1 Obstetrics and Gynecology Department, Faculty of Medicine, Kasr EL-Ainy Hospital, Cairo University, 11562 Cairo, Egypt
2 Obstetrics and Gynecology Department, Faculty of Medicine, Tanta University, 31527 Tanta, Egypt
3 Obstetrics and Gynecology Department, Mataria Teaching Hospital, Ministry of Health, 35789 Cairo, Egypt
Abstract
Background: Cesarean section (CS) is one of the most common surgical procedures worldwide, often associated with postoperative pain. Effective pain management is crucial for maternal recovery and satisfaction. Transversus abdominis plane (TAP) block and local anesthetic wound infiltration (LWI) are commonly used techniques for post-cesarean analgesia. While both methods have shown efficacy, their comparative effectiveness remains debated. This study aims to compare the efficacy of TAP block versus LWI for post-cesarean analgesia, considering pain scores and opioid consumption, to provide insights into optimizing pain management strategies for women undergoing CS. Methods: A randomized comparative double-blinded clinical trial conducted on 200 randomly selected patients aged 20 to 40 years in the university hospital from January 2021 to January 2022 after receiving approval from the ethical research committee. Patients were divided into two groups. All patients were instructed on how to use the visual analog scale (VAS), and the procedure of ultrasound-guided TAP block or local anesthetic wound infiltration was explained to them. Main Outcome Measure: The level of pain during movement (hip flexion and coughing) at 2, 4, 6, 12, and 24 hours following surgery. Pain intensity was measured using a VAS score ranging from 0 to 10 (0 representing no pain, and 10 representing the worst conceivable discomfort). Results: Patients who underwent TAP block showed significantly lower pain scores for 12–16 hours after surgery and a reduced total requirement for analgesia in the first 24 hours after surgery compared to patients who received wound infiltration. Conclusions: Our study demonstrated that bilateral TAP block was more effective in reducing postoperative pain and total 24-hour postoperative opioid and analgesic consumption after cesarean section under spinal anesthesia compared to local anesthetic wound infiltration. Clinical Trial Registration: The trial was registered in ClinicalTrials.gov registry with clinical trial: https://clinicaltrials.gov/ct2/show/NCT05447455.
Keywords
- transversus abdominis plane block
- local anesthetic wound infiltration
- cesarean section
- analgesia
The prevalence of cesarean sections has recently increased due to recognizing their correlation with improved fetal prognosis [1]. These procedures account for approximately 15% to 20% of global deliveries, with significantly higher rates in underdeveloped countries reaching about 40% [1]. Cesarean sections are commonly conducted under spinal or general anesthesia, with current research indicating no significant advantages of choosing spinal anesthesia (SA) over general anesthesia (GA). One of the avoidable consequences of surgery is postoperative discomfort, arising from the body’s natural response to tissue injury at the surgical site [2]. A 2003 survey in the USA revealed a 70% likelihood of patients experiencing severe pain after surgery [3]. Considering that postoperative pain management significantly influences patient recovery, hospital stay duration, stress response, pneumonia, thrombosis, chronic pain, slow wound healing, and depression [1], its importance cannot be overstated. Multimodal analgesia, involving the simultaneous administration of multiple drugs to alleviate pain with fewer side effects, is gaining popularity [4]. Opioids, administered intravenously (IV) or intrathecally (IT) before surgery, are the primary treatments for postoperative pain following cesarean sections [5]. Studies have shown that combining fentanyl or 100–200 µg morphine with bupivacaine enhances the efficacy and duration of spinal anesthesia [6]. However, the use of opioids is associated with postoperative side effects such as nausea, vomiting, ileus, pruritis, respiratory depression, and urinary retention. The effectiveness of non-opioid systemic analgesics in pain management is relatively lower [1]. Alternative methods like epidural analgesia require continuous monitoring [1]. Recently developed preventive analgesic techniques have the potential to reduce postoperative pain and medication usage [7]. Options include transversus abdominis plane (TAP) blocks, intravenous analgesics, and local anesthetic infusion [8]. TAP blocks involve administering a local anesthetic to block the T6-L1 nerve roots, effectively reducing pain during lower abdominal surgeries. These can be performed using anatomical landmarks or ultrasonography [9]. Local anesthetic wound infiltration is gaining attraction in clinical practice due to its simplicity, safety, and affordability [10]. Considering these factors, we formulated a hypothesis that the TAP block, within a 24-hour period, could potentially reduce surgical pain and overall opioid consumption.
Between January 2021 and January 2022, a total of 200 women undergoing cesarean
sections under spinal anesthesia participated in a prospective, double-blinded,
randomized comparative clinical trial at Kasr Al-Ainy Hospital, Cairo University,
Cairo, Egypt. The study protocol received approval from the Ethical Research
Committee under REC Number 301-2020. Before participation, written informed
consent was obtained after thoroughly informing all women about the study and its
potential implications. A thorough screening process was conducted, evaluating
245 women to ensure they met the study’s inclusion criteria. The eligibility
criteria were as follows: (1) women undergoing cesarean delivery under spinal
anesthesia, (2) aged between 20 and 40, and (3) pregnancy not less than 37 weeks.
Disqualification criteria included: (1) body mass index (BMI) exceeding 35
kg/m
Using computer-generated randomization, the study participants were evenly divided into two groups: the TAP group and the local anesthetic wound infiltration (LAWI) group. A researcher (one of the authors) assessed patient eligibility, obtained written consent, and documented baseline data before delivery. Allocation to the groups was concealed in serially numbered sealed envelopes, which were opened by the lead investigator after administering spinal anesthesia and establishing the upper sensory level of T6 or higher. Both the participants and outcome assessors were blinded to the study group. All patients underwent uncomplicated lower-segment cesarean sections performed by an experienced obstetrician.
Spinal anesthesia, conducted by a resident in anesthesiology not involved in the trial, involved administering 12.5 mg of hyperbaric bupivacaine (2.5 mL, 0.5%) and 15 mcg of fentanyl intrathecally. Surgery commenced once the pinprick test indicated an upper sensory level of T6 or higher. If the sensory level dropped below T6 within 20 minutes, the spinal anesthesia was deemed unsuccessful, and the patient was excluded from the study. A TAP block procedure was performed by an anesthesiologist with at least three years of experience in ultrasound-guided TAP blocks. After the surgery, a bilateral ultrasound-guided TAP block was performed using a 12–4-MHz linear array transducer (ClearVue 350; Philips, Bothell, WA, USA). The transducer was positioned transversely between the iliac crest and costal margin in the anterior axillary line and then moved medial-lateral to visualize the external oblique, internal oblique, and transversus abdominis muscles. The most lateral (posterior) position that provided a satisfactory ultrasound image was selected. A 22-gauge spinal needle was inserted in-plane from medial to lateral to the ultrasound probe, and 20 mL of bupivacaine 0.25% was injected under visualization into the plane between the transversus abdominis muscle and the fascia deep to the internal oblique muscle on each side. And in the infiltration group, involved injecting local anesthetic directly into the surgical wound site to provide localized analgesia. After completion of the cesarean section, the surgeon infiltrates the wound edges with the local anesthetic solution, 40 mL of bupivacaine 0.25% (20 mL in each of the upper and lower sides). The infiltration is usually performed using a small-gauge needle to ensure even distribution of the medication along the incision line.
Postoperatively, a blinded researcher assessed the patients at 2, 4, 6, 12, and
24 hours after surgery for outcomes, including opioid consumption, time to first
opioid dose, pain scores, and side effects. Standard monitors were used,
postoperative medications following cesarean section (CS) according to hospital
protocol were given as antibiotic to reduce risk of postoperative infection and
antiemetics was administered to prevent or reduce postoperative nausea and
vomiting (PONV), a common side effect of anesthesia and surgery. All women also
received postoperative standard analgesia according to the hospital protocol
(ketorolac and paracetamol), to alleviate pain and discomfort following cesarean
section surgery after 24 hours if patient felt pain. Additionally, 25–100 mg of
meperidine was given intravenously as needed (when the patient complained of pain
or when visual analog scale (VAS) score
The sample size determination was based on the mean time to the first
postoperative analgesic request following cesarean sections performed under
spinal anesthesia with either a TAP block or local anesthetic wound infiltration.
According to a prior study [11], the mean time to the first postoperative
analgesic request following cesarean sections under spinal anesthesia with a TAP
block was reported as 6.11
For our investigation, the total sample size was divided into two equal groups, each comprising 100 patients: TAP block group and LAWI group. Sample size calculation was performed using MedCalc® Statistical Software version 19.5.3 (MedCalc Software Ltd, Ostend, Belgium; https://www.medcalc.org; 2020). Data analysis was conducted using SPSS version 26.0 (IBM Corp., Armonk, NY, USA).
To assess the normality of continuous data, the Kolmogorov-Smirnov test was employed. Descriptive statistics such as mean, standard deviation, median (interquartile range (IQR)), and number (percentage) were used for normally distributed, non-normally distributed, and categorical data, respectively. Comparisons between the two groups were carried out using the independent t-test for normally distributed outcome data, specifically the cumulative meperidine ingestion at 24 hours. Non-normally distributed outcome data, such as pain scores during movement at various time points (2, 4, 6, 12, and 24 hours), were analyzed using the Mann-Whitney U test. The Fisher exact test was employed to compare categorical outcome data, including the occurrence of nausea/vomiting, pruritus, and the need for additional opioid doses. Statistical significance was defined as a two-tailed p value of 0.05.
Initially, 245 women were assessed for potential inclusion in the study. However, 45 of these women were deemed ineligible due to meeting one or more exclusion criteria or declining participation. This resulted in a final cohort of 200 women who were randomly allocated to two distinct groups: the TAP group (n = 100) and the LAWI group (n = 100), as illustrated in Fig. 1. Importantly, no significant discrepancies in initial demographic characteristics were observed between these two groups, as summarized in Table 1. Upon analysis, it was discovered that patients in the LAWI group reported higher levels of pain during movement compared to those in the TAP group, who experienced comparatively lower pain levels. This pattern was consistently observed at each follow-up interval, with statistically significant differences evident between the two groups, as presented in Table 2 and graphically illustrated in Fig. 2. The data presented in Table 3 indicate that the LAWI group required a higher number of opioid doses compared to the TAP group, with statistically significant differences observed between the groups. Additionally, the time taken for the administration of the first opioid dose was notably shorter in the LAWI group compared to the TAP group, displaying statistically significant differences, as shown in Table 4. Evaluation of the cumulative 24-hour opioid dose, as per Table 5, revealed that the TAP group exhibited the lowest dose, whereas the LAWI group had the highest dose. These disparities between the groups were found to be statistically significant. Furthermore, an assessment of side effects indicated that the LAWI group reported a higher incidence of nausea and vomiting compared to the TAP group, which experienced a lesser occurrence of these symptoms. Conversely, no significant distinctions were observed between the groups concerning pruritus, with the differences between the two groups being statistically significant, as outlined in Table 6.
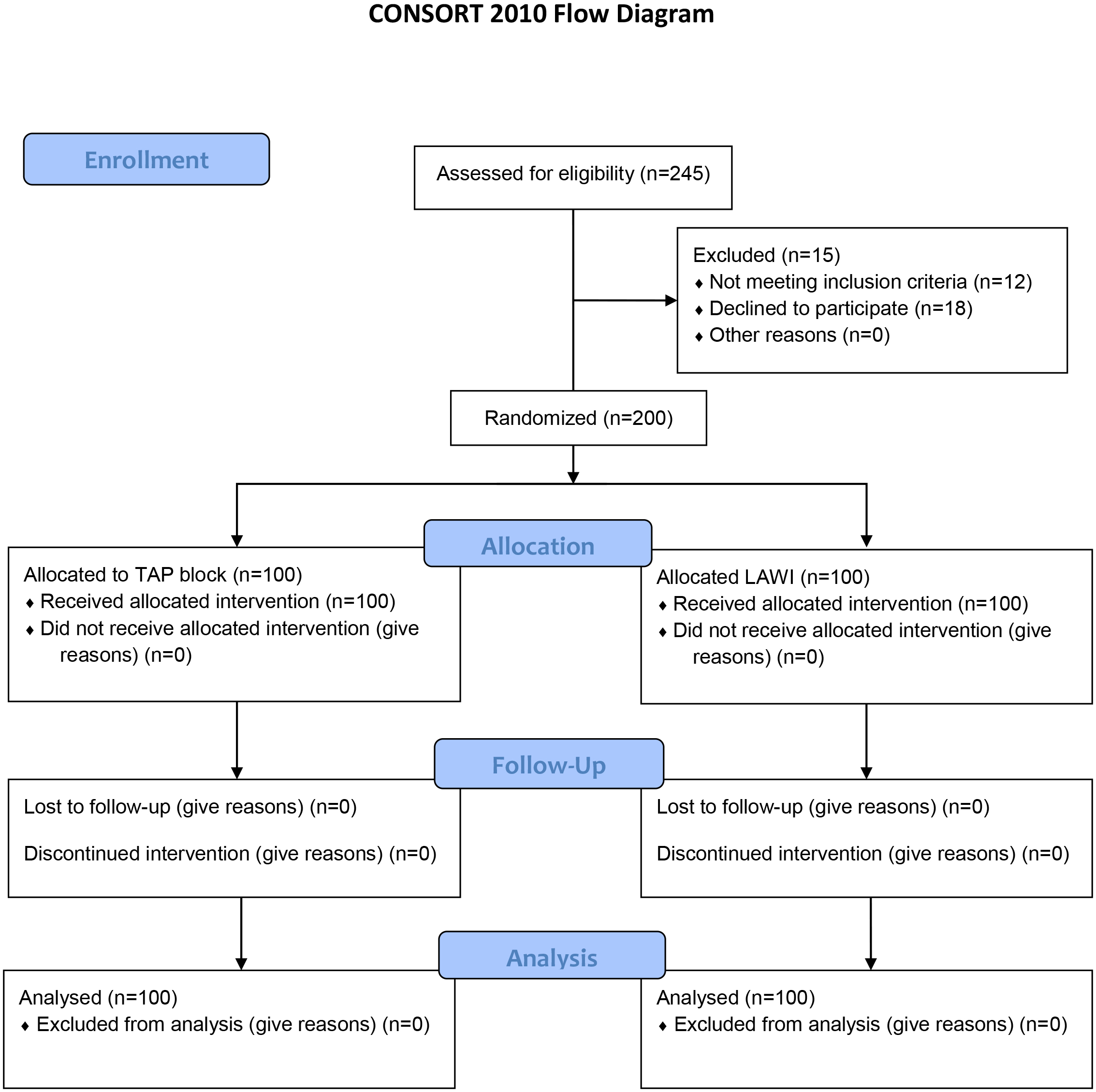 Fig. 1.
Fig. 1.Flow chart of the studied cases. TAP, transversus abdominis plane; LAWI, local anesthetic wound infiltration.
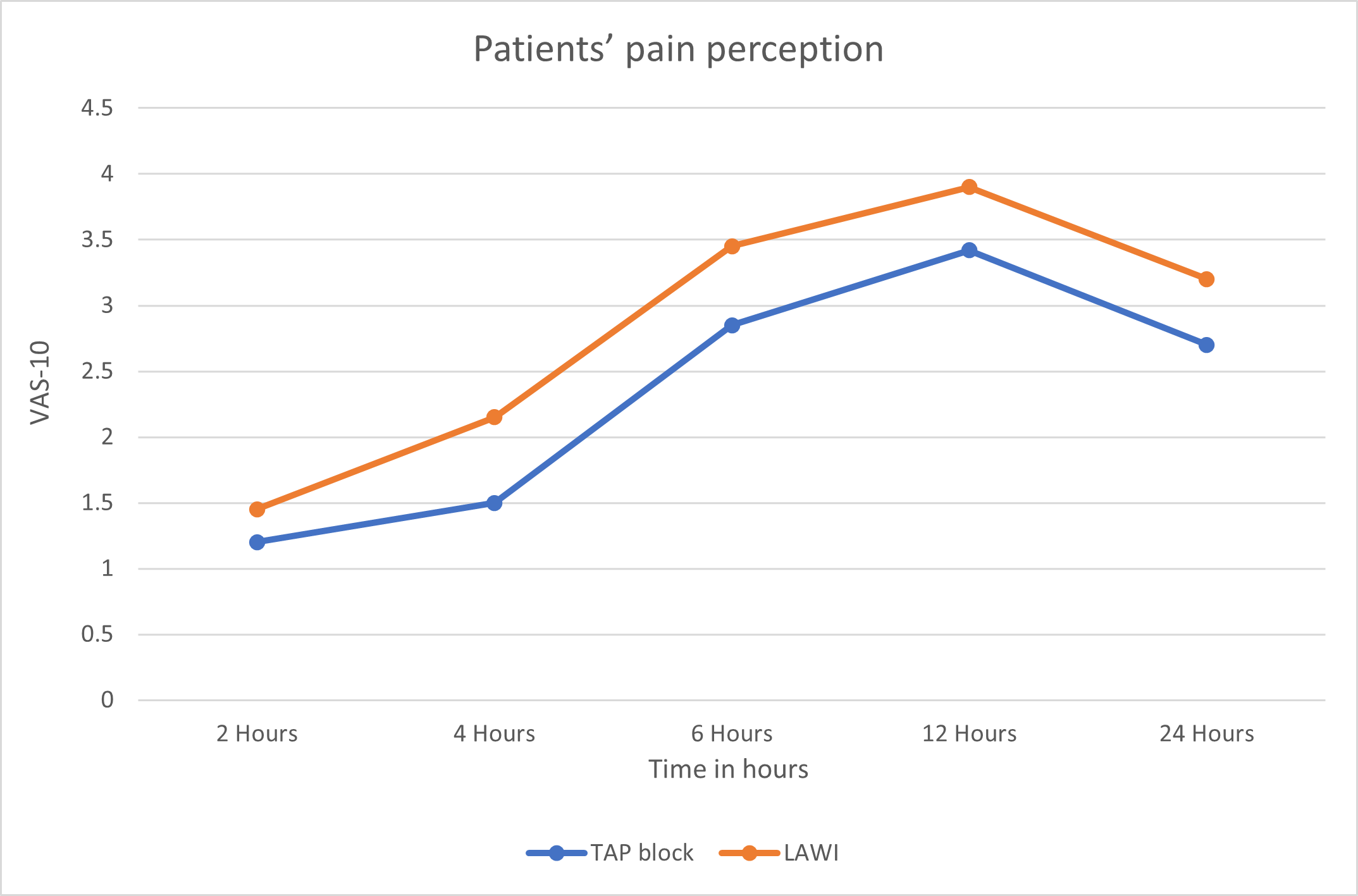 Fig. 2.
Fig. 2.Patients’ pain perception (VAS-10) among the studied groups. Each data point represents the mean of pain score reported by individual patients at each postoperative time point. VAS, visual analogue scale.
| Variables | Measures | TAP (N = 100) | Infiltration (N = 100) | p-value |
| Age (years) | Mean |
28.95 |
29 |
^0.94 |
| Range | 20–40 | 20–40 | ||
| BMI (kg/m |
Mean |
28.65 |
28.95 |
^0.38 |
| Range | 23.15–34.20 | 23.18–34.60 | ||
| Parity | Nulli | 38 (38%) | 34 (34%) | #0.66 |
| Multi | 62 (62%) | 66 (66%) | ||
| Gestational age (weeks) | Mean |
38.60 |
38.70 |
^0.41 |
| Range | 37–40 | 37–41 | ||
| Operation duration (minutes) | Mean |
56.75 |
57.25 |
^0.63 |
| Range | 41–77 | 40–72 |
^Independent t-test. #Fisher exact test.
BMI, body mass index; SD, standard deviation.
| Time | Measures | TAP (N = 100) | Infiltration (N = 100) | ^p-value |
| Hour-2 | Mean |
1.20 |
1.45 |
|
| Range | 0–2 | 0–2 | ||
| Hour-4 | Mean |
1.50 |
2.15 |
|
| Range | 0–2 | 0–3 | ||
| Hour-6 | Mean |
2.85 |
3.45 |
|
| Range | 1–3 | 2–4 | ||
| Hour-12 | Mean |
3.42 |
3.90 |
|
| Range | –4 | 3–6 | ||
| Hour-24 | Mean |
2.70 |
3.20 |
|
| Range | 1–4 | 2–5 |
^Mann Whitney U test. *Significant.
| Findings | TAP (N = 100) | Infiltration (N = 100) | #p-value |
| Needed | 9 (9%) | 42 (42%) | |
| Not needed | 91 (91%) | 58 (58%) |
#Fisher exact test. *Significant.
| Measures | TAP (N = 9) | Infiltration (N = 42) | ^p-value |
| Mean |
14.85 |
12.21 |
|
| Range | 10–24 | 5–24 |
^Independent t-test. *Significant.
| Measures | TAP (N = 9) | Infiltration (N = 42) | ^p-value |
| Mean |
44.44 |
62.62 |
|
| Range | 25–50 | 25–100 |
^Independent t-test. *Significant.
| Side effects | TAP (N = 100) | Infiltration (N = 100) | p-value |
| Nausea | 4 (4%) | 13 (13%) | § |
| Vomiting | 1 (1%) | 9 (9%) | § |
| Pruritus | 0 (0%) | 1 (1%) | §1.00 |
§Fisher’s Exact test. *Significant.
This randomized, double-blinded trial aimed to compare the efficacy of bilateral ultrasound-guided TAP block with single-shot local anesthetic wound infiltration for analgesia following cesarean section under spinal anesthesia. The study revealed notable differences in total opioid consumption at the 24-hour mark, time taken for the initial postoperative opioid dose, requirement for opioid doses, and patients’ self-assessment of pain between those receiving the two procedures. The LAWI group experienced a higher frequency of side effects, including nausea, vomiting, and pruritus, compared to the TAP group. However, no statistically significant variations in pruritus were found between the studied groups. Our findings align with prior prospective cohort studies on 60 and 62 patients, which reported reduced opioid intake in the TAP block group compared to wound infiltration within the first 24 hours post-cesarean section [12, 13]. Similarly, our results align with a randomized controlled experiment where the TAP group exhibited lower total diclofenac consumption than the local infiltration group [14]. Our research also finds support from two systematic reviews and meta-analyses, indicating that TAP block reduced 24-hour morphine consumption following cesarean delivery under spinal anesthesia [15, 16].
In contrast, a different systematic review and meta-analysis involving 268 women from five randomized controlled trials reported no significant differences in cumulative opioid consumption between TAP block and local wound infiltration at 24 and 48 hours [17]. Additionally, other randomized studies with 60 and 78 patients respectively found no dissimilarities in the total amount of fentanyl and morphine consumed between TAP block and wound infiltration within the first 48 and 24 hours [18, 19], possibly due to patient-controlled analgesia in the former trial.
Our research indicates that the TAP group required a longer time to reach their initial analgesic requirement compared to the wound infiltration group. These findings align with previous studies that reported similar results [11, 13, 14]. Notably, variations exist among studies, possibly due to factors such as methodology, local anesthetic agents used, and the timing of outcome assessments. While some trials reported no significant differences in pain scores between TAP block and wound infiltration [17], others found reduced pain scores in the TAP group at various postoperative time points [19, 20].
Furthermore, a study comparing TAP block with direct local anesthetic infiltration reported a significantly longer time to rescue analgesic with TAP block [21]. Our study highlights important distinctions in postoperative pain management and associated outcomes between TAP block and wound infiltration following cesarean section under spinal anesthesia. However, variations among studies underscore the complexity of analgesic techniques and their outcomes.
This study has several advantages. Firstly, it adopts a double-blinded, randomized comparative placebo design, and notably, there was no loss of patients during follow-up. Secondly, it stands as the first study within our hospital to compare the analgesic efficacy of TAP block to LAWI following cesarean sections under spinal anesthesia. Furthermore, it boasts a larger sample size compared to previous studies conducted in this area. However, it’s important to note that this study was limited to a single center, focusing exclusively on patients who underwent cesarean deliveries and visited the Obstetrics and Gynecology Clinics. Other types of abdominal surgeries conducted by different departments were not included in this study.
In conclusion, our results demonstrate that TAP block is superior to wound infiltration in reducing postoperative pain and total 24-hour opioid and analgesic intake in mothers who have undergone spinal anesthesia after a cesarean delivery. It provided more potent and longer-lasting analgesia.
All data generated or analyzed during this study are included in this article. For additional inquiries, please contact the corresponding author.
AS and MSS contributed to data gathering, article development, and study design. RME worked on manuscript analysis and modification. HRE and AS confirmed the analytic techniques. AEK and MR were responsible for theoretical formalism development, analytical calculations, and numerical simulations. SIM and HRE evaluated the findings and carried out the implementation. MSS assisted with data analysis and essay revision. Each author contributed ideas that refined the study, analysis, and article. All authors contributed to editorial changes in the manuscript. All authors reviewed the completed manuscript and provided approval for publication. Additionally, all authors participated in this paper with the cases they handled.
The Ethics Committee of the Department of Obstetrics and Gynecology, College of Medicine, Cairo University, granted its approval for this study (MD-301-2020) before its commencement. All participants provided their informed consent after being fully informed about the study’s objectives and methodologies. The researcher obtained written and signed informed consent from each participant before any study-related procedures were initiated. The original signed informed consent form for each participant was securely maintained by the investigator.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


