1 Department of Obstetrics and Gynecology, The Affiliated Hospital of Qingdao University, 266000 Qingdao, Shandong, China
Abstract
Background: The purpose of this meta-analysis was to assess the value of probiotics in the treatment of adult female bacterial vaginosis (BV). Methods: We looked for published randomized controlled trials in PubMed, Embase and The Cochrane Library from the inception dates of the database to January 3rd, 2022. We conducted the search focusing on the treatment of adult female BV with probiotics. Two independent researchers screened the literature, evaluated the trial quality and extracted the data according to the inclusion and exclusion criteria. The primary outcome was the ratio of patients with BV with recurrence according to Nugent score 7–10 for recurrence after treatment. After heterogeneity was assessed using Review Manager 5.4 software, meta-analysis and bias assessment were performed using Stata 17.0 software. Results: A total of 5 articles representing 425 patients were included in this meta-analysis. Compared with antibiotics alone or antibiotics combined with a placebo, probiotics or probiotics combined with antibiotics significantly reduced the rate of recurrence at around the 30th day (risk ratio (RR) 0.11; 95 % confidence interval (CI) 0.03–0.33). However, in the analysis of heterogeneity, we found that after 30 days, the therapeutic effect of probiotics decreased with the extension of follow-up time (RR 0.50; 95% CI 0.24–1.03), (RR 1.24; 95% CI 0.88–1.76). Conclusions: The short-term efficacy of probiotics in the treatment of BV in adult female patients may be satisfactory, but the long-term efficacy of probiotic therapy may be suboptimal and still requires validation by further clinical trials.
Keywords
- meta-analysis
- probiotic
- therapeutics
- vaginosis
- bacterial
Bacterial vaginosis (BV) is one of the most common reproductive diseases in adult women [1]. The worldwide incidence rate is generally over 20%, even exceeding 60% in certain parts of Africa [2]. A considerable number of patients with BV have no clinical symptoms. Fewer than 20% of patients showed obvious clinical symptoms, such as pain, itching, and burning [1, 3, 4]. While many asymptomatic and atypical patients are unaware of the harm of BV [5], BV may lead to endometritis and pelvic inflammatory disease (PID) [6, 7, 8], and may facilitate sexually transmitted infections (STIs) such as Chlamydia trachomatis, Neisseria gonorrhoeae and even human immune-deficiency virus (HIV) infection [2, 6, 9, 10]. In addition to infertility, BV may also be associated with a variety of adverse pregnancy outcomes, such as abortion, premature birth, and premature rupture of membranes (PROM) [3, 6, 11, 12].
BV can be diagnosed based on clinical or laboratory criteria [13, 14]. Due to
the complexity and cost of molecular testing, the commonly used diagnostic
standards are Amsel criteria and Nugent score [15, 16]. According to Amsel
criteria, the diagnosis of BV includes at least three of these four items: (1)
clue cell positive:
The etiology of BV is not entirely clear. The reduction of hydrogen peroxide
(H
It is worth noting that traditional metronidazole treatment has many side effects, such as nausea, and is prone to drug resistance [29]. Crucially, antibiotic therapy disregards changes of the vaginal microbial environment, and may further damage the vaginal ecosystem [30]. Studies have reported that although the cure rate of metronidazole in the treatment of BV is more than 70% within one month, over half of patients will still relapse within six months [31]. Thus, it is necessary to explore more safe and effective treatment methods.
According to the Food and Agriculture Organization (FAO) and the WHO, probiotic are defined as live micro-organisms which, when administered in adequate amounts, confer a health benefit to the host by food and Agriculture Organization (FAO) and World Health Organization (WHO) [32]. The concept of probiotics can be traced back to fermented dairy products in 1907 [33], while the term was first described in 1965 by Lilly et al. [34]. There are numerous kinds of probiotics, such as Lactobacillus, Bifidobacterium and Streptococcus [32, 35]. In light of their effectiveness and safety, probiotics are now widely used to treat various clinical diseases ranging from gastrointestinal diseases, urogenital infections, and cancer to periodontal diseases [32, 36, 37]. The application of probiotics in the treatment of BV may have the advantages of fewer side effects, lack of drug resistance and improvement of the vaginal ecological environment [15].
Despite numerous randomized controlled trials (RCTs) on probiotics in the treatment of BV, the efficacy of probiotics is still controversial. Some clinical trials have demonstrated that probiotics are beneficial to BV in adult women [20], but others dispute these claims [38, 39]. To assess the influence of probiotics on the treatment of BV, we performed a systematic review and meta-analysis of RCTs to evaluate the therapeutic value of probiotics in BV patients.
Our study strictly follows the statement guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) [40]. The protocol was registered on PROSPERO (CRD42022310132).
We searched the following databases each from inception to January 3rd, 2022: PubMed, Embase and The Cochrane Library. We also searched the MESH database for all words related to BV and probiotics. The terms associated with BV included “Vaginosis, Bacterial”, “Bacterial Vaginitides”, “Vaginitides, Bacterial”, “Bacterial Vaginosis”, “Vaginitis, Nonspecific”, “Nonspecific Vaginitis”, “Bacterial Vaginoses”, “Vaginoses, Bacterial”, “Bacterial Vaginitis” and “Vaginitis, Bacterial”. The words associated with “probiotics” included “Probiotics” and “Probiotic”. We then used subject words and free words to search the three databases. We used Boolean notation to combine subject words and free words for search strategy: “Vaginosis, Bacterial OR Bacterial Vaginitides OR Vaginitides, Bacterial OR Bacterial Vaginosis OR Vaginitis, Nonspecific OR Nonspecific Vaginitis OR Bacterial Vaginoses OR Vaginoses, Bacterial OR Bacterial Vaginitis OR Vaginitis, Bacterial” AND “Probiotics OR Probiotic”. The search strategies for RCTs included use of the RCTs filters for different databases on the Cochrane site.
Two researchers independently searched the literature and checked whether the documents were consistent. In the case of inconsistent literature search, the two authors solved it through negotiation. If it could not be solved through negotiation, the third researcher would make a decision.
Inclusion criteria: (1) study design: RCTs and two-armed studies; (2) study population: non-pregnant and non-lactating adult women diagnosed with BV by Amsel criteria or Nugent score and suffering from BV only; (3) intervention: use probiotics (regardless of the type, course and method used) only or in combination with tr aditional antibiotics; (4) controlled intervention: traditional antibiotics or a placebo or traditional antibiotics in combination with a placebo; (5) measurement of treatment outcome: Nugent score; (6) human research.
Exclusion criteria: (1) non RCTs: systematic reviews, comments, retrospective
studies, cohort studies, case reports, etc., animal experiments, one-arm studies;
(2) enrolled pregnant females or males, patients with the age
Two researchers independently read the titles and abstracts of all literature found after excluding duplicate literature. Subsequently, they eliminated the literature that obviously did not meet the requirements according to the inclusion and exclusion criteria. They then read the full text and determined the final inclusion in this study. Any disagreement was resolved through negotiation. If no agreement could not be reached through negotiation, it would be judged by the third researcher.
The following data were extracted from the final qualified literature: author, year of publication, country, sample volume (probiotic/control), age range, diagnostic standards, intervention measures (type, quantity, dosage form, frequency and course of treatment), control measures (type, quantity, dosage form, frequency and course of treatment), follow-up time and outcome assessed.
Two researchers used the same form to extract the above data independently. To ensure the accuracy and integrity of the extracted data, double check was carried out after the data extraction was completed. Disputes were settled by both researchers through discussion. If no agreement could be reached, it would be decided by the third researcher for arbitration.
The quality of the included RCTs was assessed using the Cochrane “risk of bias table” that was recommended by the Cochrane Handbook for Systematic Review. This evaluation scale consists of 7 items: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias) selective reporting (reporting bias) and other bias [41]. Each domain is divided into low risk of bias, unclear risk of bias and high risk of bias. The quality assessment was performed by two researchers independently and disagreements were adjudicated by consultation. The third researcher intervened when the negotiation could not be solved.
The RR (RR = risk of event in experimental group/risk of event in control group)
was used as an effective index, and all outcomes were expressed as RR and their
95% confidence interval (CI). RR
A total of 372 articles were identified after the initial database search according to the search strategy (PubMed: 114, Embase: 133, Cochrane: 125). Based on our inclusion and exclusion criteria, five RCTs consisting of 425 individuals were included in this study. A flow diagram of literature selection is shown in Fig. 1.
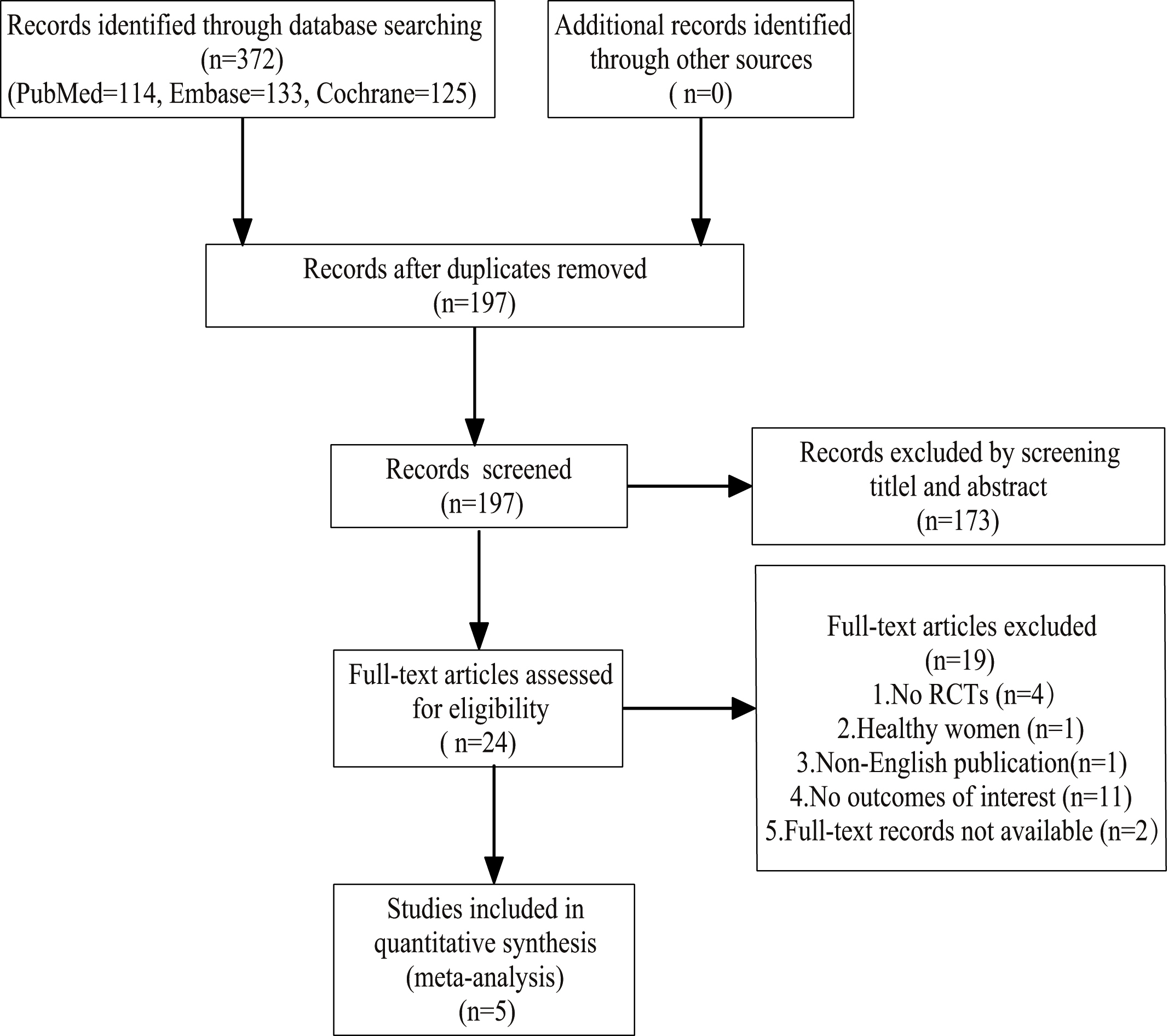 Fig. 1.
Fig. 1.Flow diagram of the study selection.
The quality assessments of these five RCTs are shown in Fig. 2,3 based on the Cochrane “risk of bias table”.
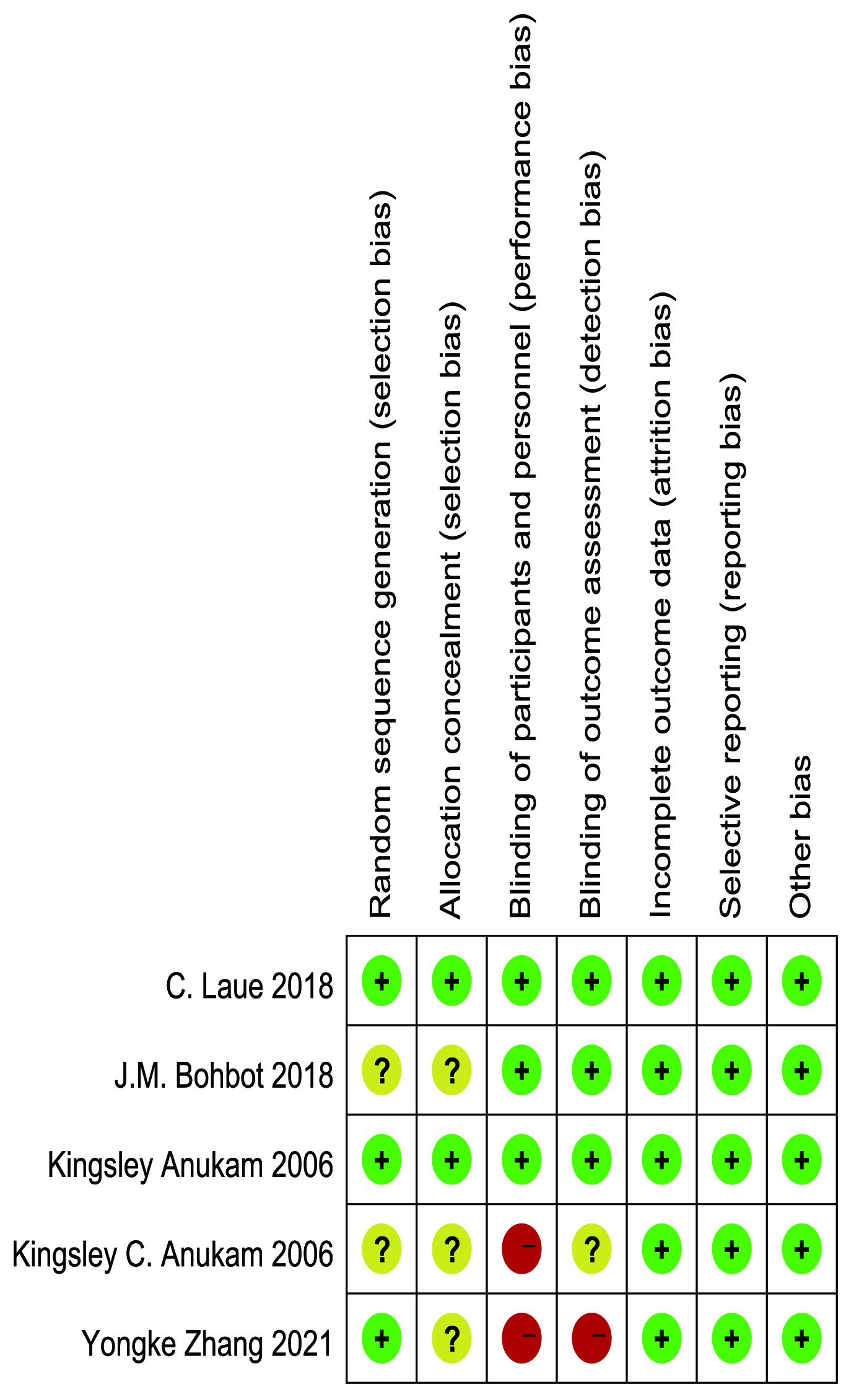 Fig. 2.
Fig. 2.Risk of bias summary of included studies.
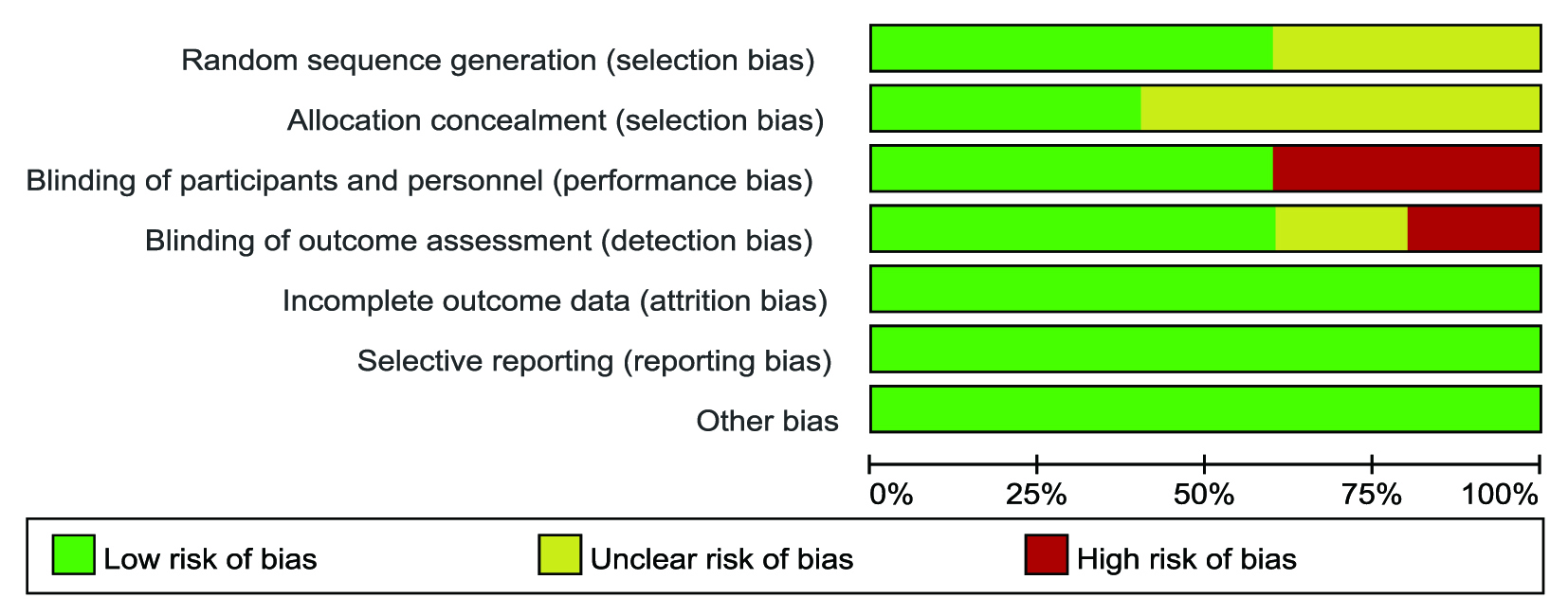 Fig. 3.
Fig. 3.Risk of bias graph of included studies.
Published between 2006 and 2021, the five studies included 425 adult female patients with BV; the main characteristics are presented in Table 1 (Ref. [39, 43, 44, 45, 46]). Two studies were conducted in Africa, two in Europe and one in China. The sample sizes ranged from 36 to 126, with 216 subjects in the experimental group and 209 subjects in the control group. As for type of intervention, three experiments applied oral, and two applied vaginal insertion. One RCT compared probiotics with antibiotics, one compared the combination of probiotics and antibiotics with antibiotics alone, while the remaining three compared the combination of probiotics and antibiotics with the combination of antibiotics with a placebo. The follow-up ranged from 4 weeks to 112 days.
| ID | Author | Publication year | Study type | Country | Sample size (probiotic/control) | Age | Diagnostic standards | Intervention measures | Dosage form and use time (probiotic) | Follow-up | Outcome assessed (interested) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Probiotic group | Control group | |||||||||||
| 1 | Kingsley Anukam [43] | 2006 | DB-RCT | Nigeria | 125 (65/60) | 18 to 44 | Symptoms and signs, a positive Nugent and BV Blue test score | Took metronidazole (1 g, days 1–7) plus Lactobacillus (days 1–30) orally | Took metronidazole (1 g, days 1–7) plus placebo (days 1–30) orally | Oral capsules, 30 days | 30 days | Nugent score |
| 2 | Kingsley C. Anukam [44] | 2006 | OL-RCT | Nigeria | 40 (20/20) | 18 to 50 | Symptoms and signs, a positive Nugent score, |
Inserted vaginally two gelatin capsules containing L. rhamnosus GR-1 and L. reuteri RC-14 (1 × 10 |
Applied 0.75% metronidazole vaginal gel to the vagina twice a day (once in the morning, once in the evening) for 5 days | Vaginal capsules insertion, 5 days | 30 days | Nugent score |
| 3 | J.M. Bohbot [45] | 2018 | DB-RCT | France | 98 (50/48) | Took metronidazole orally twice a day for 7 days and then used vaginal capsules of L. crispatus IP 174178 (10 |
Took metronidazole orally twice a day for 7 days and then used vaginal capsules of a placebo for 56 days | Vaginal capsules insertion, 56 days | 112 days | Nugent score | ||
| 4 | C. Laue [46] | 2018 | DB-RCT | Germany | 36 (18/18) | Took metronidazole for 7 days (2 × 500 mg/d) orally and then consumed twice daily verum, verum was 125 g yoghurt containing (besides Lactobacillus delbrueckii ssp. bulgaricus and Streptococcus thermophilus) living strains Lactobacillus crispatus LbV 88 (DSM 22566), Lactobacillus gasseri LbV 150N (DSM 22583), Lactobacillus jensenii LbV 116 (DSM 22567) and Lactobacillus rhamnosus LbV96 (DSM 22560), each 1 × 10 |
Took metronidazole orally for 7 days (2 × 500 mg/d) and then consumed twice daily placebo (125 g chemically acidified milk) | Oral yoghurt, 28 days | 4 weeks | Nugent score | ||
| 5 | Yongke Zhang [39] | 2021 | OL-RCT | China | 126 (63/63) | 18 to 65 | A positive Nugent score | Received orally administered probiotic drinks containing L.rhamnosus GR-1 and L reuteri RC-14 ( |
Received metronidazole vaginal suppositories only (0.2 g per day, for 7 days) | Oral drinks, 7 days | 90 days | Nugent score |
DB-RCT, double-blinded randomized controlled trial; OL-RCT, open label randomized controlled trial; BV, bacterial vaginosis.
The treatment results of all included RCTs are shown in Table 2 (Ref. [39, 43, 44, 45, 46]). The pooled results from the random-effects model combining the RR
for the recurrence rate of BV are shown in Fig. 4 (Ref. [39, 43, 44, 45, 46]).
The combined results of the five studies show that probiotics supplementation or
treatment alone can significantly reduce the recurrence rate of BV in adult
female patients (RR 0.35; 95% CI 0.11–1.14). After heterogeneity test, it was
found that the heterogeneity of the five articles was statistically significant
(The Q statistic p
| ID | Author | Publication year | Probiotic group | Control group | ||
|---|---|---|---|---|---|---|
| Number of recurrent patients (Nugent score 7–10) | Total number of patients | Number of recurrent patients (Nugent score 7–10) | Total number of patients | |||
| 1 | Kingsley Anukam [43] | 2006 | 0 | 49 | 17 | 57 |
| 2 | Kingsley C. Anukam [44] | 2006 | 2 | 17 | 9 | 18 |
| 3 | J.M. Bohbot [45] | 2018 | 8 | 39 | 16 | 39 |
| 4 | C. Laue [46] | 2018 | 0 | 16 | 4 | 17 |
| 5 | Yongke Zhang [39] | 2021 | 33 | 52 | 24 | 47 |
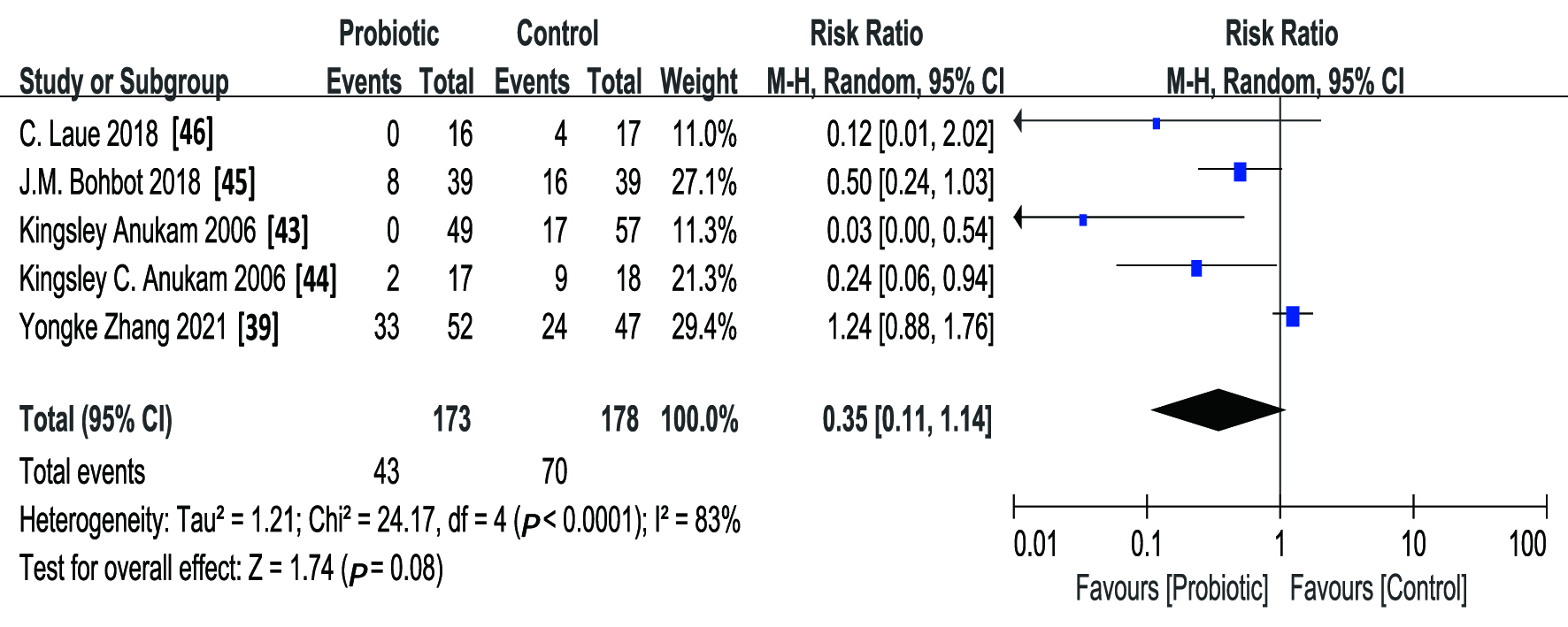 Fig. 4.
Fig. 4.Forest plot showing the effect of probiotics on the adult female patients with BV in five RCTs.
Combining the forest plot, the L’Abbe plot (Fig. 5), the Galbraith radial plot (Fig. 6) and the baseline characteristics of the studies, it appeared there was a strong possibility of heterogeneity in Bohbot’s study and Zhang’s study. The follow-up time between intervention and outcome evaluation was more than 30 days. Excessive follow-up time may affect the final efficacy evaluation. As one of the two studies showed results in complete variance from other studies (RR 1.24; 95% CI 0.88–1.76), we deleted these two RCTs (Fig. 7, Ref. [39, 43, 44, 45, 46]).
 Fig. 5.
Fig. 5.L’Abbe plot showing the heterogeneity of the five RCTs.
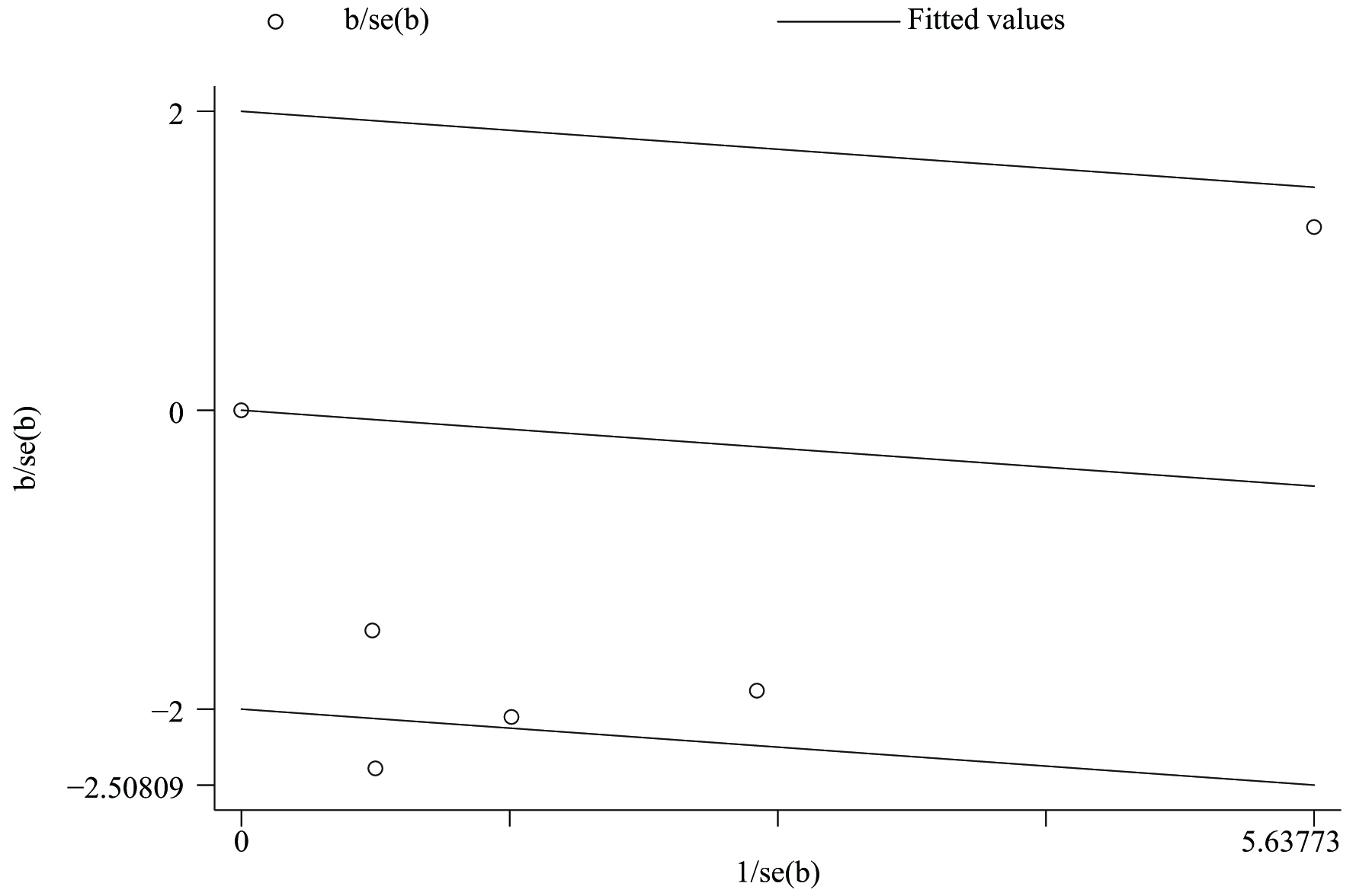 Fig. 6.
Fig. 6.Galbraith radial plot showing the heterogeneity of the five RCTs.
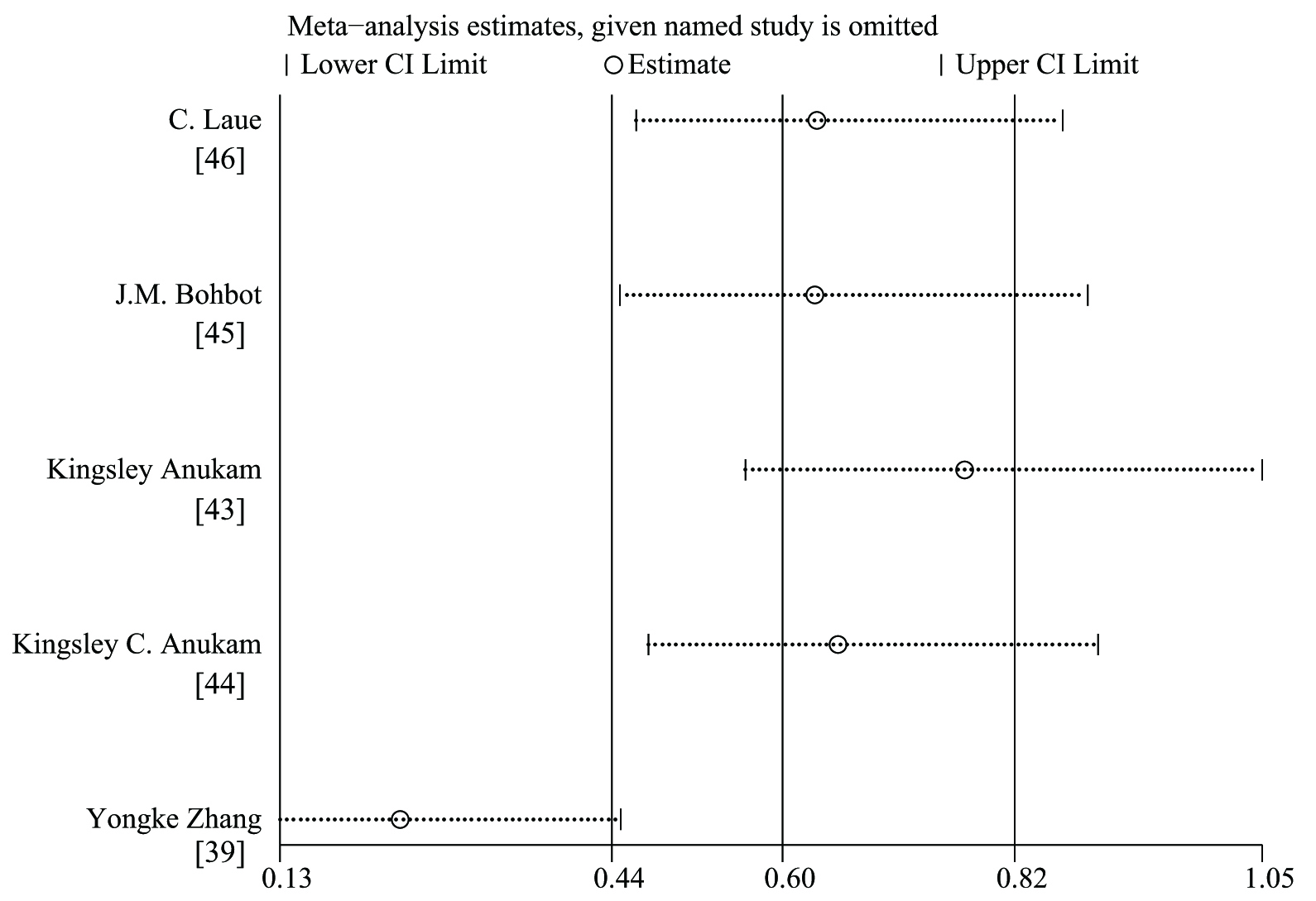 Fig. 7.
Fig. 7.Sensitivity analysis of the effect of probiotics on the adult female patients with BV in five RCTs.
After excluding these two studies, we found that the heterogeneity of the
remaining three RCTs was not statistically significant (The Q statistic
p = 0.38
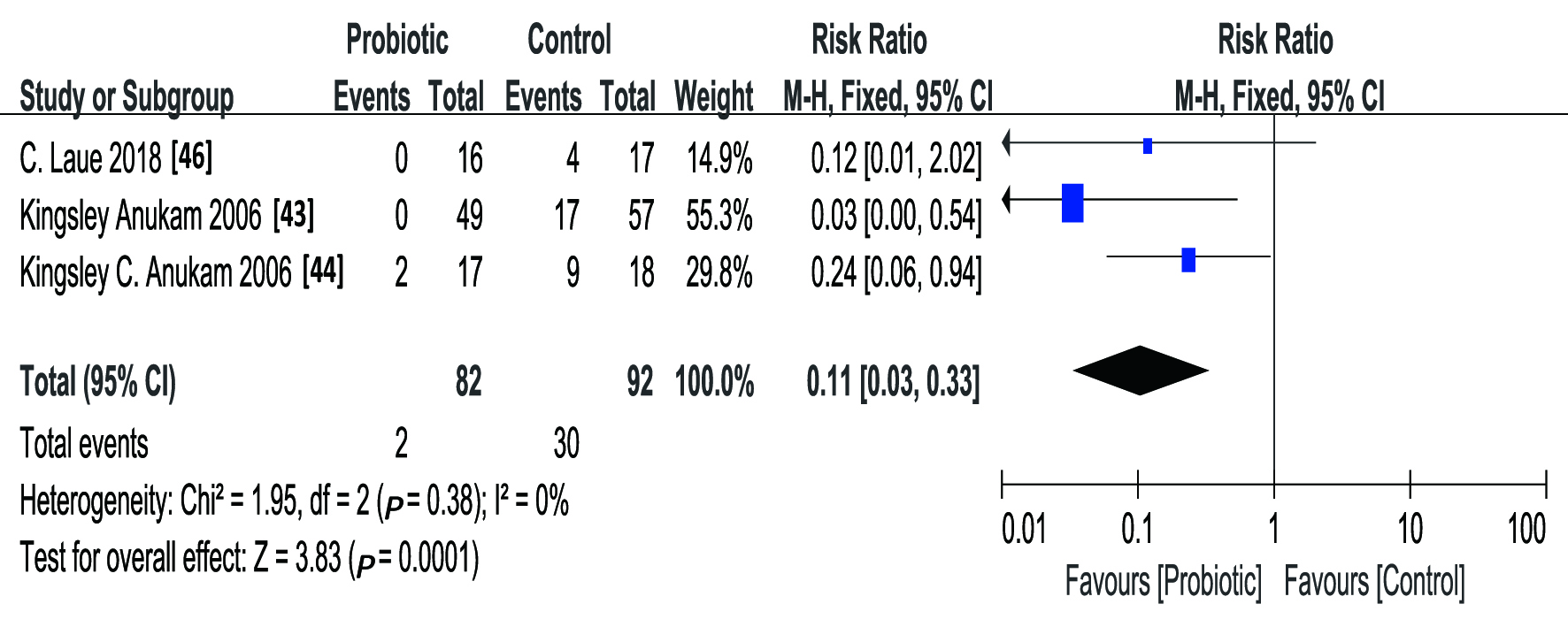 Fig. 8.
Fig. 8.Forest plot showing the effect of probiotics on the adult female patients with BV in three RCTs.
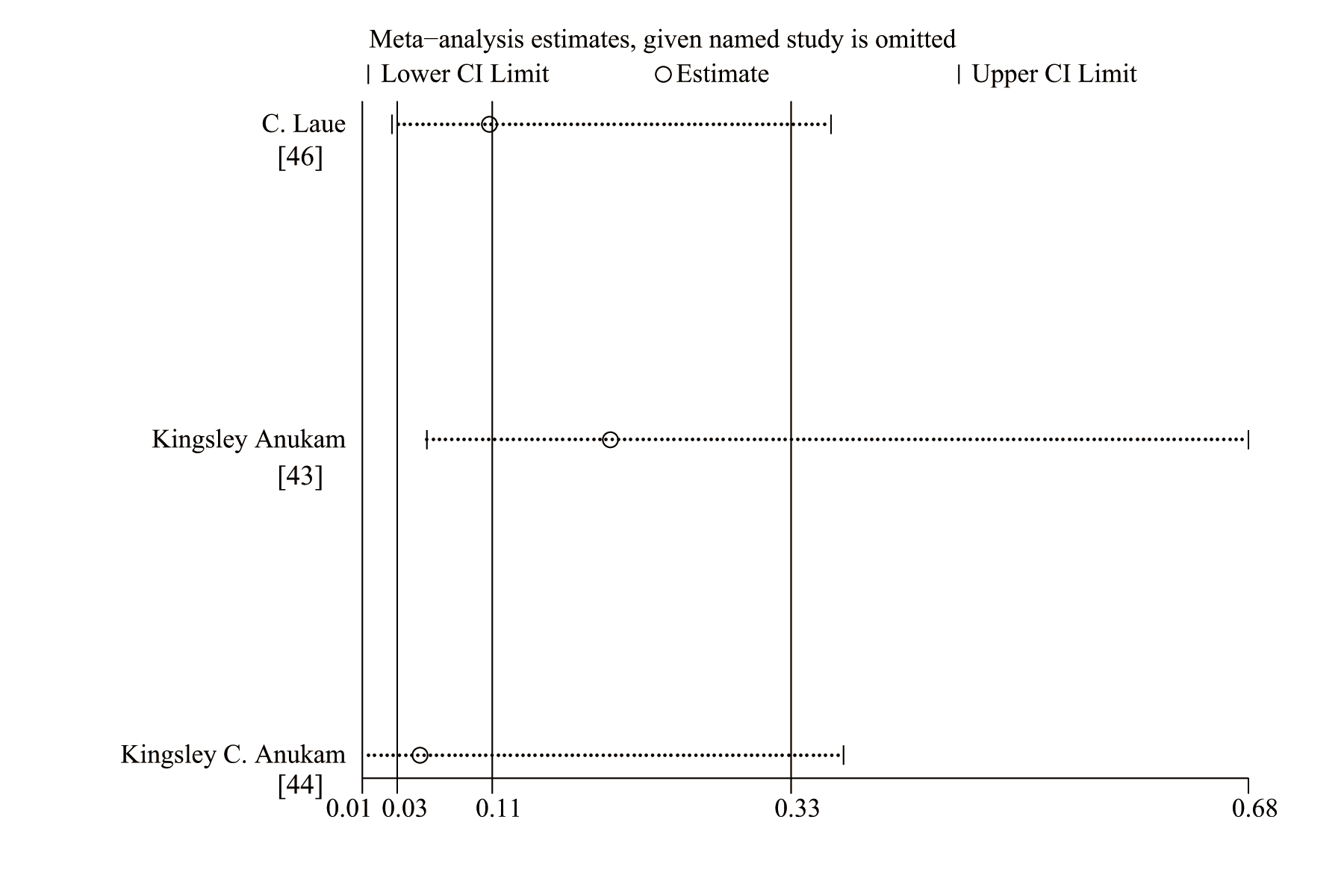 Fig. 9.
Fig. 9.Sensitivity analysis of the effect of probiotics on the adult female patients with BV in three RCTs.
The publication bias of the three RCTs was tested by funnel plot and Begg’s test. No publication bias was observed in the funnel plot (Fig. 10) and Begg’s test (Z = 0.00, p = 1.000).
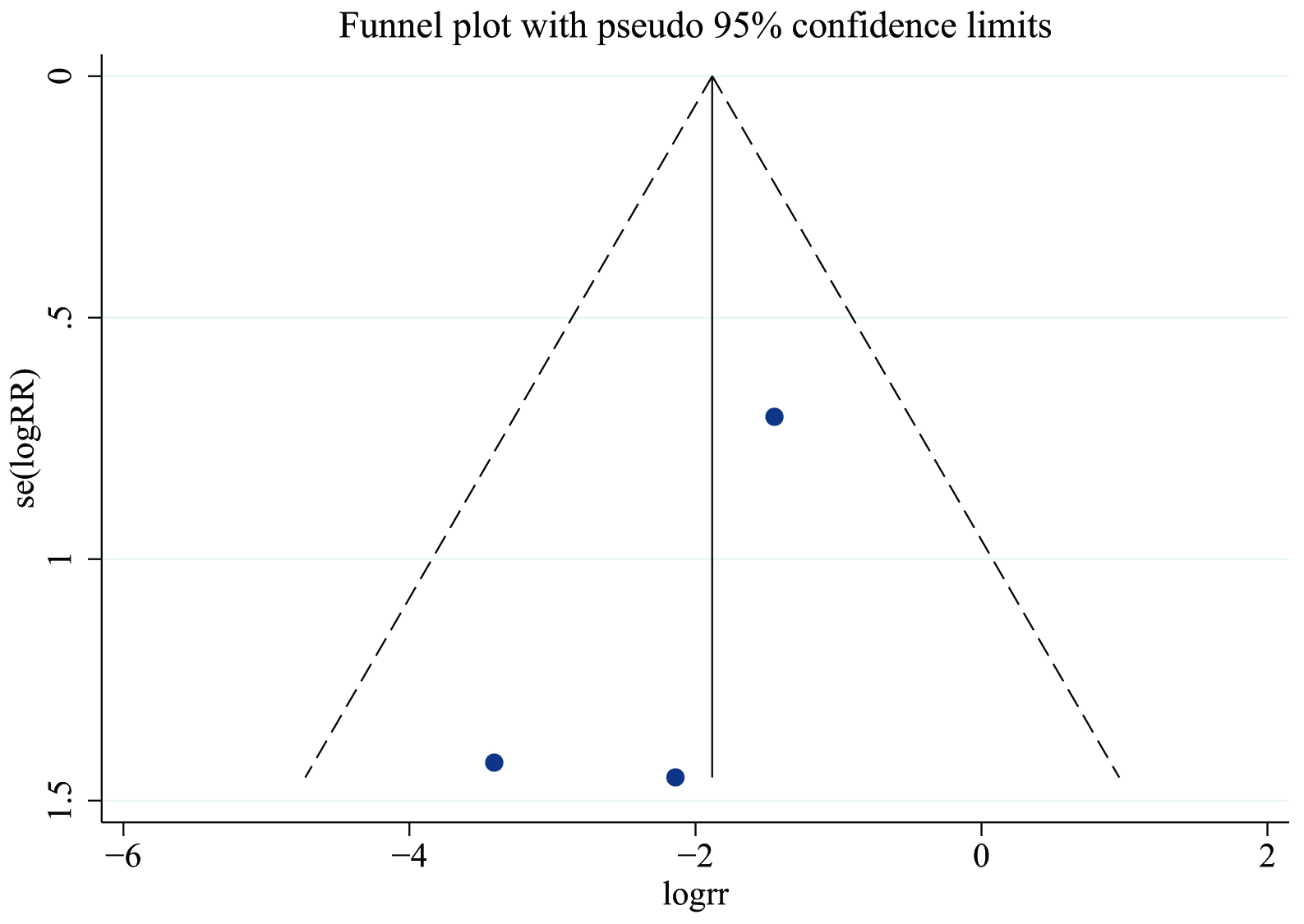 Fig. 10.
Fig. 10.Funnel plot showing the publication bias among three RCTs.
It is well known that as the antibiotics used to treat adult female BV are prone to drug resistance and lead to a high recurrence rate, probiotic treatment has risen to the fore [47, 48]. However, probiotics are still controversial in the treatment of adult female BV [15]. To evaluate the efficacy of probiotics in this context, we performed the present meta-analysis of five RCTs based on inclusion and exclusion criteria [39, 43, 44, 45, 46]. The results of our analysis show that probiotics alone or in combination with antibiotics in the treatment of adult female BV may be effective in the short term, but long-term effects appear to be less promising.
After excluding heterogeneity in this meta-analysis, it was found that patients with BV receiving probiotic supplementation or treatment alone had a lower recurrence rate than those receiving antibiotics alone or antibiotics combined with a placebo.
The efficacy of probiotics can be explained by exhibiting antagonistic knacks
against BV pathogens: antibacterial, antibiofilm, anti-colonization or
anti-adhesion, co-aggregation, and host immunomodulation [49]. Antibacterial
means probiotics can ferment the glycogen in the vaginal tract to produce lactic
acid. The lactic acid reduces the vaginal pH to
Our meta-analysis found two articles with strong heterogeneity [39, 45]. Further analysis determined that the intervention measures in both articles entailed a combination of probiotics and antibiotics. In Bohbot’s study, the control group used antibiotics plus a placebo for 56 days and was surveilled for 112 days [45]. In Zhang’s study, the control group used antibiotics alone and took probiotics orally for 7 days with surveillance for 90 days [39]. The recurrence risk of the former (RR 0.50; 95% CI 0.24–1.03) was significantly higher than that after merger (RR 0.11; 95% CI 0.03–0.33). The latter (RR 1.24; 95% CI 0.88–1.76) even appeared in contradiction with the results after the merger (RR 0.11; 95% CI 0.03–0.33). Compared with the other three studies, patients were followed up for more than 30 days from the beginning of the intervention to the evaluation of the final results in these two studies, regardless the duration of intervention was; the follow-up time of the latter exceeded 60 days. Two of the studies assessed the results immediately after stopping the intervention for 28 days and 30 days, respectively; the recurrence risk of these studies was the lowest among the five (RR 0.12; 95% CI 0.01–2.02), (RR 0.03; 95% CI 0.00–0.54) [43, 46]. The results of our analysis suggest that in the case of the same course of treatment, the shorter the follow-up time, the better the final curative effect. In the case of the same follow-up time, the longer the medication time, the better the effect may be reflected. For the probiotics plus antibiotics group, excessive follow-up time may even weaken the therapeutic effect of antibiotics, perhaps because probiotics and antibiotics may be maintained at a high level due to the short-term follow-up after the intervention, whereas long-term follow-up reduces the level of probiotics. However, antibiotics kill not only pathogens, but also inhibit the growth of remaining probiotics, and the lack of supplementation of probiotics amplifies this inhibitory effect [59]. From our point of view, the high recurrence rate of BV in these two studies may be related to the long follow-up time. This may suggest that the short-term effect of probiotics (supplemental or alone) in the treatment of BV is worth affirming, but the long-term effect is suboptimal. Strengthening and consolidating the efficacy of probiotics may require increasing the course of treatment. Further clinical trials are necessary to validate our findings.
Our study summarized RCTs with highly consistent baseline population characteristics and found via meta-analysis that probiotics had a positive effect on the treatment of BV in adult women. However, we also found that this positive effect may gradually decline with the termination of treatment, until it disappears, and even may have the opposite effect.
This discrepancy may lead to the design and implementation of more RCTs to determine the exact efficacy and optimal course of probiotics in the treatment of BV in adult women. Several limitations should be acknowledged. (1) Due to the heterogeneity of patients as well as quantities and administration methods of probiotics and antibiotics used in each study, and due to the limitation of the number of studies, we were unable to conduct a detailed subgroup analysis. (2) Other factors related to probiotic consumption, such as health level and self-care awareness, may affect the research results. (3) Most studies observed only the short-term efficacy of patients within 30 days, without long-term dynamic efficacy evaluation to further analyze the efficacy changes of probiotic supplementation or treatment alone in adult female patients with BV. (4) The antibiotics used in the included studies were metronidazole, and there was a lack of comparative study on the use of other kinds of antibiotics. (5) Language bias may arise from the inclusion of only English literature.
In the future, more high-quality research must focus on standardizing the types, routes, doses, time and treatment of probiotics and antibiotics. In addition, attention should be paid to excluding the influence of other confounding factors related to intervention. The evaluation of research results can be dynamic and continuous for an extended period. Further studies can also compare differences in race, types of antibiotics, probiotic strains. Lastly, future research should address the side effects of probiotics, an area that has been neglected.
Currently, the limited evidence suggests that probiotics alone or as a supplement to antibiotics in adult women with BV is beneficial in the short term, though long-term effects may be unsatisfactory. However, given the limitations of this study, the results should be treated with caution. Additional large-sample, well-designed and high-quality RCTs are urgently needed to further explore the short-term and long-term efficacy of probiotics in the treatment of adult female BV.
YPS designed the study, performed the literature search, screened literature, extracted data, assessed quality, analyzed the data and drafted the manuscript. ZCC performed the literature search and screened literature. ZXD extracted data and assessed quality. YSY analyzed the data and drafted the manuscript. APC evaluated methodological quality and revised the manuscript. The final version was confirmed by all authors for submission. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
We would like to express our gratitude to all those who helped us during the writing of this manuscript.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.










