1 Department of Obstetrics and Gynecology, Affiliated Matern & Child Care Hospital of Nantong University, 226000 Nantong, Jiangsu, China
Abstract
Although cervical cerclage has improved with the rapid development of medical technology, there remains a distinct probability of adverse pregnancy outcomes. To investigate the associations of changes in the high mobility group box 1 (HMGB1), soluble tumor necrosis factor receptor 1 (sTNFR-1) and peripheral blood neutrophil-to-lymphocyte ratio (NLR) with premature delivery secondary to infection in pregnant patients undergoing cervical cerclage.
Sixty-seven pregnant patients with premature delivery after cervical cerclage, who were treated at the Affiliated Matern & Child Care Hospital of Nantong University from January 2022 to October 2023, were enrolled, including 43 with premature delivery secondary to infection (infectious group) and 24 with idiopathic premature delivery (non-infectious group). The pre-delivery serum levels of HMGB1, sTNFR-1 and the peripheral blood level of NLR were compared between the 2 groups. Further, the clinical value of these 3 indicators in predicting premature delivery secondary to infection among pregnant patients undergoing cervical cerclage was assessed by receiver operating characteristic (ROC) curve analysis.
The infectious group exhibited significantly higher serum levels of HMGB1, sTNFR-1 and peripheral blood level of NLR compared to those in the non-infectious group, demonstrating significant differences (p < 0.05). Logistic regression analysis revealed that HMGB1 and NLR were independent influencing factors for premature delivery (p < 0.05). According to the ROC curve analysis results, the changes in HMGB1, sTNFR-1 and NLR levels may reflect the risk of premature delivery secondary to infection among pregnant patients undergoing cervical cerclage. The area under the curve (AUC), sensitivity and specificity of combined detection were all markedly higher than those of independent detection.
HMGB1, sTNFR-1 and NLR levels are risk factors for third-trimester premature delivery among pregnant patients undergoing cervical cerclage. Timely combined detection of serum HMGB1, sTNFR-1 and peripheral blood NLR during the third trimester can improve the clinical diagnostic acumen, which enables early prevention to help lower the risk of premature delivery.
Keywords
- cervical cerclage
- cervical insufficiency
- HMGB1
- sTNFR-1
- NLR
- infection
- premature delivery
Premature delivery, as a common obstetric occurrence, refers to childbirth occurring at 28–36 weeks+6 of gestation, which may lead to neonatal organ dysfunction and may seriously affect the pregnancy outcome. Cervical insufficiency (CIC), as a major risk factor for premature delivery, is defined as abnormal cervical structure and impaired cervical function caused by congenital developmental factors or acquired injury, whose clinical findings include cervical canal shortening and cervical dilation in the second and third trimesters of pregnancy [1]. Cervical cerclage is the only confirmed effective operation for treating CIC, with surgical closure of the cervical canal being performed to prevent further dilation. Thereby mitigating the fetal pressure on the internal cervical orifice and effectively prolonging the time to delivery of women with CIC [2]. However, recent research has shown that some pregnant patients still experience adverse outcomes such as premature delivery after cervical cerclage [3]. Actively searching for the risk factors of premature delivery following cervical cerclage in CIC pregnant patients is of great significance in order to lower the incidence of adverse pregnancy outcomes.
Depending on the presence or absence of infective factors, premature delivery
occurring in cervical cerclage patients can be divided into infectious and
non-infectious types. Early diagnosis of premature delivery secondary to
infection can provide a reference value in guiding early anti-infective therapy
for pregnancy prolongation [4, 5]. Research of serological cytokines can offer
reference for the clinical diagnosis of infectious diseases. High mobility group
box 1 (HMGB1) is a chemotaxis related factor, whose infiltrating effect on
uterine smooth muscle cells can induce the contraction of the cells, increasing
the risk for premature delivery [6, 7, 8, 9]. Soluble tumor necrosis factor receptor 1
(sTNFR-1) can induce the activation of downstream tumor necrosis factor alpha
(TNF-
To this end, this study demonstrated the correlations of the serum HMGB1, sTNFR-1 and peripheral blood NLR expressions with premature delivery secondary to infection in cervical cerclage patients, which provides a reference for the early diagnosis and timely intervention of premature delivery secondary to infection among pregnant patients undergoing cervical cerclage, with a view to providing a certain level of protection for maternal and fetal health for pregnant patients with CIC.
A total of 67 CIC patients, who had premature delivery at 28–36 weeks+6 of gestation after receiving transvaginal cervical cerclage at the Affiliated Matern & Child Care Hospital of Nantong University from January 2022 to October 2023, were enrolled, including 43 with premature delivery secondary to infection (infectious group) and 24 with non-infectious premature deliveries (non-infectious group). Diagnostic and inclusion criteria: (1) patients with clinically confirmed CIC who underwent elective cervical cerclage at 14–18 weeks of gestation; (2) gestational age of premature delivery ranging from 28 to 36 weeks+6; (3) single pregnancy; (4) positive pathogenic microorganisms in the cervical secretion culture of pregnant patients having premature delivery secondary to infection. Exclusion criteria: (1) diseases of the immune system; (2) abnormal fetal position; (3) multiple pregnancy; (4) pregnancy-induced hypertension, gestational diabetes mellitus; (5) intrahepatic cholestasis of pregnancy; (6) placental abruption, placenta previa; (7) hyperthyroidism; (8) chromosomal abnormality and other congenital defects.
McDonald’s cerclage was utilized [13]. The patient underwent spinal anesthesia, placed in a lithotomy position, and the vulva and vagina were disinfected. Alex forceps were used to grasp the cervix and pull it downwards. The needle was inserted at 11 o’clock in the cervix, then removed at 10 o’clock, followed by insertion at 8 o’clock and removal at 7 o’clock, insertion at 5 o’clock and removal at 4 o’clock, insertion at 2 o’clock and removal at 1 o’clock, avoiding the vascular plexus in the 3 o’clock and 9 o’clock directions. A knot was tied in the anterior fornix, leaving 3 cm for suture removal. After suturing, it is advisable to use a #4 Hegar dilator that can be accommodated in the cervical opening. For cases where the amniotic sac protrudes from the cervical canal, it is advisable to use the water bag blockage method to retract the amniotic sac and suture to close the cervical canal. After surgery, the patients received intravenous magnesium sulfate for protection of fetus, inhibition of uterine contractions and prevention of infection. Additionally, the patients were required to rest in bed and abstain from sexual activity. Ultrasound examination was performed 2 weeks after surgery, and stitches were removed before delivery or after 37 weeks of pregnancy, or at any time if signs of uncontrollable contractions or infection were present.
Blood samples (each 5 mL) were collected from patients after hospitalization due
to premature delivery, and centrifuged at 3500 r/min for 20 min with a
centrifugal radius of 10 cm. The sera were separated for detection of HMGB1 and
sTNFR-1 levels via peroxidase kit (Evermed Biomedical, SuXie Zhuzhun
20172401449, Suzhou, Jiangsu, China, model: 24 mL). Routine blood routine tests were performed with a
LH750 automatic blood cell analyzer (Beckman Coulter, Brea, CA, USA), and data
including leukocyte, neutrophil, lymphocyte counts and NLR were obtained
(reference range: HMGB1: 1.25–80 ng/mL, sTNFR-1: 0.93
SPSS 20.0 software (IBM Corp., Chicago, IL, USA) was utilized. The normality
test method employed is the Shapiro-Wilk (SW) test. Measurement data were
compared by t test and expressed as mean
No significant differences in the basic clinical data were noted between the 2
groups (p
| Group General information | Number of cases (n) | Age (years) | Delivery gestational weeks (weeks) | Number of pregnancies (times) | Number of deliveries (times) |
|---|---|---|---|---|---|
| Infectious group premature delivery | 43 | 26.98 |
32.82 |
1.98 |
1.47 |
| Non-infectious group premature delivery | 24 | 27.12 |
33.13 |
1.80 |
1.42 |
| t | –0.19 | –0.47 | 0.87 | 0.36 | |
| p value | 0.85 | 0.64 | 0.39 | 0.72 |
Comparison revealed that the pre-delivery serum HMGB1, sTNFR-1, neutrophil,
leukocyte and NLR levels were all significantly higher in the infectious group
than in the non-infectious group, showing statistical significance (p
| Group Indicator | Number of cases (n) | HMGB1 (mg/L) | sTNFR-1 (pg/mL) | Neutrophil (109/L) | Lymphocyte (×109/L) | Leukocyte (×109/L) | NLR |
| Infectious group premature delivery | 43 | 5.40 |
4.06 |
10.06 |
1.72 |
10.82 |
6.51 |
| Non-infectious group premature delivery | 24 | 3.85 |
3.69 |
6.63 |
1.94 |
9.03 |
3.60 |
| t | 4.43 | 2.05 | 5.98 | –1.91 | 2.54 | 5.97 | |
| p value | 0.06 |
HMGB1, high mobility group box 1; sTNFR-1, soluble tumor necrosis factor receptor 1; NLR, neutrophil-to-lymphocyte ratio; SD, standard deviation.
For regression analysis on the associations of HMGB1, sTNFR-1 and NLR with the
premature delivery in pregnant women undergoing cervical cerclage, a binary
regression model was created using HMGB1, sTNFR-1 and NLR as the independent
variables and the occurrence of premature delivery as the dependent variable (1 =
infectious group, 0 = non-infectious group). According to the binary logistic
regression results, the expressions of HMGB1, sTNFR-1 and NLR were upregulated,
and HMGB1 and NLR were independent risk factors for the premature delivery in
cervical cerclage patients (odds ratio (OR)
| Indicator Result | Standard error | Wald | OR | 95% CI | p value | Collinearity statistics | ||
| Tolerance | Variance inflation factor | |||||||
| Serum HMGB1 level | 1.317 | 0.532 | 6.126 | 3.730 | 1.315–10.581 | 0.013 | 0.359 | 2.786 |
| Serum sTNFR-1 level | –1.713 | 0.879 | 3.794 | 0.180 | 0.032–1.011 | 0.051 | 0.385 | 2.600 |
| NLR | 0.555 | 0.178 | 9.663 | 1.741 | 1.227–2.470 | 0.002 | 0.864 | 1.157 |
OR, odds ratio; 95% CI, 95% confidence interval.
As revealed by the ROC analysis evaluating the clinical value of HMGB1, sTNFR-1 and NLR levels in predicting the incidence of premature delivery secondary to infection among cervical cerclage patients, the area under the curve (AUC), sensitivity and specificity of prediction by serum HMGB1 were 0.740, 53.49% and 87.50%, respectively. The serum sTNFR-1 exhibited an AUC of 0.626, a sensitivity of 51.16% and a specificity of 79.17% for predicting premature delivery secondary to infection. The corresponding AUC, sensitivity and specificity for NLR were 0.825, 69.77% and 91.67%, respectively. The joint prediction with HMGB1, sTNFR-1 and NLR yielded an AUC of 0.883, a sensitivity of 79.07%, and a specificity of 100% (see Table 4 and Fig. 1).
| Indicator | Youden | Sensitivity (%) | Specificity (%) | False negative rate (%) | False positive rate (%) | AUC | 95% CI |
| Serum HMGB1 level | 0.410 | 53.49 | 87.50 | 46.51 | 12.50 | 0.740 | 0.619–0.840 |
| Serum sTNFR-1 level | 0.303 | 51.16 | 79.17 | 48.84 | 20.83 | 0.626 | 0.500–0.742 |
| NLR | 0.614 | 69.77 | 91.67 | 30.23 | 8.33 | 0.825 | 0.712–0.907 |
| Joint prediction | 0.791 | 79.07 | 100.00 | 20.93 | 0.00 | 0.883 | 0.781–0.948 |
AUC, area under the curve.
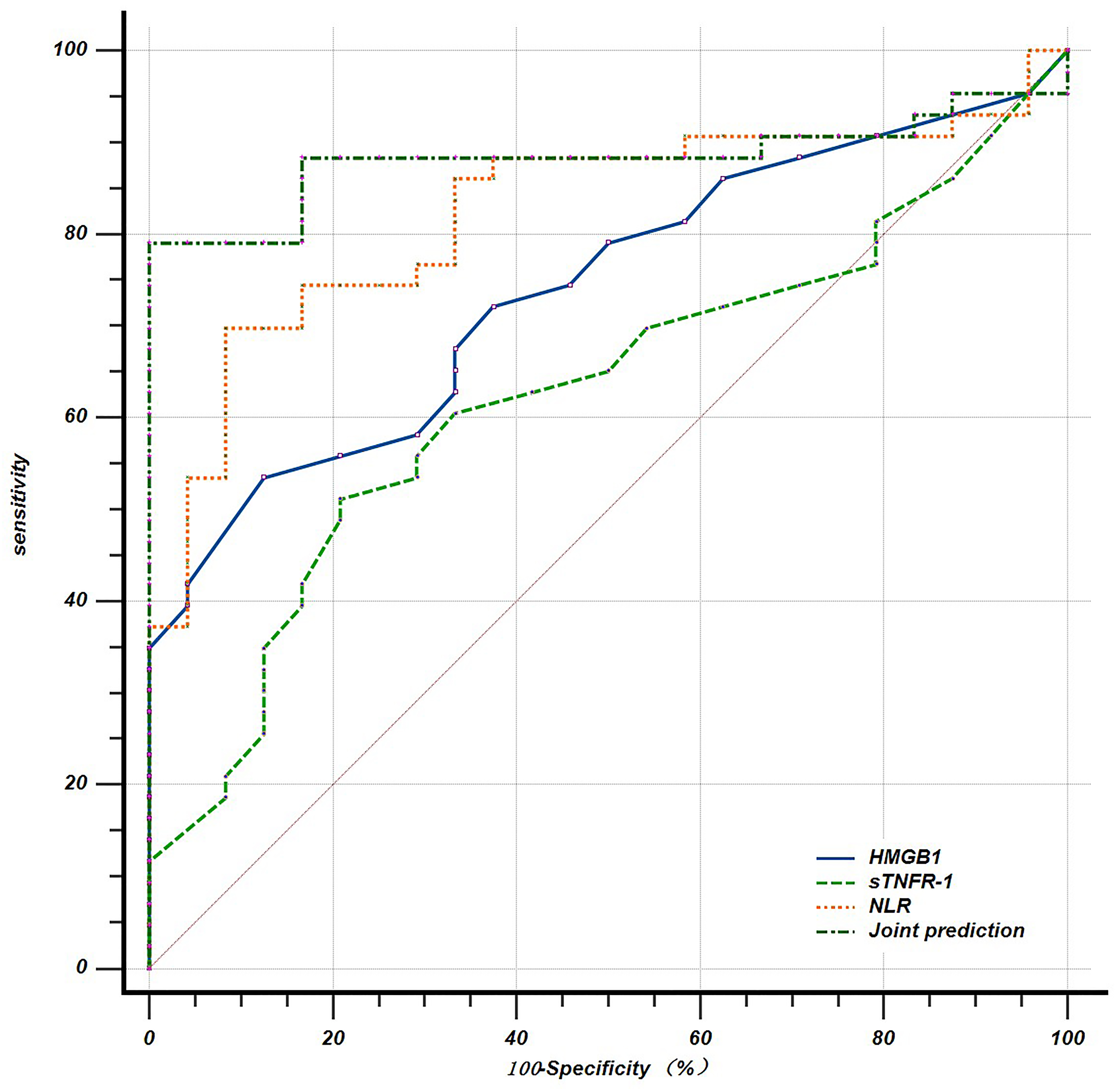 Fig. 1.
Fig. 1.
ROC curves for the independent and joint prediction of premature delivery secondary to infection by HMGB1s, sTNFR-1 and NLR in cervical cerclage patients. ROC, receiver operating characteristic.
Premature birth is one of the primary causes of perinatal mortality. While cervical cerclage can potentially prevent premature birth due to cervical laxity, a certain number of patients still experience premature birth following cervical cerclage. Infection is a significant risk factor for premature birth, particularly amniotic cavity infection triggered by pathogenic microorganisms in amniotic fluid [14]. This type of infection is an asymptomatic intrauterine infection that can only be detected during pathological examination. Therefore, actively seeking biological indicators to evaluate the occurrence of chorioamnionitis is of great significance for the diagnosis and treatment of potential infection and the prevention of premature birth caused by infection after cervical cerclage.
As a member of the chemokine family, HMGB1 induces the softening and relaxation
of the internal cervical orifice by recruiting the downstream complement or
chemokine C3a; low HMGB1 level can recruit the immune cells and promote the
proliferation and differentiation of endometrial stromal cells required for
normal pregnancy, while high HMGB1 level is associated with pregnancy failure
[15, 16]. Endotoxins generated from the metabolism of pathogenic microorganisms
after infection, along with various inflammatory factors released during the
inflammatory response, can trigger the release of HMGB1, which mediates the
inflammatory response. Conversely, HMGB1 can also stimulate monocytes to secrete
pro-inflammatory factors, such as sTNFR-1 and interleukin 1 (IL-1), thereby
exacerbating the inflammatory condition [17]. sTNFR-1 is a member of the TNFR
family, whose regulatory effects on different TNF-
In this study, based on the changes in the serum levels of HMGB1, sTNFR-1 and
NLR detected among pregnant patients with premature delivery following cervical
cerclage, those positive for pathogenic microorganisms in cervical secretion
culture were assigned to the infectious group, while those negative to pathogenic
microorganisms were assigned to the non-infectious group. It was found that the
levels of HMGB1, sTNFR-1 and NLR in the infectious group were all significantly
higher than those in the non-infectious group, suggesting that the high
expression of serum HMGB1, sTNFR-1 and NLR could all affect the occurrence of
premature delivery secondary to infection in cervical cerclage patients. Findings
of relevant cause analysis were as follows: (1) increased expression of HMGB1
could increase the degree of oxytocin receptor upregulation on the uterine smooth
muscle cell membrane by inducing the activation of downstream nuclear factor
Through the multivariate logistic regression analysis of serological indicators in pregnant patients having premature delivery after cervical cerclage, this study found that the serum HMGB1 level and NLR were independent risk factors for premature delivery, which had certain value in the independent diagnosis of premature delivery secondary to infection among pregnant patients undergoing cervical cerclage. ROC analysis revealed that the peripheral blood NLR had high sensitivity and specificity for predicting premature delivery secondary to infection in cervical cerclage patients, with an AUC of 0.825. Consistent with previous findings, close monitoring of NLR levels facilitated early prediction of premature delivery [25]. Additionally, joint prediction by serum HMGB1, sTNFR-1 and NLR exhibited the maximum AUC, which was nearly 0.9. In clinical practice, microbes are usually cultured to determine whether infection is present. However, given the long cycle of microbial culture and the fast progression of premature delivery, the optimal intervention time may be missed. Serological indicators are characterized by fast detection. Through the detection of serum HMGB1, sTNFR-1 and NLR levels in pregnant patients with signs of premature delivery, early screening of and anti-infective therapy for premature delivery secondary to infection are possible, which can control the patient’s condition as soon as possible, prolong the gestational age, and improve the maternal and fetal pregnancy outcomes. Due to the small specimen size of this study and the failure to compare preoperative serological indicators, there are still limitations in our conclusions. In future research, more clinical data should be included to reduce experimental errors, factors associated with premature delivery secondary to infection in pregnant patients undergoing cervical cerclage should be further analyzed, relevant measures should be formulated, and the pregnancy outcome of cervical cerclage patients after receiving active intervention should be included to further corroborate these findings. Although explicit external infections have been excluded, some individual patients may have latent infections outside the reproductive tract or amniotic cavity. These infections could potentially introduce bias into the research results, particularly in studies with small sample sizes.
When the pregnant patients undergoing cervical cerclage have premature delivery secondary to infection, their serum levels of HMGB1, sTNFR-1 and NLR are significantly upregulated. Joint prediction by serum HMGB1, sTNFR-1 and NLR has a crucial referential value in diagnosing premature delivery secondary to infection among the cervical cerclage patients. Early diagnosis of the risk for premature delivery secondary to infection and initiation of early anti-infective therapy are necessary, in order to avoid premature delivery in cervical cerclage patients, gain time for early treatment, control the disease as early as possible, prolong the gestational age, and improve both the maternal and fetal pregnancy outcomes.
All data generated or analysed during this study are included in this published article.
SC: experimental design, case collection and article writing. YW: case collection and data analysis. YD: cervical cerclage operator. LZ: experimental design, review of experimental process. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Affiliated Matern & Child Care Hospital of Nantong University (approval number: Y2017031).
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This study was funded by Nantong Science and Technology Bureau Social Livelihood Science and Technology Plan Project (Grant No: MSZ2022035) and Nantong Youth Talent Project (Grant No: YQY202310) and Nantong Innovation Team Project (Grant No: NTXK202302).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

