1 The Center for Reproductive Medicine, Changzhou Maternal and Child Health Care Hospital, Changzhou Medical Center, Nanjing Medical University, 213000 Changzhou, Jiangsu, China
†These authors contributed equally.
Abstract
This study aimed to compare the effects of different gamete co-incubation times on fertilization, embryo development potential, along with clinical outcomes.
The study included 530 cycles with short co-incubation times (4–6 hours), referred to as the S group, and 1653 cycles with long co-incubation times (16–24 hours), referred to as the L group, all undergoing in vitro fertilization (IVF) without rescue intracytoplasmic sperm injection (R-ICSI). The study analyzed the basic characteristics and clinical outcomes of these two groups. Additionally, the study stratified the cycles based on the number of oocytes retrieved and performed multivariate logistic regression analysis with the normal fertilization (two pronuclei, 2PN) rate as the dependent variable and co-incubation time as the main independent variable. The study also examined 79 partial short co-incubation cycles, dividing them into a short co-incubation part (10 oocytes) and a long co-incubation part (the remaining oocytes), to analyze embryo developmental parameters.
There were no significant differences in the rates of top-quality blastocyst formation, clinical pregnancy, and implantation between the two groups (p > 0.05). 2PN rate in S group was slightly lower than in L group (68.06% vs. 71.08%, p < 0.01). After stratifying by the number of oocytes obtained, multiple logistic regression analysis demonstrated no significant correlation between co-incubation time and 2PN rate (p > 0.05). In the 79 partial short co incubation cycles, there were no significant differences in the rates of 2PN oocytes, ≥3PN (3 pronuclei) oocytes, top-quality cleavage embryos, and top-quality blastocysts on Day 5/Day 6 between the two parts (p > 0.05).
Short oocyte-sperm co-incubation is an effective strategy in preventing fertilization failure. Short and long oocyte-sperm co-incubation times have similar outcomes on in vitro development.
Keywords
- short co-incubation
- long co-incubation
- fertilization rate
- reproductive outcomes
In vitro fertilization (IVF) is widely used to treat infertile couples.
Compared with intracytoplasmic sperm injection (ICSI), the combination of oocytes
and sperm in IVF more closely mimics the natural human physiological state [1, 2]. However, even with normal semen parameters, approximately 20% of IVF cycles
result in a low fertilization rate (
There is still no consensus on the optimal duration for oocyte-sperm co-incubation during IVF. Some studies suggest that short co-incubation times (1–6 hours) improve IVF outcomes compared to longer co-incubation times (16–24 hours) [8, 9]. Conversely, other studies have found no such advantage [10, 11]. A previous study indicated that prolonged exposure of oocytes to sperm might harm early embryonic development [12]. Barraud-Lange et al. [13] found that while short co-incubation of gametes reduced the fertilization rate compared to the standard overnight method, embryo quality remained comparable. Fan et al. [9] meta-analysis revealed that reduced gamete co-incubation time benefits clinical pregnancy and implantation rates compared to overnight IVF, with no significant differences in fertilization rates or embryo quality. Our study aimed to investigate these findings further and provide a reference for clinical practice.
We retrospectively analyzed the data of patients receiving IVF/ICSI assisted pregnancy at the Reproductive Center of Changzhou Maternal and Child Health Care Hospital from January 2022 to December 2023. The inclusion criteria: oocyte retrieval cycles. The exclusion criteria: (1) cycles with frozen-thawed oocytes or sperm; (2) patients with chromosomal abnormalities; (3) ICSI cycles; (4) IVF/ICSI split insemination cycles; (5) rescue ICSI cycle. This study included 530 short co-incubation cycles without rescue ICSI as S group, and 1653 long co-incubation cycles without rescue ICSI as L group (Fig. 1). All patients read and signed informed consent forms. The Ethics Committee of Changzhou Maternal and Child Health Care Hospital approved this retrospective study (2022071). All treatments followed the Declaration of Helsinki for Medical Research. We performed multivariate logistic regression analysis to identify gamete co-incubation times associated with the 2PN rate. Furthermore, we analyzed data from 79 partial short co-incubation cycles, comparing fertilization and embryo development between short and long co-incubation cycles.
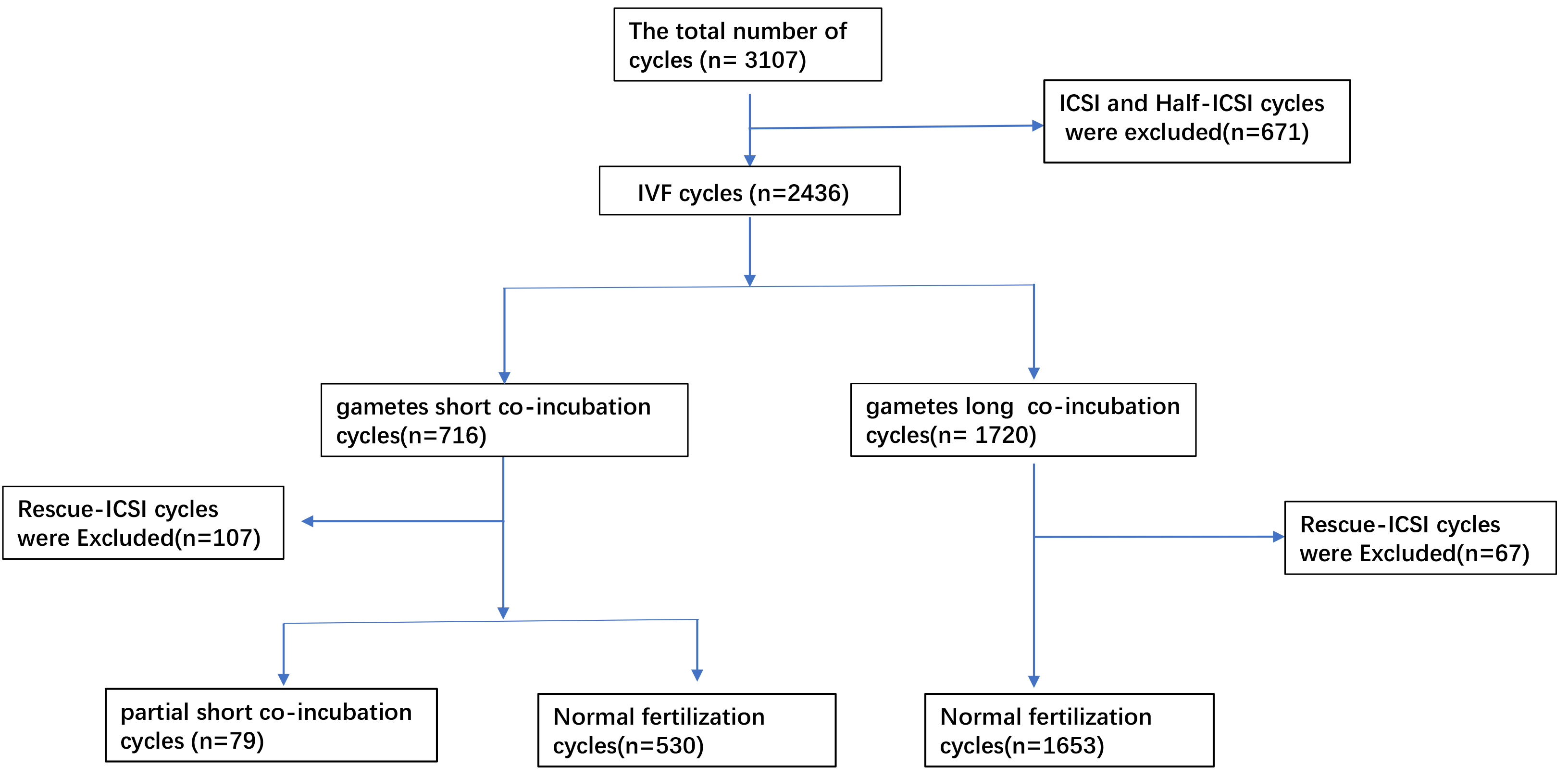 Fig. 1.
Fig. 1.
Flow of data acquisition and analysis. IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection.
Oocytes were retrieved from females undergoing controlled ovarian stimulation using gonadotropin-releasing hormone (GnRH) analogs or other agents, or during a natural cycle. During ovarian stimulation, the daily dose of recombinant follicle stimulating hormone or human menopausal gonadotropin (FSH/HMG) was adjusted according to the size of the follicles observed on ultrasound. When at least three dominant follicles reached a diameter of 18 mm, 5000 IU of human chorionic gonadotropin (HCG) was administered to trigger oocyte maturation. Oocytes were extracted approximately 36 hours after triggering.
IVF insemination was adopted when sperm concentration was
All analyses were performed using IBM SPSS Statistics 21 (IBM Corp., Chicago,
IL, USA). The Chi-square test was used to compare categorical data. Normality was
tested for continuous data. The Student’s t-test was used to compare
normally distributed data. Non-normally distributed data were compared using the
Mann-Whitney U test. Multivariate logistic regression was used to analyze the
relationship between the 2PN rate and co-incubation time. Combined odds ratios
(OR) and 95% confidence intervals (95% CI) were calculated for the data. A
p-value of
530 short co-incubation cycles with normal fertilization in the S group and 1653
long co-incubation cycles with normal fertilization in the L group were included.
There were no significant differences between the 2 groups in terms of female
body mass index (BMI), basal estrogen (E2) levels,
fallopian tube factors, or endometriosis factors (p
| S Group | L Group | p-value | ||
| No. of patients (n) | 530 | 1653 | ||
| Primary infertility (%) | 379/530 (71.51%) | 626/1653 (37.87%) | ||
| Infertility duration (years) | 3 [2, 5] | 2 [1, 4] | ||
| Age (years) | ||||
| Female | 32 [29, 35] | 33 [31, 36] | ||
| Male | 31 [28, 34] | 33 [30, 36] | ||
| BMI of female (kg/m2) | 22.8 [20.4, 25.8] | 22.8 [20.7, 25.6] | 0.82b | |
| Primary diagnosis | ||||
| Tubal factor (%) | 220/530 (41.51%) | 736/1653 (44.53%) | 0.22a | |
| Endometriosis (%) | 26/530 (4.91%) | 54/1653 (3.27%) | 0.08a | |
| DOR (%) | 34/530 (6.42%) | 349/1653 (21.11%) | ||
| Male factors (%) | 38/530 (7.17%) | 53/1653 (3.21%) | ||
| Ovulation disorders (%) | 77/530 (14.53%) | 109/1653 (6.59%) | ||
| Unexplainable factor (%) | 106/530 (20.00%) | 182/1653 (11.01%) | ||
| Female hormone levels | ||||
| Basal FSH (IU/L) | 6.04 [5.11, 7.21] | 6.55 [5.43, 8.11] | ||
| Basal E2 (ng/L) | 38.16 [29.38, 48.26] | 38.71 [28.97, 50.88] | 0.48b | |
| Basal LH (IU/L) | 5.33 [3.93, 7.31] | 4.54 [3.27, 6.19] | ||
| AMH (mg/dL) | 3.22 [2.05, 4.88] | 1.83 [0.86, 3.65] | ||
| Simulation protocol | ||||
| GnRH agonist (%) | 335/530 (63.21%) | 670/1653 (40.53%) | ||
| GnRH antagonist (%) | 172/530 (32.45%) | 385/1653 (23.29%) | ||
| Mild stimulation (%) | 23/530 (4.34%) | 598/1653 (36.18%) | ||
| Total dose of Gn | 1650.00 [1237.50, 2362.50] | 1500.00 [1050.00, 2218.75] | ||
AMH, anti-Müllerian hormone; DOR, declined ovarian reserve; Gn, gonadotropin; GnRH, gonadotropin-releasing hormone; BMI, body mass index; FSH, follicle stimulating hormone; E2, basal estrogen; LH, luteinizing hormone. Data are presented as the median [the first quartile, the third quartile] or count (percentage). a, Chi-squared test; b, Mann–Whitney U test.
The rate of
| S Group (n = 530) | L Group (n = 1653) | p-value | ||
| Average oocytes retrieved (n) | 11 [8, 14] | 7 [3, 12] | ||
| Average MII oocytes (n) | 10 [7, 13] | 6 [3, 11] | ||
| 2PN oocytes rate (%) | 4057/5961 (68.06%) | 10,048/14,136 (71.08%) | ||
| 519/5961 (8.71%) | 1125/14,136 (7.96%) | 0.08a | ||
| Top cleavage embryos from 2PN (%) | 3339/4726 (70.65%) | 7658/11,495 (66.62%) | ||
| Top blastocyst formation rate (%) | 1722/3225 (53.40%) | 3809/7331 (51.96%) | 0.17a | |
| Fresh ET cycles | 0.82a | |||
| Cleavage embryo | 155 (74.16%) | 267 (75.00%) | ||
| Blastocyst | 54 (25.84%) | 89 (25.00%) | ||
| No. of embryos transferred | 0.70a | |||
| Cleavage embryo | 274 (82.78%) | 460 (83.79%) | ||
| Blastocyst | 57 (17.22%) | 89 (16.21%) | ||
| Clinical pregnancy rate (first ET) | ||||
| Cleavage embryo | 87/155 (56.13%) | 154/267 (57.68%) | 0.76a | |
| Blastocyst | 28/54 (51.85%) | 49/89 (55.06%) | 0.70a | |
| Implantation rate (%) | ||||
| Cleavage embryo | 111/274 (40.51%) | 196/460 (42.61%) | 0.57a | |
| Blastocyst | 29/57 (50.88%) | 49/89 (55.06%) | 0.62a | |
Data are presented as the median [the first quartile, the third quartile] or count (percentage). a, Chi-squared test; b, Mann–Whitney U test; ET, embryo transfer; MⅡ, metaphase Ⅱ. 2PN, two pronuclei; 3PN, three pronuclei; 2PN oocytes rate (%) = 2PN/total number of cumulus oocyte complexes. Clinical pregnancy rate = No. of clinical pregnancy/fresh ET cycles.
We stratified the number of oocytes obtained and conducted a regression analysis
to further evaluate the association between co-incubation time and 2PN.
Multivariate logistic regression analysis was performed with the 2PN rate as the
dependent variable (
| Oocytes retrieved | Gamete co-incubation time | Adjusted OR (95% CI) | p-value |
| 1–10 | S group | Ref | |
| L group | 1.263 (0.922–1.729) | 0.146 | |
| 11–20 | S group | Ref | |
| L group | 1.340 (0.937–1.917) | 0.109 | |
| S group | Ref | ||
| L group | 0.925 (0.301–2.845) | 0.892 |
AMH, anti-Müllerian hormone; Ref, reference group; OR, odds ratio; CI, confidence interval. Multivariate logistic regression analysis for association between 2PN and co-incubation time controlling for sterility type, female age, AMH, average MⅡ oocytes, and simulation protocol.
In the partial short co-incubation cycles, the gametes were split into the short
part (10 oocytes) and the long part (the remaining oocytes) for co-incubation
(Table 4). We analyzed embryo developmental parameters in the 2 parts. The rates
of MⅡ oocytes, 2PN oocytes,
| Parameter | Value | |
| No. of cycles (n) | 79 | |
| Female age, mean |
29.59 | |
| Duration of infertility (years) | 3 [2, 5] | |
| Primary infertility (%) | 15/79 (18.99%) | |
| Average oocytes retrieved (n) | 23 [20, 27] | |
| BMI (kg/m2) | 22.60 [20.50, 27.40] | |
| Female hormone levels | ||
| AMH (mg/dL) | 5.80 [3.97, 8.79] | |
| Baseline FSH (IU/L) | 5.38 | |
| Baseline LH (IU/L) | 5.73 [4.41, 9.29] | |
| Baseline E2 (ng/L) | 38.03 [29.63, 49.71] | |
| Simulation protocol | ||
| GnRH agonist (%) | 31/79 (39.24%) | |
| GnRH antagonist (%) | 46/79 (58.23%) | |
| Diagnoses | ||
| Tubal factor, n (%) | 27/79 (34.18%) | |
| Ovulatory obstacle, n (%) | 23/79 (29.11%) | |
| Unexplainable factor, (%) | 13/79 (16.46%) | |
AMH, anti-Müllerian hormone; BMI, body mass index; FSH, follicle stimulating hormone; LH, luteinizing hormone; E2, basal estrogen; GnRH, gonadotropin releasing hormone; SD, standard deviation. Data are presented as the median [the first quartile, the third quartile] or count (percentage).
| Short co-incubation | Long co-incubation | p-value | |
| No. of oocytes (n) | 790 | 1102 | |
| MII oocytes rate (%) | 712/790 (90.13%) | 962/1102 (87.30%) | 0.060a |
| 2PN oocytes rate (%) | 570/712 (80.06%) | 761/962 (79.11%) | 0.634a |
| 73/712 (10.26%) | 98/962 (10.19%) | 0.965a | |
| Top-quality cleavage embryo rate (%) | 446/570 (78.25%) | 607/761 (79.76%) | 0.500a |
| Top-quality blastocyst embryo rate-Day 5 (%) | 207/533 (38.84%) | 305/741 (41.16%) | 0.404a |
| Top-quality blastocyst embryo rate-Day 6 (%) | 78/533 (14.63%) | 97/741 (13.09%) | 0.430a |
Data are presented as count (percentage). MⅡ, metaphase Ⅱ; a, Chi-squared test.
Conventional IVF is a widely used treatment
for female infertility factors, unexplained infertility, and some male
infertility factors. Total fertilization failure (TFF) remains challenging for
clinicians and embryologists [17]. Short-term fertilization is a preferable
method to avoid TFF, leading to the widespread use of short co-incubation of
gametes combined with early rescue ICSI. It has been suggested that early removal
of granulosa cells is beneficial for embryo quality [18], but some studies have
shown conflicting results [10, 11]. Our study found a trend toward a higher
polyspermy rate in the S group (8.71%) compared to the L group (7.96%),
although this difference was not statistically significant. The top cleavage
embryo rate was significantly higher in the S group, which can be attributed to
the younger mean age and better ovarian function compared to the L group. In
partial short fertilization, there was no discernible difference in the top
cleavage embryo rate between the two groups. The rates of
top-quality blastocyst, clinical pregnancy and implantation rates showed no
significant differences between the two groups (p
Gianaroli et al. [12] indicated that sperm enter cumulus cells within
15 minutes, interact with them within 1 hour, and reach the oocyte cortex 4 hours
later. These findings suggest that prolonged co-incubation of oocytes and sperm
may not be necessary. Additionally, some researchers have found that prolonged
co-incubation time may lead to high levels of reactive oxygen species (ROS) [5],
which can negatively affect gamete interactions and embryo quality. However, Kong
et al. [20] suggested that removing the cumulus-oocyte complex (COC) too
early may interfere with the crucial communication between oocytes and the COC.
While short co-incubation can decrease ROS levels, it may not provide sufficient
time for oocyte maturation compared to longer incubation [20, 21]. We found that
the normal fertilization rate (2PN) was slightly lower in the S group compared to
the L group, with statistical significance (p
There are some limitations to our study. Due to the nature of retrospective study, it is possible that important confounder was not taken into account in the present study. It is a single-center study with a relatively small sample size. Cycles with three or fewer eggs, overnight fertilization was used to avoid, and fresh embryo transfer is not performed in these cycles in our center, which may potentially introduce bias. The live birth rates were not included in the results. Our plan is to accumulate more cases to further verify the conclusions.
Short-term insemination does not affect the fertilization rate and embryo development [3]. Furthermore, short co-incubation may require embryologists to work at night, resulting in longer working hours. Therefore, it is unnecessary to choose short fertilization in all IVF cycles. Clinicians should identify patients at risk for low or complete fertilization failure, and the fertilization method should be based on the history of infertility, the experience of clinicians and embryology laboratory personnel, and the patient’s medical history. The inclusion of additional procedures during fertilization evaluation may pose a potential risk of adverse effects on the fertilized egg [23]. Therefore, the method of selecting only some fertilized eggs, rather than all fertilized eggs for polar body observation helps to reduce the risk. Partial short insemination may be an appropriate method to reduce IVF fertilization failure or low fertilization rates.
Short and long oocyte-sperm co-incubation times have similar in vitro developmental outcomes. It is safe to use short-term fertilization for patients who may be at risk of fertilization failure.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
FC: investigation; formal analysis; writing—original draft; data curation. LL: investigation; formal analysis; writing—original draft. XD: formal analysis; writing—original draft. XX: formal analysis; writing—original draft. LC: conceptualization; methodology; writing—review and editing; funding acquisition. YW: conceptualization; methodology; writing—review and editing; funding acquisition; project administration. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Changzhou Maternal and Child Health Care Hospital (approval number: 2022071).
We gratefully acknowledge the assistance and instruction from Dr. Zhu of the Department of Reproductive Medicine Center.
This research was funded by the China Reproductive Public Welfare Fund “Pilotage Plan” (grant SZ202412) and the Key Project of Changzhou Clinical Medical Center (grant CMCM202203).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

