1 Department of Chinese Integrative Medicine, Women’s Hospital, School of Medicine, Zhejiang University, 310006 Hangzhou, Zhejiang, China
2 Department of Epidemiology, School of Public Health, Zhejiang Chinese Medical University, 310053 Hangzhou, Zhejiang, China
3 Department of Chinese Medicine, Xinchang People’s Hospital, 312500 Shaoxing, Zhejiang, China
4 The Fourth Affiliated Hospital, School of Medicine, Zhejiang University, 322000 Yiwu, Zhejiang, China
†These authors contributed equally.
Abstract
Background: Observational studies have reported that individuals
diagnosed with polycystic ovary syndrome (PCOS) face a heightened vulnerability
to developing post-traumatic stress disorder (PTSD). However, it is unclear
whether this relationship is causal. Consequently, we implemented a bidirectional
Mendelian randomization (MR) analysis to examine the empirical causal association
of PCOS and PTSD. Methods: We acquired genetic association data for PCOS
through a comprehensive meta-analysis from several large-scale genome-wide
association studies (GWASs), which enrolled 10,074 cases and 103,164 controls.
For PTSD, we obtained data from a GWAS performed by the Psychiatric Genomics
Consortium Posttraumatic Stress Disorder (PGC-PTSD) group. The study included a
total of 23,212 cases of PTSD and 151,447 controls of European ancestry. For both
PCOS and PTSD, we carefully selected genetic instruments that met the rigorous
significance threshold (p
Keywords
- genetic instruments
- Mendelian randomization
- polycystic ovary syndrome
- post-traumatic stress disorder
Polycystic ovary syndrome (PCOS) is a widespread endocrine condition that primarily affects women during their reproductive years. It is characterized by several typical clinical features, including hyperandrogenism (acne, hirsutism, and alopecia), menstrual irregularity (long menstrual cycles, oligo/anovulation), and the presence of polycystic ovaries [1, 2]. It has been estimated that in 2019, approximately 66.0 (95% uncertainty interval (UI): 46.0–86.3) million individuals were affected by PCOS worldwide, with an overall prevalence rate of 829.6 per 100,000 [3].
Despite the precise etiology and pathogenesis of PCOS remaining uncertain,
available evidence suggests that its occurrence is shaped by a blend of genetic
and environmental factors. Established risk factors for PCOS encompass genetic
susceptibility [4], obesity [5] and insulin resistance (IR) [6, 7]. Vink
et al. [4] conducted a study revealing that the similarity of PCOS among
monozygotic twin sisters (tetrachoric r
Post-traumatic stress disorder (PTSD) is a psychiatric disorder that arises and persists after an individual personally experiences, witnesses, or is confronted with one or more traumatic events. These events can involve the individual themselves or others, and the resulting symptoms can endure for a significant period of time, affecting their mental and emotional well-being [11]. The presence of comorbid mental disorders, including major depression, is commonly observed in individuals with PTSD. Moreover, those affected by PTSD face an elevated risk of suicidal ideation, substance abuse, and impaired occupational functioning [12]. Multiple medical conditions have been associated with a heightened vulnerability for developing PTSD. A recent meta-analysis revealed that the combined prevalence rate of PTSD among children and adolescents diagnosed with cancer was about 20.90% (95% CI: 13.28–29.73%) [13]. Among adult patients, in turn, there is a higher prevalence of post-traumatic stress disorder (PTSD) observed in those with chronic heart diseases (CHDs) when compared to those without CHDs (0.8% vs. 0.5%; adjusted prevalence ratio (Apr): 1.5, 95% CI: 1.2–1.8). [14]. In addition, a study conducted by Tay et al. [15] revealed that women with PCOS had a notably higher prevalence of PTSD compared to women without PCOS (adjusted odds ratio (OR) = 1.5, 95% confidence interval (CI): 1.1–1.9). Nevertheless, as this finding stems from a conventional observational epidemiological study, it is susceptible to potential biases, including confounding and reverse causation. Therefore, the causal relationship of the observed association remains ambiguous.
Mendelian randomization (MR) is a genetic epidemiological method that leverages single nucleotide polymorphisms (SNPs) strongly associated with the exposure of interest as instrumental variables (IVs). This approach enables the estimation of potential causal relationships between the exposure and the outcome in question [16]. Assuming that genotypes are randomly allocated during gamete formation and disease onset, the application of the IV approach effectively addresses the problems of confounding and reverse causation commonly encountered in observational studies. This approach helps address these concerns to a considerable extent [17, 18]. Therefore, the present study implemented a two-sample Mendelian randomization (MR) analysis to investigate the possible causal relationship between PCOS and PTSD.
The study design, as presented in Fig. 1, used a bidirectional MR analysis to identify the causality between PCOS and PTSD. To accomplish this, we utilized summary data obtained from published genome-wide association studies (GWAS). To ensure the robustness of the MR study, it is essential to fulfill the following critical assumptions: (i) the genetic variants chosen as instrumental variables (IVs) must exhibit strong associations with the exposure of interest; (ii) the IVs employed should not be linked to any confounding factors; and (iii) the IVs should solely influence the outcome through the exposure being investigated [18].
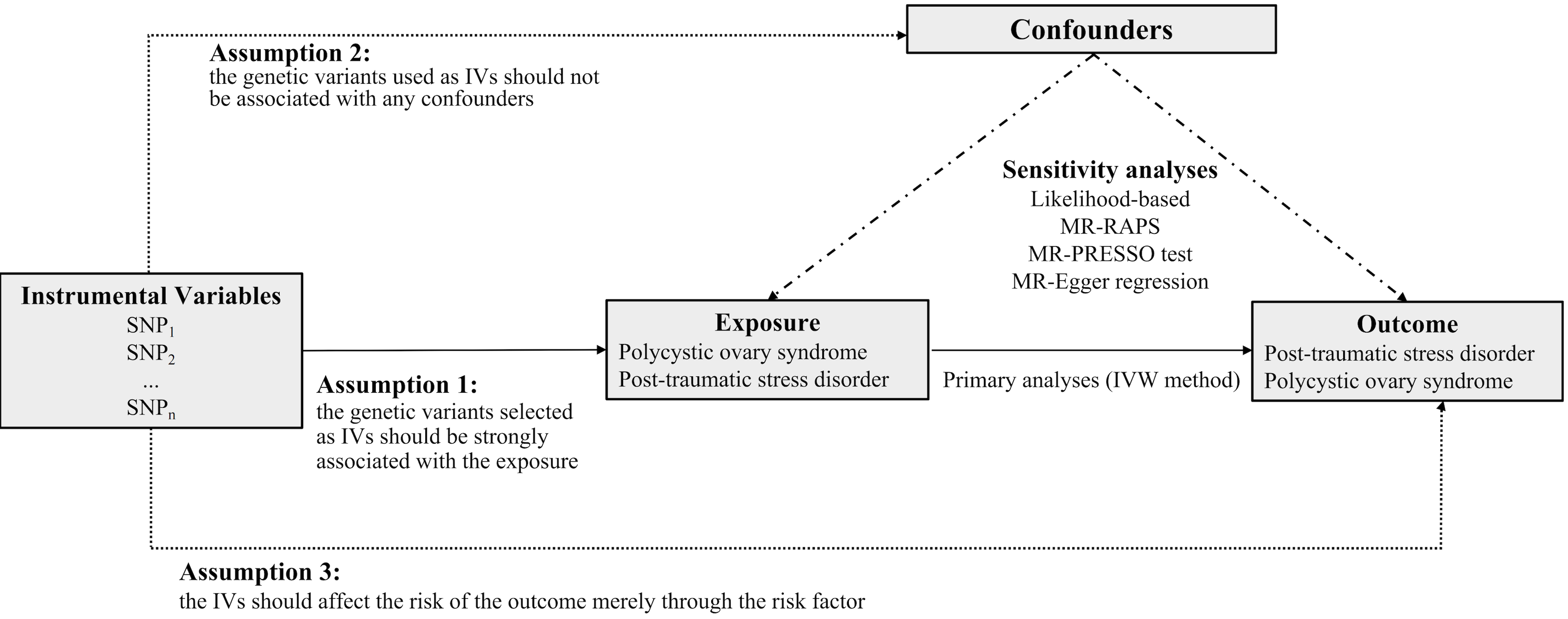 Fig. 1.
Fig. 1.The overall design of the present MR study. MR, Mendelian randomization; SNP, single nucleotide polymorphism; IVs, instrumental variables; MR-RAPS, Mendelian randomization-Robust Adjusted Profile Score; MR-PRESSO, MR Pleiotropy RESidual Sum and Outlier; IVW, inverse-variance weighted.
We acquired summary-level genetic association data for PTSD from the Psychiatric Genomics Consortium Posttraumatic Stress Disorder (PGC-PTSD) group. This dataset consisted of 23,212 individuals diagnosed with PTSD and 151,447 controls of European ancestry [19]. The determination of PTSD status was based on established criteria, while the controls primarily consisted of individuals who had experienced trauma but did not meet the diagnostic criteria for PTSD. Genotyping was carried out using Illumina genotyping arrays, and the association between SNPs and PTSD was assessed using logistic regression in PLINK 1.9 (National Institutes of Health, Bethesda, MD, USA) , incorporating the first five principal components as covariates, under an additive model.
We obtained genetic association data for PCOS from a meta-analysis of genome-wide association studies (GWAS), which encompassed a total of 10,074 cases and 103,164 controls of European ancestry [20]. This represents the most extensive GWAS meta-analysis conducted on PCOS thus far, comprising participants from seven distinct cohorts, including Rotterdam, Estonian Genome Center of the University of Tartu (EGCUT), deCODE, the UK (London/Oxford), Chicago, Boston, and 23andMe. Diagnosis of PCOS among cases was established based on the criteria outlined by the National Institute of Health (NIH) or the Rotterdam Criteria. Due to the unavailability of publicly accessible data from 23andMe, the study population ultimately consisted of 4138 individuals diagnosed with PCOS and 20,129 controls. The comprehensive details regarding the data sources utilized in this study are provided in Supplementary Table S1. As this study solely relied on published summary-level GWAS datasets, no further ethical approval was deemed necessary.
To ascertain independent SNPs associated to PCOS risk, we employed a rigorous
significance threshold (p
| SNP | Chr | Position | Effect allele | β | SE | p-value | Variance explained (r |
F-statistic | |
| PCOS | |||||||||
| rs7563201 | 2 | 43561780 | A | –0.108 | 0.017 | 3.68 × 10 |
0.000348 | 39.43 | |
| rs2178575 | 2 | 213391766 | A | 0.166 | 0.022 | 3.34 × 10 |
0.000507 | 57.45 | |
| rs13164856 | 5 | 131813204 | T | 0.124 | 0.019 | 1.45 × 10 |
0.000364 | 41.28 | |
| rs804279 | 8 | 11623889 | A | 0.128 | 0.018 | 3.76 × 10 |
0.000427 | 48.39 | |
| rs10739076 | 9 | 5440589 | A | 0.11 | 0.02 | 2.51 × 10 |
0.000275 | 31.18 | |
| rs7864171 | 9 | 97723266 | A | –0.093 | 0.017 | 2.95 × 10 |
0.000271 | 30.64 | |
| rs9696009 | 9 | 126619233 | A | 0.202 | 0.031 | 7.96 × 10 |
0.000372 | 42.19 | |
| rs11031005 | 11 | 30226356 | T | –0.159 | 0.022 | 8.66 × 10 |
0.000449 | 50.84 | |
| rs11225154 | 11 | 102043240 | A | 0.179 | 0.027 | 5.44 × 10 |
0.000382 | 43.31 | |
| rs1784692 | 11 | 113949232 | T | 0.144 | 0.023 | 1.88 × 10 |
0.000358 | 40.6 | |
| rs2271194 | 12 | 56477694 | A | 0.097 | 0.017 | 4.57 × 10 |
0.000301 | 34.14 | |
| rs1795379 | 12 | 75941042 | T | –0.117 | 0.02 | 1.81 × 10 |
0.000318 | 36 | |
| rs8043701 | 16 | 52375777 | A | –0.127 | 0.021 | 9.61 × 10 |
0.000329 | 37.28 | |
| rs853854 | 20 | 31420757 | A | –0.098 | 0.016 | 2.36 × 10 |
0.000319 | 36.15 | |
| PTSD | |||||||||
| rs34517852 | 6 | 157789333 | A | 0.11 | 0.02 | 3.10 × 10 |
0.000173 | 30.25 | |
| rs9364611 | 6 | 162163506 | T | –0.124 | 0.02 | 4.30 × 10 |
0.00022 | 38.44 | |
Chr, chromosome; PCOS, polycystic ovary syndrome; PTSD, post-traumatic stress disorder; SE, standard error; SNP, single nucleotide polymorphism.
To evaluate the strength of the instrumental variables (IVs), we calculated
F-statistics using the formula: F = r
Specifically, the IVW approach was presented, which combines the effects of IVs
and provides a weighted effect estimate. This approach assumes the validity of
all IVs and aims to yield a consistent estimate by considering the inverse of
their variances [22]. To assess the heterogeneity of the IVs used in the
analysis, we performed Cochran’s Q test to assess heterogeneity. If heterogeneity
was present (p
All statistical analyses were conducted using R software version 3.6.3 (R Project for Statistical Computing, Vienna, Austria), using the Mendelian randomization, Two-Sample MR, and MRPRESSO packages. A two-sided p-value threshold of 0.05 was considered statistically significant for all analyses.
The F-statistics for the IVs chosen for PCOS ranged from 30.64 to 57.45, while
for PTSD they ranged from 30.25 to 38.44. These values
Genetic predisposition to PCOS was found to be associated with an elevated risk
of developing PTSD (OR = 1.11, 95% CI: 1.03–1.19, p = 7.27
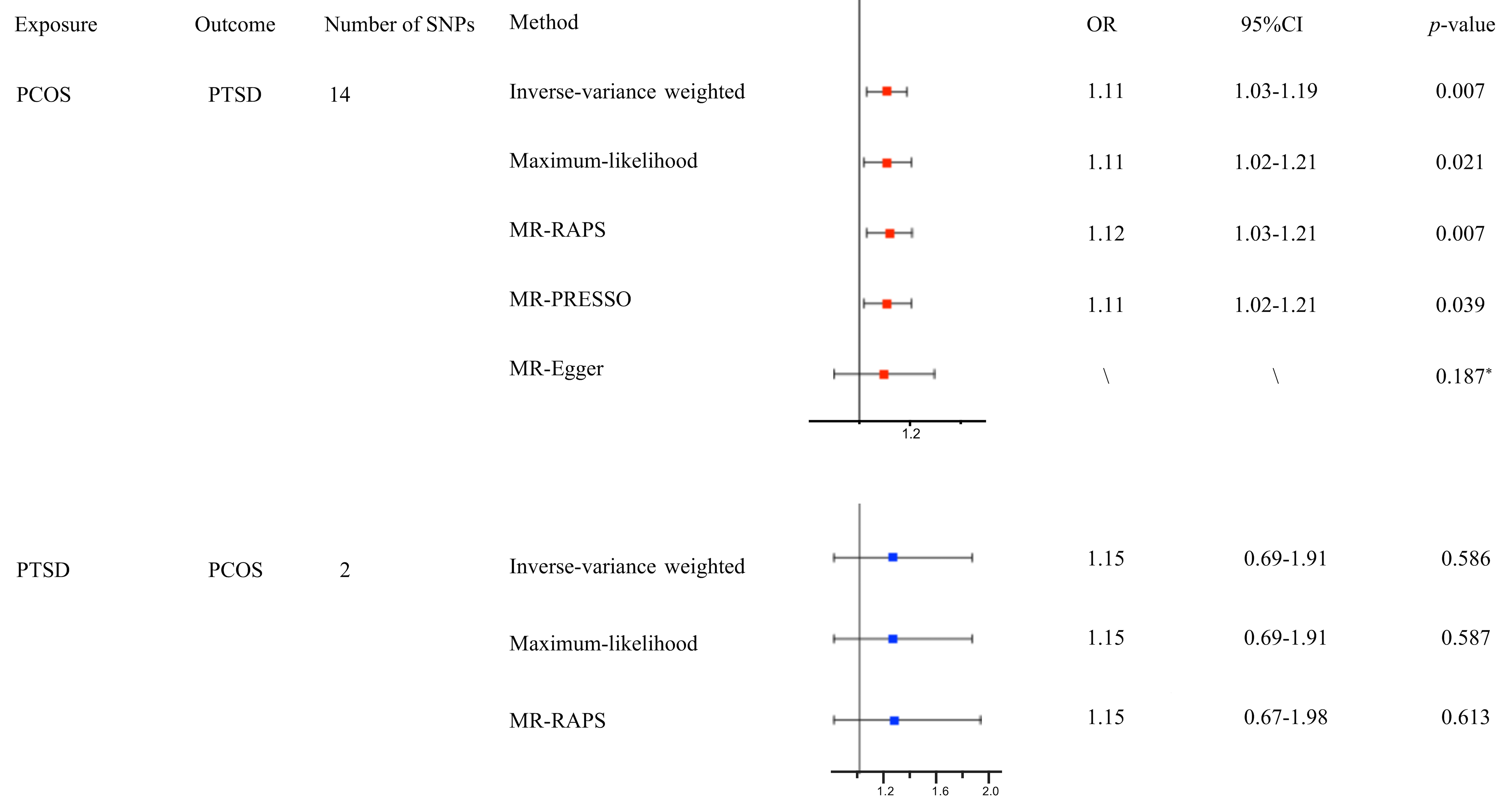 Fig. 2.
Fig. 2.MR effect estimates for associations of exposures with the risk of outcomes. *p-value of the intercept from MR-Egger regression. SNP, single nucleotide polymorphism; MR-RAPS, Mendelian randomization-Robust Adjusted Profile Score; MR-PRESSO, MR Pleiotropy RESidual Sum and Outlier; PCOS, polycystic ovary syndrome; PTSD, post-traumatic stress disorder; OR, odds ratio; CI, confidence interval.
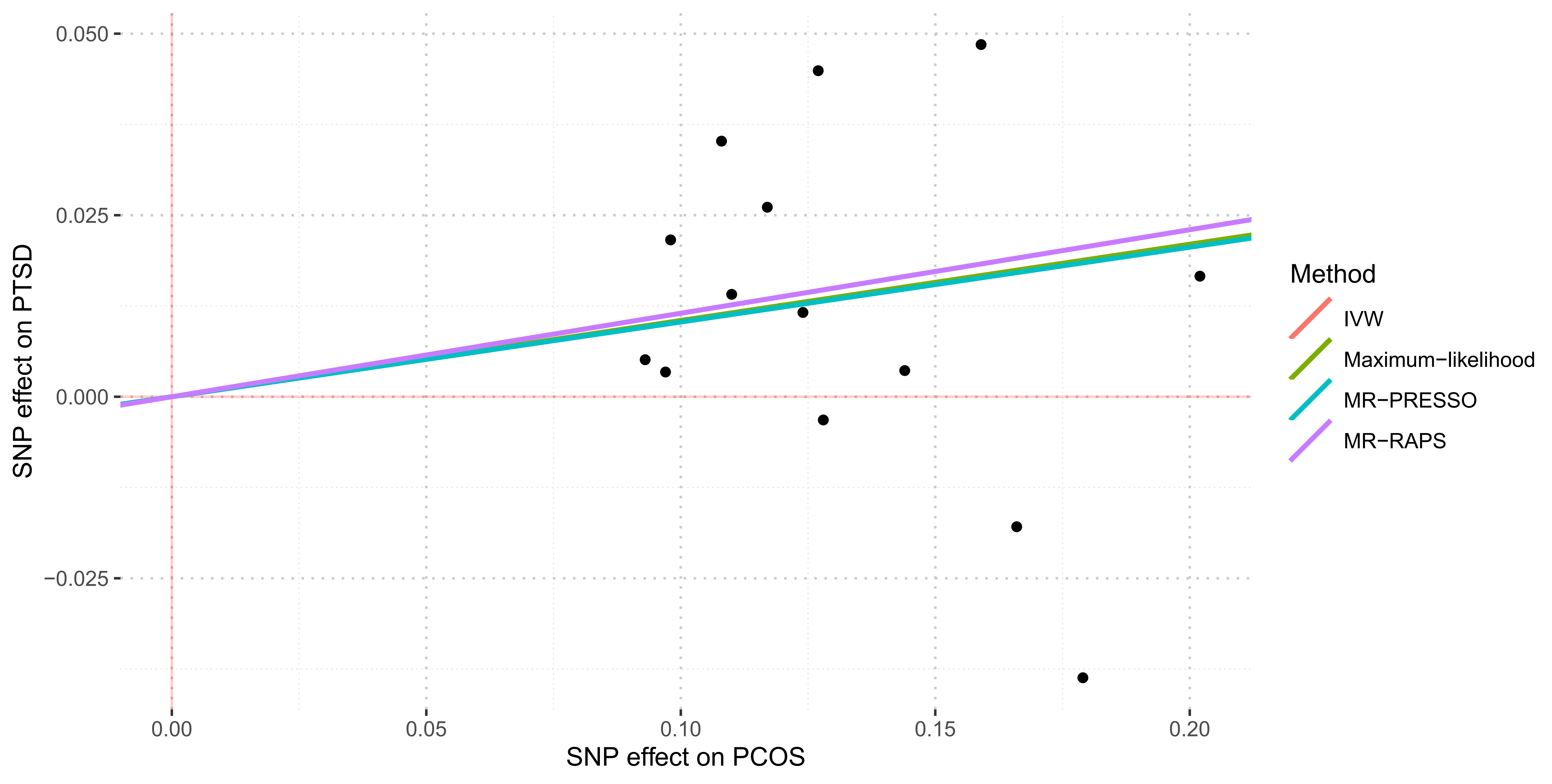 Fig. 3.
Fig. 3.Plot of the effect size and 95% CIs of each SNP on PCOS and PTSD risk. The x-axis plots the previously published b-estimate for the association of each SNP with PCOS. The y-axis plots the bestimate for the association of each SNP with risk of PTSD. Error bars indicate 95% CIs of the effect estimates. Slopes of the orange green blue and purple lines represent the combined causal effect estimates using the IVW method, maximum-likelihood method, MR-PRESSO test and MR-RAPS method. IVW, inverse-variance weighted; SNP, single nucleotide polymorphism; MR-RAPS, Mendelian randomization-Robust Adjusted Profile Score; MR-PRESSO, MR Pleiotropy RESidual Sum and Outlier; PCOS, polycystic ovary syndrome; PTSD, post-traumatic stress disorder.
During our scan of the SNPs assigned as IVs in the GWAS Catalog, we obtained three SNPs (rs11031005, rs13164856, and rs2271194) that showed associations with other traits or phenotypes (Supplementary Table S2). After excluding these SNPs, the association between PCOS and PTSD remained statistically significant (OR = 1.09, 95% CI: 1.00–1.20, p = 0.044) using the IVW method, indicating that the relationship between PCOS and PTSD is robust even without these particular SNPs.
The reverse MR analysis indicated that genetic predisposition to PTSD was not significantly associated with the risk of PCOS (OR = 1.15, 95% CI: 0.69–1.91, p = 0.586) using the IVW method. The sensitivity analyses, including the maximum-likelihood method (OR = 1.15, 95% CI: 0.69–1.91, p = 0.587) and MR-RAPS (OR = 1.15, 95% CI: 0.67–1.98, p = 0.613), consistently yielded similar causal effect estimates (Fig. 2). The results from leave-one-out method illustrated that the combined effect estimates were consistent after removing SNPs in turn (Supplementary Fig. S1).
In this study, we employed a bidirectional MR approach to examine the potential causal link between PCOS and PTSD. Our findings revealed a significant causality between genetic susceptibility to PCOS and the developing risk of PTSD. The results remained consistent and robust across various sensitivity analyses utilizing different MR methods and alternative instrumental variable sets. These results indicate the stability of our findings. Conversely, we observe non-significant evidence between genetic predisposition to PTSD and the increasing risk of PCOS.
Previous observational studies have demonstrated a significant association of
PCOS condition and PTSD onset. For instance, a cross-sectional analysis including
89 women with PCOS and 456 women without PCOS revealed that PCOS was strongly
associated with an increased risk of PTSD (OR = 2.2, 95% CI: 1.7–2.8,
p
Psychiatric disorders in individuals with PCOS are influenced by intricate mechanisms. One contributing factor is the presence of PCOS-related manifestations, including acne, hirsutism, obesity, and infertility. These physical features can potentially diminish self-esteem and amplify psychological distress among affected women. Furthermore, hormonal imbalances associated with PCOS can also impact mental well-being. A study by Marsh et al. [27] demonstrated that individuals with PCOS exhibited lower levels of positive mood and higher levels of trait anxiety in comparison to control groups. Moreover, elevated levels of total testosterone and insulin, as well as insulin resistance, were found to be associated with increased scores in trait anger and anger expression [28]. The hormonal imbalance in individuals with PCOS can also impact the microstructure of white matter, thereby affecting cognitive function [29] and work efficiency [30] in PCOS patients. Furthermore, hyperparathyroidism is a significant clinical manifestation observed in PCOS, and it can influence mood disorders and impulsivity in both women with and without PCOS [28, 31, 32]. Lastly, there is evidence suggesting that women with PCOS may exhibit an increased responsiveness of the hypothalamic-pituitary-adrenal (HPA) axis, which could contribute to the development of psychiatric disorders through disrupted stress responses [33, 34]. While these explanations offer biologically plausible connections, additional research is needed to elucidate the precise underlying mechanisms through which PCOS contributes to the development of PTSD.
The application of genetic variants as IVs in MR analyses greatly addresses the
challenge of confounding present in conventional observational studies.
Nevertheless, it is important to acknowledge several limitations in our study.
Certain limitations should be considered when interpreting the results of our
study. Firstly, the participants were predominantly of European descent, which
raises the question of generalizability to other ethnic populations. Additional
research is warranted to assess the generalizability of our findings across
diverse populations. Furthermore, it is worth noting that the lack of a
statistically significant association between PTSD and PCOS in our study may be
influenced by the limited statistical power and the relatively small size of the
instrumental variable used in the Mendelian randomization analysis. Thus,
additional studies with larger sample sizes are warranted to further investigate
the potential relationship between PTSD and PCOS. To address concerns regarding
the validity of the MR study, we took measures to ensure the selection of
appropriate instrumental variables (IVs). Specifically, we included independent
SNPs that reached the genome-wide significance level (p
Through the utilization of a genetic epidemiological approach, our study revealed a potential causality between genetic predisposition to PCOS and an increasing odd of PTSD. This finding indicates a potential causal relationship between PCOS and PTSD. Nevertheless, it is crucial to delve deeper into the underlying mechanisms that link PCOS to the development of PTSD. Further investigation in this area will provide valuable insights and contribute to a comprehensive understanding of the interplay between PCOS and PTSD.
CI, confidence interval; GWAS, genome-wide association studies; IV, instrumental variable; IVW, inverse-variance weighted; LD, linkage disequilibrium; MR, mendelian randomization; OR, odds ratio; PCOS, polycystic ovary syndrome; PGC-PTSD, Psychiatric Genomics Consortium Post-traumatic Stress Disorder; PRESSO, Pleiotropy RESidual Sum and Outlier; PTSD, post-traumatic stress disorder; RAPS, Robust Adjusted Profile Score; SNPs, single nucleotide polymorphisms.
The datasets generated and/or analyzed during the current study are available in the genome-wide association studies repository. (PCOS: https://doi.org/10.17863/CAM.36024. PTSD: https://www.med.unc.edu/pgc/download-results/)
XZ and JS performed the literature review, conducted data analysis, interpreted findings, and drafted the manuscript. BL and MCD carried on data analysis and interpreted findings. BXW, XWC and YFH mainly conducted on the data collation and checked the manuscript. YYM and FQ directed analytic strategy, supervised the study from conception to completion and revised drafts of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity.
Since this project is based on the statistical analysis of publicly available databases and published studies, ethics approval and consent to participate is not applicable.
The author sincerely thanks the researchers and participants of the original GWAS for their collection and management of large-scale data resources, as well as those who actively participated in this study.
This research was funded by National Natural Science Foundation of China (No. 82074476); Traditional Chinese Medicine Scientific Project of Zhejiang Province for the Excellent Young Scholars (No. 2021ZQ055).
The authors declare no conflict of interest. Fan Qu is serving as one of the Guest editors of this journal. We declare that Fan Qu had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Michael H. Dahan.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



