1 Department of Obstetrics and Gynecology, Universitas Airlangga, 60286 Surabaya, Indonesia
2 Division of Veterinary Clinic, Faculty of Veterinary Medicine, Universitas Airlangga, 60286 Surabaya, Indonesia
3 Department of Public Health and Preventive Medicine, Universitas Airlangga, 60286 Surabaya, Indonesia
4 Department of Obstetrics and Gynecology, Universitas Indonesia, 10430 Jakarta, Indonesia
Abstract
Vulvovaginal atrophy is part of the genitourinary syndrome of menopause caused by hypoestrogenic changes. This pathophysiological mechanism alters the concentration of collagen and elastin, which modifies the vaginal mucosa and impairs the function of the pelvic floor muscles. Quercetin is one of the flavonols found in plants, fruits, and vegetables. Quercetin helps to improve the syndrome through a variety of actions and estrogen-like effects. This study aimed to analyze the impact of quercetin on collagen I, collagen III, and elastin in a vaginal menopausal rat model.
This study's research design was an in vivo randomized control group post-test. The research was conducted at the animal laboratory, Faculty of Medicine, Universitas Airlangga. Rattus norvegicus, used in the study, were divided into 5 groups: normal rat, menopausal model without treatment, menopausal model given 17-β estradiol valerate 0.18 mg/kg, quercetin 12.5 mg/kg, and quercetin 50 mg/kg. The effectiveness of therapy was assessed from the immunohistochemical expression of collagen I, collagen III, and elastin in vaginal tissue.
The standard group in this study had the highest average expression levels of collagen I and elastin. The group of menopausal models without treatment in the study had the highest average expression of collagen III. Collagen I expression (p < 0.001), collagen III expression (p < 0.001), and elastin expression (p < 0.001) all showed significant differences. The menopausal rat model without treatment and the standard group showed the most differences in the expression of collagen I, collagen III, and elastin.
The expression of collagen I, collagen III, and elastin in the vagina of the menopausal model was affected by the administration of quercetin at a level of 50 mg/kg. Thus, quercetin can be an alternative herbal treatment option to improve vulvovaginal atrophy in menopausal conditions.
Keywords
- quercetin
- collagen I
- collagen III
- elastin
- menopause
- vulvovaginal atrophy
The condition of hypoestrogenism associated with menopause has a negative impact on the health of the vagina and urinary tract, known as the genitourinary syndrome of menopause (GSM). Genital signs and symptoms such as dryness, burning, irritation, and sexual symptoms such as discomfort or pain were previously known as vulvovaginal atrophy (VVA). GSM occurs in 15% of premenopausal women, while around 50–70% occur in postmenopausal women and will tend to get worse during menopause [1].
The hypoestrogenic environment of urogenital tissue causes changes in the thickness of the vaginal epithelium and lamina propria, smooth muscle atrophy, reduced blood flow to the vaginal area, and loss of tissue elasticity due to decreased concentrations of collagen, elastin, and hyaluronic acid. The effects of these pathophysiological mechanisms produce changes in the urogenital system, especially changes in the vaginal mucosa. In the lower urinary tract, they affect the capacity and contractile ability of the bladder, urethral sphincter, and pelvic floor muscle function [2].
Collagen I and collagen III represent the primary constituents of the extracellular matrix, exerting substantial influence on the biomechanical attributes of the vaginal tissue. Their distinct fiber diameters result in divergent mechanical properties, with collagen I predominantly contributing to tensile strength and stiffness, while collagen III confers flexibility to the tissue [3]. Elastin’s primary function is to offer resilience to tissues that experience repetitive stretching and physical strain [4].
Flavonoids are a group of natural substances often found in foods and drinks originating from plants, such as fruit, vegetables, and tea [5]. Flavonoids have anti-oxidative, anti-inflammatory, anti-mutagenic, and anti-carcinogenic properties in flavonoids and can modulate the function of vital cellular enzymes [5]. Flavonoids are widely used for the treatment of menopausal symptoms, although it is currently unclear whether they can prevent or alleviate other menopausal changes [6]. One of the most common flavonoids is quercetin. Quercetin is a phytoestrogen that has estrogenic activity. Many molecular and clinical data support the effects of estrogen receptors on normal skin homeostasis and may accelerate re-epithelialization in wound healing [7]. Several in vivo and in vitro studies have been carried out to look for evidence of the influence of quercetin, but there is no research explicitly discussing their effect on connective tissue in menopausal women. This research was carried out on experimental animals to analyze the impact of quercetin on the vagina so that it can be the first step in understanding the role of quercetin in vulvovaginal atrophy.
The study’s design was a laboratory experimental investigation with an in vivo randomized control group posttest-only design. This study focused on a Wistar rat (Rattus norvegicus) model of menopause. The study was conducted at the Experimental Animal Laboratory at Airlangga University’s Surabaya Medical Faculty from November 2022 to March 2023. The relevant Research Ethics Committee approved the protocol regarding research procedures carried out on animals. This research has been tested for ethical feasibility at the Faculty of Medicine, Airlangga University, to meet the ethical requirements for experimental animals. The study’s population included female Wistar rats (Rattus norvegicus) from the Experimental Animal Laboratory at Airlangga University’s Surabaya Medical Faculty. The sample contained a female Wistar rat weighing 200–300 grams. The total sample size in this study was 30 samples separated into five groups, each containing six samples.
Group K0 consisted of non-menopausal rats not receiving therapy and served as
the control group. Group K1 consisted of menopausal model rats who received no
treatment and served as the control group. Group K2 comprised menopausal model
rats who received oral estrogen treatment (17-
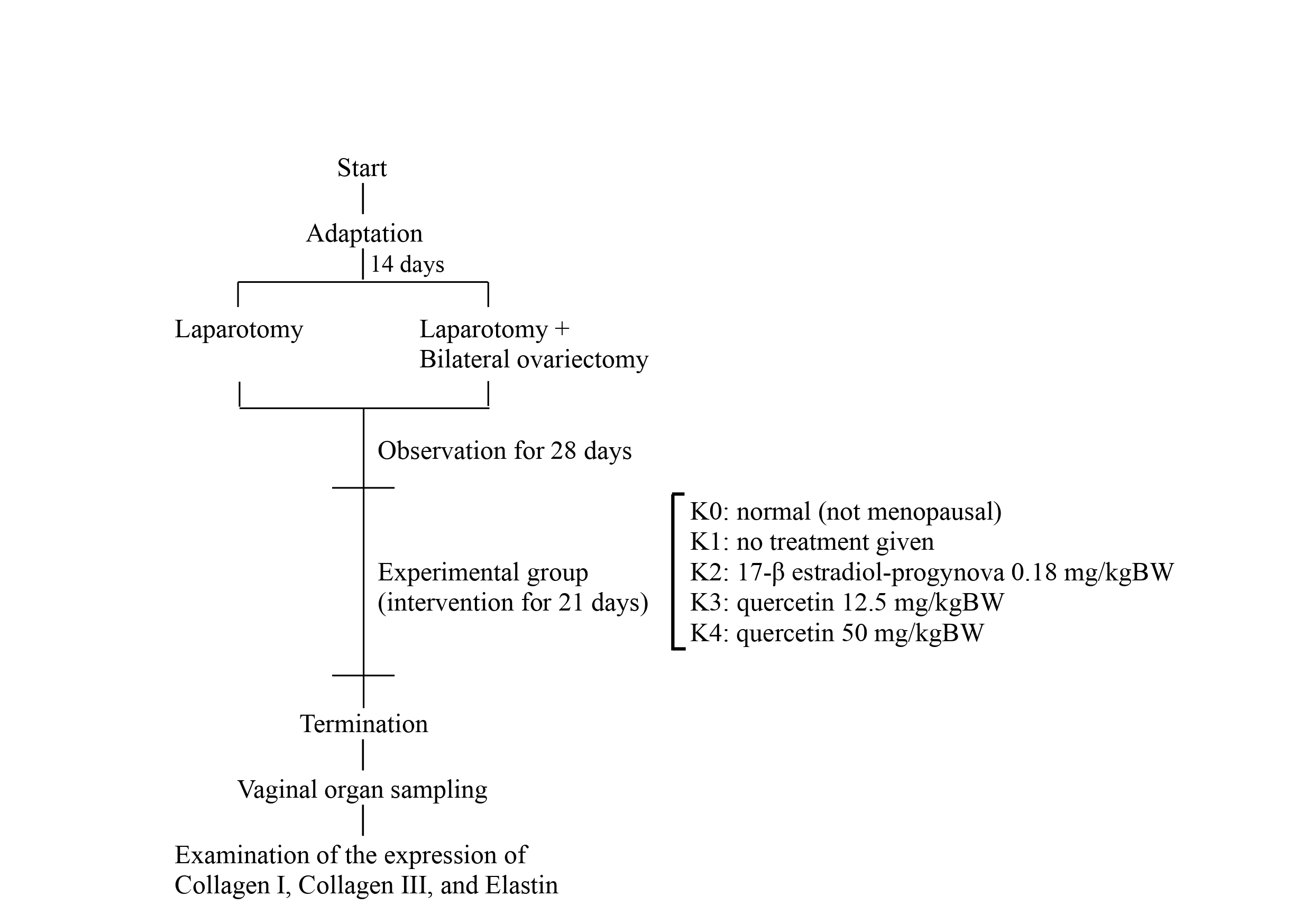 Fig. 1.
Fig. 1.
Experimental procedures for rat and quercetin administration.
Preoperative preparation involved acclimatizing rats for two weeks from the breeding source. Before the surgery, thirty rats underwent health checks, and their weight was measured using a digital weighing machine (SF-400, Zhejiang Mengxuan Electronic Co., Ltd., Jinhua, Zhejiang, China). The animals were anesthetized with a combination of 50 mg/kgBW ketamine and 10 mg/kgBW xylazine. Once anesthetized, rats underwent fur shaving and surgical site disinfection. A midline dorsal skin incision with 3 cm long preceded bilateral ovariectomy. After the muscle dissection, the peritoneal space and adipose tissue surrounding the ovary were exposed. After identifying the ovary and uterine horn, a vicryl sutures-3/0 was performed around the area of the distal uterine horns, which was sectioned after that, and the ovaries were removed. The procedure was repeated for the left ovary [8, 9]. In the K0 group, only laparotomy was performed without bilateral ovariectomy. Postoperative wound care included the application of iodine tincture, dressing replacement, as well as administration of enrofloxacin antibiotic at a dose of 5 mg/kgBW and Tolfenamic acid analgesic at a dose of 4 mg/kgBW for three days.
Flavonoids in the form of Quercetin (C15H10O7, Sigma-Aldrich Co - Merck KGaA, Darmstadt, Germany). In the in vivo therapeutic model, experimental rats received intraoral doses. Group quercetin dose 1 received 12.5 mg/kgBW, while group quercetin dose 2 received 50 mg/kgBW.
The powder of 17-
After the intervention had been completed, euthanasia was carried out on the experimental animals by previously anesthetizing them with isoflurane. Then, the implementation of euthanasia according to the Institutional Animal Care and Use Committee (IACUC) using the cervical dislocation method [10]. Vaginal organ collection was then carried out. In immunohistochemical staining, the initial step involved preparing the specimen slides by deparaffinization and rehydration using an oven and a series of xylene and ethanol solutions with appropriate concentrations. Furthermore, if necessary, the antigen retrieval procedures were performed using enzyme retrieval agents such as trypsin or protease at 37 °C for 2–30 minutes. The subsequent process involved the inactivation of endogenous peroxidase by incubating with 3% hydrogen peroxide for 5 minutes, mainly when using peroxidase-conjugated secondary antibodies or ExtrAvidin-Peroxidase. The antibodies used in this study were obtained from Genetex (Irvine, CA, USA), with batch numbers GTX26308, GTX60940, and GTX37428, and were diluted at a ratio of 1:500. Primary antibodies, including those for collagen I, collagen III, and elastin, were applied to the slides with a minimum incubation period of 60 minutes at 37 °C, followed by reaction with biotinylated secondary antibody (1:100; Starr Trek Universal HRP Detection, Biocare Medical, Pacheco, CA, USA; STUHRP700) and washing with phosphate buffered saline (PBS). A substrate mixture was prepared and utilized for color reaction development, with an incubation period of 5–10 minutes or until the desired color reaction was observed. Subsequently, counter-staining was performed using hematoxylin Mayer, with an incubation period of 0.5–5 minutes, followed by washing with distilled water. Finally, the slides were mounted using an aqueous mounting medium such as gelatin glycerol and sealed with clear nail polish.
Evaluation of collagen I, collagen III, and elastin expression was conducted using the modified Remmele method. The Remmele scoring index (Immuno Reactive Score, IRS) was obtained by multiplying the score of the percentage of positive immunoreactive cells or area with the score of color intensity in immunoreactive cells. Data from each sample represented the average IRS value observed in 5 fields of view at 1000 times magnification [11].
Research data will be recorded in the data collection form. Then, a normality
test was carried out using the Shapiro-Wilk test. If the distribution is normal
(p
The highest average expression of collagen I was observed in the control group
of untreated normal rats (10.13
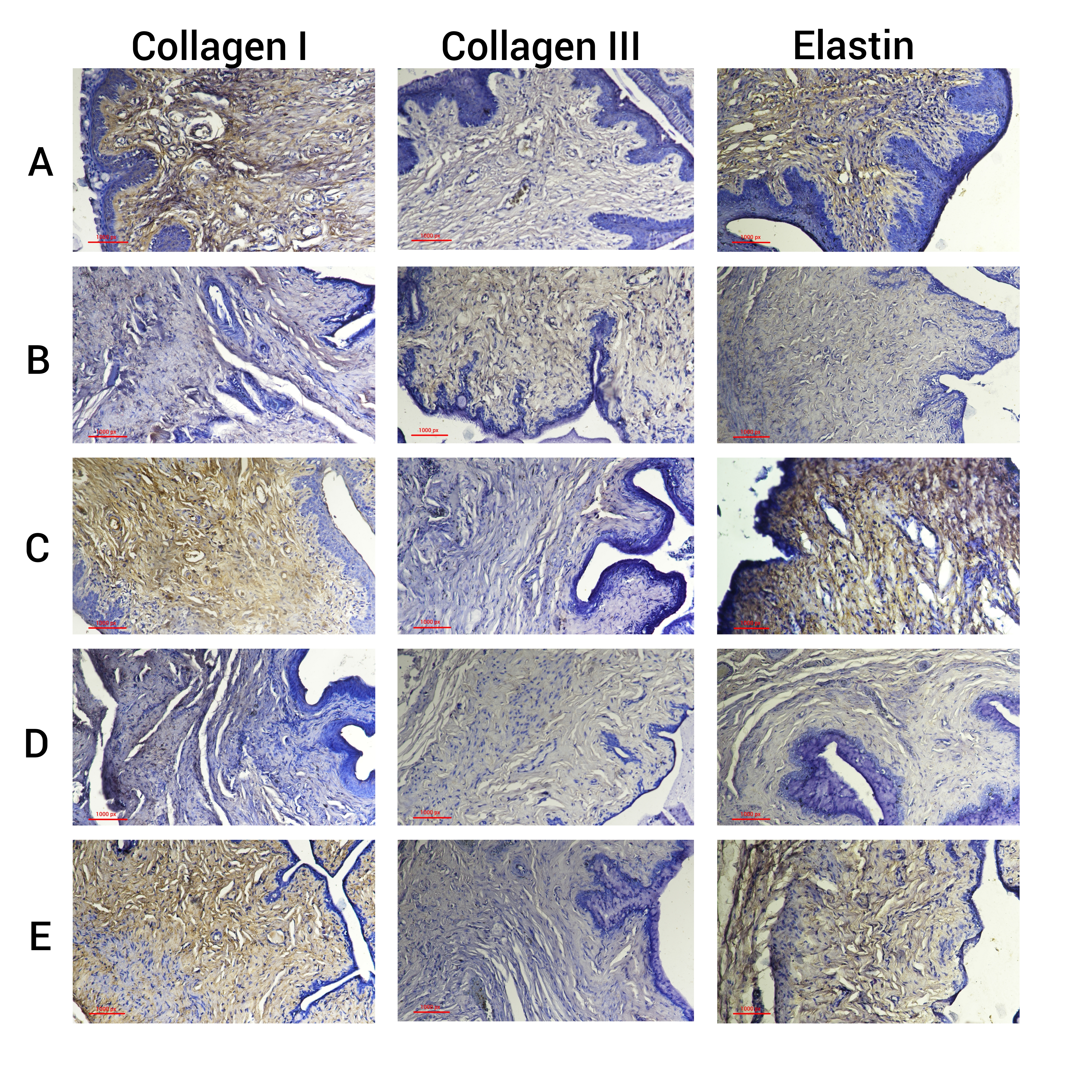 Fig. 2.
Fig. 2.
Immunohistochemical image of collagen I, collagen III, and
elastin vaginal tissue. (A) Regular rat model. (B) Menopausal rat model without
treatment. (C) Menopausal rat model with 17-
| Group | N | Mean |
One-Way ANOVA test |
| K0 | 6 | 10.13 |
p |
| K1 | 6 | 3.90 | |
| K2 | 6 | 8.70 | |
| K3 | 6 | 4.76 | |
| K4 | 6 | 6.97 |
SD, standard deviation; ANOVA, analysis of variance. ***p
| Group | p-value | ||||
| K0 | K1 | K2 | K3 | K4 | |
| K0 | 0.029* | ||||
| K1 | 0.172 | ||||
| K2 | 0.029* | 0.010* | |||
| K3 | 0.172 | 0.001* | |||
| K4 | 0.010* | 0.001* | |||
LSD, Least Significant Difference. *p
The highest average expression of collagen III was observed in the control group
of menopausal model rats without treatment (7.10
| Group | N | Mean |
One-Way ANOVA test |
| K0 | 6 | 2.83 |
p |
| K1 | 6 | 7.10 | |
| K2 | 6 | 4.00 | |
| K3 | 6 | 6.87 | |
| K4 | 6 | 4.37 |
SD, standard deviation. ***p
| Group | p-value | ||||
| K0 | K1 | K2 | K3 | K4 | |
| K0 | 0.196 | 0.093 | |||
| K1 | 0.002* | 0.793 | 0.005* | ||
| K2 | 0.196 | 0.002* | 0.003* | 0.680 | |
| K3 | 0.793 | 0.003* | 0.009* | ||
| K4 | 0.093 | 0.005* | 0.680 | 0.009* | |
*p
The highest average expression of elastin was observed in the control group of
untreated normal rats (10.10
| Group | N | Mean |
One-Way ANOVA test |
| K0 | 6 | 10.10 |
p |
| K1 | 6 | 4.13 | |
| K2 | 6 | 8.10 | |
| K3 | 6 | 4.47 | |
| K4 | 6 | 6.30 |
SD, standard deviation. ***p
| Group | p-value | ||||
| K0 | K1 | K2 | K3 | K4 | |
| K0 | 0.030* | ||||
| K1 | 0.704 | 0.019* | |||
| K2 | 0.030* | 0.048* | |||
| K3 | 0.704 | 0.045* | |||
| K4 | 0.019* | 0.048* | 0.045* | ||
*p
Vulvovaginal atrophy is a component of GSM, characterized by symptoms and signs resulting from decreased estrogen levels in various parts of the female genitalia and urinary system in menopausal women. The alterations result in troubling symptoms such as vaginal dryness, itching, painful sexual intercourse, more frequent urination, sudden need to urinate, and urine incontinence [12]. The menopausal genitourinary syndrome is associated with estrogen deficiency resulting in reduced elastin and collagen in the vaginal tissue, thinning of the vaginal epithelium, and increased vaginal pH [13]. It demonstrates how the structural integrity of vaginal tissues is significantly impacted by estrogen deficiency, which also contributes to the symptoms of GSM by reducing crucial extracellular matrix components. This research highlights the possible therapeutic use of substances like quercetin, which may improve vaginal tissue integrity and lessen symptoms of GSM by modifying the expression of collagen and elastin [14, 15].
Collagen I levels are lower in menopausal models compared to non-menopausal
models or those who have had therapy. These results show that treatment with
quercetin 50 mg/kgBW provides a significant effect compared to the menopause
model without treatment. However, it does not offer the same results as estrogen
therapy. Postmenopausal women exhibit reduced levels of collagen I expression and
thinner vaginal wall epithelium compared to premenopausal women. Menopause
without hormone medication leads to a reduction in collagen I levels [16].
Estrogen significantly impacts the production of collagen in the pelvic region.
Estrogen can stimulate the expression of type I collagen and destroy type III
collagen in fibroblasts, resulting in greater muscle strength. In addition,
estrogen can also improve total collagen cross-linking and replace old collagen
with new tissue [17]. Differences in vaginal collagen I expression were observed
among various treatment groups, including normal mice and menopausal model mice
receiving different treatments, such as 17-
The largest average of collagen III expression was located in the control group of the menopause model without treatment, and the lowest average was in the control group of the standard model without treatment. The results of this study show that quercetin at a dose of 50 mg/kgBW provides the same effect as estrogen therapy in normal mice. Collagen III expression is influenced by therapy and the beginning of menopause. Post-menopause involves a gradual decrease in ovarian function, leading to particular alterations in organs and tissues, such as atrophy of the labia, vagina, and uterus [16]. Type III collagen and type I collagen are the main constituents of the interstitial matrix. Type III collagen mutations are associated with Ehlers-Danlos syndrome, vascular deficiencies, and aortic and arterial aneurysms [19]. Changes in collagen distribution during wound healing and extracellular matrix (ECM) remodeling during injury where the ratio of collagen I and collagen III is altered with a higher amount of collagen III during healing while increased collagen I in a healed wound [20].
The highest average level of elastin expression was observed in the control group of normal mice. The lowest average level was found in the control group of menopausal model mice without any treatment. These results show that therapy with quercetin 50 mg/kgBW provides a significant effect compared to the menopause model without treatment. However, it does not give the same results as estrogen therapy. The decrease in estrogen levels results in a reduction of vaginal epithelial permeability, perfusion, and elastin levels, resulting in vaginal dryness and atrophy. Therefore, it can be assumed that one of the causes of vaginal atrophy may be an estrogen deficiency resulting from the aging process, leading to a decrease in the expression of the Lysyl oxidase (LOX) family proteins and a reduction in the level of elastin in the vagina [21]. In postmenopausal women, the expression of elastin and the thickness of the vaginal wall epithelium are reduced compared to premenopausal women. Estrogen affects the elastin metabolism in relation to the matrix metalloproteinase (MMP) enzyme. It decreases the expression of MMP-2 and MMP-9, which degrade elastin [16].
Estrogen replacement therapy is recognized as the primary intervention for managing conditions arising from estrogen deficiency, particularly in postmenopausal women. However, owing to documented adverse effects associated with this approach, there is a notable inclination toward seeking a suitable alternative [22]. A common flavonoid found in fruits and vegetables, quercetin has been extensively studied for various health benefits, including its potential in wound healing and tissue regeneration. It is well-known for its potent anti-inflammatory and antioxidant qualities and estrogen-like effect in vivo and in vitro studies [15, 23].
Our results are consistent with earlier research that demonstrated quercetin’s capacity to regulate the expression of extracellular matrix components, thereby augmenting tissue regeneration and repair [15]. Particularly, quercetin’s impact on collagen synthesis may enhance the elasticity and structural support of vaginal tissues, indicating a possible therapeutic use for disorders affecting the integrity and function of the vagina [14].
Numerous studies have been conducted on the health benefits of quercetin, which may include improving skin health by increasing the production of collagen and elastin. Recent research has highlighted several mechanisms through which quercetin achieves these effects. Quercetin is well-known for having strong antioxidant qualities. Free radicals are scavenged, and oxidative stress is decreased, which can harm skin proteins like collagen and elastin. Quercetin helps preserve the structural integrity of these proteins, which is necessary for the elasticity and firmness of skin, by reducing oxidative damage [15, 24]. Moreover, it has been demonstrated that quercetin inhibits the activity of enzymes responsible for the breakdown of collagen and elastin. In particular, it lessens the activity of the enzymes known as MMPs, which break down the skin’s collagen fibers. Quercetin preserves collagen levels by blocking MMPs, improving skin resilience, and lessening skin aging signs [15, 25].
By altering the expression of genes linked to collagen synthesis, quercetin also
encourages the synthesis of new collagen. Collagen type I and III expression are
upregulated, essential for skin elasticity and strength. This effect is
especially helpful in preventing the menopause-related loss of collagen, which
results in skin that is thinner and less elastic [24, 25]. Moreover, the
anti-inflammatory characteristics of quercetin are essential for preserving skin
health. The deterioration of collagen and elastin may be accelerated by
persistent inflammation. Quercetin protects these proteins and facilitates their
renewal and repair processes by lowering inflammation [15, 26]. A review of the
use of quercetin in osteoporosis has a similar effect of phytoestrogen on
inhibiting bone resorption by participating in the binding of estrogen receptors,
especially in estrogen receptor-
The limitation of this study is that it only takes three primary markers that occur in changes in the condition of vulvovaginal atrophy. Therefore, more research is needed, for example, by examining tissue inhibitor of metalloproteinase-1 (TIMP-1), antigen Kiel 67 (Ki-67), vascular endothelial growth factor (VEGF), aquaporin-2 (AQP-2), and MMP-1, so that this research can describe more complete changes in the condition of vulvovaginal atrophy.
In conclusion, quercetin is a promising option for enhancing skin health and reducing the effects of aging, particularly in menopausal women, because it increases the expression of collagen I, collagen III, and elastin through its antioxidant, enzyme-inhibiting, and anti-inflammatory qualities in the vagina of menopausal model especially by the administration of quercetin at a level of 50 mg/kgBW. Thus, quercetin can be an alternative herbal treatment option to improve vulvovaginal atrophy in menopausal conditions. For further research, studies can be conducted with markers related to vulvovaginal atrophy or pelvic organ prolapse with quercetin as one of the therapy options.
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
THSH, GH, EMK, ISY, HP, BU, and BIS were involved in conceptualizing and designing the research study. THSH was responsible for database management. THSH and BU conducted the statistical analysis. THSH drafted the initial manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final version of the manuscript. Additionally, all authors participated significantly in the research and agreed to take responsibility for all aspects of the work.
This research has been tested for ethical feasibility at the Faculty of Medicine, Airlangga University, to meet the ethical requirements for experimental animals. It has received approval from the research ethics committee with number 215/EC/KEPK/FKUA/2022. The research data and results are only used for knowledge purposes.
We are deeply grateful to everyone who assisted us throughout the composition of this manuscript. Furthermore, we extend our heartfelt thanks to the reviewers for their dedicated time and effort in meticulously reviewing the manuscript and offering invaluable insights to enhance its quality.
This research was funded by Universitas Airlangga Research and Community Service Institute (No. 66/UN3.15/PT/2022).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


