1 Department of Obstetrics and Gynecology, Zhongshan Hospital of Xiamen University, School of Medicine, Xiamen University, 361004 Xiamen, Fujian, China
2 Department of Basic Medicine, School of Medicine, Xiamen University, 361102 Xiamen, Fujian, China
3 Department of Endocrinology, Zhongshan Hospital of Xiamen University, School of Medicine, Xiamen University, 361004 Xiamen, Fujian, China
†These authors contributed equally.
Abstract
The effects of hydroxychloroquine (HCQ) on maternal and fetal outcomes in pregnant patients with systemic lupus erythematosus (SLE) have not been fully elucidated in recent research. This study aimed to provide a more comprehensive evaluation of the efficacy and safety of HCQ in pregnant patients with SLE.
A systematic search of relevant articles was conducted using Medline, Embase, Scopus, Web of Science, and the Cochrane Library from inception to October 4th, 2023. The risk of bias in the included studies was assessed using the Newcastle-Ottawa Scale or Cochrane's Quality Assessment Form. Data on maternal and fetal outcomes of the HCQ treatment groups (HCQ+) and control groups (HCQ–) were extracted, analyzed and evaluated. Pooled odds ratios (ORs), pooled relative risks (RRs), and weighted mean differences with 95% confidence intervals (95% CIs) were calculated for the meta-analysis using Stata/MP V.18.
This meta-analysis included 17 studies, involving a total of 2238 pregnant patients with SLE. Compared to the HCQ– groups, the HCQ+ groups exhibited a significant reduction in the incidence of preeclampsia (RR = 0.51, 95% CI: 0.37–0.71), high lupus activity (RR = 0.77, 95% CI: 0.64–0.92), gestational hypertension (OR = 0.41, 95% CI: 0.18–0.91), premature delivery (RR = 0.71, 95% CI: 0.60–0.84), and fetal growth restriction (OR = 0.61, 95% CI: 0.43–0.85). Additionally, the HCQ+ groups exhibited a significant increase in fetal birth weight of 0.21 kg (95% CI: 0.09–0.33). No significant differences were revealed in the other 15 pregnancy outcomes between the two groups.
Despite the presence of a certain heterogeneity among the included studies, this study provides a more comprehensive evaluation of the efficacy and safety of HCQ in pregnant patients with SLE.
Keywords
- systemic lupus erythematosus
- pregnancy
- hydroxychloroquine
- maternal outcomes
- fetal outcomes
- meta-analysis
Systemic lupus erythematosus (SLE) is a serious and chronic autoimmune disease involving multiple organs [1]. It particularly affects women of childbearing age [2]. Multiple large-scale populationbased studies and an extensive metaanalysis of pregnant patients with SLE have demonstrated an increased risk of a range of maternal and fetal adverse pregnancy outcomes (APOs), including hypertensive disorders of pregnancy (HDP), premature delivery, and fetal growth restriction (FGR) [3, 4, 5, 6, 7, 8, 9]. Therefore, the management of pregnant women with SLE is of vital importance during pregnancy [3].
In 2020, the American College of Rheumatology Guideline recommended that all pregnant patients with SLE should take hydroxychloroquine (HCQ) if possible [10]. Moreover, several clinical studies have investigated its impact on maternal and fetal outcomes in pregnant patients with SLE [11]. In the literature to date, four meta-analysis studies have partially summarized the effects of HCQ [11, 12, 13, 14].
In 2018, Guillotin et al. [11] reported that HCQ was ineffective at preventing prematurity and FGR during SLE pregnancies. In 2021, Duan et al. [12] demonstrated that HCQ treatment resulted in a decrease in the incidence of preeclampsia, gestational hypertension, and prematurity among pregnant patients with SLE. A study conducted by Clowse et al. [13] in 2022 revealed a decrease in lupus activity among women who received HCQ during pregnancy. In 2023, Hu et al. [14] reported that the use of HCQ during pregnancy reduced the risk of high lupus activity and preeclampsia in SLE patients.
However, the effects of HCQ on maternal and fetal outcomes in pregnant patients with SLE have not been fully elucidated. This systematic review and meta-analysis aimed to provide a more comprehensive evaluation of the efficacy and safety of HCQ during pregnancy among individuals with SLE.
This study was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines [15]. The relevant articles were identified through a comprehensive literature search conducted across multiple databases, including Medline, Embase, Scopus, Web of Science, and the Cochrane Library. The search process included articles published from the inception of the databases through October 4th, 2023. During the search process, specific terms such as “systemic lupus erythematosus”, “pregnancy” and “hydroxychloroquine” were utilized (Supplementary File 1). Two authors (AHL and YWY) worked independently on this step, while another author (XWL) resolved any discrepancies between their results. This systematic review has been registered on PROSPERO (CRD42023485388).
We included studies that met the following criteria: (1) case-control studies, cohort studies, or randomized controlled trials; (2) studies comparing pregnant patients with SLE who received HCQ treatment (HCQ+ groups) versus those without HCQ treatment (HCQ– groups); (3) studies reporting maternal and/or fetal outcomes in both groups; and (4) studies with a minimum sample size of 10 patients in each group.
We excluded studies that met the following criteria: (1) case reports, case series, reviews, or in vivo/vitro studies; (2) studies lacking a control group; (3) studies without available data on maternal and/or fetal outcomes; and (4) duplicate studies.
The following 10 maternal outcomes were measured: preeclampsia, high lupus
activity, gestational hypertension, premature rupture of membrane, spontaneous
abortion, oligohydramnios, thrombotic disease, gestational diabetes mellitus,
hemolysis, elevated liver enzymes, and low platelet count syndrome (HELLP)
syndrome, and immune thrombocytopenia. Additionally, we investigated the
following 11 fetal outcomes: premature delivery, FGR, birth weight, miscarriage,
stillbirth, congenital malformation, intrauterine distress, gestational age at
delivery, five-minute Apgar score
The activity of lupus is commonly assessed using the SLE Disease Activity Index
(SLEDAI) [16]. According to the given definition, a pregnant patient with a
SLEDAI score of
The data from eligible studies were independently extracted by two authors (AHL and YWY) and organized into a two-by-two table. Any discrepancies were resolved through discussions with another author (XWL). The information collected included the author’s name, region, period, design, pregnancy outcomes, sample size of HCQ+ and HCQ– groups, HCQ dose, mean maternal age and body mass index (BMI), proportion of renal involvement history, proportion of patients with antiphospholipid syndrome/antiphospholipid antibody (APS/aPL), and concurrent medications in each group.
The quality of the observational studies was assessed using the Newcastle-Ottawa Scale (NOS), and the quality of the randomized trials was evaluated using the Cochrane’s Quality Assessment Form (CQAF) [22, 23]. A star rating system of items in the NOS and CQAF was utilized to evaluate each study. An observational study was considered to be of high quality when it received a rating of at least 6 stars, and a randomized trial was considered to be of high quality when it received a rating of at least 6 stars [12, 24].
The certainty of evidence for each study outcome was assessed using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) handbook [25]. Based on the study design and the factors influencing certainty, the study outcomes were classified as having very low, low, moderate or high certainty.
Pooled odds ratios (ORs) and weighted mean differences (WMDs) with 95%
confidence intervals (95% CIs) were calculated for selected maternal and fetal
outcomes. Additionally, pooled relative risks (RRs) with 95% CIs were calculated
in some of the meta-analyses after excluding the case-control studies. p
values
Heterogeneity was assessed using the Q-test and I2 statistic, and
p values
To evaluate the presence of publication bias, this study employed visual funnel plots and Egger’s test [27]. The absence of publication bias was determined when the funnel plot exhibited a symmetrical distribution and/or Egger’s test yielded a p value greater than 0.10.
A sensitivity analysis (leave-one-out analysis) was conducted to assess the robustness of the outputs [28]. One study was excluded at each stage, and the results of all stages were compared to those obtained from the complete analysis. In order to reflect the effect of HCQ on APOs more effectively, subgroup analyses were conducted from three dimensions (different populations, HCQ doses and sample sizes).
All the statistical analyses were conducted using Stata/MP 18.0 software (StataCorp LLC, 4905 Lakeway Drive, College Station, TX, USA). Review Manager 5.4 (Cochrane Collaboration’s Information Management System, London, UK) and GraphPad Prism 9.5 (GraphPad Software LLC, Boston, MA, USA) were utilized for certain aspects of the meta-analysis.
In the initial search across five databases, a total of 1332 studies were identified based on the search strategies determined in the protocol. During the second phase, a total of 456 duplicate articles were subsequently excluded. After conducting a thorough screening of titles, abstracts, and full texts, 17 studies were ultimately selected for meta-analysis [17, 21, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43]. The flow chart is presented in Fig. 1.
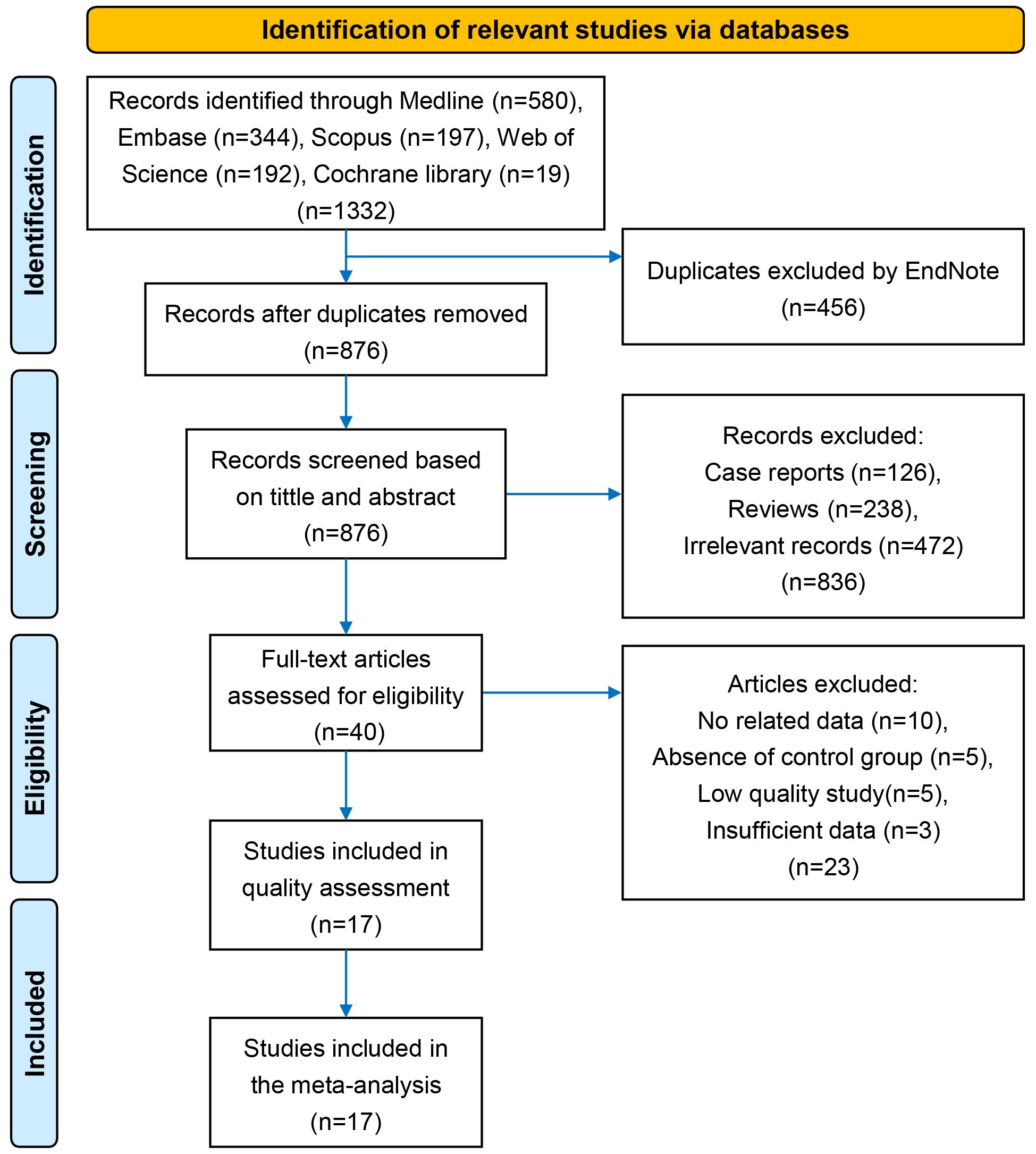 Fig. 1.
Fig. 1.
The preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow chart. PRISMA, preferred reporting items for systematic reviews and meta-analyses.
The design types and the selected maternal and fetal outcomes in each study are summarized in Table 1 (Ref. [17, 21, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43]). This meta-analysis included 17 studies, which consisted of 2 randomized trials, 11 cohort studies, and 4 case-control studies. These studies involved a total of 2238 pregnancies with SLE. Among these pregnancies, 1165 patients were in the HCQ+ groups and 1073 patients were in the HCQ– groups. The patients were enrolled from 12 regions worldwide, including the United States of America, the United Kingdom, Italy, Germany, China, and others during the period spanning from 1987 to 2020. The HCQ dose ranged from 100 to 400 mg/d, while some studies did not provide precise dose information. Based on the available data, there were no significant differences observed in most studies between the HCQ+ and HCQ– groups regarding mean maternal age (11/12) or BMI (4/4), proportion of renal involvement history (8/9), APS/aPL+ (6/7), or concurrent medications (8/13) (Table 2, Ref. [17, 21, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43]).
| Study | Selected maternal outcomes | Selected fetal outcomes | Design |
| Baalbaki et al. 2020 [29] | PE, GH, TD | PM, BW, FGR | |
| Buchanan et al. 1996 [30] | PE, HLA, GH, SA, TD | PM, BW, Mis, FGR, CGM, IUD | |
| Canti et al. 2021 [31] | PE, GH | PM, BW, Mis, GA, FGR, IUD | ▲ |
| Clowse et al. 2006 [17] | HLA | PM, Mis, Still, FGR, CGM, NL | ▲ |
| Do et al. 2020 [32] | PE, HLA | PM, BW, FGR | |
| Haase et al. 2020 [33] | PE, HLA | PM, FGR, CGM | ▲ |
| Jiang et al. 2021 [21] | — | Fetal APOs | ● |
| Kroese et al. 2017 [34] | PE, HLA, SA, HELLPs | PM, Mis, Still, GA, FGR, CGM, NL | |
| Leroux et al. 2015 [35] | PE, HLA, GH, PROM, OH, TD, ITP | PM, Mis, Still, FGR, CGM, NL, APGAR |
|
| Levy et al. 2001 [36] | HLA | — | ■ |
| Liu E. et al. 2018 [37] | HLA, PROM, OH | PM, IUD, FGR, BW | ■ |
| Liu Y. et al. 2021 [38] | PE, PROM | PM, GA, IUD | |
| Louthrenoo et al. 2021 [39] | HLA, GH, PROM | PM, FGR | ● |
| Phillips et al. 2023 [40] | PE | — | ● |
| Abd Rahman et al. 2020 [41] | PE, GH, ITP, GDM | PM, BW, Mis, Still, GA, FGR, APGAR |
|
| Seo et al. 2019 [42] | PE, SA, HELLPs, OH, GDM | PM, BW, Mis, Still, FGR, APGAR |
|
| Zhang et al. 2022 [43] | — | Fetal APOs | ● |
PE, preeclampsia; GH, gestational hypertension; TD,
thrombotic disease; HLA, high lupus activity; SA, spontaneous
abortion; HELLPs, hemolysis, elevated liver enzymes, and low platelet count
syndrome; PROM, premature rupture of membrane; OH,
oligohydramnios; ITP, immune thrombocytopenia; GDM, gestational
diabetes mellitus; PM, premature delivery; BW, birth weight;
FGR, fetal growth restriction; Mis, miscarriages; CGM,
congenital malformation; IUD, intrauterine distress; Still,
stillbirths; GA, gestational age at delivery; NL, neonatal
lupus; Fetal APOs, fetal adverse pregnancy outcomes; APGAR
| Study | Region | Period | HCQ+ | HCQ– | Dose (mg/d) | Maternal Agea (years) | BMI (kg/m2) | Renal involvement history (%) | APS/aPL (%) | Concurrent medications (%) |
| Baalbaki et al. 2020 [29] | USA | 2006–2013 | 47 | 30 | — | 26.2/30.5b | 29.0/29.0 | — | — | Pred (83/40)b, Asp (70/67) |
| Buchanan et al. 1996 [30] | UK | 34 | 53 | 200–400 | comparable | — | — | — | Pred (comparable), Aza (21/13) | |
| Canti et al. 2021 [31] | Italy | 2003–2019 | 45 | 29 | 300 | 32.7/32.0 | — | 40.0/24.0 | 13.0/21.0 | Corti (60/48), Aza (36/14), Asp (76/59), LMWH (24/31) |
| Clowse et al. 2006 [17] | USA | 1987–2002 | 56 | 163 | — | — | — | 36.0/40.0 | 5.0/14.0 | Pred (63/67), Aza (14/13) |
| Do et al. 2020 [32] | USA | 2000–2017 | 53 | 76 | — | 33.0/32.1 | 24.7/27.3 | 28.3/26.3 | 1.9/5.3 | Pred (87/87), Aza (13/3)b, Asp (19/24), Antico (8/11) |
| Haase et al. 2020 [33] | Germany | 77 | 107 | — | 30.0/31.0 | — | 33.8/23.4 | — | Aza (26/17), Asp (53/33)b | |
| Jiang et al. 2021 [21]* | China | 2010–2018 | 405 | 108 | — | NA | NA | NA | NA | Gluco, Immun, Asp, LMWH (NA) |
| Kroese et al. 2017 [34] | Netherlands | 2000–2015 | 30 | 80 | 200–400 | 32.5/30.6 | — | — | 7.0/13.0 | Pred (60/54) |
| Leroux et al. 2015 [35] | France | 2001–2011 | 41 | 77 | 400 | 31.2/30.1 | 24.1/23.3 | 19.5/6.5b | 9.8/20.7 | Pred (76/64), Aza (5/3), Asp (61/44), LMWH (32/43) |
| Levy et al. 2001 [36] | Brazil | 10 | 10 | — | 29.0/29.0 | — | — | — | Pred (70/90), Asp (50/20) | |
| Liu E. et al. 2018 [37] | China | 2016–2017 | 39 | 39 | 400 | 29.1/29.1 | — | 12.8/15.4 | — | Metaco (0/100)b |
| Liu Y. et al. 2021 [38] | China | 2004–2019 | 44 | 44 | — | 29.8/28.7 | — | 18.2/27.3 | — | Gluco (75/66), Asp (16/5), LMWH (9/7), Immun (14/2) |
| Louthrenoo et al. 2021 [39]* | Thailand | 1993–2017 | 37 | 53 | — | NA | NA | NA | NA | Pred, Immun (NA) |
| Phillips et al. 2023 [40]* | Australia | 2010–2020 | 51 | 44 | 100–150 | NA | NA | NA | NA | Pred, Aza, Asp, Other Immun (NA) |
| Abd Rahman et al. 2020 [41] | Malaysia | 2007–2017 | 47 | 35 | — | 32.0/31.5 | — | 48.9/62.9 | 76.6/60.0 | Ster (96/89), Immun (53/68) |
| Seo et al. 2019 [42] | Korea | 1995–2018 | 80 | 71 | — | 32.8/31.8 | 24.3/23.0 | 18.8/16.9 | 28.8/8.5b | Ster (53/61), Aza (6/9), Asp (20/6)b, Hepa (15/6) |
| Zhang et al. 2022 [43]* | China | 2014–2020 | 69 | 54 | 100–400 | NA | NA | NA | NA | Pred, Asp, Cyc, LMWH (NA) |
*Case-control study. aData are presented by mean, median or percentage. bData reveal significant differences between the HCQ+ and HCQ– groups. BMI, body mass index; APS, antiphospholipid antibody syndrome; aPL, antiphospholipid antibody; —, not available; NA, not applicable: these data were usually not analyzed in the case-control studies when considering HCQ as one of the influence factors; Pred, prednisone; Asp, aspirin; Aza, azathioprine; Corti, corticosteroid; LMWH, low molecular weight heparin; Antico, anticoagulation; Gluco, glucocorticoid; Immun, immunosuppressive agent; Metaco, metacotandracin; Ster, steroid; Hepa, heparin; Cyc, cyclosporine.
The NOS scores for cohort and case-control studies ranged from 7 to 8, while the CQAF scores for randomized trials were between 6 and 7 (Table 3, Ref. [17, 21, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43]). Consequently, all the studies included in the quality assessment were deemed to possess a high level of quality and thus were incorporated into the meta-analysis.
| Study/Assessment | Item 1 | Item 2 | Item 3 | Item 4 | Item 5 | Item 6 | Item 7 | Item 8 | Total scores | |
| ▲/ |
A* | B | C | D | E | F | G | H | NOS scores for cohort studies | |
| Baalbaki et al. 2020 [29] |
★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| Buchanan et al. 1996 [30] |
★ | ★ | ★ | ✩ | ★✩ | ★ | ★ | ★ | 7† | |
| Canti et al. 2021 [31] ▲ | ★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| Clowse et al. 2006 [17] ▲ | ★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| Do et al. 2020 [32] |
★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| Haase et al. 2020 [33] ▲ | ★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| Kroese et al. 2017 [34] |
★ | ★ | ★ | ✩ | ★✩ | ★ | ★ | ★ | 7† | |
| Leroux et al. 2015 [35] |
★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| Liu Y. et al. 2021 [38] |
★ | ★ | ★ | ✩ | ★✩ | ★ | ★ | ★ | 7† | |
| Abd Rahman et al. 2020 [41] |
★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| Seo et al. 2019 [42] |
★ | ★ | ★ | ✩ | ★★ | ★ | ★ | ★ | 8† | |
| ● | a | b | c | d | e | f | g | h | NOS scores for case-control studies | |
| Jiang et al. 2021 [21] ● | ★ | ★ | ✩ | ★ | ★★ | ★ | ★ | ★ | 8† | |
| Louthrenoo et al. 2021 [39] ● | ★ | ★ | ✩ | ★ | ★★ | ★ | ★ | ★ | 8† | |
| Phillips et al. 2023 [40] ● | ✩ | ★ | ✩ | ★ | ★★ | ★ | ★ | ★ | 7† | |
| Zhang et al. 2022 [43] ● | ★ | ★ | ✩ | ★ | ★★ | ★ | ★ | ★ | 8† | |
| ■ | S | T | U | V | W | X | Y | Z | CQAF scores for RCTs | |
| Levy et al. 2001 [36] ■ | ✩ | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 7† | |
| Liu E. et al. 2018 [37] ■ | ★ | ★ | ✩ | ✩ | ★ | ★ | ★ | ★ | 6† | |
*Items of quality assessment methods. A, representativeness of the exposed
cohort. B, selection of the non-exposed cohort. C, ascertainment of exposure. D,
demonstration that outcome of interest was not present at start of study. E,
comparability of cohorts on the basis of the design or analysis. F, assessment of
outcome. G, was follow-up long enough for outcomes to occur. H, adequacy of
follow up of cohorts. a, is the case definition adequate. b, representativeness
of the cases. c, selection of controls. d, definition of controls. e,
comparability of cases and controls on the basis of the design or analysis. f,
ascertainment of exposure. g, same method of ascertainment for cases and
controls. h, non-response rate. S, randomized selection in the population. T,
adequate concealment of allocation to intervention. U, blinding trial
participants and researchers from knowledge of which intervention a participant
received. V, blinding of outcome assessment. W, validated completeness of outcome
data. X, adequate selective outcome reporting. Y, report of drop-outs during
follow-up. Z, clear definition of inclusion/exclusion criteria. NOS, the
Newcastle-Ottawa scale; CQAF, the Cochrane’s Quality Assessment Form; RCT,
randomized controlled trial.
The meta-analysis revealed statistically significant differences in the
incidence of preeclampsia (OR = 0.45, 95% CI: 0.32–0.65, p
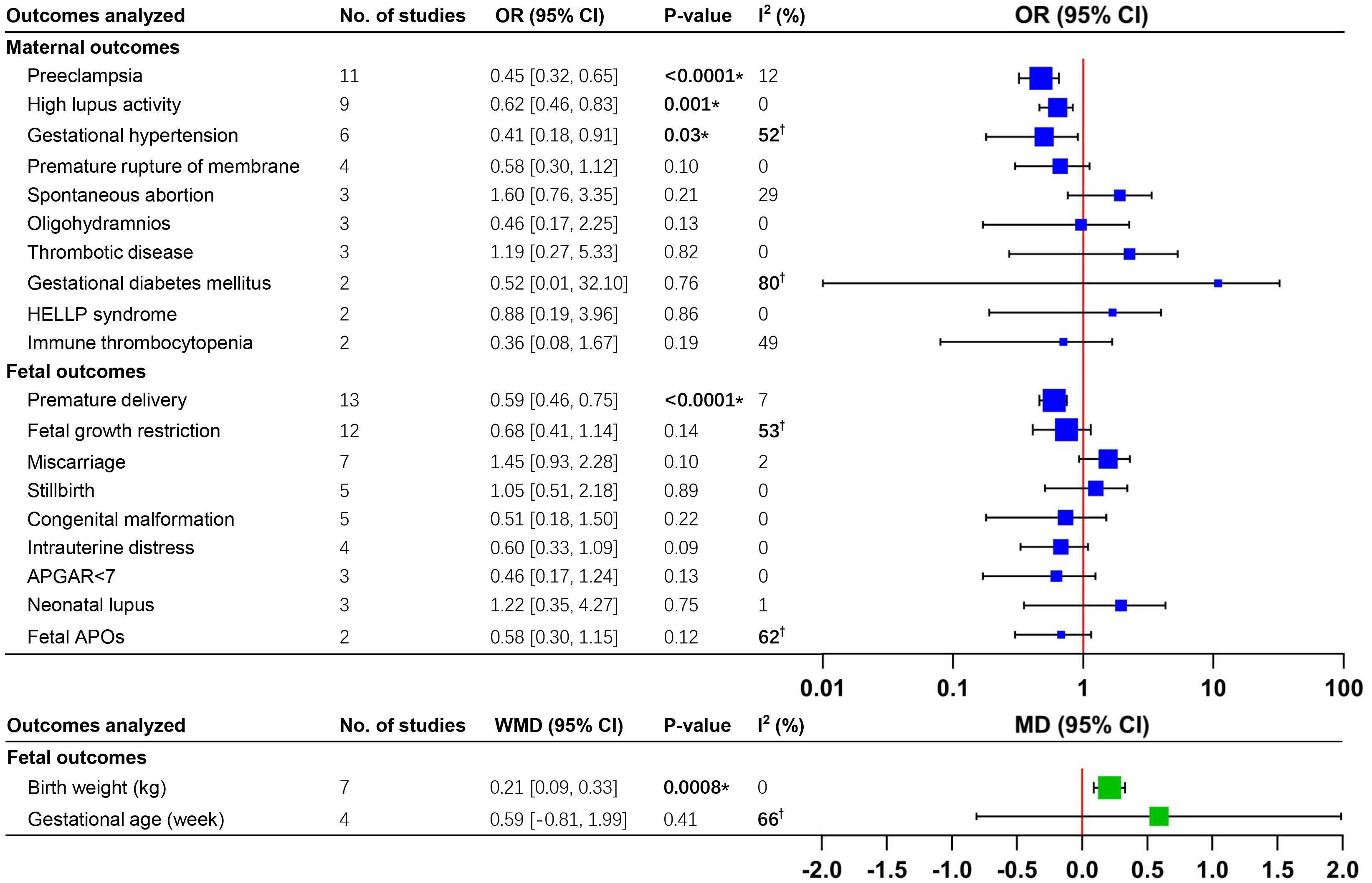 Fig. 2.
Fig. 2.
Meta-analyses of adverse pregnancy outcomes in pregnant patients
with SLE receiving HCQ vs. controls. *p
For further analysis, the RRs were calculated for adverse maternal outcomes with
positive results, after excluding the case-control studies. Significant
differences were observed for preeclampsia (RR = 0.51, 95% CI: 0.37–0.71,
p
The meta-analysis revealed significant differences in the incidence of premature
delivery (OR = 0.59, 95% CI: 0.46–0.75, p
After excluding the case-control studies, significant differences in the
incidence of premature delivery (RR = 0.71, 95% CI: 0.60–0.84, p
Visual inspection of the funnel plots did not reveal any significant asymmetry among all maternal or fetal outcomes (Supplementary File 3). Egger’s test was used for meta-analyses involving more than 10 studies and indicated no significant publication bias in the analyses of preeclampsia, premature delivery, or FGR (Fig. 3A,C,E) [44].
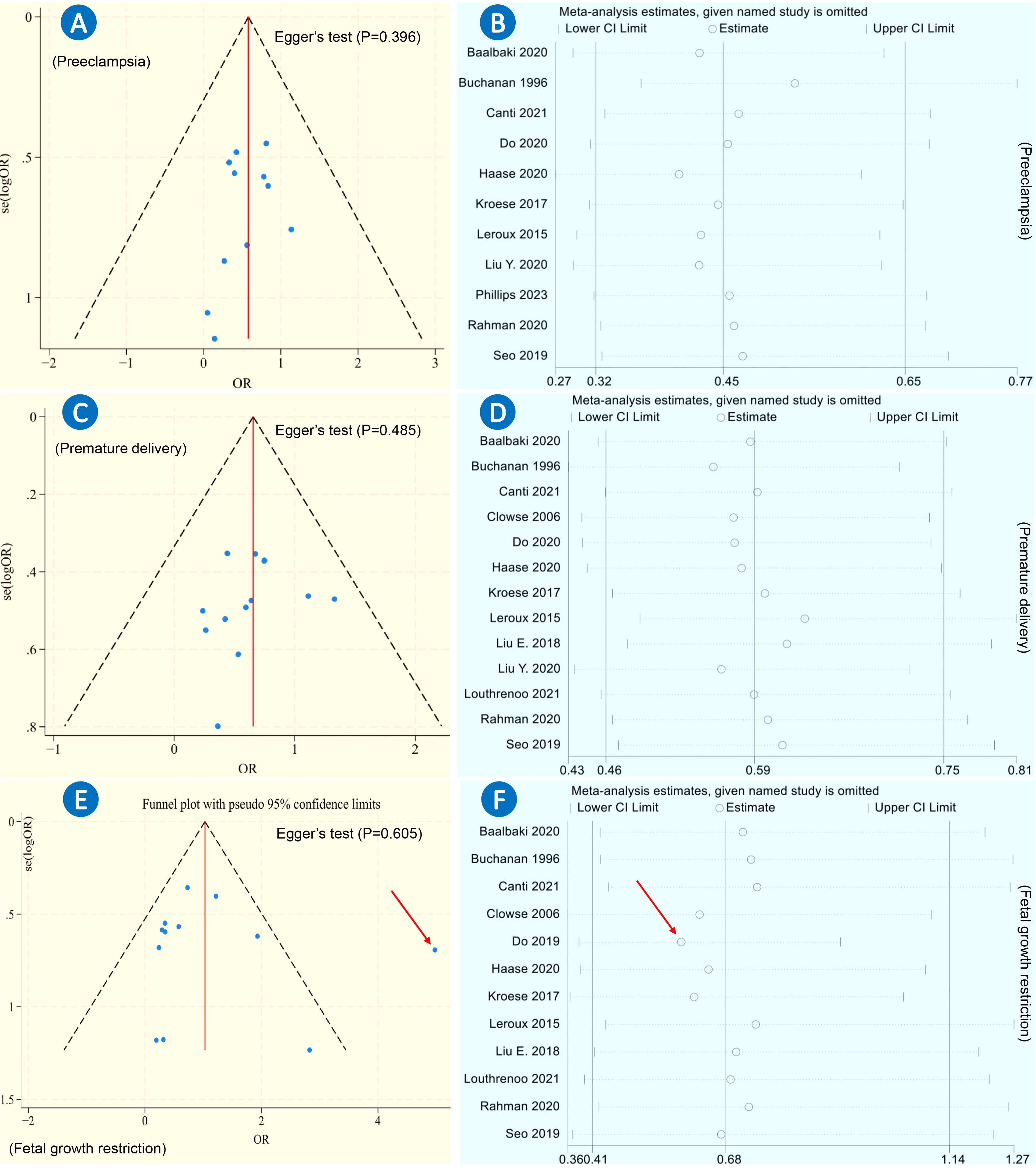 Fig. 3.
Fig. 3.
Funnel plots and sensitivity analyses of three adverse pregnancy outcomes. (A) Funnel plots of preeclampsia with Egger’s test (p = 0.396). (B) Sensitivity analysis (leave-one-out analysis) of preeclampsia. (C) Funnel plots of premature delivery with Egger’s test (p = 0.485). (D) Sensitivity analysis (leave-one-out analysis) of premature delivery. (E) Funnel plots of fetal growth restriction with Egger’s test (p = 0.605). (F) Sensitivity analysis (leave-one-out analysis) of fetal growth restriction. Red arrow: the study reported by Do et al. in 2019 [32].
For meta-analyses that included more than 10 studies, robustness was assessed through leave-one-out analysis. The analysis demonstrated the robustness of the findings regarding preeclampsia and premature delivery (Fig. 3B,D). However, the analysis of FGR revealed a significant instability (Fig. 3F).
Through the examination of funnel plots and full-texts, a potential problem was identified in the results of FGR in the study reported by Do et al. (Fig. 4A). [32]. The baseline characteristics revealed that the occurrence rates of most categories of previous SLE manifestations in the HCQ+ group were higher than those in the HCQ– group (12/15 categories). Therefore, the SLE diseases were considered to be more severe in the HCQ+ group to a certain extent. This probably concealed the true impacts of HCQ on FGR in pregnant patients with SLE.
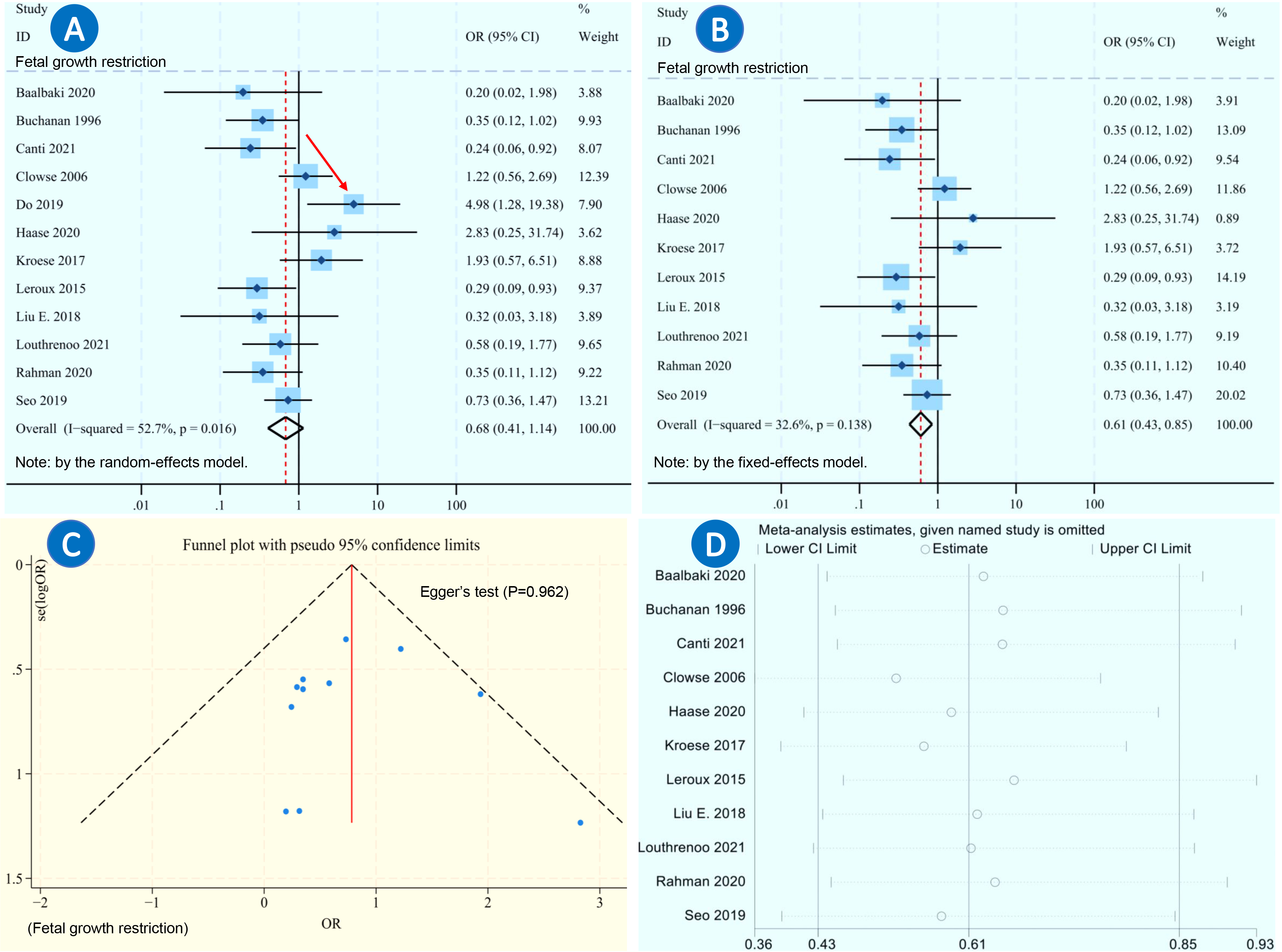 Fig. 4.
Fig. 4.
Meta-analysis of fetal growth restriction after excluding one study. (A) Forest plot meta-analysis of fetal growth restriction in pregnant patients with SLE receiving HCQ vs. controls, using the random-effects model. (B) Forest plot meta-analysis of fetal growth restriction in pregnant patients with SLE receiving HCQ vs. controls using the fixed-effects model, after excluding the study reported by Do et al. in 2019 [32]. (C) Funnel plots of fetal growth restriction with Egger’s test (p = 0.962), after excluding Do et al’s study. (D) Sensitivity analysis (leave-one-out analysis) of fetal growth restriction, after excluding Do et al’s study. Red arrow: the study reported by Do et al. in 2019 [32].
After excluding the study by Do et al. [32], the meta-analysis revealed significant differences in the incidence of FGR (OR = 0.61, 95% CI: 0.43–0.85, p = 0.003) between the HCQ+ and HCQ– groups (Fig. 4B). The funnel plot and Egger’s test showed no apparent evidence of publication bias (p = 0.962, Fig. 4C). Additionally, the sensitivity analysis demonstrated significant stability in the analysis of FGR (Fig. 4D).
Due to the limited number of included studies, subgroup analyses were conducted for maternal and fetal outcomes that included more than 10 studies (Supplementary Figs. 45–62). The effects of HCQ on three APOs (preeclampsia, premature delivery, and FGR) were similar in Asian populations as well as in European and North American populations. For preeclampsia and premature delivery, there were no apparently different effects of HCQ shown between two dose groups (200–400 mg/d group, and group with no precise dose provided). Similarly, no significant differences in the effects of HCQ were observed between two sample-size groups (20–100 group and 100–200 group).
However, the subgroup meta-analysis of FGR revealed no significant effects of
HCQ in the group with no precise HCQ dose provided (Supplementary Fig.
59), as well as in the sample-size group of 100–200 or
The final certainty analysis revealed that the results of preeclampsia, high lupus activity, premature delivery and birth weight yielded high-quality evidence. Moderate quality evidence was found for gestational hypertension, premature rupture of membranes, spontaneous abortion, miscarriage, stillbirth and intrauterine distress. After excluding the study by Do et al. [32], high quality evidence was also obtained for FGR. Other APOs received low or very low scores for certainty (Table 4).
| Outcomes analyzed | Study design | Initial certainty | Reasons for certainty up or down | Final certainty | |
| Maternal outcomes | |||||
| Preeclampsia | ▲/●×11 | Low | ↑A, ↑B | High* | |
| High lupus activity | ▲/●×7, ■×2 | Low | ↑A, ↑B | High* | |
| Gestational hypertension | ▲/●×6 | Low | ↑A, ↑B, ↓D | Moderate* | |
| Premature rupture of membrane | ▲/●×3, ■×1 | Low | ↑B | Moderate* | |
| Spontaneous abortion | ▲/●×3 | Low | ↑B | Moderate* | |
| Oligohydramnios | ▲/●×2, ■×1 | Low | ↑B, ↓C | Low | |
| Thrombotic disease | ▲/●×3 | Low | ↑B, ↓C | Low | |
| Gestational diabetes mellitus | ▲/●×2 | Low | ↑B, ↓C, ↓D | Very Low | |
| HELLP syndrome | ▲/●×2 | Low | ↑B, ↓C | Low | |
| Immune thrombocytopenia | ▲/●×2 | Low | ↑B, ↓C | Low | |
| Fetal outcomes | |||||
| Premature delivery | ▲/●×12, ■×1 | Low | ↑A, ↑B | High* | |
| Fetal growth restriction† | ▲/●×10, ■×1 | Low | ↑A, ↑B | High* | |
| Birth weight (kg) | ▲/●×6, ■×1 | Low | ↑A, ↑B | High* | |
| Miscarriage | ▲/●×7 | Low | ↑B | Moderate* | |
| Stillbirth | ▲/●×5 | Low | ↑B | Moderate* | |
| Congenital malformation | ▲/●×5 | Low | ↑B, ↓C | Low | |
| Gestational age at delivery (week) | ▲/●×4 | Low | ↑B, ↓C, ↓D | Very Low | |
| Intrauterine distress | ▲/●×3, ■×1 | Low | ↑B | Moderate* | |
| APGAR |
▲/●×3 | Low | ↑B, ↓C | Low | |
| Neonatal lupus | ▲/●×3 | Low | ↑B, ↓C | Low | |
| Fetal APOs | ▲/●×2 | Low | ↑B, ↓D | Low | |
*GRADE rating showing high or moderate certainty. †After
excluding the study reported by Do et al. [32], high-quality evidence
was observed for fetal growth restriction. ▲/● observational
study. ■ randomized trial. ↑A, upgraded one level due to large magnitude
of effect. ↑B, upgraded one level due to all plausible confounding would reduce a
demonstrated effect or suggest a spurious effect when results show no effect. ↓C,
downgraded one level due to imprecision of results: wide confidence intervals. ↓D,
downgraded one level due to serious inconsistency: heterogeneity in interventions
(I2
To date, the effects of HCQ on APOs in pregnant patients with SLE have been poorly and incompletely summarized by meta-analyses [11, 12, 13, 14]. This is a more comprehensive systematic review and meta-analysis based on 17 included studies, involving a total of 2238 pregnancies with SLE. It reveals the beneficial effects of HCQ on prophylaxis for preeclampsia, high lupus activity, gestational hypertension, premature delivery, and FGR in pregnant patients with SLE. The administration of HCQ also increases fetal birth weight. Moreover, this study does not provide any apparent evidence that HCQ has an adverse impact on other APOs (Fig. 5).
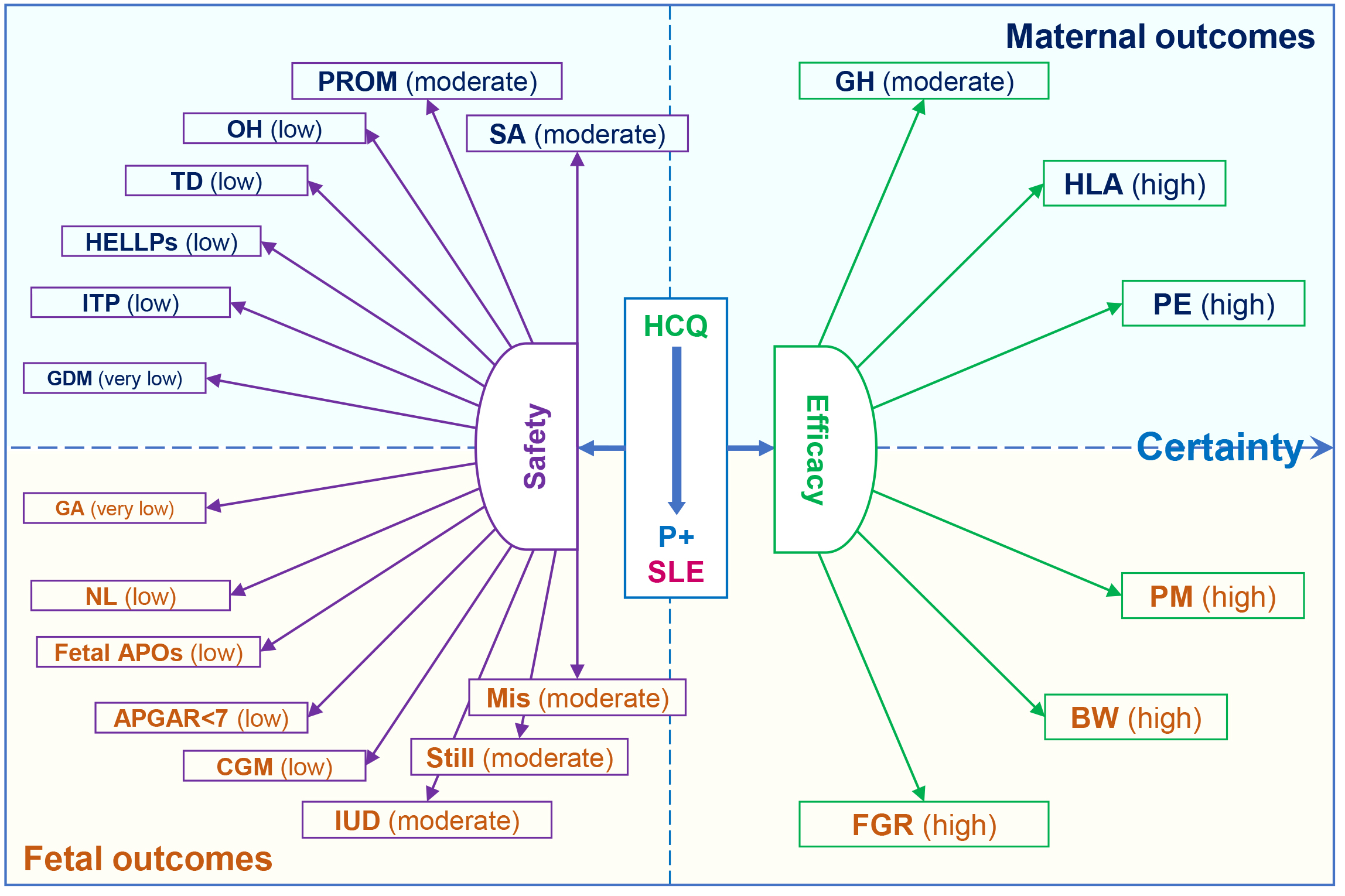 Fig. 5.
Fig. 5.
Effects of hydroxychloroquine (HCQ) on maternal and fetal
outcomes in pregnant patients (P) with systemic lupus erythematosus (SLE). High,
high certainty; Moderate, moderate certainty; Low, low certainty; Very low, very
low certainty; PE, preeclampsia; GH, gestational hypertension; TD, thrombotic
disease; HLA, high lupus activity; SA, spontaneous abortion; HELLPs,
hemolysis, elevated liver enzymes, and low platelet count syndrome; PROM, premature
rupture of membrane; OH, oligohydramnios; ITP, immune thrombocytopenia; GDM,
gestational diabetes mellitus; PM, premature delivery; BW, birth weight; FGR,
fetal growth restriction; Mis, miscarriages; CGM, congenital malformation; IUD,
intrauterine distress; Still, stillbirths; GA, gestational age at delivery; NL,
neonatal lupus; Fetal APOs, fetal adverse pregnancy outcomes; APGAR
In 2018, Eudy et al. [18] reported an increased incidence of flares during pregnancy in SLE patients. Subsequently, Clowse in 2022 and Hu in 2023 detected a reduction in lupus activity among pregnant patients who received HCQ, as evidenced by two meta-analyses [13, 14]. Our study supported this finding based on a larger analysis that included 9 studies. As an immunomodulatory agent, HCQ exhibits a moderate anti-inflammatory effect on the treatment of various rheumatic diseases through multiple immune pathways, such as the Toll-like receptor 7 (TLR7) and Toll-like receptor 9 (TLR9) signaling pathways [45, 46]. This may demonstrate its prophylactic effects on high lupus activity at the molecular level.
Many studies including ours revealed the beneficial effects of HCQ on
prophylaxis for preeclampsia and gestational hypertension [12, 41, 42]. There is
growing evidence that HDP, including preeclampsia and gestational hypertension,
primarily involves endothelial dysfunction and immune response dysregulation
[47]. HCQ improves physiopathological mechanisms through multiple pathways, such
as reducing the production of tumour necrosis factor alpha (TNF-
The incidence of premature delivery in pregnant women with SLE was found to be greater (16–50%) than that in the general population (11%) [52]. Although based on limited studies, our results revealed no differences in the incidence of gestational age at delivery between the HCQ+ and HCQ– groups (very low certainty), with the pooled data supporting that the administration of HCQ could reduce the incidence of premature delivery [12].
Several studies have indicated an increased risk of FGR during pregnancy in individuals with SLE [3, 4, 5, 6, 7, 8, 9]. After excluding 1 study with heterogeneity, this meta-analysis firstly summarizes that the use of HCQ during pregnancy can reduce the incidence of FGR. In addition, our study demonstrates the positive impact of HCQ on increasing fetal birth weight (+0.21 kg) in pregnant patients with SLE. The primary mechanisms for FGR are various, but they usually share a common final pathway of inadequate fetal nutrition and uteroplacental perfusion [53, 54]. HCQ is widely believed to have a vascular protective effect, and to exert regulatory control over aberrant autoimmunity in SLE through diverse molecular and cellular mechanisms [45]. Although there is a lack of in vitro experiments on the effects of HCQ on FGR in SLE models, it is possible that HCQ could improve uteroplacental perfusion and fetal nutrition through these pathways.
Historical researches indicated that HCQ might cause congenital malformations, such as vision loss, ototoxicity, and cochleovestibular dysfunction, as well as stillbirths and spontaneous abortions [55, 56]. However, more recent studies have supported the safety of HCQ in pregnant patients with SLE [57, 58]. Similarly, this study did not find any apparent evidence indicating an increased incidence of congenital malformations, stillbirths, spontaneous abortions, or miscarriages in the HCQ+ groups. In addition, there were no significant differences detected in the occurrence of the other 11 APOs between the HCQ+ and HCQ– groups. Based on these findings, we have gained more confidence in the safety of using HCQ during pregnancy among individuals with SLE [45].
In addition, it is widely acknowledged that there exists a significant association between APS and SLE [59]. Whereas, the absence of sufficient APS/aPL data limited further analysis in this study. It has been reported that HCQ may enhance the live birth rate and reduce the prevalence of pregnancy complications in APS patients in a retrospective study [60]. The data on the use of HCQ in APS are still insufficient, and more studies are needed [59].
There are three key strengths in our study. First, compared with previous studies, this meta-analysis provides a more comprehensive evaluation of the efficacy and safety of HCQ for preventing APOs among pregnant patients with SLE. Second, our findings reveal that the administration of HCQ during pregnancy can also increase fetal birth weight and reduce the incidence of FGR, which has not been identified in prior studies [11, 12, 13, 14]. Third, based on the limited available data, we did not detect any obvious evidence of a harmful impact of HCQ on the other 15 APOs, which has not been fully elucidated by previous meta-analyses. Conversely, there are several limitations. First, this meta-analysis was not completely based on randomized controlled trials. Second, some of the included studies did not provide complete baseline characteristics, and there were differences in a few characteristics between the HCQ+ and HCQ– groups in several studies. This suggests a certain degree of heterogeneity among the included studies; therefore, the results should be interpreted with caution.
Despite these limitations, the findings derived from this meta-analysis provide a more comprehensive assessment of the effects of HCQ on maternal and fetal outcomes in pregnant patients with SLE, thereby establishing a basis for more precise and further research.
This study provides a more comprehensive evaluation of the efficacy and safety of HCQ in pregnant patients with SLE. Therefore, we have more compelling grounds to advocate for the use or adjunctive use of this drug for managing pregnancies complicated with SLE.
SLE, Systemic lupus erythematosus; APO, Adverse pregnancy outcome; HCQ, Hydroxychloroquine; HDP, Hypertensive disorders of pregnancy; FGR, Fetal growth restriction; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; APGAR
All data are available within the manuscript and its supplementary files.
All authors contributed to this piece of work. AHL participated in the study design, literature search, data extraction, data analysis, interpretation of findings, manuscript drafting and paper revision. YWY and XWL participated in the literature search, data extraction, data analysis and interpretation of findings. BX participated in the data analysis, interpretation of the findings, manuscript drafting and paper revision. RL and CR participated in the data analysis and interpretation of the findings. XRR, LLT, YPM and MZY participated in the interpretation of the findings and manuscript drafting. XMH participated in the study design and contributed analysis tools. XC conceived and designed the study, contributed analysis tools, and participated in the manuscript drafting and paper revision. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to thank the staff of the library of Xiamen University for providing us with the full texts of some articles.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.ceog5110220.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





