1 College of Veterinary Medicine, Hunan Agricultural University, 410128 Changsha, Hunan, China
2 Department of Obstetrics, The People's Hospital of Longhua Shenzhen, 518109 Shenzhen, Guangdong, China
3 Department of Orthopaedics and Traumatology, The People's Hospital of Longhua Shenzhen, 518109 Shenzhen, Guangdong, China
4 Department of Gynaecology, The People's Hospital of Longhua Shenzhen, 518109 Shenzhen, Guangdong, China
5 Department of Obstetrics and Gynaecology, Center for Reproductive Medicine, Key Laboratary for Major Obstetric Diseases of Guangdong Province, The Third Affiliated Hospital of Guangzhou Medical University, 510150 Guangzhou, Guangdong, China
6 Guangdong and Shenzhen Key Laboratory of Male Reproductive Medicine and Genetics, Peking University Shenzhen Hospital, Shenzhen Peking University-Hong Kong University of Science and Technology Medical Center, 518036 Shenzhen, Guangdong, China
Abstract
Background: TBC1 domain family member 20 (TBC1D20) is important in male
reproductive, eye lens function and neural system development. However, the role
of TBC1D20 on reproduction in female mice remain unclear. Methods: As a
prospective laboratory-based study, a mouse model with spontaneous functional
mutations of Tbc1d20 (Tbc1d20
Keywords
- TBC1D20
- uterine development
- decidualization
- endoplasmic reticulum stress
The uterus is a part of the female reproductive tract and comprises the endometrial and myometrial layers. Uterine endometrium is organized and constructed by many different cell populations which cooperated playing important roles in embryonic development, implantation, and placentation [1, 2, 3, 4]. The Uterine development including stratification of the uterine stroma, myometrial differentiation and growth, and the differentiation and development of endometrial glands, was accomplished in rodents, domestic animals and humans at the postnatal [5, 6, 7]. The processes of uterine development and uterine functions are exact regulated by intrinsic and/or extrinsic regulatory factors. Previous studies have demonstrated that loss of function mutations of certain genes cause defect related to female fertility [6, 8, 9]. The ovarian steroid hormones, estrogen and progesterone, play pivotal roles in regulating uterine functions for embryonic implantation, development, and pregnancy. Mice lacking functional estrogen receptors (ERs) are infertile and have hypoplastic uterus [10]. Various functional and morphological changes with disorder phenotype are observed in the endometrium of ERs mutation mice, relating leukocyte recruitment and the differentiation of endometrial stromal fibroblast cells (ESCs) into decidual stromal fibroblast cells (DSCs), to form the decidual lining during the implantation of blastocysts [11]. Proper decidualization controls conception and the exact time course of pregnancy, and is a critical determinant of the success of pregnancy in humans and mice. Progesterone receptor (PR) gene knockout resulted in uterine hyperplasia and loss of decidualization, which render the uterus non-receptive for blastocyst implantation in mice [12].
The blind sterile (bs) mouse model reported in 1983 exhibits the
typical phenotypes of Warburg Micro syndrome (WARBM), including congenital
cataract, male infertility and gonadal dysplasia [13]. A causative gene mutation
of the TBC1 domain family member 20 (TBC1D20) is response for the abnormal of
phenotype of bs mice, which is characterized by congenital cataracts and
male infertility [14, 15]. Our previous study also found that TBC1D20 is
essential for maintaining the integrity of the blood-testis barrier postnatally
and viability of Sertoli cells by modulating the structure and function of the
endoplasmic reticulum and Golgi apparatus [16, 17]. Furthermore, during the
breeding of bs mice, we also realized the phenomena of reducing
reproductive capacity of female mouse with TBC1D20 deficiency. However, the exact
role and cellular pathway of Tbc1d20 in female fertility remains to be
elucidated to date. In this study, we used the female bs mouse (defined
as Tbc1d20
This research is a prospective laboratory-based study. The bs mouse
(Tbc1d20
Ovaries and fallopian tubes were collected during the estrus phase for
subsequent morphological analysis. Ovariectomized Tbc1d20
The ovariectomized mice were injected with E2 for three days (100 ng/day) and
the uterine stromal cells were isolated at D4. Briefly, uterine horn from mice
was cut into 3–5 mm pieces and dissected longitudinally to expose the uterine
lumen. Tissue was washed with phosphate buffered saline (PBS) and then immersed
in 0.25% trypsin (Sigma, St. Louis, MO, USA) for 1 h at 4 °C followed by
1 h at room temperature. PBS containing 10% charcoal-stripped fetal bovine serum
(Invitrogen, Carlsbad, CA, USA) was used to terminate digestion and mixed gently
to remove epithelial clumps by pipetting. The remaining tissues were reserved,
then was washed twice in PBS and finally placed in PBS containing 0.5%
collagenase Type II (Sigma, St. Louis, MO, USA) for 30 min at 37 °C. 10%
fetal bovine serum (FBS) was used to stop the digestion. After being washed twice in PBS, the tissues
were mixed thoroughly by pipetting up and down to disperse stromal cells. The
stromal cells were collected by centrifugation. The isolated primary endometrial
stroma cells were first cultured in vitro for establishing a stromal
cell proliferation model in 6-well cell culture plates seeded at a density of
with 2.5
Sections of uterine tissue were deparaffinized and rehydrated before assigning to hematoxylin and eosin (H&E) staining or immunohistochemical assays. Immunohistochemical assays were performed using a Streptavidin-Peroxidase Histostain-Plus Kit (SAP-9100, Beijing Zhong Shan Goldenbridge Biotech., Beijing, China), according to the manufacturer’s instructions. Briefly, sections were treated with 3% hydrogen peroxide for 15 min, washed thrice with PBS, permeabilized with 0.05% Triton-X 100 for 2 min, and subsequently rinsed with PBS. Then sections were blocked with a blocking buffer for 1 h and incubated overnight at 4 °C with primary antibodies (anti-TBC1D20, 1:100 diluted, NBP1-92478, Novus Biologicals, Inc., Littleton, CO, USA; Ki67, 1:200 diluted, ab16667, Abcam, Cambridge, MA, USA; Cytokeratin, 1:200 diluted, Z0622, DAKO, Carpinteria, CA, USA; Vimentin, 1:100 diluted, ab92547, Abcam, Cambridge, MA, USA; and anti-estrogen receptor alpha antibody, 1:100 diluted, IR657, DAKO, Carpinteria, CA, USA). Section incubated with rabbit IgG was employed as negative control. Sections were subsequently incubated with the secondary antibody for 1 h at room temperature. The peroxidase reaction was performed with a SignalStain® DAB Substrate Kit (8058s, CST), and the signals were measured by light microscopy (Nikon ECLIPSE Ci-L, Nikon, Tokyo, Japan).
For immunofluorescence assay, the dewaxed uterine tissue sections were subjected
to antigen retrieval in citrate antigen retrieval solution (pH 6.0), and
subsequently incubated with 5% bovine serum albumin for 1 h followed by
overnight incubation with the primary antibodies (anti-alpha smooth muscle
actin, ab5694, Abcam, Cambridge, MA, USA; anti-CD34 antibody, PA5-89536, Thermo
Fisher Scientific, Cleveland, OH, USA) at 4 °C. The tissue sections were
washed with PBS and incubated with the fluorescent secondary antibody (Thermo
Fisher Scientific, Cleveland, OH, USA) for 1 h at room temperature. The tissue
sections were stained with 4
For postnatal uterine morphological and histological measurement, the results from H&E assay, immunohistochemistry analysis and immunofluorescence assay of each individual, were average valued based on three tissue sections from the proximal cervical segment, the middle segment of the uterine horn and the tail segment of the uterine horn of the same uterus, respectively. To measure the thickness of the longitudinal and circular layers of the myometrium, starting from the side of the uterine mesentery, lines perpendicular to the circumference of the longitudinal and circular layers of the uterus at multiple evenly distributed locations was drawn and valued by image J (https://imagej.net/ij/), a public domain Java image processing program inspired by National Institutes of Health (NIH) Image for the Macintosh. The calculate average value was deemed as the thickness of thickness of the longitudinal and circular layers of the myometrium.
The total RNA was extracted and purified from tissues/cells using TRIzol™
reagent (Invitrogen, CA, USA). The concentration of the RNA was quantitated using
an UV5Nano Spectrophotometer (Mettler-Toledo, LLC, Columbus, OH, USA). The RNA
was reverse transcribed to the complementary DNA (cDNA) using a PrimeScript RT
Master Mix kit (Takara, Dalian, Liaoning, China) with 1 µg of total RNA per
reaction in a final volume of 20 µL was used. Quantitative RT-PCR (qRT-PCR) was
performed using the SYBR Premix Ex Taq II PCR Kit (Takara, Dalian, Liaoning,
China), according to the manufacturer’s 185 instructions with an Applied
Biosystems™ 7500 Real-Time PCR System (Thermo Fisher Scientific,
Cleveland, OH, USA). The protocol of qRT-PCR was as follows: pre-denaturation at
95 °C for 30 sec, denaturation at 95 °C for 5 sec, and 35
cycles of annealing at 60 °C for 34 sec, following which the reaction
was halted at 4 °C. The results of PCR were analyzed using the
comparative threshold cycle (
The proteins were extracted from whole cells using lysis buffer (50 mM Tris-HCl (pH 7.6) and 2% sodium dodecyl sulfate (SDS)) supplemented with proteinase inhibitor cocktail (P8340, Sigma-Aldrich, St. Louis, MO, USA). The concentrations of the proteins were measured using the bicinchoninic acid (BCA) method (Thermo Fisher Scientific, Waltham, MA, USA). The protein extracts (30 µg per sample) were separated by 12% SDS-PAGE (polyacrylamide gel electrophoresis) and electrophoretically transferred onto polyvinylidene difluoride (PVDF) membranes (0.45 µm; Millipore, Bedford, MA, USA). The PVDF membranes were blocked with 5% skim milk in Tris-buffered saline with 0.05% Tween-20 (TBST) for 1 h at 24 °C, and sequentially incubated overnight with the primary antibodies at 4 °C. The membranes were then incubated with a horseradish peroxidase (HRP)-conjugated secondary antibody for 1 h at 24 °C. The signals were measured using a MilliporeSigma™ Immobilon™ Western Chemiluminescent HRP Substrate (Billerica, MA, USA) and the results were recorded with an Amersham ImageQuant™ 800 Western Blot Imaging System (Cytiva, Marlborough, MA, USA). Information regarding the primary antibodies used for western blotting are provided in Supplementary Table 2.
Blood samples were collected from two-month-old Tbc1d20
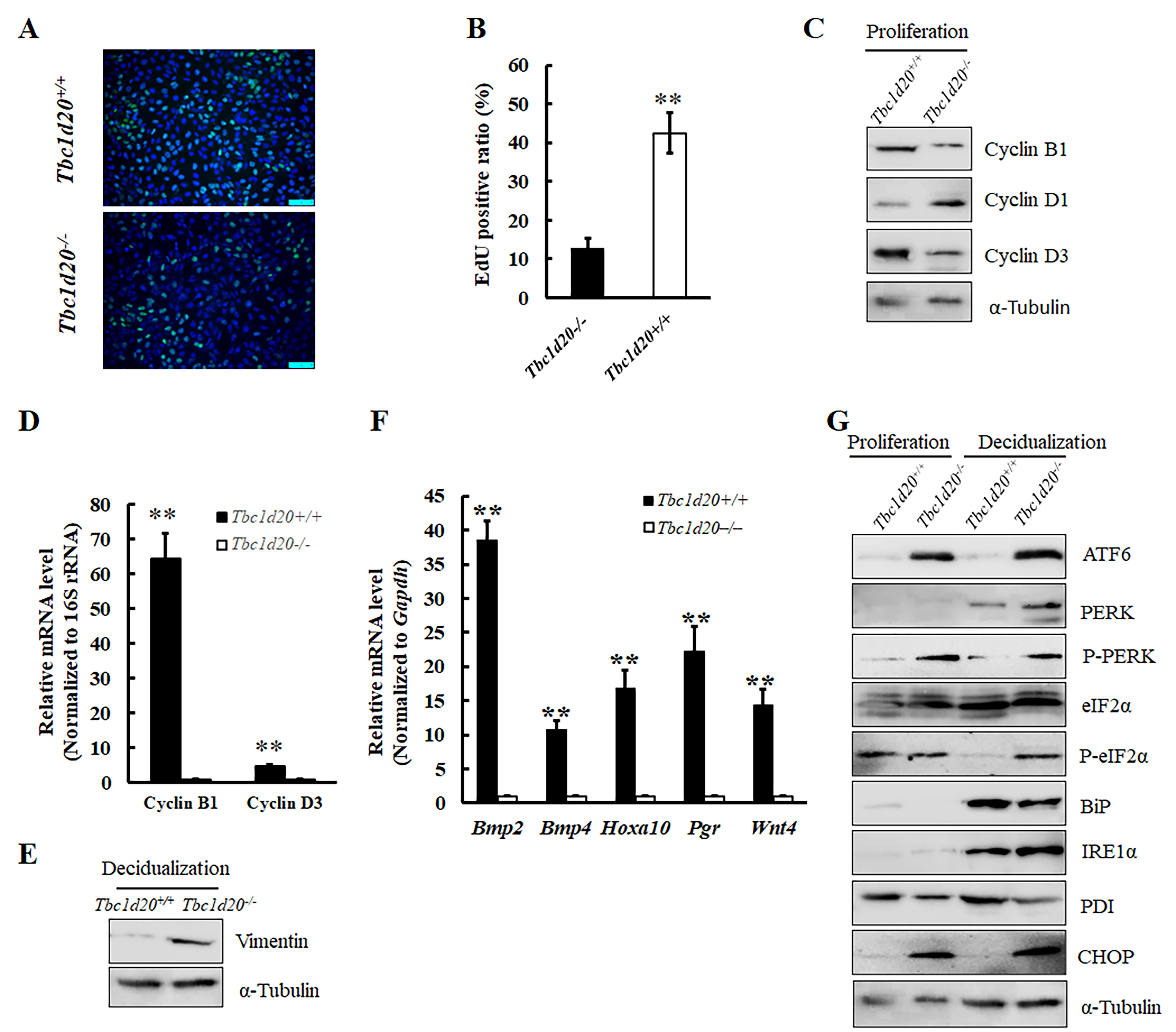
Cell proliferation was detected using a Cell-Light™ EdU (5-ethynyl-2-deoxyuridine) Apollo567 In Vitro Kit (RiboBio, Guangzhou, Guangdong, China) according to the Kit Operation Manual. Briefly, cell was seeded in 96-well plate. Culture medium containing EdU was added to well and incubation with cells for 1 h. Then the cells were fixed with polyformaldehyde (PFA) for 10 min at room temperature. The cell nuclei were double dyed with EdU and DAPI for assessing cell proliferation. The images were captured with a laser confocal microscope (Leica TCS SP8), and the cells were counted from six different microscopic fields.
The data were presented as the mean
The mutation in Tbc1d20 results in a p.Phe231Met substitution followed
by an in-frame p.Arg232_Val235 deletion in the Tbc1d20 protein
(Supplementary Fig. 1A), which disrupts the highly conserved amino acids
in the TBC domain of Tbc1d20. Result of statistical assay of
reproductive capacity showed that Tbc1d20
| Female | Male | Couples of matings | Females having offspring (%) | Average litter size |
| Tbc1d20 |
Tbc1d20 |
20 | 0 | 0 |
| Tbc1d20 |
20 | 100 | 7.263 | |
| Tbc1d20 |
20 | 100 | 7.409 |
In order to explore the causes of infertility in Tbc1d20
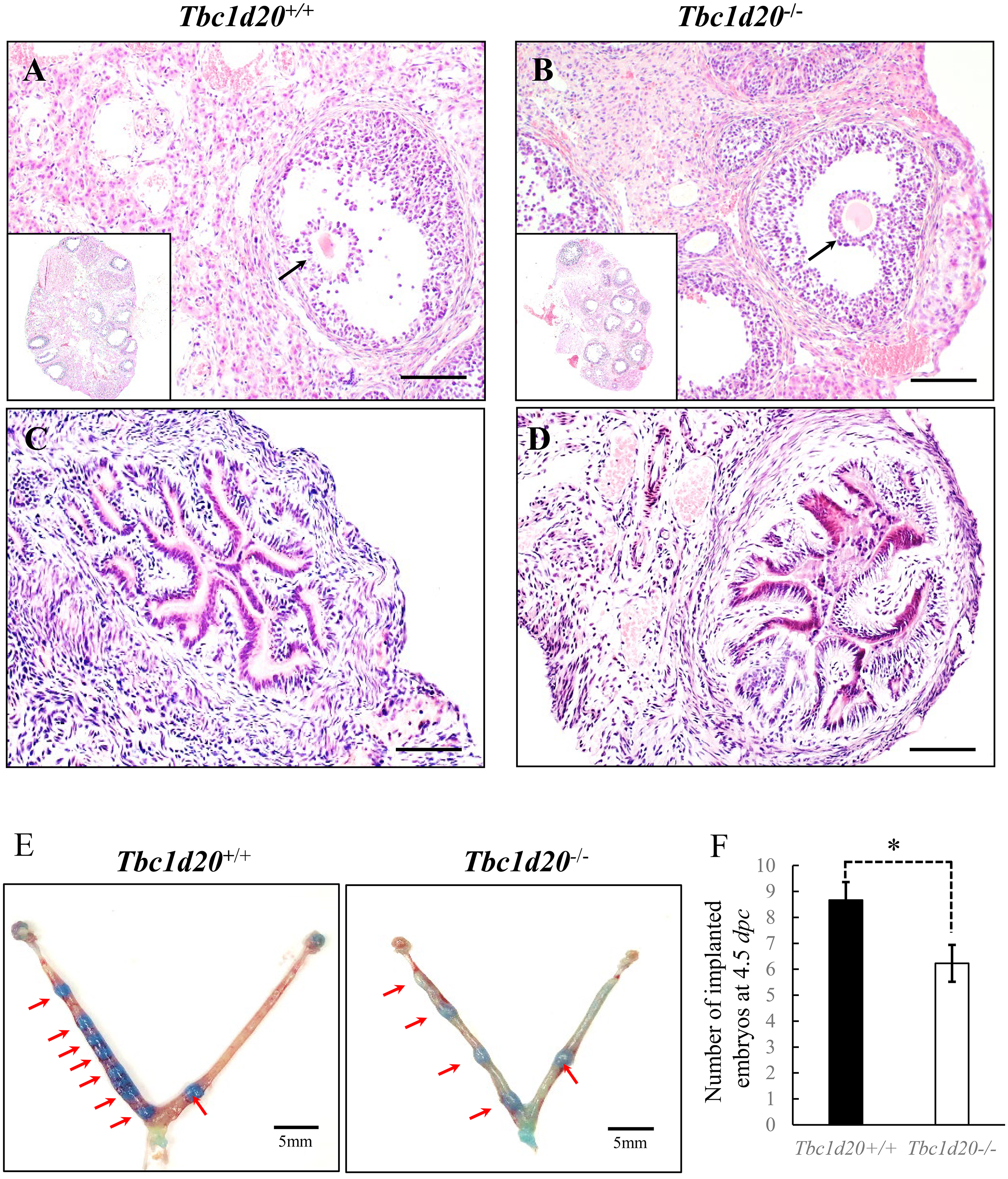
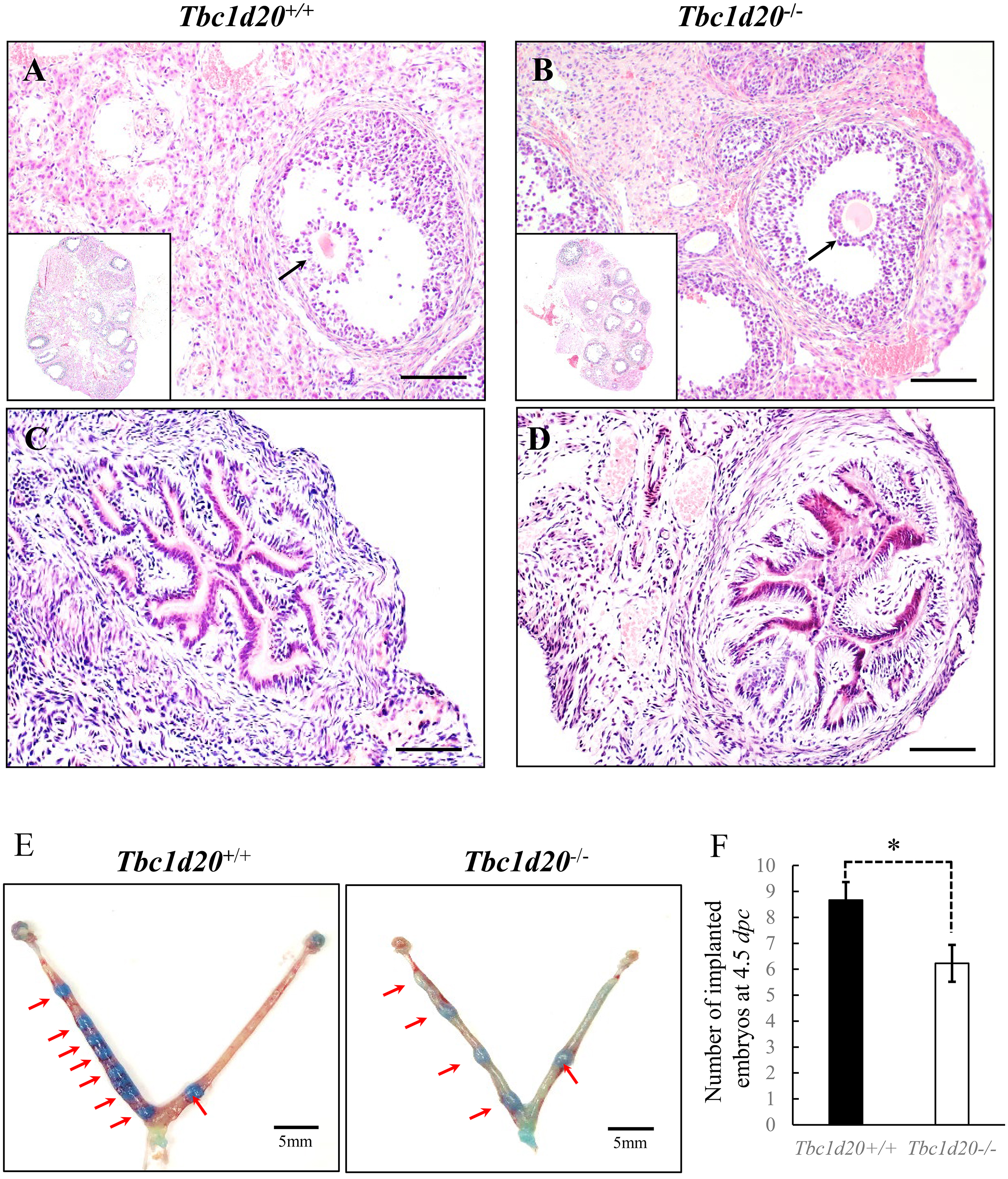 Fig. 1.
Fig. 1.Female mice with loss-functional mutations in Tbc1d20
exhibited normal ovulation and successful embryonic implantation. (A–D)
Hematoxylin and eosin (H&E) staining of ovaries and uterine samples during the
estrus phase in Tbc1d20
The uterine histomorphology of the mutant Tbc1d20
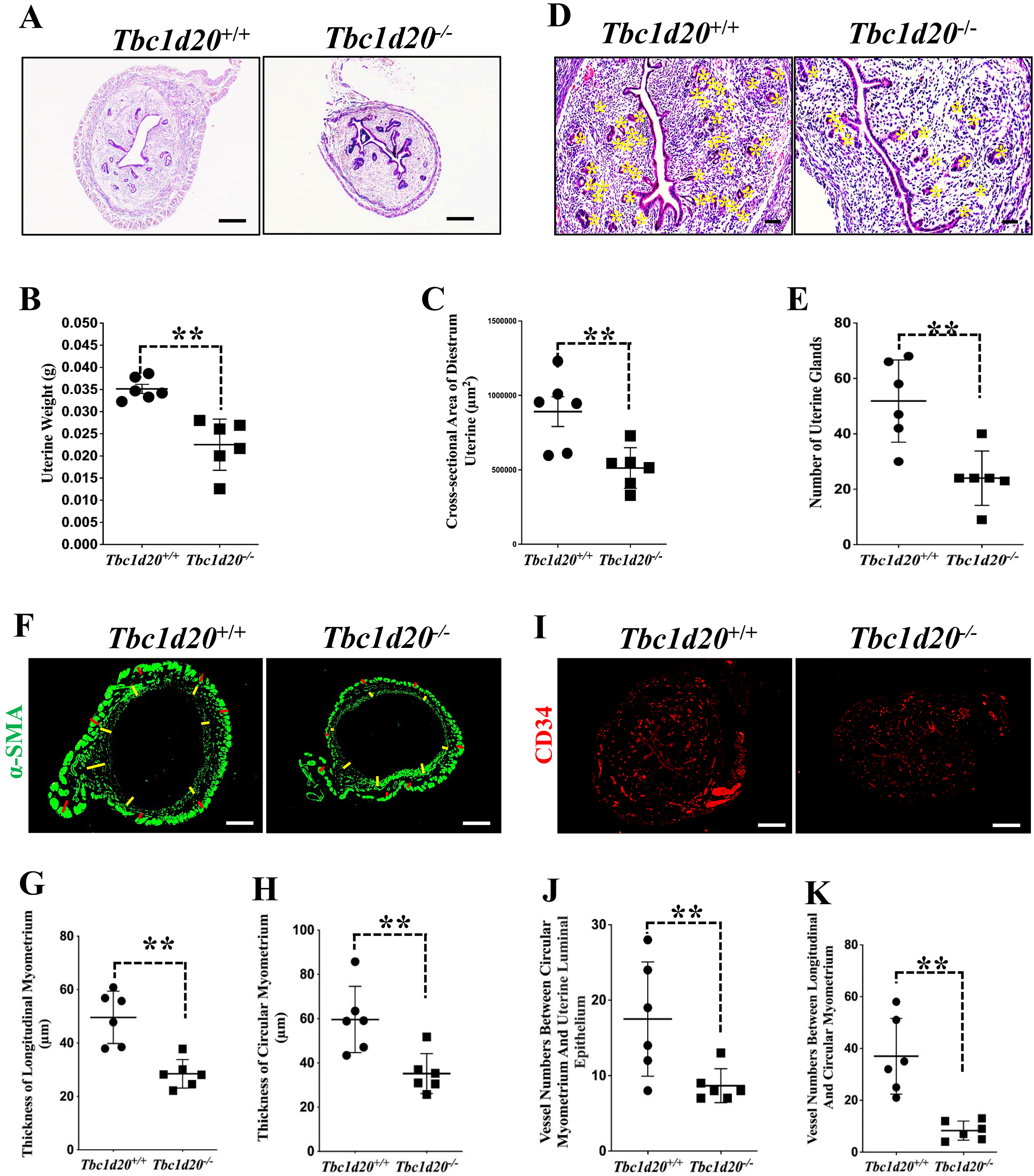
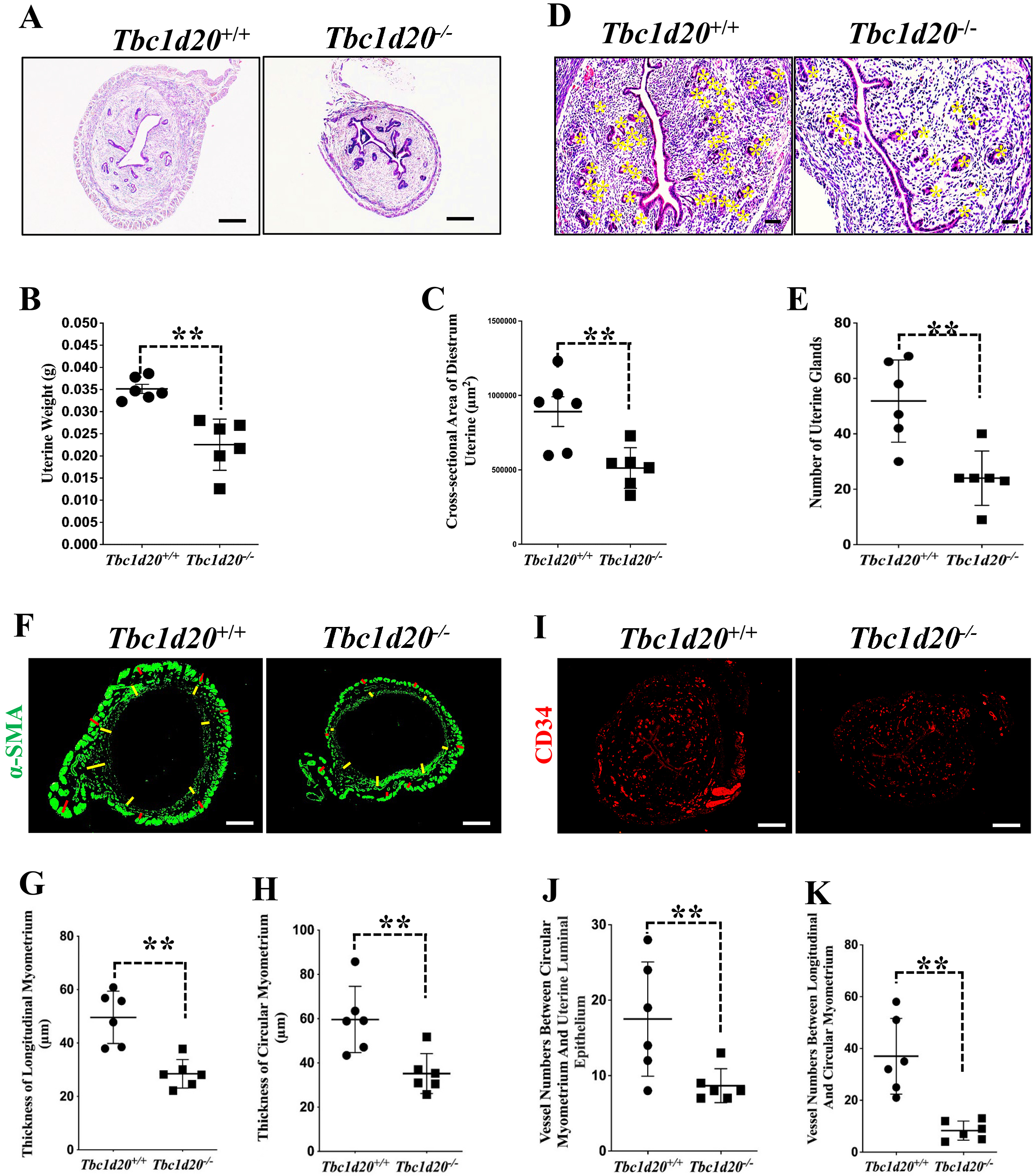 Fig. 2.
Fig. 2.Loss-functional mutations in Tbc1d20 impair postnatal
uterine development in mice. (A–C) H&E staining revealed a reduction in the
size of the uterus in Tbc1d20
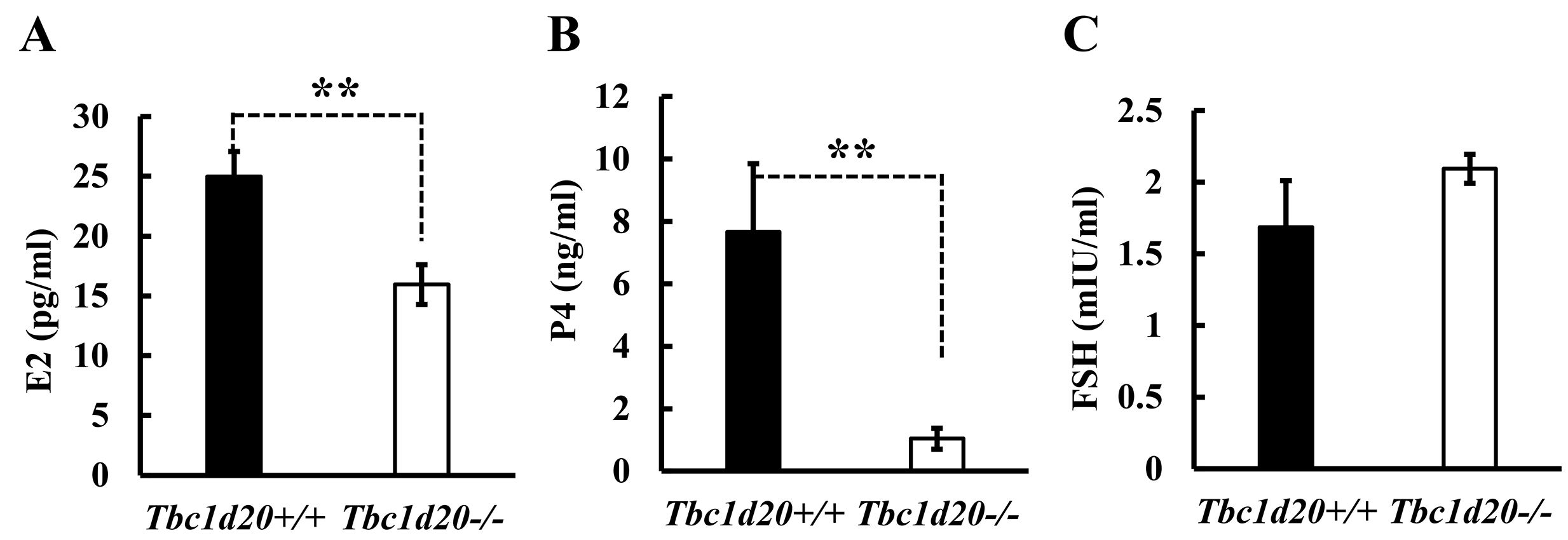
Concentration of sex hormones, the E2, P4 and FSH in maternal serum at the
diestrus stage, were determined by RIA. Results from RIA indicated that the
levels of E2 (p = 0.0095, n = 6; Fig. 3A) and P4 (p = 0.0133, n
= 6; Fig. 3B) decreased significantly in female Tbc1d20
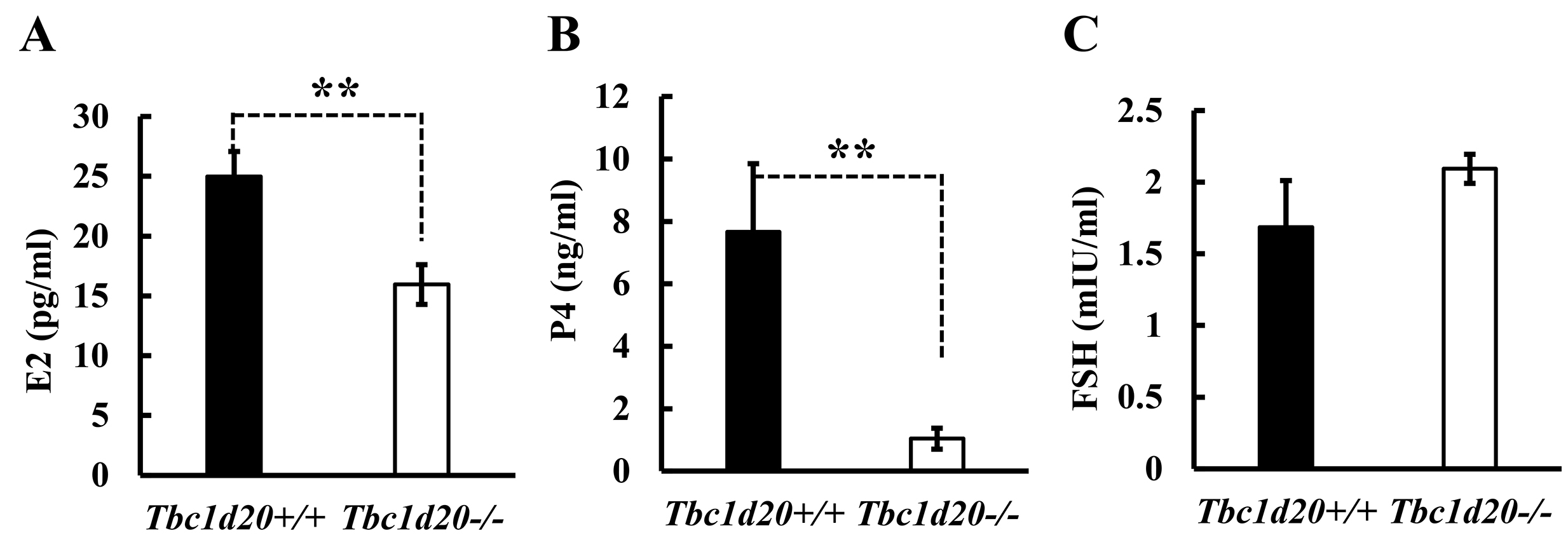 Fig. 3.
Fig. 3.Loss of function mutations in Tbc1d20 affect the
expression of steroidal sex hormones in female mice. The concentrations of
steroidal sex hormones, including estrogen (E2) (A), progesterone (P4) (B), and
follicle-stimulating hormone (FSH) (C) were measured by radioimmunoassay (RIA) (n
= 6). The expression levels of these hormones in Tbc1d20
Results from our research indicated that the affected ovary function and embryo
implantation ability does not seem to be the main cause of infertility in
Tbc1d20
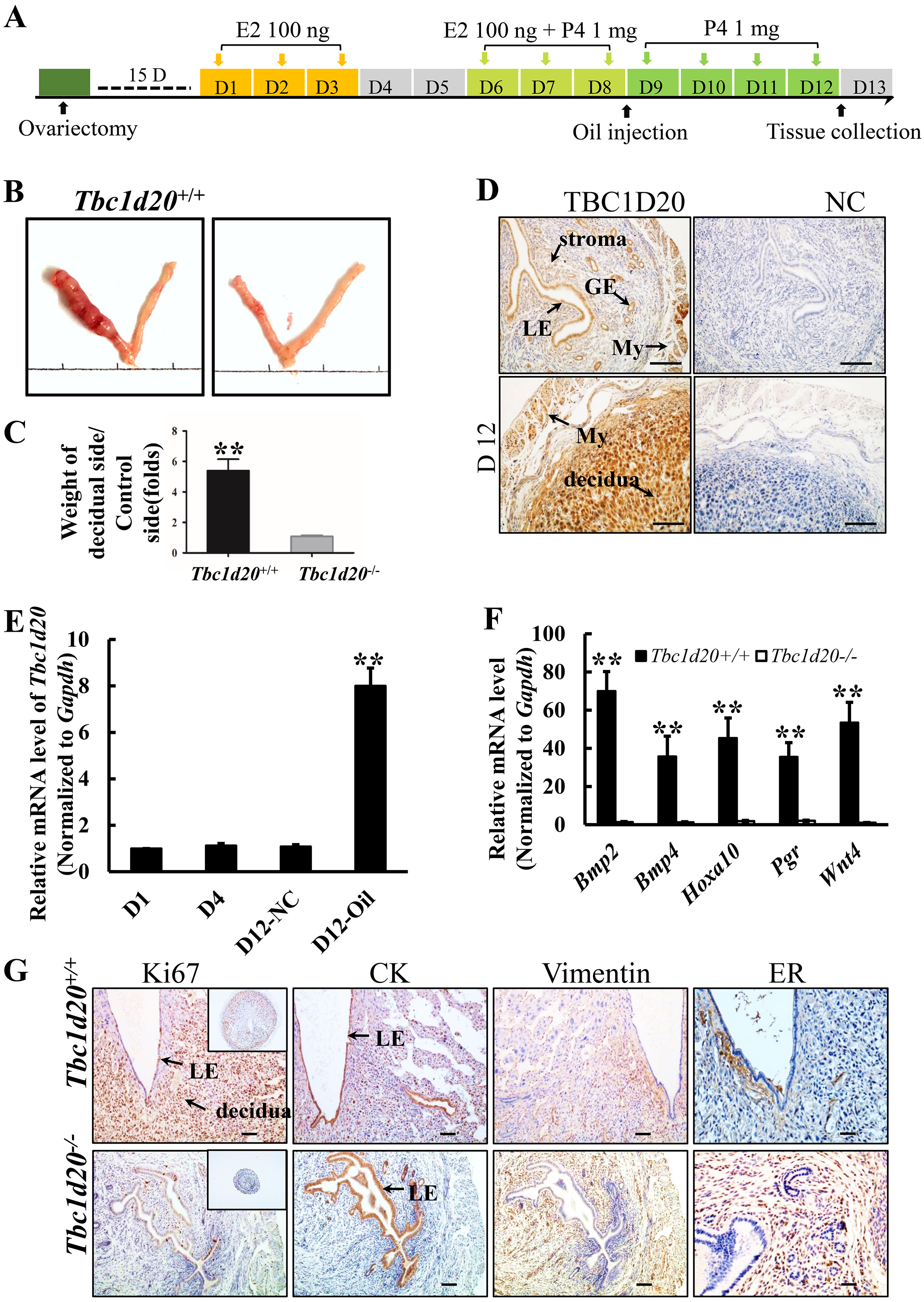
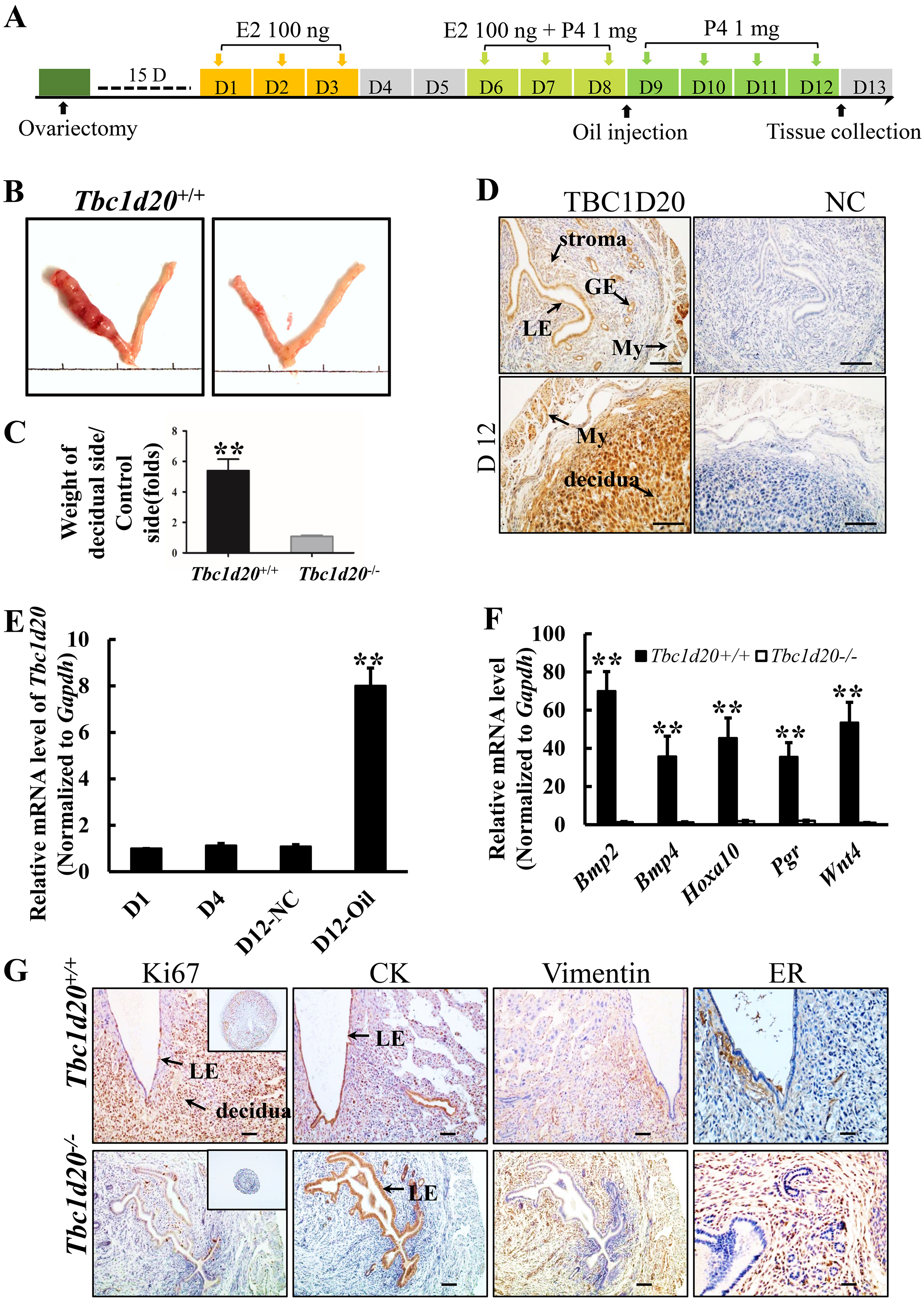 Fig. 4.
Fig. 4.Loss-functional mutations in Tbc1d20 results in failure
of uterine decidualization. (A) Procedure of artificial decidualization
in vivo. (B) Morphology of the uterine tissues in which the left horn
was stimulated with sesame oil and the right horn was left untreated as the
normal control. (C) Ratio of the average weights of the stimulated uterine horns
to those of the unstimulated uterine horns from WT and Tbc1d20
Stromal cells were isolated from the uterus of diestrous mice and cell
proliferation was detected with 5-Ethynyl-2’-deoxyuridine (EdU) assays, for 1 h in vitro. The
findings revealed a significant decrease in the number of EdU-positive uterine
stromal cells lacking functional TBC1D20 (Fig. 5A,B). The expression levels of
cyclin B1 and cyclin D3 decreased significantly in the proliferative uterine
stroma cells of WT mice while the expression levels of cyclin D1 increased
significantly (Fig. 5C,D) compared to those of the mutant mice. These findings
suggested the occurrence of impaired DNA synthesis in the uterine stromal cells
of Tbc1d20
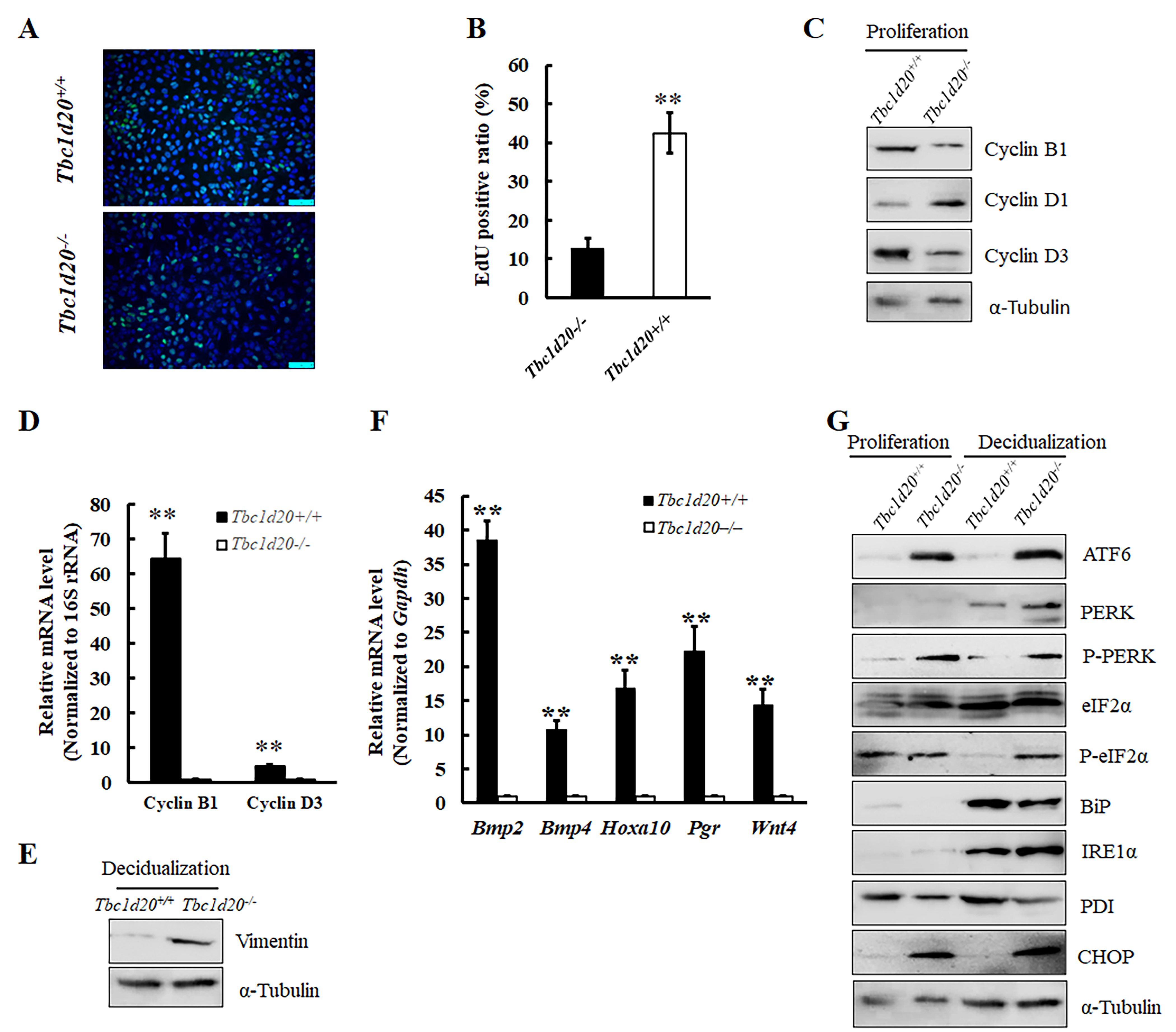 Fig. 5.
Fig. 5.Mutations in Tbc1d20 retarded the proliferation and
decidualization of uterine stromal cells in vitro due to the disrupted
function of endoplasmic reticulum. (A,B) The 5-Ethynyl-2’-deoxyuridine (EdU) incorporation assay revealed
that Tbc1d20 depletion significantly inhibited the proliferation of
endometrial stromal cells. (C) Western blotting was used to detect the levels of
cyclin B1, cyclin D1, and cyclin D3 proteins in murine endometrial stromal cells
during the proliferative phase in vitro. (D) The results of quantitative
reverse transcription polymerase chain reaction (qRT-PCR)
revealed that the deletion of Tbc1d20 inhibited the expression of cyclin
B1 and cyclin D3 mRNA in murine endometrial stromal cells during the
proliferative phase in vitro. (E) Western blotting was used to detect
the levels of vimentin protein in murine endometrial stromal cells that underwent
decidualization following artificial induction. (F) The expression level change
of five marker genes associated with uterine stroma cell decidualization
differentiation were detected by qRT-PCR. (G) Western blotting was used to detect
the expression levels of endoplasmic reticulum stress-related proteins, namely,
CHOP, BiP, IRE
In our previous study, we demonstrated that the deletion of TBC1D20 resulted in
endoplasmic reticulum stress in mouse Sertoli cells, and excessive endoplasmic
reticulum stress induced DNA damage and promoted cell death. We therefore
examined the expression of the key proteins that regulate endoplasmic reticulum
stress. The results demonstrated that the expression levels of C/EBP homologous
protein (CHOP), inositol-requiring 1 alpha (IRE1
Normal anatomical and histological structure of postnatal uterus is a
prerequisite for successful gestation in mice and humans. The development of the
uterus in humans and mice is not fully accomplished until the embryo implantation
stage [18, 19]. Prenatal uterine development, which is referred to as
organogenesis, begins at the embryonic stage and comprises the formation,
patterning, and fusion of the Müllerian ducts. Postnatal uterine development
requires further morphogenesis and differentiation into functional adult
components. Serial genes have been found that are important in postnatal uterine
development in mice and humans [5, 8, 19, 20]. However, most key genes related to
uterine growth and development and their exact function pattern that are still
unknown [5]. In this study, we found Tbc1d20 is essential for normal
postnatal uterine development and decidualization in mice. Causative mutation of
the Tbc1d20 gene in bs mouse model markedly limited postnatal
uterine morphogenesis and resulting in failed decidualization. Expression pattern
of TBC1D20 in uterine indicated the involvement of this gene in regulating the
cellular activity of the stromal cells, myometrium, blood vessel endothelium, and
the glandular and luminal uterine epithelia of prepubertal mice. The thickness of
the endometrial stromal layer, longitudinal and circular layers of the
myometrium, and the number of uterine glands and vessels were significantly
reduced in the postnatal uterus of Tbc1d20
Postnatal uterine morphogenesis in mice and humans is governed by a variety of
hormonal, cellular, and molecular mechanisms [18, 19, 23]. In this study, we
observed that the concentration of E2 and P4 in the peripheral blood of female
Tbc1d20
In this study, successful embryo implantation was observed in TBC1D20 deficiency
female mice. But the number of implantation embryo was reduced when was compared
to WT (Fig. 1E,F), which may be attributed to the negative effects of
Tbc1d20 mutation on the viability of embryos. The speculation that the
Tbc1d20
Endoplasmic reticulum stress seems to play a role in female reproductive
processes [28]. The phenomena that moderate elevation of endoplasmic reticulum
stress markers during the decidualization of human endometrial stromal cells was
reported [29]. Thus, appropriate and finely regulated endoplasmic reticulum
stress in time course may be critical for uterine decidualization or to meet the
requirement of accommodate protein synthesis during decidualization [30], whereas
excessive and disorder endoplasmic reticulum stress leads to DNA damage and
inhibits the proliferation and decidualization of stromal cells [28]. We
previously identified that TBC1D20 assembles in the Golgi apparatus and
endoplasmic reticulum of Sertoli cells, which induces irreversible endoplasmic
reticulum stress, leading to the disruption of organelles [16]. The present
findings revealed that the loss of function in TBC1D20 disrupted the metabolism
of steroid hormones and the corresponding receptors in female mice. This could
also be attributed to the structural and functional interference resulting from
the endoplasmic reticulum and Golgi apparatus. In this research, checkpoint
markers of the three typical signaling pathway in endoplasmic reticulum stress
were detected by western blotting. And results indicate the activation of
endoplasmic reticulum stress in the proliferative uterine stromal cells and
differentiated decidual cells caused by Tbc1d20 mutations, is mainly regulated on
PERK-eIF2
Our findings suggested that TBC1D20 is involved in the regulation of postnatal uterine development and decidualization, and possibly controls the proliferation of endometrial stromal cells by regulating endoplasmic reticulum stress. However, further studies are necessary for well understanding the cellular and molecular mechanisms of TBC1D20 in regulating of postnatal uterine growth and development.
The data are available from the corresponding author on reasonable request.
WM conducted the whole experiments and prepared the manuscript. XL, JZ and WY performed parts of the experiments and analyzed the data. QY, XJ and BL supervised the data and revised the manuscript. LL, YH, LC and WLC designed the study, reviewed the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was performed with the Guide for the Care and Use of Laboratory Animals by the Institute of Laboratory Animal Resources for the National Research Council. The protocol was approval by the Animal Ethics Committee of Shenzhen Peking University-Hong Kong University of Science and Technology Medical Center (Ethics approval number: 2019-205).
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their sincerely opinions and suggestions.
This work was supported by grants Guangdong Basic and Applied Basic Research Foundation (2019A1515110884; 2021A1515010515), Medical Scientific Research Foundation of Guangdong Province of China (A2023182), Shenzhen Science and Technology Innovation Program (JCYJ20190809090803702; JCYJ20210324140600001) and the Scientific Research Projects of Medical and Health Institutions of Longhua District Shenzhen (2020001; 2021008; 2021020).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.