1 The First Clinical Medical College, Nanjing University of Chinese Medicine, 210023 Nanjing, Jiangsu, China
2 Center for Diagnosis and Treatment of Cervical Diseases, Changzhou Maternity and Child Health Care Hospital, Changzhou Medical Center, Nanjing Medical University, 213004 Changzhou, Jiangsu, China
3 Department of Gynecology, Beigang Street Community Health Service Center, 213023 Changzhou, Jiangsu, China
4 International Medical College, Chongqing Medical University, 400016 Chongqing, China
5 Chinese Medicine Gynecology Department, Jiangsu Province Hospital of Chinese Medicine Affiliated Hospital of Nanjing University of Chinese Medicine, 210004 Nanjing, Jiangsu, China
†These authors contributed equally.
Abstract
Background: This real-world study aims to investigate clinical
efficacy and safety of CO
Keywords
- chronic cervicitis
- clinical efficacy
- Kushen gel
- CO2 cryotherapy
One of the most prevalent gynecologic conditions is chronic cervicitis, which primarily affects women who have previously given birth. Approximately 60% of married women experience persistent cervicitis [1]. The clinical symptoms of chronic cervicitis mainly include increased vaginal discharge, mucous purulence, pruritus of the vulva and contact bleeding in severe cases [2, 3].
The are several main treatments of chronic cervicitis [4]. Surgical therapy is
the most commonly used method and comprises laser, microwave, cryotherapy, or
loop electrosurgical excision procedure (LEEP) [5]. Among these, CO
Following surgical therapy, there usually follows adverse reactions, such as
continuous vaginal drainage, vaginal bleeding and cervical stenosis, the latter
resulting in secondary infertility and cervical dystocia [4, 5]. Kushen gel is a
semisolid gel made of a mixture of matrine (C15H24N2O) and carbopol. Matrine is
an alkaloid from the traditional Chinese herb medicine sophora flavescens
reported to exhibit anti-inflammatory, anti-bacterial, and protective effects
[9]. It is a topical preparation that is absorbed through the tissue mucosa
without any irritation. Because it is in gel form, the drug can flow evenly to
the cervix and every corner of the vagina after being applied [10]. This study
aimed to investigate the clinical efficacy and safety of CO
This real-world study was conducted in the Center for Diagnosis and Treatment of
Cervical Diseases of Changzhou Maternity and Child Health Care Hospital,
Changzhou, China in 2021. After obtaining the approval of the ethics committee
(Changzhou Maternal and Child Health Care Hospital, NO. 2020), and obtaining
signed consent, 363 women with chronic cervicitis were enrolled. Among them, 208
patients were treated with CO
 Fig. 1.
Fig. 1.Study flowchart.
Inclusion criteria included normal cytology with the absence of human
papillomavirus (HPV), cleanliness of vaginal secretions is I–II, and consent to
undergo cervical CO
Two hundred and eight patients agreed to CO
One hundred and fifty-five cases only received cervical CO
The women received follow-up visits one, two, and three months post-treatment and their symptoms were reassessed using the four-stage scoring evaluation (Supplementary Table 1).
The scores for symptoms and signs increased as cervical inflammation worsened.
Efficacy was evaluated according to changes in patients’ symptoms and signs and
through laboratory examination. Therapeutic effect calculation method: Efficacy
index = (total score before treatment – total score after treatment)/(total score
before treatment)
Chi-square tests were used to assess the measured data. SPSS 23.0 (IBM Corp, Armonk, NY, USA) was utilized for all data analysis in this study, with a two-sided significance level of 0.05.
Of the 363 women enrolled in the study, there was no significant difference in
age between the study and control group (30.0
| Variables | Study group | Control group | p-value | |
| Age | 30.0 |
29.2 |
||
| Age of first sexual intercourse | 22.0 |
21.7 |
||
| Number of abortions | 0.4 |
0.3 |
||
| Severity of cervicitis | 0.949 | |||
| Mild | 45 | 34 | ||
| Moderate | 89 | 66 | ||
| Severe | 74 | 55 | ||
Significant difference within groups (p
A total of 208 patients were treated with CO
| Degree of inflammation | Case number (N) | Therapeutic effects | p | ||||
| Recovery (n (%)) | Significantly effective (n (%)) | Effective (n (%)) | Total effective rate (n (%)) | ||||
| Mild | 79 | 79 (100) | |||||
| Study group | 45 (100) | 0 | 0 | 100 | - | ||
| Control group | 34 (100) | 0 | 0 | 100 | |||
| Moderate | 155 | 108 (69.7) | |||||
| Study group | 68 (76.4) | 20 (22.5) | 1 (1.1) | 100 | 0.035 | ||
| Control group | 40 (63.6) | 23 (33.9) | 3 (2.5) | 100 | |||
| Severe | 129 | 46 (35.7) | |||||
| Study group | 31 (41.9) | 39 (52.7) | 4 (5.4) | 100 | 0.013 | ||
| Control group | 15 (27.3) | 28 (50.9) | 12 (21.8) | 100 | |||
| Total | 363 | 233 (64.2) | 110 (30.3) | 20 (5.5) | 100 | ||
| Study group | 208 | 144 (69.2) | 59 (28.4) | 5 (2.4) | 100 | 0.004 | |
| Control group | 155 | 89 (57.4) | 51 (32.9) | 15 (9.7) | 100 | ||
Significant difference within groups (p
Among the 363 patients, they were classified as mild, moderate or severe
cervicitis according to the degree of inflammation, and their cure rates
gradually decreased with the increasing amount of inflammation. Seventy nine
cases with mild inflammation had a 100% cure rate; 108 cases with moderate
inflammation had a 69.7% cure rate; and 46 cases with severe inflammation had a
35.7% cure rate. Among the patients with moderate and severe inflammation, the
treatment effect after 3 months was significantly better in the study group than
in the control group (p = 0.035, p = 0.013) (Table 2).
Significant difference was found between groups (p
Before treatment, there was severe columnar epithelium migration on the cervical
surface, covering two–thirds of the entire cervix (Fig. 2A and Fig. 3A). One
month after CO
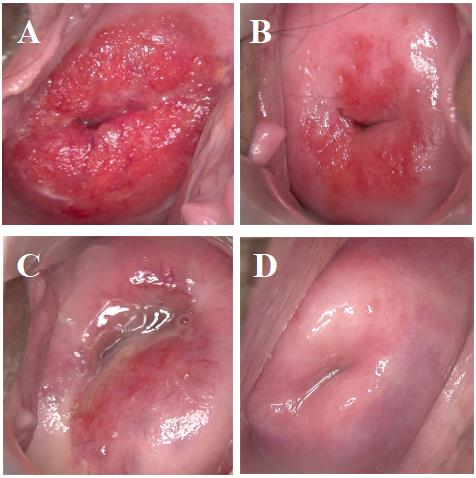 Fig. 2.
Fig. 2.Comparison before and after therapy in study group. (A) prior treatment. (B) 1 month after treatment. (C) 2 months after treatment. (D) 3 months after treatment.
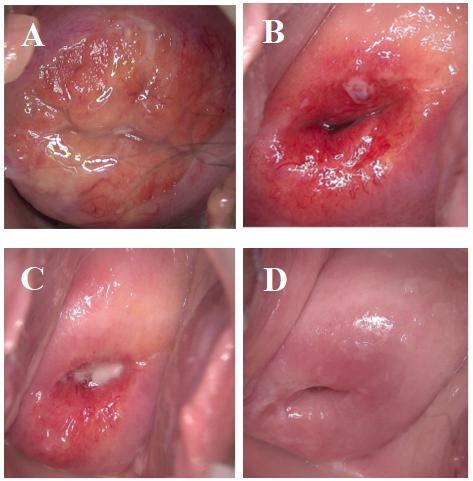 Fig. 3.
Fig. 3.Comparison before and after therapy in control group. (A) prior treatment. (B) 1 month after treatment. (C) 2 months after treatment. (D) 3 months after treatment.
With mild inflammation, 26.5% of patients in the control group had vaginal
discharge for more than 2 weeks, which was 3.96 times more than that in the study
group (6.7%) (p = 0.015); with moderate inflammation, 48.5% of
patients in the control group had vaginal discharge for more than 2 weeks, which
was 1.54 times more than that in the study group (31.5%) (p = 0.031);
with severe inflammation, the proportion of patients with vaginal discharge for
Among the patients in the study group, 12 cases had various amounts of vaginal bleeding 1–3 weeks after treatment, most of which were minimal and lasted less than 2 weeks, with 1 patient with severe cervicitis had increased postoperative bleeding (less than menstrual volume). In contrast, in the control group, 22 patients had vaginal bleeding; 1 with moderate inflammation and 2 with severe inflammation for more than 2 weeks; 3 patients with severe cervicitis had moderate vaginal bleeding (less than menstrual volume), which was 3.93 times more than the study group (Table 3).
| Degree of inflammation | Case number (N) | Vaginal discharge time (d) | Vaginal bleeding time (d) | Vaginal bleeding volume | p | |||||
| 1–15 | 16–20 | 7–15 | 16–20 | Mild | Moderate | Severe | ||||
| (n (%)) | (n (%)) | (n (%)) | (n (%)) | (n (%)) | (n (%)) | (n (%)) | ||||
| Mild | 79 | |||||||||
| Study group | 42 (93.3) | 3 (6.7) | 1 (2.2) | 0 | 1 (2.2) | 0 | 0 | 0.015 | ||
| Control group | 25 (73.5) | 9 (26.5) | 1 (2.9) | 0 | 1 (2.9) | 0 | 0 | |||
| Moderate | 155 | |||||||||
| Study group | 61 (68.5) | 28 (31.5) | 3 (3.4) | 0 | 3 (3.4) | 0 | 0 | 0.031 | ||
| Control group | 34 (51.5) | 32 (48.5) | 5 (7.6) | 1 (1.5) | 6 (9.1) | 0 | 0 | |||
| Severe | 129 | |||||||||
| Study group | 40 (54.1) | 34 (45.9) | 8 (10.8) | 0 | 7 (9.5) | 1 (1.4) | 0 | 0.009 | ||
| Control group | 17 (30.9) | 38 (69.1) | 13 (23.6) | 2 (3.6) | 12 (21.8) | 3 (5.5) | 0 | |||
| Total | 363 | |||||||||
| Study group | 143 (68.8) | 65 (31.2) | 12 (5.8) | 0 | 11 (5.3) | 1 (0.5) | 0 | 0.000 | ||
| Control group | 76 (49.0) | 79 (51.0) | 19 (12.3) | 3 (1.9) | 19 (12.3) | 3 (1.9) | 0 | |||
Significant difference within groups (p
Many women experience repeated or sustained symptoms of cervicitis. These symptoms can affect the quality of life and marital relationships and consequently, affect the health of the family atmosphere [11]. According to Sukru’s research, there was a statistically significant improvement in vaginal discharge, pelvic pain, and postcoital bleeding symptoms after the cryotherapy [12]. However, Shabnam’s findings highlighted the equal effect of tranexamic acid and cryotherapy for treating cervical ectropion [13, 14]. Chinese traditional medicine believes that cervicitis belongs to leukorrheal diseases, which is mainly due to unsteadiness of Ren and belt channel dysfunction by dampness and heat. The treatment should be based on clearing heat and dehumidification [15]. Kushen gel is a pure Chinese medicine preparation, and its main component is matrine, which has heat-clearing and detoxifying effects as well as antibacterial and insecticidal properties and removes saprophytic muscles [16, 17].
In this study, CO
The postoperative use of Kushen gel makes up for the vulnerability of wound
infection after simple CO
There are some limitations of this study. First, randomized clinical trials (RCTs) can draw more reliable conclusions, but we were unable to conduct RCT studies because of the inability to achieve double blinding. Second, we cannot avoid the selective bias due to the patient’s own condition.
The combined treatment of CO
The data that support the findings of this study are available from the corresponding author upon reasonable request.
LZ and YZ were involved in the analysis and interpretation of study data, drafting and critical revision of the manuscript; YL and WP were involved in the collection of research subject information; JL was involved in the analysis and interpretation of data; XX and MX were involved in the acquisition of data; SL was involved in the conception and interpretation of study data, drafting and critical revision of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The studies involving human participants were reviewed and approved by the research program of the Ethics Committee of Changzhou Maternal and Child Health Care Hospital, NO. 2020. The patients/participants provided their written informed consent to participate in this study. The authors received participants’ consent to publish photographs. This study was conducted in accordance with the Helsinki Declaration of 1964 and its later amendments.
The authors thank Changzhou Maternity and Child Health Care Hospital for their support for this study, and we thank the participants of the study.
Scientific Research Project of Maternal and Child Health of Jiangsu Province (F202166). Key talents in maternal and child health care in Jiangsu Province and Leading Talent of Changzhou “The 14th Five-year Plan” High-Level Health Talents Training Project (2022CZLJ030). 2023 Changzhou Policy Guidance Project (International Science and Technology Cooperation) (CZ20230020), Clinical Research Project of Changzhou Medical Center (CMCC202314), and 2023 Jiangsu Province Traditional Chinese Medicine Science and Technology Development Plan (MS2023089).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



