1. Introduction
Ovarian cancer (OC) is the foremost cause of mortality from
gynecological malignancies. Typically diagnosed at advanced stages with extensive
peritoneal metastasis due to insidious onset and atypical early symptoms, OC
exhibits a 5-year overall survival (OS) of approximately 30% [1, 2]. Although
cytoreductive surgery and platinum-based combination chemotherapy remain the
first-line treatment option, the high risk of lymphatic metastasis and recurrence
persists [3, 4]. Adjuvant therapies like intraperitoneal infusion chemotherapy
and hyperthermic intraperitoneal chemotherapy offer improvements [5, 6, 7] but pose
risks of drug resistance and serious complications [8]. Therefore, exploring
novel treatment approaches is imperative for improving the prognosis of OC.
In view of the radiosensitivity of OC [9], molecular targeting
technology offers a means of delivering precise internal irradiation to tumors
and metastatic lesions after intravenous injection of radionuclides [10].
Radionuclide imaging aids in visualizing OC pathogenesis and progression [11, 12], showing diagnostic and therapeutic potential. Shabani et al. [13]
synthesized Ru template gold nanoparticles and assessed the therapeutic effect of
these nanoparticles on breast cancer cells. The results indicated the selective
and effective anticancer function of the nanoparticles, with low cytotoxicity and
superior biocompatibility [13]. Furthermore, intraperitoneal injection of
radioactive agents for internal irradiation radiotherapy can generate a high
local drug concentration similar to intraperitoneal chemotherapy, thereby
reducing recurrence and improving survival [14, 15, 16]. Therefore, efforts focus on
enhancing irradiation delivery via developing new and effective carriers for the
irradiation agents, aiming to mitigate adverse events and enhance outcomes for
OC.
The number and activity of folate receptor (FR), as well as the affinity of
folic acid (FA) conjugates (Kd: 10–10 M) have been reported to be
much higher on the surface of 90% of OC tumor cells than those in normal cells
[17]. Therefore, FR-targeted therapies have attracted great attention in recent
years for treating OC [18, 19, 20]. However, in vivo studies on FR-targeted
carriers labeled with radionuclides (Tc, Re, Ga,
Cu) [21, 22, 23, 24] revealed limited targeting effectiveness for FA conjugated
carriers such as liposomes and nanoparticles [21, 22]. Herein, we used FR as the
targeting molecule, the biodegradable material polyethyleneglycol-polylactic
acid-co-glycolic acid (PEG-PLGA) [25] as the carrier matrix, and diethylene
triamine pentaacetic acid (DOTA) as the metal chelating agent to prepare
lutetium labeled nanoparticles, named Lu-FA-DOTA-PEG-PLGA
nanoparticles. In addition, both labeling yield and radiochemical purity of the
nanoparticles are essential for clinical applications, they were therefore
evaluated in this study. Moreover, the efficacy and safety of nanoparticles in
the two treatment modes including tail vein injection and intraperitoneal
infusion were evaluated utilizing mouse models bearing subcutaneously
transplanted SKOV3 OC tumors or intraperitoneal metastatic SKOV3 OC tumors,
respectively. The Lu-FA-DOTA-PEG-PLGA nanoparticles prepared in this
study possess targeting, degradability and nuclide internal irradiation
therapeutic properties. Notably, intraperitoneal infusion of the
Lu-FA-DOTA-PEG-PLGA nanoparticles for treating OC metastatic lesions
represents a novel approach. This method holds promise for collaboration with
comprehensive OC treatment, potentially improving efficacy and reducing drug
resistance.
2. Materials and Methods
2.1 Animals
Bagg albino strain C (BALB/c) nude mice (female, 4 weeks old, weight: 18–20 g)
bearing subcutaneously transplanted SKOV3 human OC tumors and healthy Institute
of Cancer Research (ICR) mice (female, 4 weeks old, weight: 18–20 g) were
provided by Huajing Molecular Imaging & Drug Research Institute (Nanjing,
Jiangsu, China). BALB/c nude mice (female, 4 weeks old, weight: 18–20 g) bearing
intraperitoneal metastatic SKOV3 OC tumors were purchased from Yunqiao Purui
Biotech (Nanjing, Jiangsu, China). Animals were housed in plastic cages in a
specific pathogen-free (SPF) room with a light/night (12/12 h)
cycle and a temperature of 24 2 °C. During the experiments, the
animals had free access to food and water. All mice were administered a
folate-free diet (TP6020, Trophic Animal Feed High-tech Co. Ltd.,
Nantong, Jiangsu, China). All animal care and
experimental protocols were approved by the Ethical Committee of Xuzhou Medical
University Experimental Animal Center (No. 202101w015).
2.2 Structural Characterization of the Nanoparticle
Precursor FA-DOTA-PEG-PLGA
The nanoparticle precursor, designated as FA-DOTA-PEG-PLGA, was prepared by
Nanoeast Biotech (Nanjing, Jiangsu, China). The morphology and size of
nanoparticles were assessed using a JEM-2100 transmission electron microscopy
(TEM; JEOL, Tokyo, Japan) and a Zeta plus dynamic light scattering (DLS;
Brookhaven Instruments, Holtsville, NY, USA).
2.3 Preparation of the Lu-FA-DOTA-PEG-PLGA Nanoparticles
FA-DOTA-PEG-PLGA (50 µL, 50 nmol) and Lu (50 µL; Atom High
Tech, Beijing, China) were added to a centrifuge tube and vortexed for 10 sec,
followed by heating at 40 °C for 60 min. The Lu-FA-DOTA-PEG-PLGA
nanoparticles were isolated by three rounds of ultrafiltration
at 12,000 rpm for 5 min each. The labeling yield and radiochemical purity of the
nanoparticles were subsequently determined.
2.4 Histological Distribution of the Nanoparticles
Twelve healthy ICR mice, administered 18.5 Mbq of Lu-FA-DOTA-PEG-PLGA
nanoparticles via tail vein or intraperitoneal injection, were randomly divided
into 4 groups (3 mice per group). Subsequently, the mice were sacrificed by
cervical dislocation under anesthesia at 4 h, 24 h, 72 h, and 168
h post-injection, followed by radioactivity measurement in
blood, brain, heart, liver, spleen, lung, kidneys, stomach, muscle (hind limb),
and bone (femoral segment) using a radioisotope dose calibrator
(CRC®-55tR, Capintec, Ramsey, NJ, USA). The data were presented as the
percentage injected dose per gram tissue (%ID/g of tissue) after decay
correction.
2.5 Micro-Single-Photon Emission Computed Tomography/Computed Tomography (Micro-SPECT/CT)
Imaging
Twenty BALB/c nude mice bearing subcutaneously transplanted SKOV3 human OC
tumors were treated with 18.5 Mbq of Lu-FA-DOTA-PEG-PLGA nanoparticles
via tail vein injection and were randomly divided into four groups (5 mice per
group). The mice in the four groups underwent micro-SPECT/CT imaging using a
four-head SPECT/CT system (U-SPECT/CT, MI Lab, Houten, Netherlands) at 4 h, 24 h,
72 h, and 168 h, respectively. The tumor-to-muscle uptake ratio (T/M) was
calculated using PMOD software (4.4 version, PMOD Technology, Fallanden,
Netherland). T/M signified the ratio of radioactive uptake counts between the
tumor and muscle, reflecting the specific uptake ability of radiopharmaceuticals
in tumors.
2.6 Maximum Tolerated Dose (MTD) Determination
Fifteen BALB/c nude mice were randomly divided into 3 groups (5 mice per group)
treated with 18.5, 37.0 and 55.5 Mbq of
Lu-FA-DOTA-PEG-PLGA nanoparticles via tail vein
injection or intraperitoneal injection, respectively. The mice were observed for
behavioral changes every 2 days, and their body weight was
measured over a 30-day period. MTD was defined as the dose at which the body
weight of mice decreased by more than 20% or at least one mouse succumbed to the
treatment.
2.7 Antitumor Efficiency of the Nanoparticles
2.7.1 Antitumor Efficiency in the Subcutaneously Transplanted
Tumors
Twelve BALB/c nude mice bearing subcutaneously transplanted tumors were randomly
assigned to control, chemotherapy, and Lu-FA-DOTA-PEG-PLGA nanoparticle
groups (n = 4 per group). The control group received 0.1 mL saline solution, the
chemotherapy group received 3 mg/kg cisplatin twice per week, and the
Lu-FA-DOTA-PEG-PLGA nanoparticle group received 18.5 Mbq nanoparticles
via tail vein injection. Tumor volumes were evaluated every 2 or 3 days using a
caliper. Tumor growth inhibition (TGI) was calculated using the formula: TGI (%)
= (1 – Ti/Vi) 100%, where Ti and Vi represented the mean tumor
volume in each treatment group (chemotherapy or nanoparticle) and the control
group, respectively.
2.7.2 Antitumor Efficiency in the Intraperitoneal Metastatic
Tumors
Twelve BALB/c nude mice bearing metastatic tumors were randomly assigned to
control, chemotherapy, and nanoparticle groups (n = 4 per group). The control
group received 0.2 mL saline solution, the chemotherapy group received 3 mg/kg
cisplatin twice per week, and the Lu-FA-DOTA-PEG-PLGA nanoparticle group
received 18.5 Mbq nanoparticles intraperitoneally. In vivo fluorescence
imaging was performed before treatment and on day 7 after treatment. Then
abdominal tumor fluorescence intensity was analyzed using PerkinElmer software
(version 4.4, PerkinElmer, Waltham, MA, USA). TGI was calculated with the formula: TGI (%) = (1 – T/C)
100%, where T and C represented the relative fluorescence intensity of the
tumors in each treatment group (chemotherapy or nanoparticle) and the control
group, respectively. Subsequently, the tumor-bearing mice in all three groups
were sacrificed for the comparison of ascitic fluid volume.
2.8 Safety Evaluation
Hematoxylin and eosin (HE) staining was performed to evaluate the safety of
Lu-FA-DOTA-PEG-PLGA nanoparticles. Specifically, small intestine and
colon tissues of nude mice bearing intraperitoneal metastatic tumors were
subjected to HE staining in the control, chemotherapy, and nanoparticle groups.
2.9 Statistical Analysis
SPSS 23.0 version (IBM Corp., Armonk, NY,
USA) was used for the statistical analysis. Quantitative data with a normal
distribution were expressed as mean standard deviation. Multiple-group
comparisons were carried out using one-way analysis of variance (ANOVA), and
pairwise comparisons were performed using the Tukey multiple comparison test. A
statistically significant difference was defined as a p value less than
0.05.
3. Results
3.1 Structural Characteristics of the FA-DOTA-PEG-PLGA
TEM images showed that nanoparticle precursors FA-DOTA-PEG-PLGA were spherical
with a diameter of approximately 20 to 60 nm (Fig. 1). DLS results showed a
uniform size distribution of FA-DOTA-PEG-PLGA, with a polydispersity index (PDI)
of 0.182. The average zeta potential of FA-DOTA-PEG-PLGA was –15 mv, indicating
the good stability of FA-DOTA-PEG-PLGA nanoparticles.
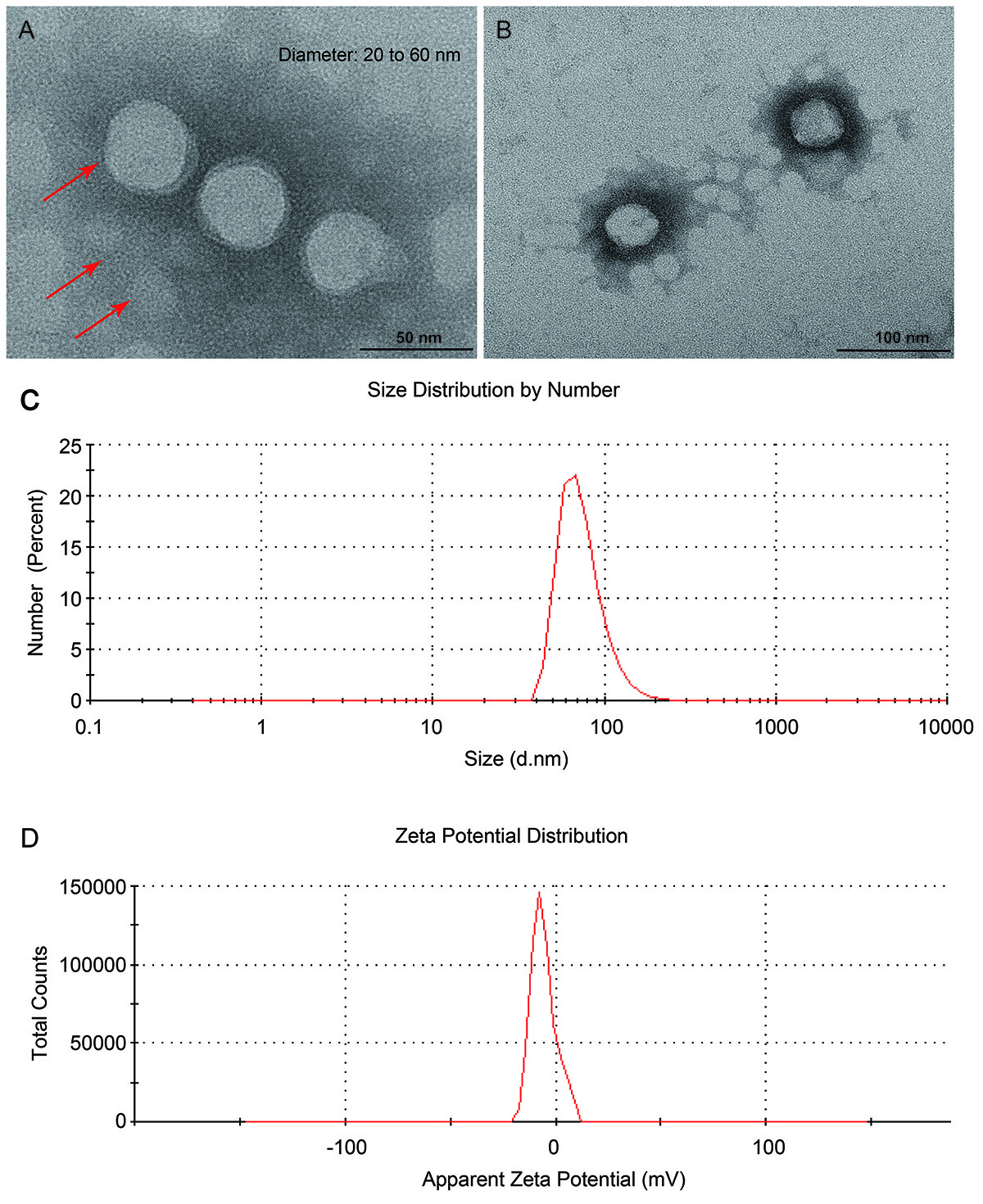 Fig. 1.
Fig. 1.
Structural characteristics of the FA-DOTA-PEG-PLGA
nanoparticles. (A,B) Transmission electron microscopy (TEM) images with
different magnifications. (C) Size distribution profile. (D) Zeta potential
profile.
3.2 Labeling Yield and Radiochemical Purity of the
Lu-FA-DOTA-PEG-PLGA Nanoparticles
The labeling yield of the Lu-FA-DOTA-PEG-PLGA nanoparticles was 97% to
98%. The radiochemical purity of the nanoparticles ranged from 96% to 98%.
3.3 MTD of the Lu-FA-DOTA-PEG-PLGA Nanoparticles
All nude mice injected with 18.5 Mbq and 37.0 Mbq Lu-FA-DOTA-PEG-PLGA
nanoparticles showed no significant weight loss or mortality. Consequently, the
dose of 18.5 Mbq was selected for subsequent experiments.
3.4 Histological Distribution of the Lu-FA-DOTA-PEG-PLGA
Nanoparticles
The distributions of Lu-FA-DOTA-PEG-PLGA nanoparticles
injected through tail vein in various tissues were shown in Fig. 2A. The blood
showed low-level radioactivity uptake at 4 h, 24 h and 72 h
post-nanoparticle injection. The radioactivity uptake at 168 h in the blood was
at the background level and was basically eliminated. Among the vital organs, the
liver and spleen showed the highest levels of radioactivity uptake, maintaining a
consistently high level within 168 h. The kidneys exhibited stable radioactivity
uptake levels, significantly lower than those observed in the liver and spleen.
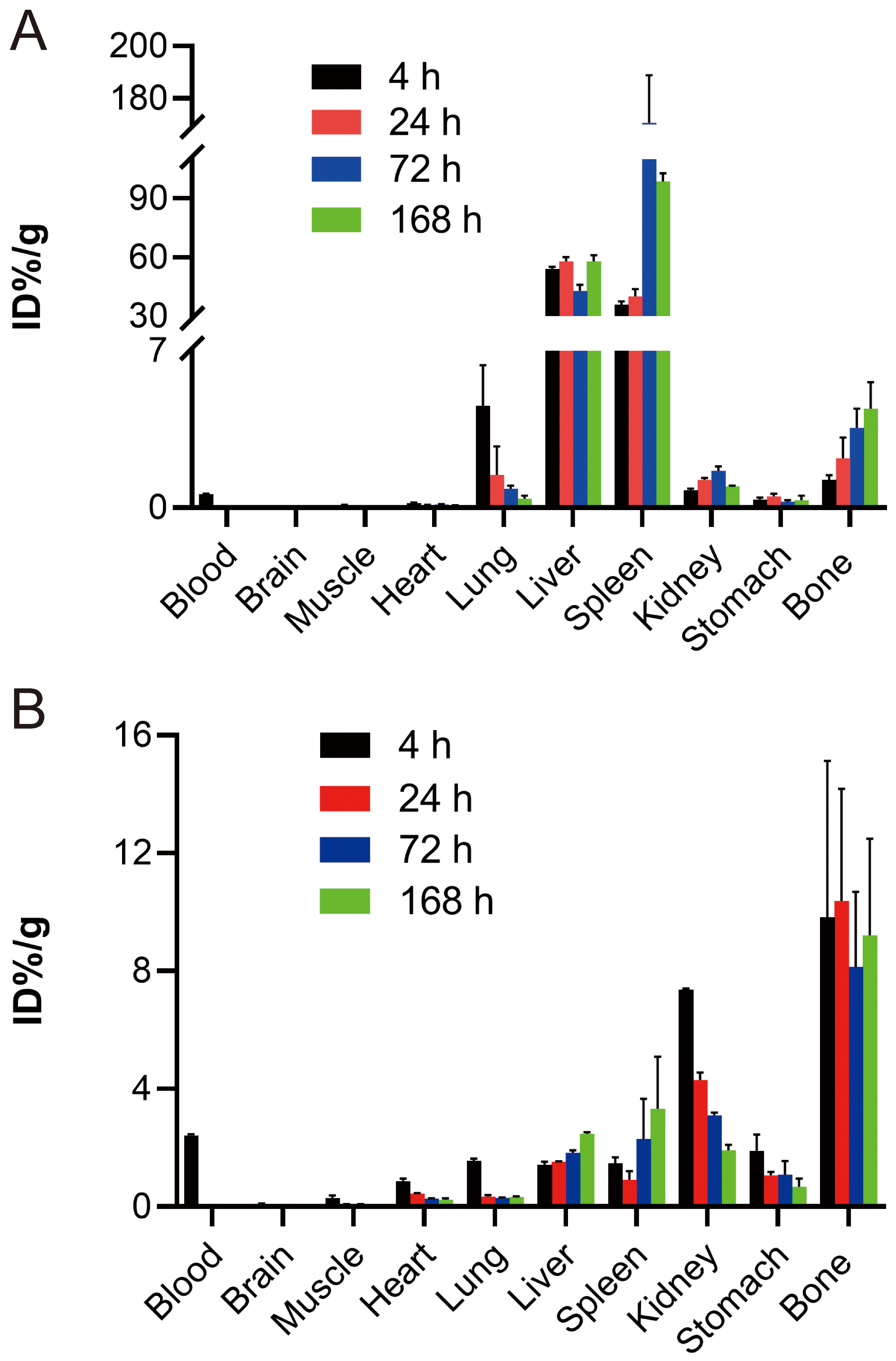 Fig. 2.
Fig. 2.
The distribution of Lu-FA-DOTA-PEG-PLGA
nanoparticles in various tissues at 4 h, 24 h, 72 h, and 168 h after tail vein
injection (A) and intraperitoneal injection (B).
In the case of intraperitoneal injection, the distributions of the nanoparticles
in various tissues were presented in Fig. 2B. The blood exhibited the highest
radioactivity uptake after 4 h of nanoparticle injection and low-level
radioactivity uptake after 24 h and 72 h. Additionally, the radioactivity uptake
was reduced to background level and essentially eliminated at 168 h. Among the
vital organs, the bone and kidneys exhibited the highest radioactivity uptake
levels. Interestingly, the bone showed a sustained high level of radioactivity
uptake, while the kidneys showed a gradual decrease in radioactive uptake within
168 h. Additionally, the radioactivity uptake levels of the liver and spleen were
significantly lower than those observed in the bone and kidneys.
3.5 Micro-SPECT/CT Imaging
The micro-SPECT/CT imaging results of the nude mice bearing subcutaneously
transplanted tumors were shown in Fig. 3. The mass-like radioactive accumulation
was observed in the transplanted tumors localized in the right lower limb at 4 h,
24 h, 72 h and 168 h post-nanoparticle injection. The mean T/M values at these
time points were 2.18 0.26, 2.81 0.49, 1.84 0.31 and 1.65
0.27, with the peak observed at 24 h.
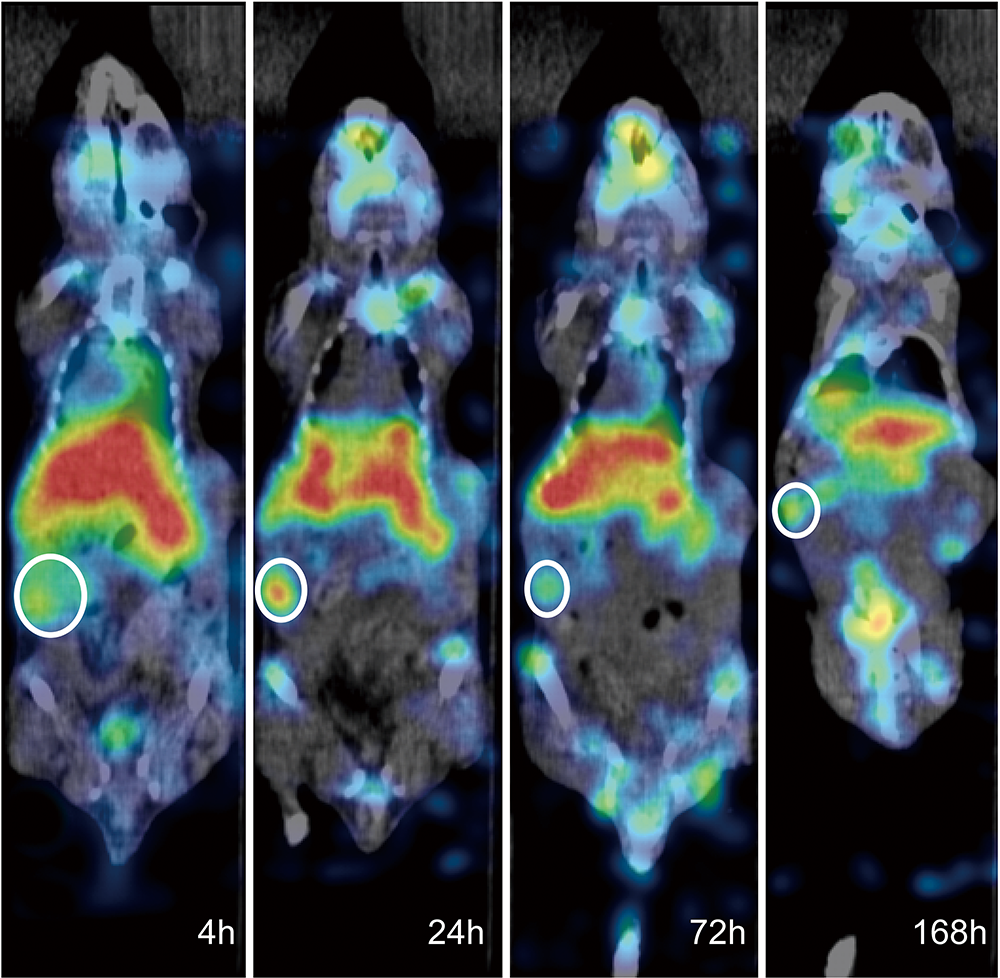 Fig. 3.
Fig. 3.
The micro-single-photon emission computed tomography/computed
tomography (micro-SPECT/CT) imaging results of the nude mice bearing
subcutaneously transplanted tumors after 4 h, 24 h, 72 h, and 168 h of
Lu-FA-DOTA-PEG-PLGA nanoparticle injection. The regions circled using
white circles were the subcutaneously transplanted tumors in the right lower limb
of the mice.
3.6 Antitumor Efficiency of the Lu-FA-DOTA-PEG-PLGA
Nanoparticles
Tumor growth was inhibited in both the chemotherapy and
Lu-FA-DOTA-PEG-PLGA nanoparticle groups compared with the control group
(Fig. 4). The TGI values of the chemotherapy group and nanoparticle group on day
7 of the treatment were 20.31% and 27.28%, respectively. However, after 12 days
of the treatment, the antitumor efficacy of the nanoparticles showed a tendency
to decrease.
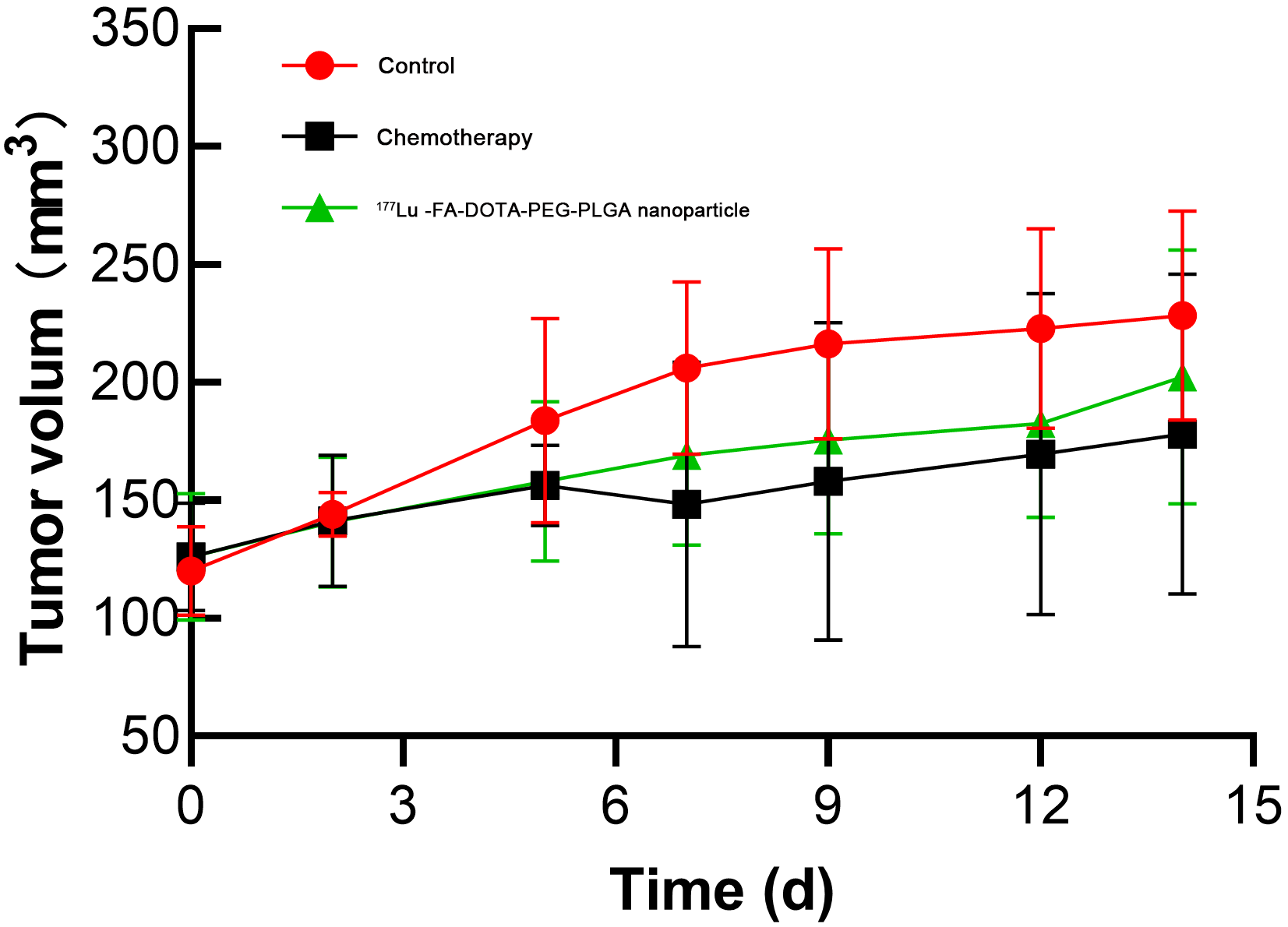 Fig. 4.
Fig. 4.
Tumor volume changes of the nude mice in the control,
chemotherapy, and Lu-FA-DOTA-PEG-PLGA nanoparticle groups at different
time points after treatment.
The fluorescence intensities of the
intraperitoneal metastatic tumors in the control, chemotherapy and
Lu-FA-DOTA-PEG-PLGA nanoparticle groups were (2.63 0.79)
10, (2.21 0.36) 10, and (1.45
0.19) 10, respectively, showing a statistical difference
(F = 6.09, p = 0.029, Fig. 5). The tumor fluorescence intensity
in the nanoparticle group was significantly lower than that in the control group
(p = 0.025). TGI values of the chemotherapy and nanoparticle groups were
18.6% and 35.6%, respectively.
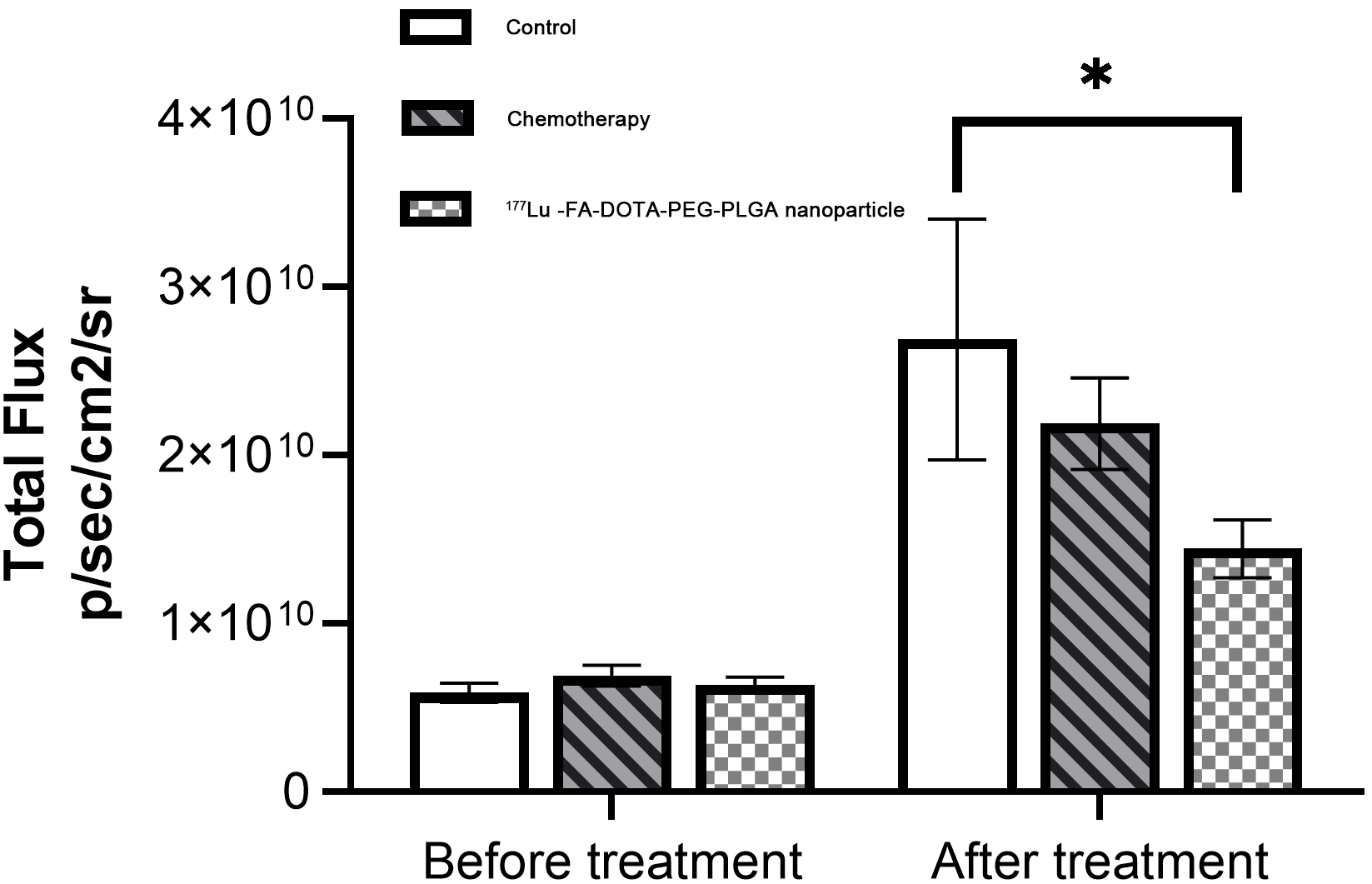 Fig. 5.
Fig. 5.
Fluorescence intensity of the tumors in the control,
chemotherapy and Lu-FA-DOTA-PEG-PLGA nanoparticle groups before and after
intraperitoneal injection treatment. *p 0.05.
The ascitic fluid volumes in the control, chemotherapy and nanoparticle groups
after intraperitoneal injection treatment were 0.77 0.09, 0.31
0.14, and 0.34 0.11 mL, respectively (Fig. 6). The ascitic fluid volumes
in the chemotherapy and nanoparticle groups were significantly lower than that in
the control group (F = 13.43, p = 0.006).
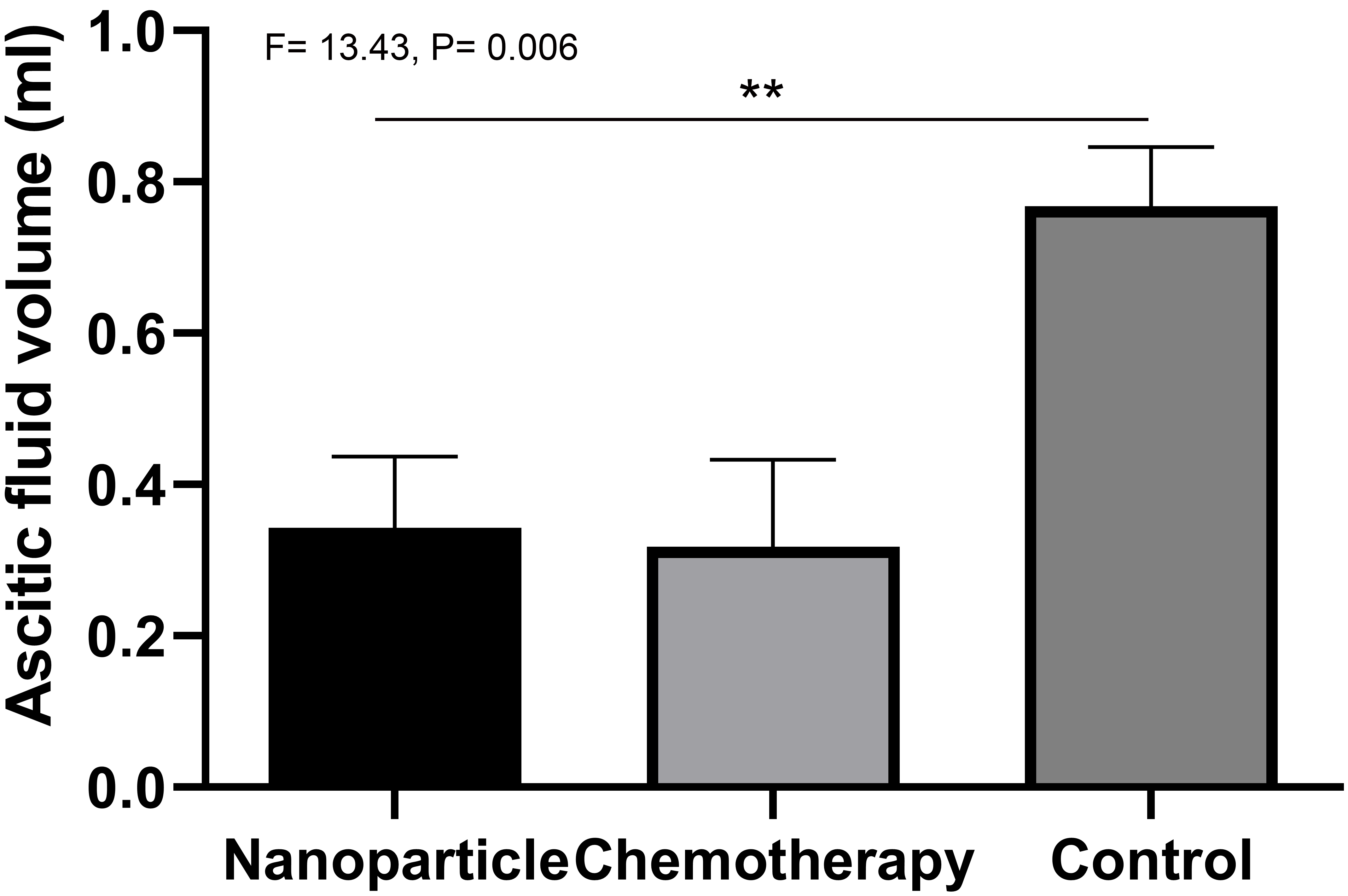 Fig. 6.
Fig. 6.
The ascitic fluid volumes in the control, chemotherapy
and nanoparticle groups after intraperitoneal injection treatment. **p 0.01.
3.7 Safety of the Lu-FA-DOTA-PEG-PLGA Nanoparticles
HE staining results were shown in Fig. 7. Compared with the control group, there
were no obvious abnormalities in the small intestine and colon tissues of the
mice in the nanoparticle group after intraperitoneal injection treatment. No
significant signs of apoptosis or necrosis in enteric mucosal crypt cells and
lymphocytes, epithelial cell shedding, and vascular expansion or bleeding were
found in the nanoparticle group.
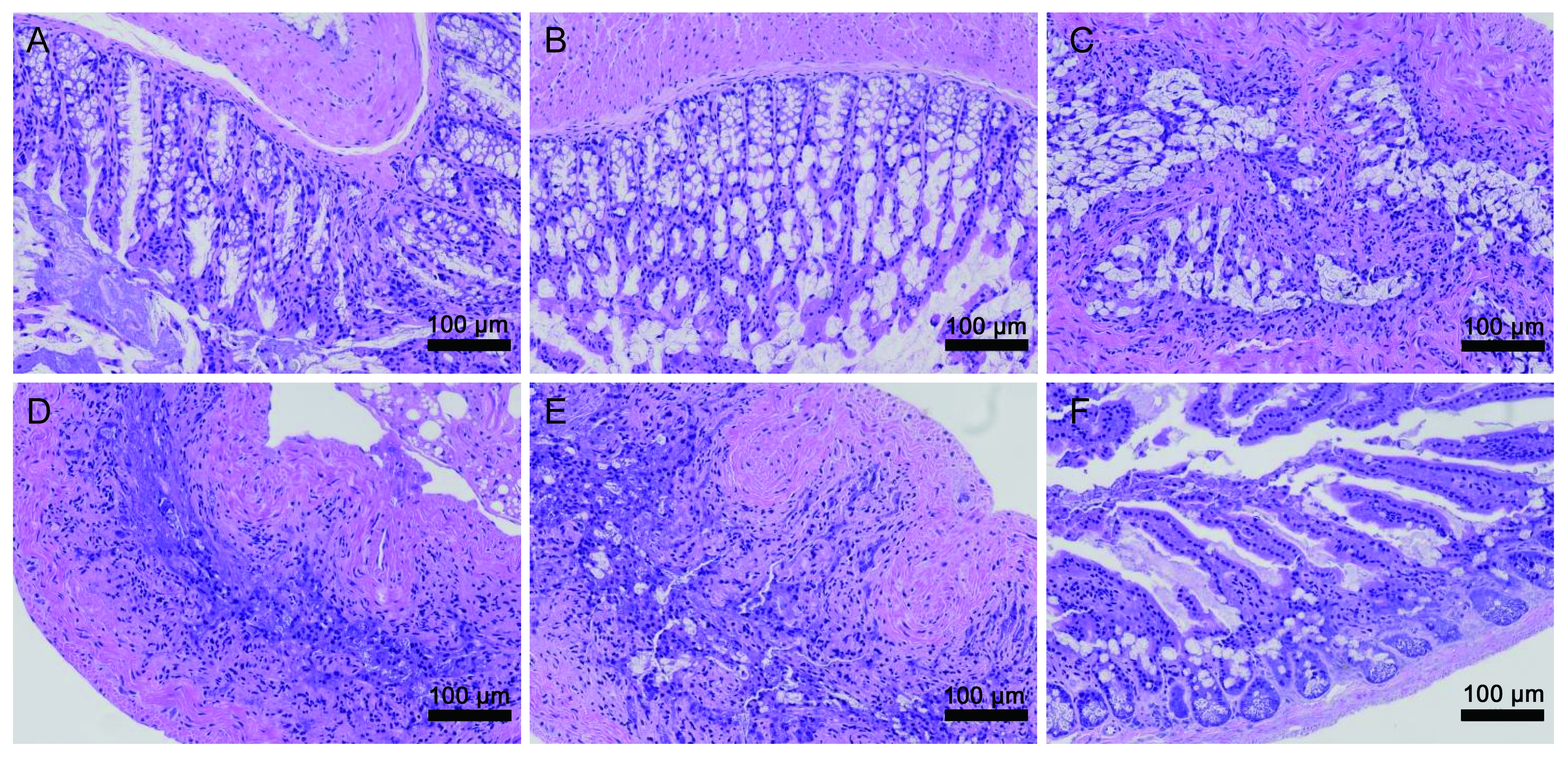 Fig. 7.
Fig. 7.
Hematoxylin and eosin (HE) staining images (200) of
the colon tissues ((A) control group; (B) chemotherapy group; (C)
Lu-FA-DOTA-PEG-PLGA nanoparticle group) and small intestine tissues ((D)
control group; (E) chemotherapy group; (F) Lu-FA-DOTA-PEG-PLGA
nanoparticle group) in the mice after intraperitoneal injection treatment.
4. Discussion
FR is a glycosylphosphatidylinositol-linked protein comprising four subtypes
(, , and ). Previous study has reported
significantly higher numbers and activity of FR, along with enhanced
affinity of folate conjugates on the surface of 90% of OC tumor cells compared
to normal cells [17]. It has emerged as a promising candidate for imaging and
targeted therapy of OC due to its marked expression in OC cells
[26]. However, the small molecular weight of FR results in a short blood
circulation time and rapid clearance from the bloodstream, leading to reduced
tumor uptake.
Several strategies have been explored to overcome these limitations. Some
studies have utilized small molecule albumin conjugates to noncovalently couple
antibody fragments and folate conjugates with plasma proteins, effectively
increasing the blood circulation time and drug concentration in tumors. However,
this approach often led to high radioactivity uptake in the kidneys [27, 28].
Additionally, pre-administration of antifolates has been employed to reduce the
reabsorption of In-DTPA-FA in the proximal renal tubules, but the
underlying mechanism remains unclear, and results are often poorly reproducible
[27]. Based on the property of PEG to
covalently bind to folate ligands and their analogues [29], Bao et al.
[30] prepared FA-DOTA-PEG-PLGA nanocarriers. These nanoparticles were designed to
modify the nanoparticle surface using the hydrophilic polymer material PEG,
creating long-circulating nanoparticles, also known as stealth nanoparticles
(SNP) [30]. This modification aimed to reduce recognition and phagocytosis by the
liver and spleen reticuloendothelial system, enhancing the blood circulation time
of the nanoparticles. To address the concern about nanoparticle retention in the
kidneys, we utilized PLGA as the nanocarrier, known to significantly reduce renal
drug distribution. Additionally, we controlled the size range of nanoparticles to
be greater than 20 nm, a parameter shown to reduce renal drug excretion [31].
Histological distribution analysis in this study revealed partial radioactivity
uptake in blood circulation 72 h after tail vein injection of nanoparticles, with
elimination of radioactivity uptake in the blood at 168 h. This suggested a
prolonged retention time of Lu-FA-DOTA-PEG-PLGA nanoparticles in the
blood circulation. Furthermore, the peak value of radioactivity uptake in the
kidneys was only 1.646 %ID/g, significantly lower than that observed in previous
study on FR radionuclide drug [32].
Micro-SPECT/CT imaging revealed a notable increase in radioactivity uptake in
subcutaneous tumors of the mice at 4 h after the tail vein injection of
nanoparticles, with peak uptake observed at 24 h. These findings suggested an
active targeting effect and rapid tumor entry of the nanoparticles. While there
was no significant difference in tumor volume between the nanoparticle group and
the control group, nanoparticle-treated tumors displayed a discernible trend of
suppression. It is noteworthy that the tumor-suppressing effect of the
nanoparticles weakened from day 9 to day 14, potentially attributed to the
diminished antitumor efficacy of the Lu after multiple decays. This
implied that the optimal therapeutic time of a single administration of the
nanoparticles spans approximately one week. An escalation in the
radiopharmaceutical dose may enhance the antitumor efficacy of the nanoparticles.
Intraperitoneal injection of radiopharmaceuticals has been under development for
nearly 50 years as a treatment for peritoneal metastasis and ascites of OC. Early
studies utilized P colloid, which could be attached to the inner wall of
the body cavity or the surface of organs for radiotherapy. However, the lack of a
targeting effect limited its maximum dose [14, 16]. Subsequent studies attempted
to use radionuclide-labeled antibody agents for targeted therapy, but their
transient residence and action time in the abdominal cavity resulted in limited
advantages for local treatment [33, 34]. Moreover, P colloid demonstrated
uneven distribution in the body after administration, leading to potential
intestinal toxicity and adverse events [15]. On this basis, we performed
further in vivo experiments to explore the therapeutic efficacy of
intraperitoneal injection of nanoparticles in treating mice with peritoneal
metastasis.
In this study, the histological distribution results revealed persistent
radioactivity uptake in the blood within 72 h after intraperitoneal injection of
nanoparticles, indicating a continuous influx of nanoparticles into the
bloodstream from the abdominal cavity during this period. The peritoneum,
characterized by abundant capillaries and lymphatic vessels, serves as a
bidirectional semipermeable membrane, permitting the passage of water,
electrolytes, and some small molecular substances. The
Lu-FA-DOTA-PEG-PLGA nanoparticles, being macromolecules with a size of
20–80 nm, have the capability to traverse the abdominal cavity but not the
peritoneum. With the gradual dissolution and decomposition of the matrix PLGA
[31], the nanoparticles underwent subsequent breakdown into small molecular
fragments, which were then absorbed by peritoneal capillaries and lymphatic
vessels. Some of these fragments entered tumors under the influence of targeting
effect, while the remaining fragments were excreted in urine. This slow
degradation process of nanoparticles ensured prolonged residence time of
radiopharmaceuticals in the abdominal cavity. The absorption into the bloodstream
post-degradation mitigated the impact of uneven distribution of
radiopharmaceuticals, especially in some patients with intestinal adhesions [35],
where encapsulated fluid may cause uneven drug dispersion, resulting in residual
lesions and suboptimal therapeutic effects.
Both the nanoparticle group and chemotherapy group exhibited tumor suppression
and a reduction in ascitic fluid volume, especially the nanoparticle group. In
addition, side effects such as intestinal perforation may be induced after
intraperitoneal injection of the nanoparticles due to the direct contact between
nanoparticles and organs. This concern was addressed through HE staining results,
which showed the absence of side effects in the small intestine and colon tissues
of the mice, indicating the safety of the Lu-FA-DOTA-PEG-PLGA
nanoparticles. Our findings indicated that, in comparison with P colloid
[15], Lu-FA-DOTA-PEG-PLGA nanoparticles exhibited reduced intra-abdominal
local retention and a diminished potential for adverse reactions. Collectively,
these findings provide a novel perspective for the treatment and recurrence
reduction of OC.
In addition to their targeting and intraperitoneal retention properties,
Lu-FA-DOTA-PEG-PLGA nanoparticles may exhibit other therapeutic
advantages. Firstly, the -ray range of Lu extends up to 2 mm,
possessing a specific range of action and penetration (about 0.2–0.3 mm in soft
tissues). This characteristic makes it more likely to exert the ‘cross-effect’ of
radiation in confined spaces such as the abdominal cavity, particularly in
discreet sites. Conventional chemotherapy drugs, in contrast, are limited to
surface perfusion on abdominal tissues and lack the local penetration capability
of ionizing radiation. For instance, the capsules of liver and spleen are common
sites for peritoneal metastasis in OC patients. Achieving a high drug
concentration in these local areas is challenging due to their elevated position
and deep narrow cavities. In addition to the therapeutic role in intraabdominal
tumors, the nanoparticles absorbed by the liver and spleen can effectively target
tumors in the capsules and adjacent areas. Furthermore, the PEG long chains on
the nanoparticle surface can bind a significant number of water molecules. This
property contributes to maintaining the small size, monodisperity, and stable
state of the nanoparticles, consequently enhancing their tumor-targeted
aggregation. In addition, PEG plays a crucial role in stabilizing the
nanoparticles, ensuring their persistent retention in tumors [36].
This study has several limitations. We did not explore the potential benefits of
combining systemic and intraperitoneal administration of the nanoparticles for
treatment. Besides, the optimal dosage, long-term efficacy, and safety of
the Lu-FA-DOTA-PEG-PLGA nanoparticles have not been
thoroughly investigated. Furthermore, our understanding on the controlled release
dynamics of nanoparticles in the body remains limited. These aspects merit
increased attention in future research endeavors.
5. Conclusions
We successfully engineered PEG surface-modified Lu-FA-DOTA-PEG-PLGA
long-circulating nanoparticles with a prolonged blood circulation time of 72 h
and a remarkably low renal radioactivity uptake of only 1.646% ID/g. This uptake
was significantly lower than that of previous FR radionuclide drugs. Notably,
these nanoparticles had targeting, degradability, and nuclide internal
irradiation therapeutic properties. Moreover, our findings indicated that the
nanoparticles showed no damage to intestinal tissues, reducing systemic toxicity
and side effects commonly associated with conventional chemotherapy. The
nanoparticles demonstrated notable antitumor efficacy against subcutaneous tumors
via FR targeting. Furthermore, their sustained presence in the peritoneal cavity
enables effective targeting of intraperitoneal metastatic OC tumors, leading to a
reduction in ascitic fluid volume. In summary, the unique properties of the
Lu-FA-DOTA-PEG-PLGA nanoparticles offer significant promise for improving
therapeutic outcomes of OC by addressing challenges associated with drug
delivery.
Availability of Data and Materials
The original data presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Author Contributions
ZG and YD: methodology, formal analysis, data curation, writing. JZ and SS:
investigation, software, writing. JW: conceptualization, supervision,
writing-review & editing. All authors read and approved the final manuscript.
All authors have participated sufficiently in the work and agreed to be
accountable for all aspects of the work.
Ethics Approval and Consent to Participate
All animal care and experimental protocols were approved by the Ethical
Committee of Xuzhou Medical University Experimental Animal Center (No.
202101w015).
Acknowledgment
Not applicable.
Funding
This study was support by the Jiangsu Provincial Health Committee Key scientific
research projects (No. ZD2021053).
Conflict of Interest
The authors declare no conflict of interest.
 Fig. 1.
Fig. 1. Fig. 2.
Fig. 2. Fig. 3.
Fig. 3. Fig. 4.
Fig. 4. Fig. 5.
Fig. 5. Fig. 6.
Fig. 6. Fig. 7.
Fig. 7.






