1 Radiation Oncology, Ankara Bilkent City Hospital, 06800 Ankara, Turkey
2 Radiation Oncology, Ankara Yıldırım Beyazıt University, 06145 Ankara, Turkey
3 Gynecology and Obstetrics, Ankara Medicana International Hospital, 06510 Ankara, Turkey
Abstract
Background: Although the radiotherapy (RT) effect is reversible in some tissues, it is progressive and permanent for the ovary. Ovary radiation exposure during breast cancer radiotherapy (BCR) may cause concern in patients and obstetricians. In this study, we evaluated ovarian radiation doses in patients who received adjuvant radiotherapy for breast cancer prospectively. Methods: Ovary doses were calculated in patients diagnosed with breast cancer (BC) and receiving RT at the Radiation Oncology clinic of Ankara Bilkent City Hospital. Helical intensity modulated radiotherapy (IMRT) planning and three-dimensional (3D) planning were performed for each patient. Results: The data of 8 patients who received BCR at 8 August 2023 and 19 September 2023 were evaluated prospectively. The median age of the patients was 57 (range 39–65), 4 (50%) had breast conserving surgery (BCS), and 4 (50%) patients underwent modified radical mastectomy (MRM). Four (50%) patients were left sided, while four (50%) patients were right sided. According to our results, the ovarian doses in adjuvant radiotherapy of breast cancer patients who received RT with the 3D technique were zero. In helical IMRT plans, the ipsilateral ovary dose was median 12 (range 10–30) centi Gray (cGy); the contralateral ovary dose was median 8 (range 5–13) cGy. In patients with MRM, ipsilateral ovary dose in helical IMRT plans was median 10 (range 10–13); in patients with BCS, it was median 15 (12–30) cGy. Conclusions: This is the first study to examine ovarian doses in breast cancer radiotherapy on patients. In our study, in which we evaluated eight patients with 2 different RT techniques, the dose values of ovaries in breast cancer radiotherapy were found to be negligible and no measurable dose was noted in 3DRT technique.
Keywords
- radiotherapy
- breast cancer
- fertility
- ovary dose
Every year, 2.3 million women are diagnosed with breast cancer (BC) worldwide
and it has now become a public health problem [1]. Survival rates have increased
with effective treatments for breast cancer, and today long-term side effects of
treatments such as fertility have become more important. Radiotherapy (RT) is an
important component of breast cancer treatment (BCT) and the relationship between
RT and ovary is a well-studied topic, especially for patients receiving pelvic
RT. RT-related ovary damage is often progressive and permanent [2]. There are
many studies in the literature based on animal experiments on a safe radiation
threshold value for the ovary, however additional human studies are needed.
Considering these studies, the dose that kills 50% of ovarian cells is accepted
as
The risk of radiation-induced infertility is related to the location of the radiation field, RT technique, RT dose, presence of concurrent systemic therapy, and the age of the patient. Considering the typical radiation fields in breast cancer radiotherapy (BCR), it is predicted that the ovary and uterus do not receive significant doses of radiation [3, 4, 5]. BC patients have two major surgical options; modified radical mastectomy (MRM) and breast conserving surgery (BCS). In patients undergoing MRM, the entire breast is removed, and adjuvant RT is usually not required for early-stage BC. BCS is the removal of only the localization of the breast mass, cosmetic results are better than MRM and adjuvant RT is usually indicated in patients undergoing BCS [6]. However, according to our clinical observations, some early-stage breast cancer patients who desire fertility MRM, instead of BCS to avoid adjuvant RT. The percentage of patients who have preferred MRM over BCS due to RT concerns is not clearly known [7]. Although as radiation oncologists, we think that it is unnecessary for patients with early-stage breast cancer to consider MRM, a more aggressive surgery, instead of BCS to avoid RT, there is no study in the literature evaluating the ovarian dose of patients in breast cancer radiotherapy. In this study, we aimed to analyze the dose received by the ovaries in breast cancer radiotherapy.
Patients diagnosed with breast cancer and treated with adjuvant RT at Ankara Bilkent City Hospital Radiation Oncology Clinic were evaluated, prospectively. Patients received standard treatment and there were no study-specific changes in treatment plans. Data from patient files, electronic system information, patient interview information, radiotherapy dose volume histograms were used to obtain data. Patients’ pathologic diagnoses, chemotherapy data, surgery details, radiotherapy information, and ovarian dose values were noted.
Adult female patients with breast cancer who underwent adjuvant radiotherapy for curative purposes were included in the study. In breast cancer radiotherapy, a ‘simulation computerized tomography (CT)’ image is obtained for RT planning and the targeted risk areas are contoured on these CT images. Patients who consented to a wider CT imaging were included in the study. Informed consent form was obtained from each patient. Patients who did not volunteer for the study, male breast cancer patients and who underwent RT for palliative purposes were excluded from the study.
Patients were laid in the supine position on a simulation bed called a ‘breast board’. The breast board was fixed at a patient-specific inclination between 7–15 degrees to optimize patients’ chest walls/chests for the RT plan. Patients were positioned with the ipsilateral shoulder in abduction and the head slightly rotated to the contralateral side. The operation scars and drainage areas were marked with lead wire. CT simulation limits are as follows for standard breast simulation CT scans; upper limit is C3 vertebra and lower limit is 5 cm below the lower border of the contralateral breast. In our study, the CT field was expanded inferiorly. The CT scan area of the study was extended until the femoral heads were seen. The CT scan images were obtained at 2.5-mm slice thickness without contrast media.
Radiation oncologists identify/contour target and organ at risk areas on simulation CT. Standard target volumes were defined in accordance with guidelines [8, 9]. Organ at risk (OAR) refers to healthy structures that are close to the target volume. Because of this proximity, OARs are exposed to radiation. The dose limits for each OAR are defined by guidelines [10, 11] and radiation doses to OAR are controlled to avoid side effects. Radiation oncologists check both the target volume and OAR doses after RT planning by medical physicists and initiate the treatment process if all are optimal.
The following structures are contoured in breast cancer as organ at risk: lungs, spinal cord, esophagus, heart, left anterior descending artery, thyroids, plexus brachialis, humeral head. In this study, unlike the standard, the patients’ ovaries were also contoured. In our study, patients received a total dose of 50 Gy with a daily fraction dose of 2 Gy in 5 weeks.
In recent years, there have been significant improvements in breast cancer radiotherapy techniques. This change is due to developing technologies and the production of more qualified RT-devices. With modern techniques especially with intensity modulated radiotherapy (IMRT), better target volume coverage is achieved, but an increase is observed in low dose areas [12].
In this study, two different techniques were planned for each patient. Helical IMRT technique plans were obtained using Radixact Accuracy Precision version 1.1.1.0 device (Accuracy, Madison, WI, USA). Three-dimensional (3D) technique plans were obtained using Varian Eclipse Version 15.6 device (Varian, Palo Alto, CA, USA). In our study, in addition to measuring ovarian dose values, we will also determine which technique provides a lower ovarian dose. The contours of the patients’ ovaries were contoured on simulated CT, as OAR. In both plans, the doses received by the ovaries were calculated.
The data of 8 patients who received BCR at 08.08.2023 and 19.09.2023 were evaluated prospectively. The median age of the patients was 57 (range 39–65), 4 (50%) were BCS, and 4 (50%) patients underwent MRM. Four (50%) patients were left sided, while four (50%) patients were right sided.
According to the 3D planning evaluation of the patients, the Varian Eclipse Vers 15.6 planning algorithm could not detect measurable doses in both the ipsilateral and contralateral ovaries. According to our results, the ovarian doses in adjuvant radiotherapy of breast cancer patients who received RT with the 3D technique were zero (Fig. 1A). In the helical IMRT planning system, ovarian doses were very low and these doses were expressed as cGy, which is one-hundredth of a Gy (Fig. 1B). In helical IMRT plans, the ipsilateral ovary dose was median 12 (range 10–30) cGy; the contralateral ovary dose was median 8 (range 5–13) cGy (Table 1). In patients with MRM, ipsilateral ovary dose in helical IMRT plans was median 10 (range 10–13); in patients with BCS, it was median 15 (12–30) cGy. In patients with MRM, contralateral ovary doses in helical IMRT plans was median 6 (range 5–8) cGy; in patients with BCS, it was median 8 (range 7–13) cGy (Table 2, Fig. 2).
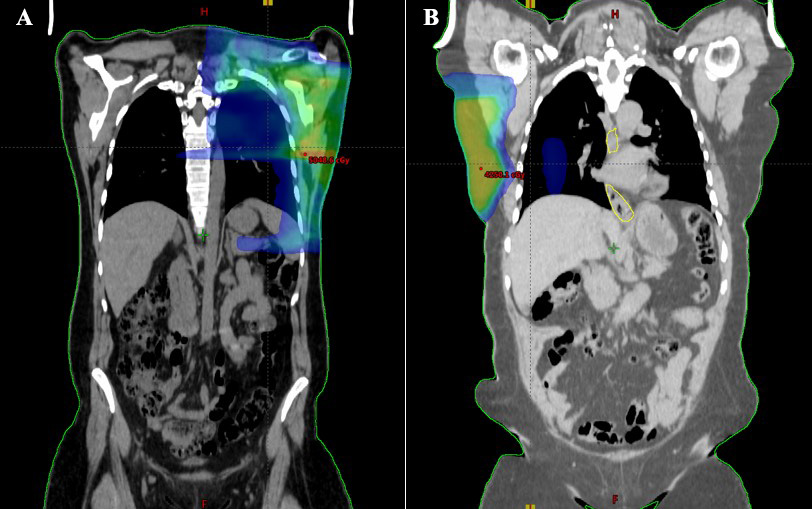 Fig. 1.
Fig. 1.Computerized tomography (CT) field size image of patients simulating radiotherapy. The ovarian doses in adjuvant radiotherapy of breast cancer patients who received radiotherapy (RT) with the three-dimensional (3D) technique were zero (A). In the helical intensity modulated radiotherapy (IMRT) planning system, ovarian doses were very low (B).
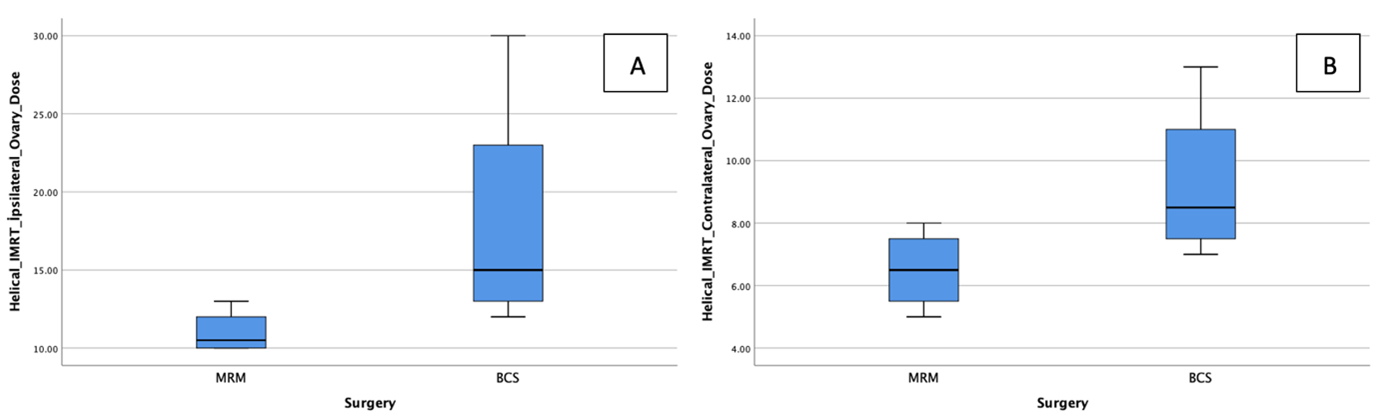 Fig. 2.
Fig. 2.Relationship between surgery and ovarian dose. Evaluation of ipsilateral (A) and contralateral ovarian doses according to surgery (B). MRM, modified radical mastectomy; BCS, breast-conserving surgery.
| Patient | Demographics | Clinical information | Radiotherapy | Surgery and laterality | Helical IMRT ipsilateral ovary dose (cGy) | Helical IMRT contralateral ovary dose (cGy) |
| 1 | 39 yo, H: 166 cm, W: 60 kg, BMI: 21.8 | cT2N1M0, Stage 2B, IDC | 50 Gy | MRM | 10 | 7 |
| Left | ||||||
| 2 | 39 yo, H: 166 cm, W: 77 kg, BMI: 27.9 | cT2N1M0, Stage 2B, IDC | 50 Gy | MRM | 11 | 6 |
| Left | ||||||
| 3 | 77 yo, H: 150 cm, W: 71 kg, BMI: 31.6 | cT2N1M0, Stage 2B, IDC | 50 Gy | MRM | 10 | 5 |
| Left | ||||||
| 4 | 50 yo, H: 160 cm, W: 82 kg, BMI: 32 | cT3N1M0, Stage 3A, IDC | 50 Gy | MRM | 13 | 8 |
| Right | ||||||
| 5 | 60 yo, H: 160 cm, W: 78 kg, BMI: 30.5 | cT1N0M0, Stage 1, IDC | 40 Gy | BCS Right | 30 | 13 |
| + 10 Gy | ||||||
| boost | ||||||
| 6 | 65 yo, H: 165 cm, W: 78 kg, BMI: 28.7 | pT1cN0M0, Stage 1, IDC | 40 Gy | BCS | 12 | 9 |
| Right | ||||||
| 7 | 80 yo, H: 155 cm, W: 68 kg, BMI: 28.3 | cT2N0M0, Stage 2A, IDC | + 10 Gy | BCS | 14 | 8 |
| boost | Left | |||||
| 8 | 54 yo, H: 160 cm, W: 115 kg, BMI: 44.9 | pT1cN0M0, Stage 1, IDC | 40 Gy | BCS | 16 | 7 |
| Right |
IMRT, intensity modulated radiotherapy; Gy, Gray; cGy, centi Gray; MRM, modified radical mastectomy; BCS, breast-conserving surgery; H, height; W, weight; BMI, body mass index; IDC, invasive ductal carcinoma.
| Ipsilateral Ovary dose (cGy) | Contralateral Ovary dose (cGy) | |
| MRM | 10 (range 10–13) | 6 (range 5–8) |
| BCS | 15 (range 12–30) | 8 (range 7–13) |
cGy, centi Gray; MRM, modified radical mastectomy; BCS, breast-conserving surgery.
In current study, the ovaries doses of breast cancer patients were evaluated, as predicted, it was observed that they received a very low dose in the IMRT planning system, and no dose value could be detected in the 3DRT planning system. No dramatic difference was detected in MRM and BCS patient arms in terms of ovary doses, which are two important techniques in breast cancer surgery. BCS is preferred over MRM, especially in early-stage patients, and adjuvant RT is routinely applied to patients after BCS. Patients considering fertility may prefer MRM over BCS, which is a more aggressive surgery and cosmetically less acceptable, to avoid RT. However, according to the data of our study, ovary doses are quite low in both BCS and MRM patients. In the patient with the highest measured value of 30 cGy, the field size was larger and more inferiorly located due to the high breast volume. Moreover, the contralateral ovarian dose for the same patient was 13 cGy.
Each technique has its own advantages and disadvantages, and the choice of RT technique is patient-specific. IMRT provides better dose distribution in the target volume. In the 3DRT technique, body volumes receiving low doses are generally lower [12]. In general, scattered doses are higher in IMRT techniques than in 3DRT techniques [13]. In current study, higher doses were measured with helical IMRT than 3DRT. We believe that this difference is due to the RT technique. Ovarian doses in the IMRT technique may be due to scattered doses, which are inherently high in the IMRT technique. However, in all our cases, ovary doses were below the nearly all ovarian threshold values in the literature.
Women have limited ovarian reserve, which is maximized during intrauterine life.
After childbirth, it decreases with age and ovarian function ceases with
menopause [14]. This usual decline in ovarian reserves can become dramatic with
RT. With RT atrophy of follicles in the ovary occurs, the number of oocytes and
production of ovarian hormones decreases. As a result, these can lead to early
menopause and infertility [2, 4]. Ovaries are radiosensitive [15, 16], the LD50
dose (dose to destroy 50% of oocytes) is estimated to be
The ovarian threshold doses for premature ovarian failure (POF) and infertility are very high doses of such as 2000–3500 cGy, which are far from the doses received by the ovary in breast cancer radiotherapy [17, 23]. Another important question is the risk of fetal anomalies in pregnancy after RT. In related animal experiments, an increase in congenital anomalies was observed in pregnancies following 100–500 cGy radiation exposure in the ovary. However, these animal data do not correlate with human studies [24, 25]. Breast cancer patients are followed for at least five years after the end of their treatment. Although it varies depending on the stage of the disease and the patient’s complaints, CT is frequently used in patient follow-up. Patients’ ovaries are already exposed to low doses of radiation during both diagnosis and follow-up imaging processes. In our opinion, the radiation doses received by the ovaries during breast cancer radiotherapy are not high.
Since it is estimated by radiation oncologists that ovarian doses will not be high, no studies on ovarian doses in breast cancer radiotherapy have been conducted so far. However, with prolonged survival [26] and more patients desiring pregnancy after cancer treatment, this issue has become a current question for both patients and clinicians other than radiation oncologists [4]. Ovarian radiation doses have mostly been studied in pelvic irradiation and total body irradiation [27]. There are no studies directly addressing ovarian dose in breast cancer. Only, there are data on patients who received RT for breast cancer accidentally during pregnancy. Ngu et al. [28] treated a third trimester breast cancer patient using a 9 mm lead shield over the abdomen. The estimated fetal dose was 14–18 cGy for a total dose of 5000 cGy delivered to the breast [28]. A total dose of 46 Gy was given in 20 fractions over a 4-week period in Antypas et al. case [29]. The patient was aware of her pregnancy in the 2nd week of radiotherapy. Calculated unprotected fetal dose was 3.9 cGy [29]. In both cases healthy babies were born [28, 29]. Although not including ovary doses, there are also studies testing fetal doses on artificial systems called Phantoms. Van der Giessen performed an evaluation on a phantom and calculated fetal doses between 3–201 cGy according to gestational week and RT preference (Co-60 vs. 6–25 MV) [30]. In the Mazonakis et al. [31] phantom model, the fetal dose was measured as 0.021–0.076 Gy, 0.022–0.246 Gy and 0.022–0.586 Gy in the first, second and third trimesters, respectively. Although these studies give an idea about ovarian dose, ovarian dose was not directly measured, and this is an important gap in the literature. This is the first study on this topic and ipsilateral ovaries received 10–30 cGy and contralateral ovaries received 5–13 cGy in IMRT technique. In our eight-patient dose evaluation, no radiation exposure that could cause infertility or ovarian failure was observed in our patients.
The study has some limitations. First, our premenopausal patients generally did not volunteer for the study. Since the majority of the patients who volunteered for our study were elderly, anti mullerian hormone values before and after RT were not measured, which was planned at the beginning of the study. Secondly, only dosimetric evaluation was performed. Six of our patients were elderly and postmenopausal, while the other 2 premenopausal patients did not want pregnancy. Therefore, long-term follow-up and clinical evaluation could not be performed. Finally, dosimetric evaluation was performed on only 8 patients. However, we believe that our data provide valuable information on ovarian doses in breast cancer radiotherapy.
This is the first study to examine ovarian doses in breast cancer radiotherapy on patients. In our study, in which we evaluated eight patients with 2 different RT techniques, the dose values of ovaries in breast cancer radiotherapy were found to be negligible and no measurable dose was found in 3DRT technique.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
İPA designed the research study. İPA, GAİ, HB, MSE literature review and drafted the manuscript. İPA and HGK analyzed the data interpreted data. İPA and HGK did the contouring. EA made radiotherapy physical plans. GAİ and YT checked the contouring and plans. İPA, MSE and GAİ wrote the article. YT and MSE supervised the whole study. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
The study was conducted in accordance with the Helsinki Declaration. The ethical suitability of the study was approved by Ankara Bilkent City Hospital Ethics Committee No. 1 with the number E1-23- 3778 at 12/07/2023. Informed consent form was obtained from each patient.
We would like to thank all our patients who volunteered in our study. We would also like to thank everyone who contributed to the writing of this article.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


