to avoid omitting unpublished, newly published or internet resources.
The literature was systematically retrieved by combining medical sub-headings (MeSH) and specific keywords, as follows: (endometriosis) or (endometrioses) or (endometrioma) or (endometriomas) and (indocyanine green) or (wofaverdin) or (green, indocyanine) or (vophaverdin) or (cardio-green) or (vofaverdin) or (ujoveridin) or (ICG) or (cardiogreen) or (cardio green) or (fluorescence) or (near-infrared). Clinical studies comparing diagnostic values between WL and ICG imaging were selected for screening without any form of geographic and language limitation. Pathological diagnosis was regarded as the gold standard for EMs. We included prospective randomized controlled trials (RCTs), case-control studies, quasi-RCTs, cohort studies, and observational studies. We extracted the required information from all studies eligible for inclusion medical records and files, including the authors, year of publication, sample sizes, baseline characteristics, intraoperative results, diagnostic sensitivity, diagnostic specificity, positive likelihood ratio (LR+), negative likelihood ratio (LR–), and adverse events.
Two authors (YZ and YX) independently reviewed the identified publications and applied these to the specific inclusion and exclusion criteria. First, we used the titles and abstracts to exclude obvious unmatched research, including reviews, case reports, comments, letters, and in vivo and/or in vitro studies. Second, the full-text of each selected publication was acquired and further screened according to the inclusion criteria, as follows: (1) clinical studies relating to EMs, (2) diagnostic value was considered as an outcome, (3) comparative outcomes between WL and ICG imaging, (4) pathological diagnostic outcomes, and (5) full text available. Any differences between the two reviewers were moderated by a third reviewer (HY).
Two authors (YX and HO) independently evaluated the risk of bias of the enrolled
studies by applying nine key criteria in Stata/MP version 14.0 (Stata Corp LP,
College Station, TX, USA): (1) prospective design, (2) sample size
Publication bias is presented as a funnel plot, defined as a scatterplot of the odds ratio (OR) of each study on the X-axis, and the standard error (SE) of the log OR on the Y-axis. A symmetrical inverted funnel plot was considered to indicate a low risk of publication bias, whereas a asymmetrical funnel plot was considered as a potential risk of publication bias. We also conducted sensitivity analysis to evaluate whether the results were robust by eliminating each study in a step-by-step manner.
True-positives (TPs) were defined as when lesions were pathologically confirmed as EMs, and when WL/ICG imaging correctly identified the lesions as EMs. False-positives (FPs) were identified as when lesions were pathologically confirmed as non-EMs, and when WL/ICG imaging incorrectly identified the lesions as EMs. True-negatives (TNs) were defined as when lesions were pathologically confirmed as non-EMs, and when WL/ICG imaging correctly identified the lesions as non-EMs. False-negatives (FNs) were defined as when lesions were pathologically confirmed as EMs, and when WL/ICG imaging incorrectly identified the lesions as non-EMs.
Sensitivity was calculated as TP/(TP + FN) while specificity was calculated as TN/(TN + FP). The positive predictive value (PPV) was calculated as TP/(TP + FP) while the negative predictive value (NPV) was calculated as TN/(FN + TN). The positive likelihood ratio (LR+) was calculated as sensitivity/(1-specificity) while the negative likelihood ratio (LR–) was calculated as (1-sensitivity)/specificity. The diagnostic odds ratio (DOR) was calculated as LR+/LR–.
Heterogeneity was evaluated by Higgins I
This meta-analysis was conducted using Review Manager version 5.3 (The Nordic
Cochrane Centre for The Cochrane Collaboration, Copenhagen, Denmark) and Stata/MP
version 14.0 (Stata Corp LP, College Station, TX, USA), statistical significance
was set as p
By systematically searching multiple databases, we retrieved 2476 published articles. By applying the exclusion and inclusion criteria, we selected six comparative studies for formal analysis. Supplementary Table 1 depicts the search strategies applied for each database while Fig. 1 depicts the method used to screen the literature. The main characteristics of the six studies included in our analysis are described in Table 1 (Ref. [14, 15, 16, 17, 18, 29]). In total, these six studies included 223 patients with suspected EMs; 1271 and 1240 lesions were evaluated by WL and ICG imaging, respectively, and all lesions were removed for pathological diagnosis.
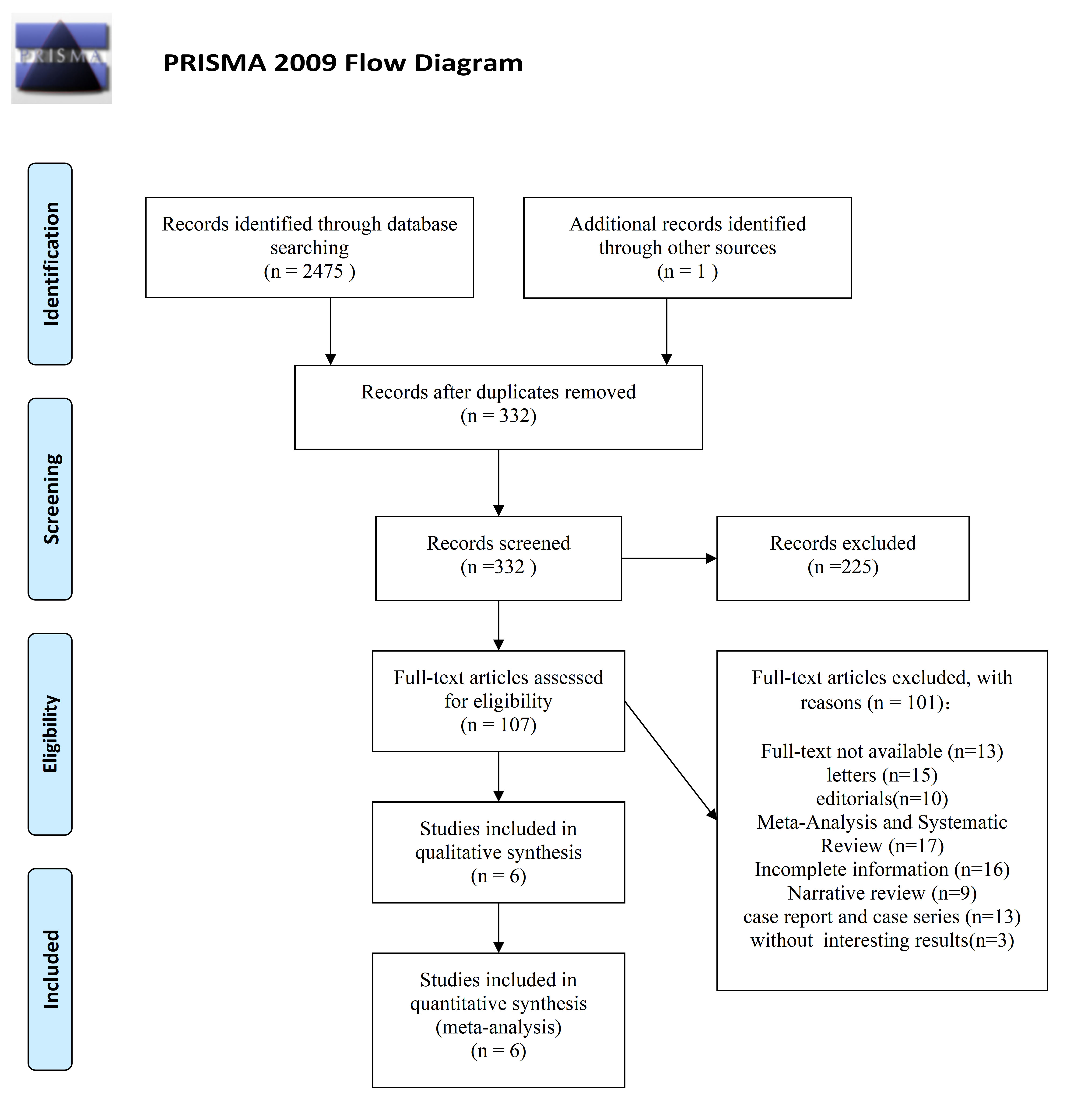 Fig. 1.
Fig. 1.Flow diagram depicting how evidence was acquired for systematic review and meta-analysis.
| Author | Year | Laparoscopic equipment | Study type | No.of patient | WL | ICG | ||||||||
| TP | FP | TN | FN | Total | TP | FP | TN | FN | Total | |||||
| Lier MCI, et al. [14] | 2020 | 2D WL, NBI, ICG, 3D | prospective | 20 | 69 | 13 | 72 | 22 | 176 | 30 | 9 | 73 | 53 | 165 |
| Cosentino F, et al. [15] | 2018 | 2D WL, ICG | prospective | 27 | 95 | 5 | 99 | 16 | 215 | 91 | 2 | 89 | 20 | 202 |
| Al-Taher M, et al. [16] | 2022 | 2D WL, ICG | prospective | 15 | 42 | 4 | 24 | 2 | 72 | 25 | 11 | 17 | 19 | 72 |
| Vizzielli G, et all. [29] | 2020 | 2D WL, ICG, 3D | retrospective | 47 | 158 | 0 | 2 | 28 | 188 | 159 | 2 | 3 | 22 | 186 |
| Siegenthaler F, et al. [17] | 2020 | 2D WL, ICG | prospective | 63 | 150 | 16 | 1 | 6 | 173 | 22 | 10 | 10 | 131 | 173 |
| Turco LC, et al. [18] | 2021 | 2D WL, ICG | prospective | 51 | 200 | 7 | 207 | 33 | 447 | 203 | 3 | 206 | 30 | 442 |
WL, white light; ICG, indocyanine green; TP, true-positives; FP, false-positives; TN, true-negatives; FN, false-negatives; NBI, narrow-band imaging.
Overall, our analysis of WL imaging included six comparative studies featuring
223 patients and 1271 lesions. In total, 821 lesions were pathologically
confirmed as EMs and 450 lesions were pathologically confirmed as non-EMs. Our
analyses indicated that the diagnostic sensitivity was 0.88 (95% confidence
interval [95% CI]: 0.81–0.93) while the diagnostic specificity was 0.85 (95%
CI: 0.49–0.97) (Fig. 2A). The diagnostic score was 3.74 (95% CI: 2.28–5.21)
and the diagnostic odds ratio (DOR) was 42.18 (95% CI: 9.76–182.27) (Fig. 2B).
The LR+ was 5.8 (95% CI: 1.4–24.5) and the LR– was 0.14 (95% CI: 0.09–0.20)
(Fig. 2C). The area under cure (AUC) of receiver operating characteristic curve
(ROC) was 0.92 (95% CI: 0.89–0.94) (Fig. 2D). Asymmetry testing found no
evidence of publication bias (p = 0.26) (Fig. 2E). Collectively, these
results indicate that WL imaging has satisfactory diagnostic value for EMs
although there was significant heterogeneity between studies (I
 Fig. 2.
Fig. 2.The diagnostic value of white light (WL) imaging for endometriosis (EMs). (A) Sensitivity and specificity. (B) Diagnostic score and diagnostic odds ratio (DOR). (C) Positive likelihood ratio (LR+) and negative likelihood ratio (LR–). (D) Area under cure (AUC). (E) The asymmetry test for publication bias. SENS, sensitivity; SPEC, specificity; SROC, summary receiver operating characteristic; ESS, effective sample size.
Overall, our analysis of ICG imaging included six comparative studies featuring
223 patients and 1240 lesions. In total, 805 lesions were pathologically
confirmed as EMs and 435 lesions were pathologically confirmed as non-EMs. Our
analyses showed that the diagnostic sensitivity was 0.64 (95% CI: 0.36–0.84)
and the diagnostic specificity was 0.88 (95% CI: 0.66–0.97) (Fig. 3A). The
diagnostic score was 2.58 (95% CI: 0.33–4.82) and the DOR was 13.15 (95% CI:
1.39–124.19) (Fig. 3B). The LR+ was 5.4 (95% CI: 1.2–24.1) and the LR– was
0.41 (95% CI: 0.18–0.94) (Fig. 3C). The AUC of the ROC was 0.84 (95% CI:
0.81–0.87) (Fig. 3D). Asymmetry testing found no evidence of publication bias
(p = 0.10) (Fig. 3E). Collectively, these results suggest that ICG
imaging has satisfactory diagnostic value for EMs although there was significant
heterogeneity between studies (I
 Fig. 3.
Fig. 3.The diagnostic value of indocyanine green (ICG) imaging for EMs. (A) Sensitivity and specificity. (B) Diagnostic score and diagnostic odds ratio (DOR). (C) Positive likelihood ratio (LR+) and negative likelihood ratio (LR–). (D) AUC. (E) The asymmetry test for publication bias. ESS, effective sample size.
Next, we used Review Manager version 5.3 to compare the diagnostic value between
WL and ICG imaging; Fig. 4A shows a forest plot of the studies included in our
analysis. Direct comparison revealed that the diagnostic value of ICG imaging was
not superior to WL imaging (Fig. 4B). Sensitivity analysis, involving the
systematic exclusion of individual studies did not result in any significant
changes in the final results, thus proving that the results are reliable. Quality
assessments of the six studies included in our meta-analysis are summarized in
Fig. 4C. The six studies were associated with low bias in terms of prospective
design, a sample size
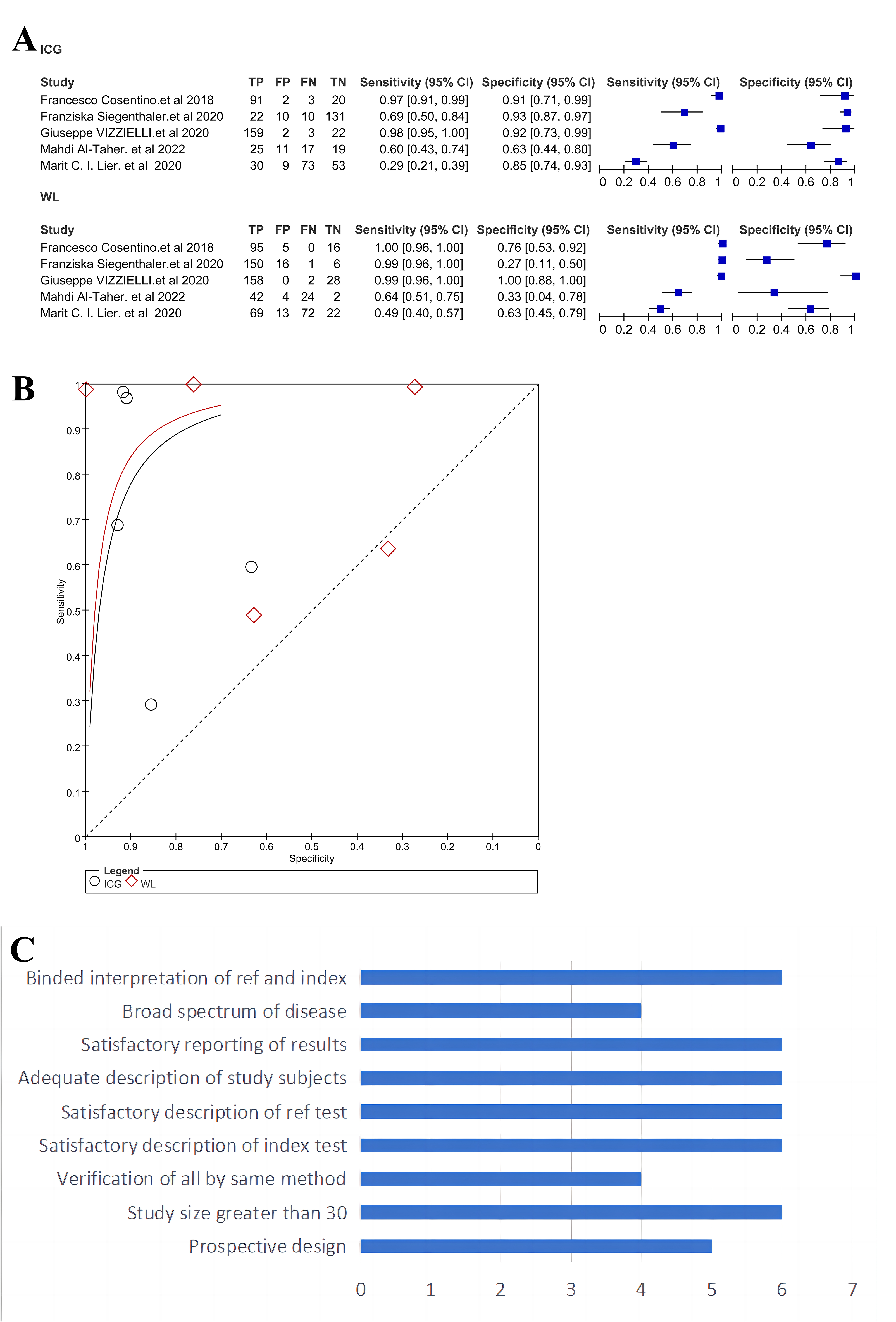 Fig. 4.
Fig. 4.A comparison of diagnostic value between white light (WL) and ICG imaging for EMs. (A) A forest plot generated from the studies included in this meta-analysis. (B) A direct comparison of receiver operating characteristic curves (ROCs) between WL and ICG imaging. (C) The quality assessment of the studies included in this analysis.
In this study, we performed a systematic review and meta-analysis of the published literature comparing WL and ICG imaging for the diagnosis of EMs. First, we report robust outcomes based on the largest series of patients investigated thus far (N = 223 patients); moreover, this represents the first meta-analysis to evaluate the specific diagnostic effect of ICG imaging for EMs to our knowledge. Second, we found that the diagnostic effect of ICG imaging was not superior to conventional WL imaging. Thus, laparoscopic WL imaging with histological confirmation remains the gold standard to define EMs.
EMs is a benign gynecological disorder with a malignant biological behavior that may lead to chronic pelvic pain (CPP), an impaired quality-of-life and fertility issues. This condition affects approximately 10% of women of childbearing age [30]. Despite recent advances in medication and long-term management, a significant number of patients with EMs require surgery because they do not respond well to medication or are experiencing fertility problems. The radical removal of all lesions can relieve pain and improve fertility; however, this practice can be challenging due to the intra-operative identification of EMs. Indeed, the intra-operative diagnosis of EMs remains highly subjective and the rate of diagnostic accuracy is highly dependent on the appearance of EMs, the extent of the lesions, and the experience of the surgeon involved. Missed diagnoses and misdiagnosis are frequent, even by experienced surgeons. Previous studies suggested that up to 25% of EMs lesions may be missed during laparoscopic surgery with WL imaging, and 16% to 53% of suspicious EMs lesions are not pathologically confirmed to be EMs [31, 32, 33].
In view of the high risk of recurrence, repeated episodes of surgery, and the potential impact of removing healthy tissue, there is an urgent need to develop an enhanced form of laparoscopic imaging that can improve the intra-operative and real-time visualization of EMs lesions. Several enhanced imaging techniques have been investigated during EMs surgery to improve the intra-operative diagnosis of EMs, including three-dimensional (3D) white light [34], 5-aminolevulinic acid imaging [35], autofluorescence imaging (AFI) [36] and narrow-band imaging (NBI) [37]. These techniques led to a certain improvement in diagnostic ability but with an unsatisfactory diagnostic accuracy when compared to conventional WL imaging. ICG imaging is a new and developing technique in which intravenous ICG is bound to plasma proteins, thus serving as an ideal angiographic agent. EMs is characterized by chronic inflammation, neovascularization, and hyperangiogenesis; these conditions may be identifiable by ICG imaging. ICG imaging for the detection of peritoneal EMs was first described by Levey et al. in 2014 [10]. In a subsequent study, Park and Farnam [38] reported the first clinical application of ICG imaging for the intraoperative identification of the ureter during resection of deep infiltrating EMs. In another study, Guan et al. [39] reported the advantage of robot-assisted ICG imaging for the resection of rectal deep infiltrating EMs.
Several studies have compared the diagnostic effect of ICG imaging to conventional WL imaging for EMs; however, the results arising from these studies were inconsistent. Lier et al. [14] compared the diagnostic efficiency of conventional two-dimensional WL imaging to NBI, ICG, and 3D imaging when treating 20 patients with advanced EMs (stage III-IV) and found that NBI and ICG imaging did not improve the intra-operative diagnosis of EMs. In another study, Cosentino et al. [15] compared the diagnostic efficiency of conventional WL imaging to ICG imaging when treating 27 patients with EMs and found that ICG imaging might represent an efficient tool for identifying occult EMs, with a sensitivity and specificity of 82% and 97.9%, respectively. Al-Taher et al. [16] compared the diagnostic efficiency of conventional WL imaging to ICG imaging when treating 15 patients with EMs and reported that the advantage of ICG imaging was limited, with a positive predictive value (PPV) of 64% for WL imaging, 69% for ICG imaging, and 61% when using both imaging tools, respectively. Vizzielli et al. [29] evaluated the diagnostic effect of ICG imaging coupled with robotic technical advances during the treatment of 47 patients with EMs; analyses supported the advantage of ICG imaging as a diagnostic tool for EMs. However, Siegenthaler et al. [17] reported that the diagnostic value of ICG imaging was minimal, with a PPV of 89.8% for WL imaging, 68.8% for ICG imaging, and 86.7% for the combination of WL and ICG imaging. In another study, Turco et al. [18] compared the diagnostic efficiency of conventional WL imaging to ICG imaging during the treatment of 51 patients with EMs and found that both ICG imaging, and the combination of WL and ICG imaging, resulted in significant improvements in the rate of EMs detection. These inconsistent results mean that the diagnostic effect of ICG imaging remains uncertain; only the quantitative synthesis of existing published data can yield a more reliable conclusion. This was the main purpose of the present systematic review and meta-analysis. Our analysis showed that the diagnostic effect of ICG imaging is not superior to WL imaging, with an AUC of 0.92 (95% CI: 0.89–0.94) and 0.84 (95% CI: 0.81–0.87) for WL and ICG imaging, respectively.
The ultimate aim of our meta-analysis as to solve clinical confusion with regards to the potential value of ICG imaging for the intra-operative detection of EMs. However, the intraoperative diagnosis effect is highly dependent on several factors, including the appearance of EMs, the extent of the lesions, and the clinical experience of the surgeon. In addition, the infusion time, fluorescence assessment, type of camera (2D, real 3D, or robotic) and the specific type of EMs, may also exert influence on the diagnostic effect. Our present meta-analysis revealed significant heterogeneity between the six studies identified by our literature search. By systematically evaluating these six studies, we identified significant differences relating to the selected patients, stages and types of EMs, surgical procedures, control methods, biopsies of the normal peritoneum as a control, ICG doses, and the interval between intravenous ICG injection and exploration. These factors may represent the sources of heterogeneity and may have influenced the reliability of our conclusion. The potential effects of these factors represent the main limitations in the present study. Another important limitation to consider is the limited number of cases, this may also have influenced the robustness of our findings.
The main strength of our study is that this represents the first meta-analysis of published studies relating to the diagnostic effect of ICG imaging for the detection of EMs. Although, our results did not identify a significant difference between ICG and WL imaging, it is important to consider a serious issue. All six of the original studies reported that certain EMs lesions could only be identified by ICG imaging, and were not detectable by WL imaging, thus suggesting that ICG imaging may be helpful for the visualization of occult EMs. Consequently, ICG imaging could represent a vital tool for the radical removal of EMs lesions, the relief of chronic pelvic pain, the improvement of fertility, and in reducing disease recurrence and the need for repeat episodes of surgery.
As a novel intra-operative form of real-time diagnostic technology, ICG imaging needs to be investigated further in order to fulfil its maximal potential. Future research should evaluate the long-term clinical outcomes of ICG-guided surgery for EMs, including re-intervention rates, recurrence rates, quality-of-life and pain-free intervals. This research should involve homogeneous, randomized controlled studies in multiple centers with large sample sizes if we are to fully validate the diagnostic value of ICG imaging.
Although ICG may facilitate the visualization of occult EMs, the diagnostic effect of ICG imaging is not superior to that of WL imaging. However, our findings should be considered cautiously owing to the limited number of research studies in this field and the small sample sizes of the existing literature. There is an urgent need to validate our findings using reliable evidence from studies performed at multiple centers with large sample sizes.
All data generated or analysed during this study are included in this article. Further enquiries can be directed to the corresponding author.
HY and YZ designed the research study. HO and YX performed the research. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This work is supported by the Medical Science and Technology Research Foundation of Guangdong Province (A2022317).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




