1 Department of Anesthesiology, Fuxing Hospital, Capital Medical University, 100038 Beijing, China
Abstract
Background: The aim of this study was to determine whether
dexmedetomidine can reduce the circulatory response in laparoscopic uterine
fibroids (UF) surgery. Our purpose was to investigate the effect of
dexmedetomidine on the circulatory response in laparoscopic UF surgery.
Methods: This was a randomized controlled study of 214 patients age
range of 40–60 years, American Society of Anesthesiologists (ASA) 1–2 grade,
undergoing elective laparoscopic UF surgery at Fuxing Hospital, Capital Medical
University from January 2020 to October 2023. Patients were randomly allocated to
the non-dexmedetomidine group (Group1) and dexmedetomidine group (Group2). The
mean arterial pressure (MAP) and heart rate (HR) were measured: when entering the
operation room (MAP1, HR1), immediately at the time of local pituitrin injection
(MAP2, HR2), and at 5 minutes after injection of pituitrin (MAP3, HR3). The
medications and dosages (propofol, urapidil hydrochloride, fluid) used during the
operation were recorded as well as the recovery (drowsiness, chills, nausea,
vomiting, dysphoria) after the operation. Total mean time from the beginning of
the operation to pituitrin injection (Time1) and recovery time after peaking
blood pressure with pituitrin injection (Time2) were also recorded.
Results: There were no significant differences in age, height, weight,
size and number of UFs, and the location of uterine fibroids between the two
groups. There were no differences between the groups for MAP1, MAP2, HR1, HR2,
HR3, and Time1 (all p
Keywords
- dexmedetomidine
- laparoscopy
- leiomyoma
- uterine fibroids
- blood circulation
- hemodynamics
Uterine fibroids (UF) are common gynecological benign tumors, with the most common symptoms being pelvic or abdominal pressure, bladder symptoms, and/or abnormal uterine bleeding [1, 2, 3]. Approximately 20%–50% patients with uterine fibroids require surgery [4, 5, 6, 7, 8]. Hemodynamic instability often occurs during laparoscopic UF surgery. Reasons include the carbon dioxide pneumoperitoneum and the head-low lithotomy position. The wide use of pituitrin in gynecologic surgery induces stress that can regulates the sympathetic nervous system [9, 10, 11], leading to increase plasma levels of norepinephrine, epinephrine, vasopressin, dopamine, cortisol, and angiotensin [12, 13, 14, 15], all resulting in circulatory response [16]. In particular, the use of pituitrin during laparoscopic surgery can significantly increase blood pressure [16]. Excessive hemodynamic fluctuations can present many challenges to the anesthesiologist during UF surgery [14].
Dexmedetomidine, a selective
The present study aimed to investigate the effect of dexmedetomidine on the hemodynamic response in laparoscopic UF surgery. The results have the potential to provide evidence for the improvement of the safety of anesthesia during UF surgery.
This was a pilot prospective interventional study from January 2020 to October 2023. This study was approved by the ethics review committee of Fuxing Hospital, Capital Medical University (Ethics approval number: 2020FXHEC-KY036). Trial registration number: NCT03524950.
The inclusion criteria were: age 40–60 years; ASA grade 1–2; scheduled to undergo elective laparoscopic UF surgery.
The exclusion criteria were: patients with unstable cardiovascular disease; hepatic and renal insufficiency; obesity; alpha-blocker use; severe obstructive sleep apnea hypopnea syndrome (OSAHS); severe allergies, history of specific drug use; history of psycho-psychological disorders and emergency operation.
The 214 patients who received laparoscopic UF surgery in the Fuxing Hospital, Capital Medical University were randomly divided into two groups, the non-dexmedetomidine group (Group1) or the dexmedetomidine group (Group2), via a computer generated random number table. The random numbers were sealed in envelopes by a trial coordinator. The coordinator opened the envelopes to assign the treatment and prepared the study drugs according to the group assignment (the Group1: normal saline 50 mL; the Group2: dexmedetomidine, Yangzijiang Pharmaceutical Company, Taizhou, Jiangsu, China. 200 µg, diluted with normal saline to 50 mL). The coordinator was not involved in anesthesia management or data collection. All providers were blinded to the individual patient assigned group.
Before anesthesia induction, both groups were injected with the study drug pump for 10 minutes (the injection rate was 1 µg/kg/h of dexmedetomidine when calculating the pump rate). During anesthesia induction, the two groups received sufentanil 0.5 µg/kg, propofol initiated at 6 µg/mL via target-controlled infusion (TCI), and rocuronium 0.6 mg/kg. During the maintenance phase, the two groups received continuous remifentanil 0.1–0.3 µg/kg/min and propofol. The dosage of propofol was adjusted to maintain the bispectral index (BIS) at 45–55. In addition, the two groups received the study pump (the injection rate was 0.7 µg/kg/h of dexmedetomidine when calculating the pump rate). The monitor was an Infinity Vista XL (Dräger, Lubeck, Germany), and the anesthesia machine was a Fabius 2000 (Dräger, Lubeck, Germany).
The other procedures were same in both groups. The BIS value was maintained at 45–55 through adjusting the dosage of propofol. The end-expiratory carbon dioxide partial pressure was maintained at 35–45 mmHg during the operation. If the mean arterial pressure increased by 30%, urapidil hydrochloride 10–30 mg was used. Repeat administration could occur after 5–10 min. If the mean arterial pressure decreased by 30%, ephedrine hydrochloride 6–10 mg was used, with repeat after 5–10 min. There was no significant decrease in blood pressure in this study, resulting in ephedrine hydrochloride not being used.
During the operation, the surgeon performed the operation through three perforations performed in the patient’s abdomen, injecting a specific dose of pituitrin in the surgical site, and then removed the uterine fibroids. The surgeons are all senior doctors who performed laparoscopic UF surgery for over 500 patients.
Demographic information, including age, height, weight, number of the UF, size and location of the UF, were obtained from the medical records. The mean arterial pressure (MAP) and heart rate (HR) were used as the indicators of hemodynamic status at multiple time points: when entering the operating room (MAP1, HR1), immediately at the time of local pituitrin injection (MAP2, HR2), and at 5 min after injection of pituitrin (MAP3, HR3). The medications and dosages (propofol, urapidil hydrochloride, fluid) used during the operation, and the recovery (drowsiness, chills, nausea, vomiting and dysphoria) after the operation were recorded. Total mean time from the beginning of the operation to pituitrin injection (Time1) and recovery time after peak blood pressure with pituitrin injection (Time2) were recorded. Data recording was done by the anesthesiologist.
SPSS 26.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. The
continuous variables conforming to the normal distribution (Kolmogorov-Smirnov
test) were presented as means
There were 214 patients in each group. Four cases of conversion to laparotomy and 2 cases of voluntary abandonment from the study were excluded. The remaining 210 patients underwent surgery successfully. Their baseline characteristics are shown in Table 1. There were no significant differences in age, height, weight, size and number of UF, and the location of uterine fibroids between the two groups (Fig. 1).
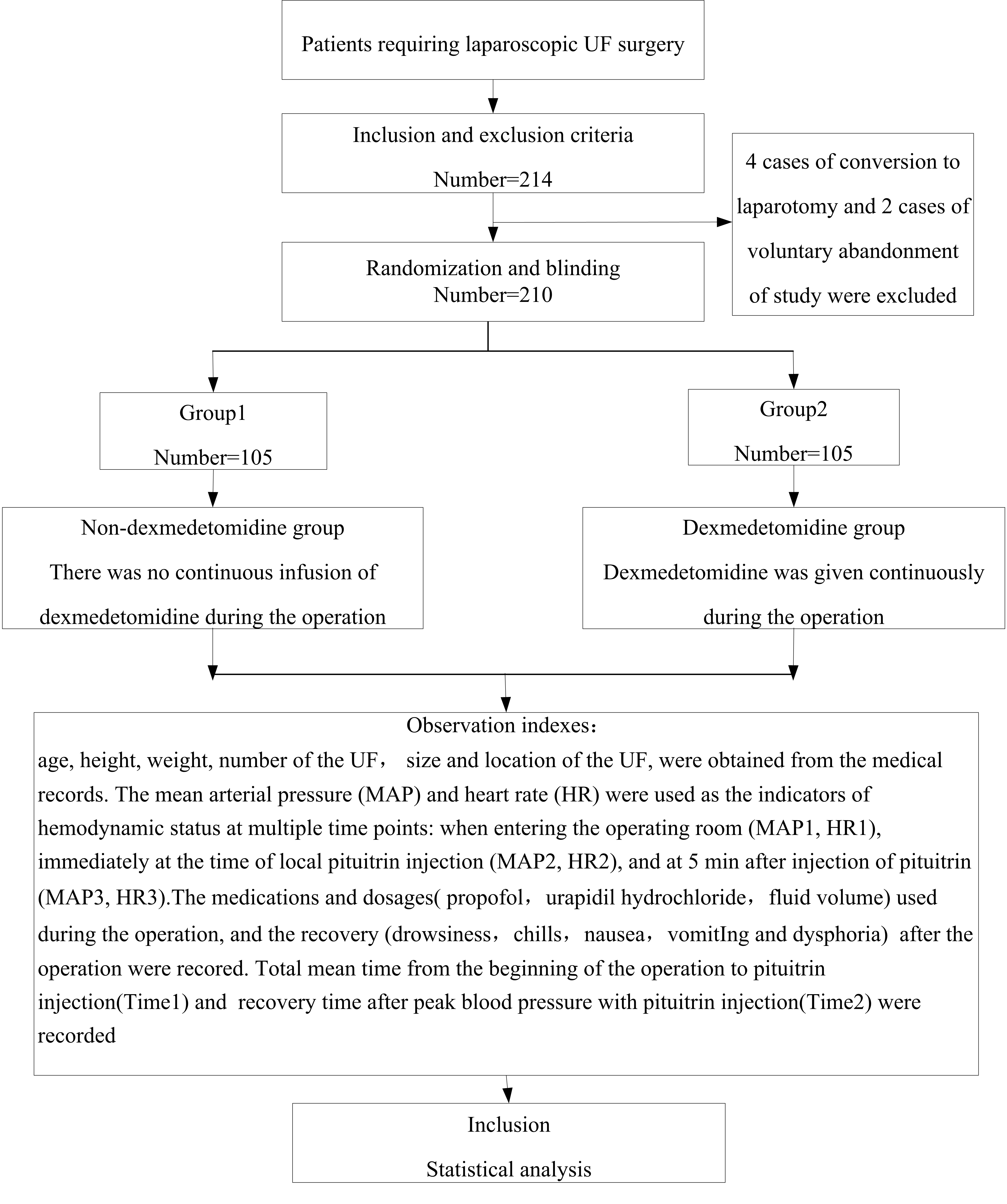 Fig. 1.
Fig. 1.Flow chart. UF, uterine fibroids.
| Characteristics | All (n = 210) | Group1 (n = 105) | Group2 (n = 105) | t/ |
p | |
| Age (years) | 39.75 |
39. 92 |
39.57 |
0.414 | 0.919 | |
| Height (cm) | 161.65 |
161.67 |
161.63 |
0.055 | 0.094 | |
| Weight (kg) | 60.75 |
60.86 |
60.64 |
0.184 | 0.218 | |
| The number of uterine fibroids | 1.68 |
1.75 |
1.61 |
0.979 | 0.091 | |
| The size of uterine fibroids (cm) | 6.47 |
6.68 |
6.25 |
2.007 | 0.818 | |
| The Location of uterine fibroids, n (%) | 5.485 | 0.064 | ||||
| Anterior | 94 (44.8%) | 39 (37.1%) | 55 (52.4%) | |||
| Posterior | 84 (40.0%) | 46 (43.8%) | 38 (36.2%) | |||
| Lateral and fundal | 32 (15.2%) | 20 (19.0%) | 12 (11.4%) | |||
SD, standard deviation.
Table 2 presents the surgery-related data. There were no differences between the
groups for MAP1, MAP2, HR1, HR2, HR3, Time1 (all p
| Characteristics | All (n = 210) | Group1 (n = 105) | Group2 (n = 105) | t | p |
| MAP1 | 90.80 |
89.96 |
90.99 |
0.671 | 0.495 |
| MAP2 | 80.68 |
80.53 |
80.83 |
0.191 | 0.992 |
| MAP3 | 104.74 |
107.37 |
102.12 |
2.460 | 0.041 |
| HR1 | 76.05 |
76.96 |
75.13 |
0.979 | 0.130 |
| HR2 | 66.73 |
68.13 |
65.32 |
1.701 | 0.665 |
| HR3 | 63.40 |
63.91 |
62.89 |
0.566 | 0.965 |
| Time1 (min) | 15.74 |
16.24 |
15.24 |
0.774 | 0.085 |
| Time2 (min) | 16.94 |
18.86 |
15.00 |
1.822 | 0.000 |
MAP, mean arterial pressure; HR, heart rate.
Table 3 presents the changes in hemodynamic index data. There were no
differences between the groups for MAP2-MAP1, MAP3-MAP2, HR2-HR1, HR3-HR1, and
HR3-HR2 (p
| Characteristics | All (n = 210) | Group1 (n = 105) | Group2 (n = 105) | t | p |
| MAP2-MAP1, (mmHg) | 9.79 |
9.43 |
10.16 |
0.367 | 0.463 |
| MAP3-MAP1, (mmHg) | 14.27 |
17.41 |
11.12 |
2.779 | 0.025 |
| MAP3-MAP2, (mmHg) | 24.06 |
26.84 |
21.28 |
2.709 | 0.218 |
| HR2-HR1, (bpm) | 9.32 |
8.83 |
9.81 |
0.440 | 0. 544 |
| HR3-HR1, (bpm) | 12.65 |
13.05 |
12.25 |
0.345 | 0.249 |
| HR3-HR2, (bpm) | 3.33 |
4.22 |
2.44 |
0.911 | 0.790 |
MAP, mean arterial pressure; HR, heart rate.
Table 4 presents the surgery-related parameters data. There were no differences
between the two groups for fluid volume (p
| Characteristics | All (n = 210) | Group1 (n = 105) | Group2 (n = 105) | t | p |
| Propofol (mg) | 599.19 |
670.01 |
528.36 |
7.178 | 0.011 |
| Urapidil hydrochloride (mg) | 0.54 |
0.95 |
0.12 |
1.980 | 0.000 |
| Fluid volume (min) | 712.33 |
715.62 |
709.05 |
0.255 | 0.573 |
Table 5 presents the relevant indicators of postoperative recovery data. There
were no differences between the groups for drowsiness (p
| Characteristics | All (n = 210) | Group1 (n = 105) | Group2 (n = 105) | p | |
| Drowsiness | 6 (2.9%) | 3 (2.9%) | 3 (2.9%) | 0.000 | 1.000 |
| Chills | 13 (6.2%) | 12 (11.4%) | 1 (1.0%) | 9.922 | 0.002 |
| Nausea | 15 (7.1%) | 14 (13.3%) | 1 (1.0%) | 12.130 | 0.000 |
| Vomiting | 4 (1.9%) | 4 (3.8%) | 0 (0.0%) | 4.078 | 0.043 |
| Dysphoria | 6 (2.4%) | 5 (95.2%) | 0 (0.0%) | 5.122 | 0.024 |
This study aimed to investigate the effect of dexmedetomidine on the hemodynamic stability in laparoscopy UF surgery. The results suggest that dexmedetomidine stabilizes the hemodynamics during laparoscopic UF surgery.
Takahashi et al. [18] review summarized the cardio protective effects for dexmedetomidine. The study by Xin Li et al. [19] showed that dexmedetomidine could reduce the hemodynamic response in patients undergoing surgery. We demonstrated similar results. The MAP3 and MAP3-MAP1 in the dexmedetomidine group were significantly lower than those in the non-dexmedetomidine group. This was in accordance with the known impact of dexmedetomidine on blood pressure [20]. In a previous randomized trial, administration of intraoperative and postoperative dexmedetomidine reduced the incidence of posttraumatic stress disorder [21]. Similarly, the recovery time after peaking blood pressure with pituitrin injection was shortened in the dexmedetomidine group, indicating that the cycle fluctuation time was reduced and the adverse effects of the cycle fluctuation on patients were reduced.
Our study revealed that the doses of propofol and urapidil hydrochloride were reduced in the dexmedetomidine group, and postoperative complications (chills, nausea, vomiting and dysphoria) were also reduced. This may be because dexmedetomidine combined with propofol is more conducive to postoperative recovery of patients.
The study of Shehabi et al. [22] indicated dexmedetomidine and propofol combination was associated with decreased mortality. The study by Persson et al. [23] showed dexmedetomidine could serve as a sedative hypnotic drug to enhance clearance of harmful waste from the brain. This may also be the reason for the decrease in adverse reactions after the use of dexmedetomidine during surgery. In the review by Takahashi et al. [18], it was pointed out that heart rate changes occur at the end of the surgery and after extubation. In this study, no difference in HR was found, which is consistent with the study by Fu et al. [24], which demonstrated that adding dexmedetomidine to remifentanil did not change HR. There was no change in HR in this study, which could be related to the point when heart rate was recorded. Therefore, dexmedetomidine maintains hemodynamic stability during laparoscopic UF surgery.
This study has limitations as it was a single center study. Additional studies are necessary with expansion of sample size and utilization of other surgical centers.
In conclusion, our results suggest that dexmedetomidine maintain hemodynamic stability and is beneficial to postoperative recovery in laparoscopic UF surgery.
MAP, mean arterial pressure; HR, heart rate; UF, uterine fibroids; ASA, American Society of Anesthesiologists; OSAHS, obstructive sleep apnea hypopnea syndrome; TCI, target-controlled infusion; BIS, bispectral index; BP, blood pressure.
The data sets used in this article are available through the corresponding author upon reasonable request.
XC—data collection, extraction, drafting of the manuscript, analysis of data, manuscript revision; XD—data collection; HHS—data collection; LHZ—analysis of data; XL—analysis of data. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work. All authors read and approved the final manuscript.
This study was approved by the ethics review committee of Fu Xing Hospital, Capital Medical University. Ethics approval number: 2020FXHEC-KY036. Trial registration: NCT03524950. All subjects gave their informed consent for inclusion before they participated in the study.
We would like to express my gratitude to all those who helped me during the writing of this manuscript.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

