1 Collaborative Innovation Center for Birth Defect Research and Transformation of Shandong Province, Jining Medical University, 272067 Jining, Shandong, China
2 College of Second Clinical Medical, Jining Medical University, 272067 Jining, Shandong, China
3 Lin He's Academician Workstation of New Medicine and Clinical Translation, Jining Medical University, 272067 Jining, Shandong, China
4 College of Basic Medicine, Jining Medical University, 272067 Jining, Shandong, China
†These authors contributed equally.
Abstract
Objective: This review aims to provide some theoretical guidance for the precise treatment of ovarian cancer and the development of estrogen-related drugs. Mechanism: Ovarian cancer is one of the leading causes of death in gynecological cancer patients, mainly affecting middle-aged and elderly women. It has the characteristics of hidden location, strong heterogeneity and lack of specific symptoms in the early stage. Numerous studies have shown that estrogen receptor (ER) plays an important role in different types of cancer, including ovarian cancer. Accordingly, the study of ER signaling pathways and related regulatory factors in ovarian cancer cells should help us understand the pathogenesis of ovarian cancer. Findings in Brief: The expression of estrogen receptor subtypes is related to ovarian cancer gene and leads to ovarian cancer. Estrogen receptor modulators appear to be an important factor in the prognosis of patients with ovarian cancer after hormone therapy. Conclusions: This review summarizes the regulatory mechanism of ER in the occurrence and development of ovarian cancer and outlines the specific role of estrogen receptor modulators (SERMs) in the treatment and prevention of ovarian cancer.
Keywords
- ovarian cancer
- estrogen
- estrogen receptor
- receptor modulators
- precise treatment
Ovarian cancer, one of the most common cancers in women, is evaluated as one of the leading causes of death in the world. A cancer survey report in Poland in 2019 showed that the incidence of ovarian cancer in women in the country ranked fifth, lower than breast cancer and cervical cancer, but higher than gastric cancer, colon cancer and pancreatic cancer [1]. Due to its late diagnosis, it is typically diagnosed at an advanced stage. In recent years, the overall survival rate has not significantly improved, while treatment efficacy remains unsatisfactory; it is therefore necessary to intensify the study of the occurrence and development of ovarian cancer [2, 3]. New findings on estrogen have uncovered its association with ovarian cancer, highlighting the pivotal role of estrogen receptors (ER) as its primary target. These investigations propose that ER could serve as a crucial contributing factor in the development of ovarian cancer [4, 5]. Drawing upon recent research advancements, this comprehensive review highlights the intricate relationship between ovarian cancer, estrogen and its receptors. It thoroughly examines the regulatory influence exerted by ERs and selective estrogen receptor modulators (SERMs) on the initiation and progression of ovarian cancer. Furthermore, this review anticipates the promising potential of SERMs in the treatment of ovarian cancer, paving the way for a broader spectrum of therapeutic avenues [6].
Estrogen (E), a class of steroid hormones that includes estradiol and estrone,
is mainly secreted by the ovaries. It plays an important role in reproductive
organs such as the ovaries, testes, uterus, and non-reproductive organs such as
skeletal muscles and mammary glands [7]. ER is a protein receptor found in many
tissues such as ovary that binds to estrogen. It can be divided into nuclear
receptor and membrane receptor: Among them, ER
As a common cancer affecting women, ovarian cancer can be divided into different types. Based on the classification of the primary anatomical location, ovarian cancer is divided into two main types: primary ovarian cancer (POC), which originates within the ovary itself, and secondary metastatic ovarian cancer (SMOC), which is characterized by the spread of cancer cells from other tissues to the ovary. POC can be divided into epithelial, germ cell and specific sex cord interstitial sources, among which epithelial ovarian cancer accounts for the highest proportion [14]. Kajiyama et al. [15] found that there are roughly four primary sites of SMOC, ranked in descending order of occurrence: the gastrointestinal tract, appendix, breast and pancreas. The incidence of gastrointestinal ovarian cancer is the highest, far more than the sum of the other three, and the harm range is the most extensive [16].
ERs have different mechanisms and outcomes depending upon the types of ovarian cancer, and the same ER can have different outcomes in different types of ovarian cancer. At present, most of the clinical literature focuses on the study of ER on a few types of high-risk ovarian cancer, such as serous ovarian cancer, mucinous ovarian carcinoma. However, some types of ovarian cancer with a low incidence and a high degree of malignancy are rarely mentioned in the current literature. The reasons are as follows: such patients have a high degree of malignancy, low 5-year survival rate, low incidence, few research cases, and lack of funding and research. It is thus of great significance to study how this type of ovarian cancer plays a role in disease progression by affecting ER, and to increase the research on the lesser studied types of ovarian cancer to elucidate the pathogenesis of ovarian cancer and subsequent new drug development. In several rare cases of ovarian cancer, the literature only describes the clinical manifestations, while the relationship with ER has not been studied thoroughly or at all. Simultaneously, owing to the heterogeneity of ovarian cancer, the efficacy of drug therapy varies. Each drug exhibits distinct effects in the treatment of specific ovarian cancer subtypes. While certain drugs may play a role in one ovarian cancer subtype or in a variety of ovarian cancer subtypes, the role of currently known modulators is not fully elucidated. Exploring the detailed effects of various modulators on different types of ovarian cancer and achieving accurate treatment of drugs will be crucial for the treatment of ovarian cancer. Accordingly, we summarize the degree and source of benign and malignant ovarian cancer according to the type of ovarian cancer (Table 1, Ref. [17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32]), in an effort to supplement the content of this review.
| Type of ovarian cancer | Benign/Malignant | Primary/metastatic (source) | References |
| Fibroma | Benign | Primary (Ĥ specific sex cord stroma) | [17] |
| Mucinous tumors | Benign | Primary (ovarian germinal epithelium) | [18] |
| Serous cystadenoma | Benign | Primary (ovarian germinal epithelium) | [18, 19] |
| Immature Teratoma | Benign | Primary (germ cell) | [20] |
| Asexual cell tumor | Malignant | Primary (germ cell) | [21] |
| Choriocarcinoma | Malignant | Primary (germ cell) | [22] |
| Clear cell carcinoma | Malignant | Primary (ovarian germinal epithelium) | [23] |
| Embryonal carcinoma | Malignant | Primary (germ cell) | [24] |
| Endodermal sinus tumor | Malignant | Primary (germ cell) | [25] |
| Endometrioid tumor | Malignant | Primary (ovarian germinal epithelium) | [26] |
| Granulosa cell tumor | Malignant | Primary (Ĥ specific cord stroma) | [27] |
| Mixed germ cell tumor | Malignant | Primary (germ cell) | [28] |
| Metastatic carcinoma of digestive tract | Malignant | Metastatic (digestive tract) | [29] |
| Metastatic carcinoma of breast | Malignant | Metastatic (Mammary gland) | [30] |
| Metastatic carcinoma of genital tract | Malignant | Metastatic (genital tract) | [31] |
| Serous Ovarian Carcinoma | Malignant | Primary (ovarian germinal epithelium) | [18, 19] |
| Sex cord stromal tumor | Malignant | Primary (Ĥ specific cord stroma) | [32] |
| Testicular blastoma of ovary | Malignant | Primary (Ĥ specific cord stroma) | [31] |
Estrogen, as a fat-soluble steroid hormone, can enter the cell membrane and bind
to nuclear receptors in a free diffusion state, that is, the two combine to form
an E-ER polymer complex on the nuclear membrane [33]. After estrogen binds to
nuclear receptors, the quaternary spatial structure of the complex will change
under the induction of heat shock protein (Hsp90), and then binds to an
ER
Cyclin D1 gene is a kind of proto-oncogene downstream of AP-1 sequence.
Cyclin D1 promotes the proliferation of serous ovarian carcinoma (SOC) cells by
regulating cyclin-dependent kinases (CDKs) to phosphorylate specific target
proteins. The combination of cyclin D1 and CDKs is the rate-limiting step in
regulating the cell cycle [44]. Studies have shown that ER
ER
Several studies have shown that the combination of estrogen and GPER1 can activate MAP kinase pathway by releasing heparin-binding epidermal growth factor (HB-EGF). Overexpression of mitogen-activated protein kinase kinase kinase 3 (MAP3K3) is recognized as an indicator of poor prognosis in ovarian cancer. High expression of phosphorylated p38 MAPK in EOC leads to an increased risk of death in ovarian cancer patients [65, 66]. The increase of GPER1 expression is accompanied by the increase of matrix metalloproteinase-9 (MMP9) (r = 1.000, p = 0.002) [67]. The high expression of MMP9, MMP10, MMP12 and MMP25 is positively correlated with the overall survival rate of ovarian cancer [67, 68]. Based on these findings, GPER1 may be involved in the invasion and metastasis of EOC, and is a potential indicator for early diagnosis and malignant degree prediction of EOC. Schüler-Toprak et al. [69] demonstrated that treatment with GPER1 agonist G-1 led to transcriptome responses related to growth inhibition and reduced the growth of OVCAR-3 and OAW-42 ovarian cancer cell lines.
Nuclear and membrane receptors both play an independent role and can
synergistically interfere with ovarian cancer: hormones bind to a small number of
nuclear receptors in the cytoplasm, which dimerize and migrate to the nucleus and
bind to transcription factors phosphorylated by GPER1, and then bind to ERE and
other sites through nuclear receptors to regulate transcription. ER
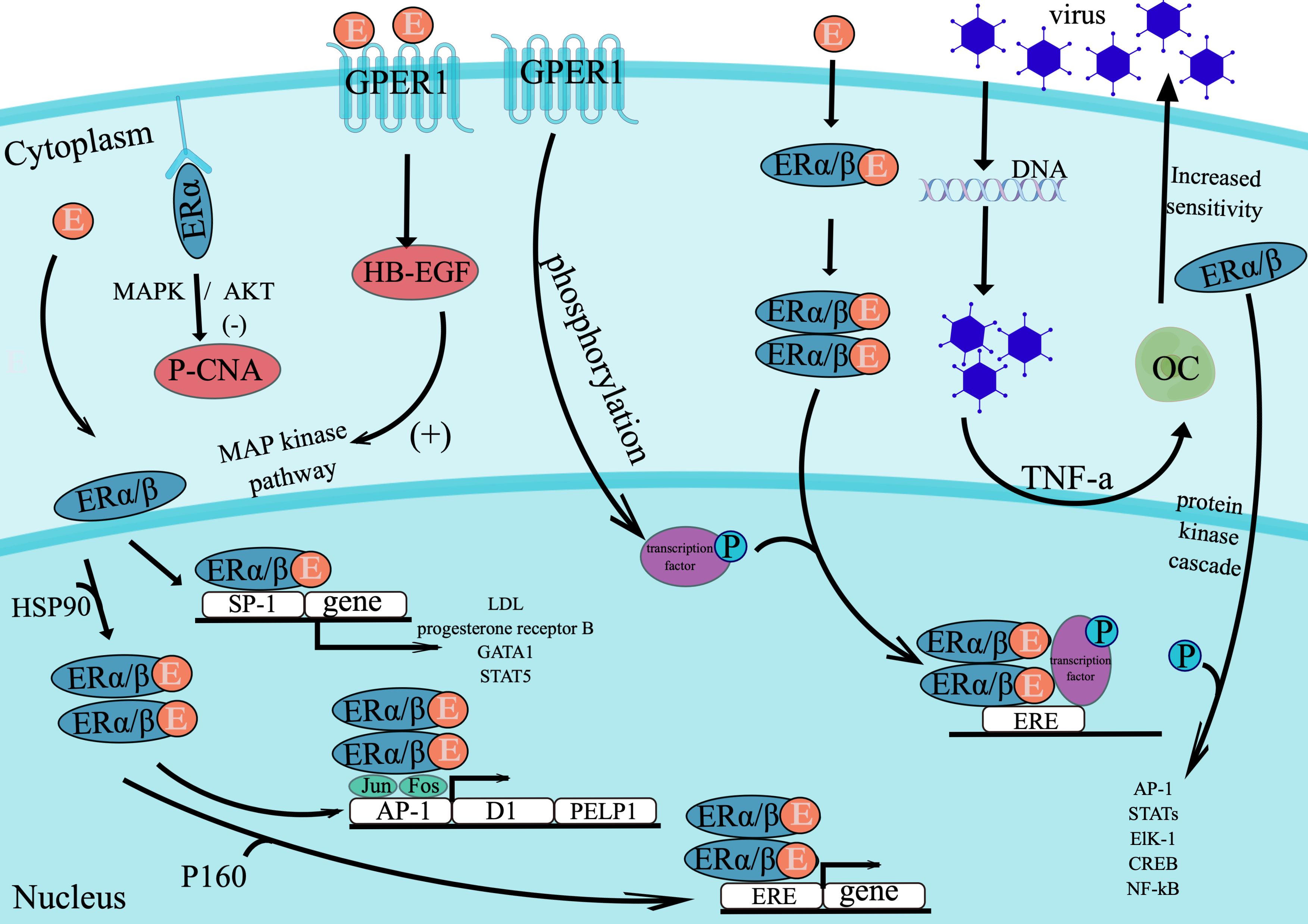 Fig. 1.
Fig. 1.This figure shows the effect of estrogen on cell gene expression by acting on nuclear receptors and membrane receptors. By Figdraw. E, estrogen; ER, estrogen receptor; OC, ovarian cancer cell; virus, oncolytic adenoviruses; P-CAN, proliferating cell nuclear antigen; HB-EGF, heparin-binding epidermal growth factor; ERE, estrogen-response element; P, phosphorylation.
We summarized the risk factors of ovarian cancer and the different effects of ER
modulators on ovarian cancer. Our summary of the risk factors of ovarian cancer
should enhance women’s awareness of prevention and help protect women from
ovarian cancer. Our focus on SERMs should contribute to researchers’ further
exploration of non-toxic and efficient anti-cancer drugs [76, 77]. Risk factors
of ovarian cancer include postmenopausal estrogen therapy, unmarried infertility,
smoking, obesity, and age. [77]. The ovaries of unmarried infertile women will
continue to ovulate, causing damage to the ovarian epithelium, which may lead to
mutations in tumor suppressor genes such as TP53 and BRCA1/2.
Bilateral salpingectomy and delayed salpingectomy (BS/DO) have been found to
reduce the risk of ovarian cancer in BRCA1/2 mutation carriers,
Meanwhile, a study investigated that loss of paired box 2 (PAX2) in murine
oviductal epithelium (MOE) implies a precursor of high-grade serous ovarian
cancer (HGSOC) with secretory cell proliferation (SCOUT), deletion of which
generates a messenger RNA expression pattern [78, 79]. It is closely related to
the enrichment of estrogen signaling genes and the increase of estrogen receptor
Chan et al. [80] reported that MPP inhibits the growth of SKOV3 ovarian
cancer cells by targeted regulation of ER
The combination of ER modulator and receptor can not only regulate ovarian
cancer through PI3K/AKT signal pathway, but also through NF-
| Regulator | CAS number | ER |
ER |
ER |
ER |
Mechanism of action | References |
| Tamoxifen | 10540-29-1 | √ | √ | Tamoxifen blocks the mitosis of estrogen by competitively binding to ER and exerts its antiproliferative effect. | [57] | ||
| curcumin | 458-37-7 | √ | √ | Curcumin promotes the expression of apoptosis promoting protein and Circ-PLEKHM3, inhibit the expression of proliferation protein and AKT/mTOR/p70S6 pathway. | [82, 83] | ||
| Bisphenol AF | 1478-61-1 | √ | √ | BPAF can exert a higher estrogenic effect than BPA through the GPER pathway. | [101] | ||
| Bisphenol C | 79-97-0 | √ | √ | Due to the enlarged halogen bond, BPC has a strong binding ability to the two ERs, and is very active for ER |
[102] | ||
| Bisphenol AP | 1571-75-1 | √ | √ | Halogen atom-based dispersion is the main driving force for the activity of ER |
[103] | ||
| Bispheno B | 77-40-7 | √ | √ | Combination of BPB and GPER leads to activation of subsequent signaling pathways. | [101] | ||
| Bisphenol Z | 843-55-0 | √ | √ | Halogen atom-based dispersion is the main driving force for the activity of ER |
[103] | ||
| Apigenin | 520-36-5 | √ | √ | Apigenin competitively binds to E2 and has weak selectivity for ER |
[104] | ||
| Liq | 578-86-9 | √ | Liq significantly down-regulation of PTCH2 gene expression and decrease of NF-κB activity in SKOV3 and other ovarian cancer cells. | [89] | |||
| ERB-041 | 524684-52-4 | √ | ERB-041 reduces migration and invasion of ovarian cancer cells by mediating FAK and PI3K / AKT signaling pathways to induce cell cycle arrest and apoptosis. | [87] | |||
| WAY200070 | 440122-66-7 | √ | Downregulation of PTCH2 gene expression regulated by ER |
[89] | |||
| 3 |
unk | √ | 3 |
[89] | |||
| OSU-ERb-12 | unk | √ | Increased expression of tumor suppressors FOXO1 and FOXO3a inhibits epithelial-mesenchymal transition (EMT). | [92, 93] | |||
| LY500307 | 533884-09-2 | √ | LY500307 upregulates cell cycle arrest gene CDKN1A to promote ovarian cancer stem cell apoptosis. | [94] | |||
| Diaryl propionitrile | 1428-67-7 | √ | Diaryl propionitrile reduced phosphorylated AKT expression by 80 %. | [80] | |||
| Cyclic Ketoximes | unk | √ | Cyclic Ketoximes recruits coactivators to form active complexes with ER |
[105] |
ER, estrogen receptor;
This review provides an overview of ovarian cancer, estrogen and its receptors,
highlighting the mechanisms underlying ovarian cancer development, the
involvement of estrogen receptors, and the significance of ER modulators. Current
evidence suggests that ER signaling is associated with the development of ovarian
cancer. However, the underlying mechanism of how ER and related signals regulate
the growth of ovarian cancer needs to be further elucidated; further prospective
research is warranted. Goals of such research should be to clarify the pathway of
ER activity, find suitable and convenient therapeutic targets, and to choose
efficient ER
YG and KM contributed to the study conception and design. KM, MZ and HX performed the research. MZ, HX, ZL and WM wrote the manuscript. YG and KM revised the manuscript critically for important intellectual content. ZL, WM, HX, YZ, JX and WL contributed to the acquisition, analysis, or interpretation of data for the work. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This study was supported by grants from Research Fund for Lin He’s Academician Workstation of New Medicine and Clinical Translation in Jining Medical University (JYHL2021MS13), Teachers Research Support Fund in Jining Medical University (JYFC2019KJ001) and College Students’ Innovation Training Program of Jining Medical University (cx2021038).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

